1 Center for Reproductive Medicine, Department of Obstetrics and Gynecology, West China Second University Hospital of Sichuan University, 610000 Chengdu, Sichuan, China
2 Key Laboratory of Birth Defects and Related Diseases of Women and Children (Sichuan University), Ministry of Education, 610000 Chengdu, Sichuan, China
†These authors contributed equally.
Abstract
Background: To evaluate whether the
addition of human menopausal gonadotropin (HMG) during the early follicular phase
of controlled ovarian stimulation improves clinical outcomes in patients
classified as group 4 on the Patient-Oriented Strategy Encompassing
IndividualizeD Oocyte Number (POSEIDON) classification. Methods: A
prospective, randomized, and non-blind controlled trial was conducted, involving
172 patients seeking infertility treatment with an indication for in vitro
fertilization (IVF) or intracytoplasmic sperm injection (ICSI). Among them, 78
patients were randomly assigned to the HMG/follicle stimulating hormone (FSH)
group, receiving 75 IU of HMG for FSH on either day 2 or day 3 of menstruation,
while 94 patients were assigned to the FSH monotherapy group. Results:
The total dose of gonadotropin used in the HMG/FSH group (2510.77
Keywords
- assisted reproduction technology
- controlled ovarian stimulation
- HMG
- POSEIDON
In vitro fertilization and embryo transfer (IVF-ET) is widely applied for treating infertility. However, a significant proportion of patients, ranging from 9% to 24%, experience poor ovarian response (POR) during controlled ovarian stimulation (COS) in IVF treatment. POR is characterized by a low number of oocytes, high cycle cancellation rates, and reduced live birth rates [1]. To address the need for more homogeneous patient populations in clinical studies, the concept of Patient-Oriented Strategies Encompassing IndividualizeD Oocyte Number (POSEIDON) was introduced in 2016 [2]. According to the POSEIDON criteria, group 4 refers to women aged 35 years or older with poor ovarian reserve parameters, such as an antral follicle count (AFC) below 5 or anti-Mullerian hormone (AMH) levels below 1.2 ng/mL [2, 3]. This particular group accounts for approximately 55% of patients with POR [4] and poses a significant challenge for reproductive clinicians. According to the “two cell, two gonadotropins” hypothesis, follicle stimulating hormone (FSH) and luteinizing hormone (LH) both play critical roles during the growth and maturation of oocytes in natural menstrual cycles. LH is generally not administered in standard COS protocols, as exogenous FSH is sufficient to generate an adequate follicular response. However, in recent years, exploration of LH supplementation in patients with POR has yielded controversial results. LH supplementation may reduce the apoptosis of granulosa cells, increase FSH receptor expression and oocyte quality, and promote embryo implantation by the decidualization of endometrial stromal cells [5, 6, 7]. Recombinant LH (rLH) supplementation appears to be beneficial for elderly women [8, 9]. However, the largest randomized controlled trial performed to date did not find an increased number of retrieved oocytes (primary efficacy endpoint) when rLH was added to FSH for COS in patients with POR compared with FSH monotherapy [10]. Further, a related systematic review and network meta-analysis did not show better clinical outcomes by LH supplementation in patients with POR [11].
Urinary human menopausal gonadotropin (HMG) is a type of gonadotropin that combines the activities of both FSH and LH. It is commonly used in COS for infertility treatment. HMG has been suggested to have similar benefits as rLH supplementation in POR patients. A recent retrospective study demonstrated that commencing HMG during the early follicular phase resulted in increased live birth rates in POSEIDON groups 3 and 4, which are classified as poor responders [12]. However, an observational case-control study did not find any improvement in IVF outcomes when HMG was added to FSH compared to rLH supplementation in long GnRH agonist protocols. Currently, there are limited prospective studies that have investigated the use of HMG supplementation initiated during the early follicular phase. Therefore, this prospective, randomized, controlled pilot study aimed to evaluate whether the addition of HMG to FSH on days 2–3 of menstruation would enhance IVF outcomes in patients classified as POSEIDON Group 4.
This single-center, non-blind, randomized
clinical pilot trial (https://www.chictr.org.cn identifier: ChiCTR2100043040) was conducted at the West
China Second University Hospital of Sichuan University. As a feasibility study,
we estimated the number of participants required as around 20% of the number
required for future definitive randomized controlled trial (RCT). A total of 177
patients with indications for IVF/intracytoplasmic sperm injection (ICSI) seeking
infertility treatment were enrolled between April 2021 and July 2022. Patients were invited to participate if they
were between the ages of 35 and 44 years and were classified as having poor
ovarian reserve parameters (AFC
On Day 2 (D2) or Day 3 (D2) of menstruation, participants were randomized into the HMG/FSH or FSH alone groups using a computer-generated randomization list. The physicians and the participants were not blinded to the treatments.
A long gonadotropin-releasing hormone (GnRH) agonist or antagonist protocol was implemented. The GnRH agonist (H20140298, Triptorelin Acetate for Injection, Ipsen Pharma Biotech, Paris, France) was administered in the previous mid-luteal phase until the human chorionic gonadotropin (hCG) trigger. A GnRH antagonist (H20140476, Cetrorelix, Merck Serono, Amsterdam, the Netherlands) was administered in a fixed or flexible manner. On D2 or D3 of menstruation, women who were randomized to the HMG/FSH group were administered HMG of menopausal origin (H10940097, Menotropins for Injection, Lizhu Group, Zhuhai, China) 75 IU per day in addition to FSH until hCG was triggered. The control group was administered FSH alone. The starting doses of FSH ranged from 225 to 300 IU by experienced physicians based on these individual patient characteristics including the patient’s age, ovarian reserve, body mass index (BMI), hormone levels, and previous response to fertility treatments. FSH doses were adjusted according to the ovarian response based on monitored follicle development and serum sex steroids. When 1–3 leading follicles reached a size of 18 mm or greater, either 5000–10,000 IU of hCG (H44020674, Chorionic Gonadotrophin for Injection, manufactured by Lizhu Group, Zhuhai, China) or 0.2 mg of Triptorelin (H20160237, Triptorelin Acetate for Injection, produced by Ipsen Pharma Biotech, Paris, France) was administered. Approximately 35–36 hours after the administration of the trigger, follicular aspiration was performed under sedation. Fertilization of the retrieved oocytes was carried out using either conventional IVF or ICSI, depending on the results of the semen analysis and previous fertilization outcomes. Embryos were subsequently cultured in vitro until they reached the cleavage or blastocyst stage. Their quality was assessed based on blastomere morphology and the proportion of detached nuclear fragments [13]. A good-quality embryo on day 3 was defined as having 6–10 cells with less than 20% fragmentation. The embryos deemed suitable were then cryopreserved through vitrification.
Luteal progesterone support was administered in the form of progesterone gel (H20140552, Crinone 8%, Fleet Laboratories Limited, Hertfordshire, UK) 90 mg once daily, plus dydrogesterone tablets (HJ20170221, Solvay Pharmaceuticals B.V, OLST, the Netherlands) 20 mg once daily, or progesterone injection (H31021401, Shanghai General Pharmaceutical, Shanghai, China) 40 mg twice daily for fresh cycles on the day of oocyte retrieval.
Patients were recommended to undergo freezing of all embryos if they exhibited a
high progesterone concentration (
The primary focus of this study was to determine the ongoing pregnancy rate per
first embryo transfer cycle. Ongoing pregnancy was specifically defined as the
presence of fetal heart motion at 11–12 weeks of gestation. Secondary outcomes
examined in the study included biochemical pregnancy, clinical pregnancy rate
(CPR), and early miscarriage occurring before the 12th week of gestation.
Biochemical pregnancy was defined as a human chorionic gonadotropin level
exceeding 30 mIU/mL, measured 14 days following embryo transfer. Clinical
pregnancy was determined by the presence of an intrauterine gestational sac
observed on ultrasound after 6–7 weeks of gestation. Early miscarriage (
Descriptive statistics were utilized to provide an overview of the participants’ biological, socioeconomic, and lifestyle characteristics. Continuous variables are presented as means with standard deviations, while categorical variables are presented as counts and percentages. To assess the significance of the differences observed, appropriate statistical tests such as the Chi-square test, Student’s t-test, and Fisher’s exact test were performed. These tests were chosen based on the nature of the variables being compared. All statistical analyses were conducted using SPSS version 26.0 software (SPSS Inc., Chicago, IL, USA). A p-value less than 0.05 was considered statistically significant, indicating a significant difference between groups or variables under investigation.
This study was conducted in adherence with the ethical principles outlined in the Declaration of Helsinki for Medical Research involving Human Subjects. The research protocol received approval from the Ethics Committee of West China Second Hospital, Sichuan University, with the ethical permit number 2021029. Prior to their involvement in the study, all participants provided informed consent, demonstrating their understanding of the study’s objectives, procedures, and potential risks or benefits. To safeguard patient privacy, all data collected during the study were anonymized to ensure confidentiality.
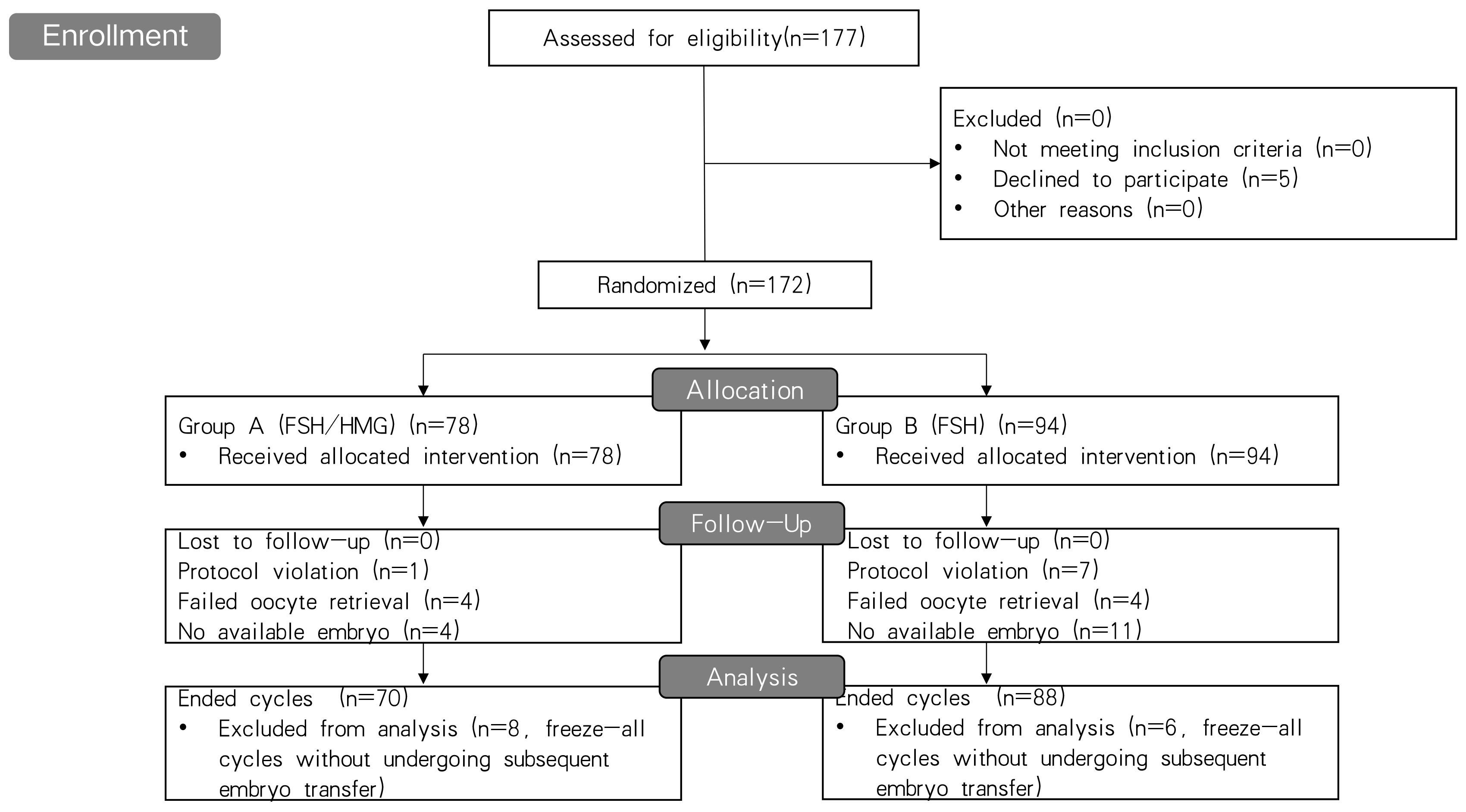
A flowchart of the patient recruitment process for this study is shown in Fig. 1. A total of 172 patients randomized into two groups were included in the analysis; 94 were assigned to the FSH monotherapy group and 78 to the HMG/FSH group. No patients were lost to follow-up. Eight cycles violated the assigned protocols and there were 14 cases freezing all embryos without undergoing subsequent frozen embryo transfer for personal or medical reasons in the two groups. Finally, 23 cycles in the two groups were cancelled because of either oocyte retrieval failure or a lack of available embryos and one 135 cases underwent embryo transfer. The cycles with embryo transfer and cancelled cycles are defined as ended cycles.
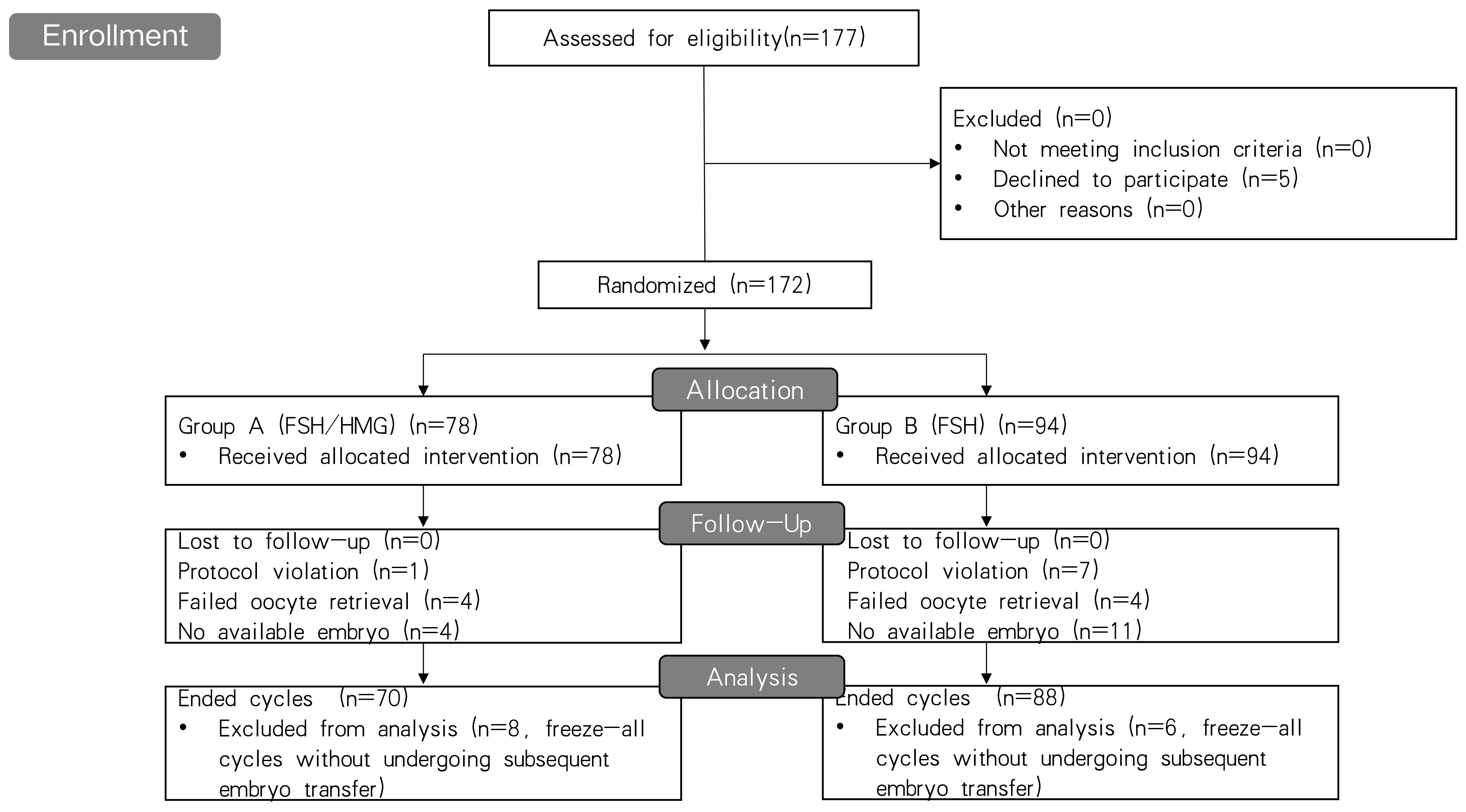 Fig. 1.
Fig. 1.Flowchart of the patient recruitment process for this study. FSH, follicle stimulating hormone; HMG, human menopausal gonadotropin.
Table 1 presents the baseline characteristics of the study participants. The average age of the patients was 38.1 years, with a mean AMH level of 0.67 ng/mL and a mean antral AFC of 4.58. The groups were comparable in terms of age, body mass index, blood pressure, type of infertility, infertility duration, AMH level, basal hormone level, and AFC measured on Day 2–3.
| Total | FSH | FSH/HMG | ||
| N = 172 | n = 94 | n = 78 | ||
| Age (years) | 38.07 |
38.04 |
38.10 | |
| BMI (kg/m |
22.51 |
22.56 |
22.45 | |
| Blood pressure (mmHg) | ||||
| Systolic | 114.80 |
114.87 |
114.72 | |
| Diastolic | 74.67 |
74.86 |
74.44 | |
| Type of infertility n (%) | ||||
| Primary | 51 (29.7) | 29 (30.9) | 22 (28.2) | |
| Secondary | 121 (70.3) | 65 (69.1) | 56 (71.8) | |
| Duration of infertility (years) | 3.62 |
3.63 |
3.61 | |
| AMH level (ng/mL) | 0.67 |
0.67 |
0.67 | |
| AFC | 4.74 |
4.86 |
4.58 | |
| E2 level on D2–3 (ng/mL) | 41.29 |
40.99 |
41.65 | |
| FSH level on D2–3 (mIU/mL) | 10.17 |
10.14 |
10.20 | |
| LH level on D2–3 (mIU/mL) | 3.57 |
3.69 |
3.45 | |
Data are reported as mean
Table 2 shows the COS outcomes of the two
groups. The majority of the COS protocols in both groups were antagonist
protocols (97%). The total dose of gonadotropin in HMG/FSH group was
significantly higher than that in FSH alone group (2510.77
| FSH | FSH/HMG | p | ||
| n = 94 | n = 78 | |||
| Start dose of Gn (IU) | 283.78 |
285.58 |
0.423 | |
| Total Gn dose (IU) | 2310.98 |
2510.77 |
||
| Duration of COS (days) | 8.71 |
9.36 |
0.074 | |
| Antagonist protocol | 8.70 |
9.32 |
||
| Agonist protocol | 9.50 |
11.00 |
||
| E2 level on the day of hCG (pg/mL) | 1053.07 |
1098.95 |
0.951 | |
| P4 level on the day of hCG (ng/mL) | 0.64 |
0.68 |
0.807 | |
| LH level on the day of hCG (mIU/mL) | 3.54 |
3.58 |
0.796 | |
| Endometrium thickness on the day of hCG (mm) | 9.41 |
9.63 |
0.033 | |
| No. of developed follicles |
3.59 |
3.39 |
0.180 | |
| No. of retrieved oocytes | 3.44 |
3.79 |
0.594 | |
| Fertilization rate (%) | 67.3 | 63.3 | 0.930 | |
| No. of fertilized oocytes | 2.41 |
2.41 |
0.284 | |
| IVF | 2.34 |
2.17 |
0.016 | |
| ICSI | 2.64 |
3.25 |
0.577 | |
| No. of good quality embryos | 1.16 |
1.43 |
0.280 | |
| Cancelation rate | 15 (16.0) | 8 (10.3) | 0.247 | |
| Failed oocyte retrieval | 4 | 4 | ||
| No available embryo | 11 | 4 | ||
| Cycles reaching embryo transfer | 73 | 62 | 0.741 | |
| D3 | 68 | 58 | ||
| D5–6 | 6 | 4 | ||
| No. of embryos transferred | 1.53 |
1.58 |
0.247 | |
| Fresh embryos | 1.54 |
1.55 |
0.975 | |
| Frozen embryos | 1.47 |
1.67 |
0.186 | |
Data are reported as mean
The clinical outcomes of all participants are shown in Table 3. There were no statistically significant differences between the groups in terms of ongoing pregnancy rates, biochemical pregnancy rates, or clinical pregnancy rates per end-cycle or embryo transfer cycle.
| rFSH | rFSH/HMG | p | |
| n = 94 | n = 78 | ||
| Biochemical pregnancy rate/ended cycles |
28/88 (31.8) | 22/70 (31.4) | 0.958 |
| Clinical pregnancy rate/ended cycles | 26/88 (29.5) | 20/70 (28.6) | 0.894 |
| Early pregnancy loss rate/ended cycles | 3/88 (3.4) | 1/70 (1.4) | 1 |
| Ongoing pregnancy rate/ended cycles | 23/88 (26.1) | 19/70 (27.1) | 0.887 |
| Biochemical pregnancy rate/embryo transfer | 28/73 (38.4) | 22/62 (35.5) | 0.731 |
| Clinical pregnancy rate/embryo transfer | 26/73 (35.6) | 20/62 (32.3) | 0.682 |
| Early pregnancy loss rate/embryo transfer | 3/73 (4.1) | 1/62 (1.6) | 1 |
| Ongoing pregnancy rate/embryo transfer | 23/73 (31.5) | 19/62 (30.6) | 0.914 |
Data are reported as number/number (percentage). rFSH, recombinant follicle stimulating hormone.
In this prospective randomized study, we compared the efficacy of HMG supplementation with FSH and FSH alone during COS in patients classified as POSEIDON Group 4. Overall, our study showed that there were no significant beneficial effects of HMG addition.
Based on the two-cell two-gonadotropin concept, LH supplementation plays a crucial role in stimulating the conversion of cholesterol into androgens in theca cells during the early follicular stage. This increased intraovarian androgen production stimulates the expression of FSH receptors in granulosa cells, working synergistically with insulin-like growth factor 1 (IGF1) to promote follicle growth. Animal models have shown that androgens play a role in stimulating early primate follicle development [14]. During the mid-follicular phase, LH binds to LH receptors on granulosa cells to sustain FSH-dependent activity, which includes induction of aromatase, release of growth factors, and regulation of final follicle and oocyte maturation [15, 16]. Therefore, LH serves dual roles during the process of folliculogenesis.
In clinical practice, LH preparations are supplemented during the early or mid-phase of follicular development for various purposes. In this prospective study, we added HMG at an early stage to improve the reaction and outcomes of COS. The number of retrieved oocytes and good-quality embryos showed an increasing trend in the HMG supplementation group compared to the FSH alone group, but did not reach statistical significance. A similar trend was observed in Musters et al.’s [17] RCT, in which they added rLH to FSH at the start of stimulation in women with poor ovarian reserve, and found that the number of retrieved oocytes and top-quality embryos per woman showed an increasing trend, although not statistically significant compared with FSH alone. However, another large RCT conducted by Humaidan et al. [10] did not show similar results when rLH was supplemented with FSH in patients.
HMG is a common type of gonadotropin widely used in COS which exhibits both FSH and LH activities. LH activity in the HMG is induced by the addition of human chorionic gonadotropin (hCG) glycoprotein, which shares a common alpha subunit with LH and stimulates the LH receptor [18]. In the current prospective study, HMG was supplemented as an LH preparation in the early follicular phase, which has not been performed in previous prospective studies and did not show beneficial effects. In two prior retrospective studies, HMG supplementation showed fewer benefits than recombinant-human luteinizing hormone (r-hLH) [18, 19]. Currently, randomized controlled trials (RCTs) on rLH supplementation of FSH in the early follicular phase do not yield consistent results. In Bosch E et al.’s [20] RCT study, they found that rLH administration in an antagonist protocol significantly increased the implantation rate in women aged 36–39 years, while no benefit was found in patients younger than 36 years. Griesinger et al. [21] did not find better COS results in women aged 20–39 years when rLH was added to FSH compared with FSH monotherapy in an antagonist protocol. Another large RCT did not show beneficial primary and secondary endpoints, such as the number of oocytes retrieved and live birth rate, when rLH was supplemented with FSH in poor responders to a long agonist protocol. However, post-hoc analysis showed a lower rate of total pregnancy outcome failure and a higher live birth rate in patients with moderate and severe POR who received rLH [10].
In the present study, eight cases violated the assigned protocols, including seven in the FSH alone group and one in the HMG/FSH group. All these cycles resulted in a hyporesponse, which represents an initial slow response or stagnation in follicle growth [8], and were treated by LH addition to the FSH alone group or higher LH supplementation to the HMG/FSH group after sufficient FSH doses were administered. The rescued treatments resulted in higher total gonadotropin (Gn) doses, but avoided cycle cancellation, which is in accordance with the ethical spirit.
This study has several limitations. First, this was a single-center pilot study and its limited sample size may have underpowered the ultimate effect of HMG on pregnancy outcomes. Another limitation is the nature of a pilot study necessitates certain compromises regarding study design and analysis. We acknowledge that there are confounding factors that could influence IVF outcomes in POSEIDON Group 4 patients including the heterogeneity in COS protocols, the type of embryo transfer (ET), and the stage of transferred embryos. We intentionally included both long GnRH agonist and antagonist protocols to capture the variability in clinical practice and investigate the effectiveness of different COS approaches, all of which play crucial roles in successful embryo implantation and subsequent pregnancy establishment. Therefore, caution should be exercised when directly comparing the outcomes in our study.
Overall, this study showed no benefit of HMG addition to FSH in the early follicular phase in COS for IVF/ICSI on pregnancy outcomes in POSEIDON Group 4 patients. Further prospective multicenter studies with large sample sizes are required to add rLH to specific difficult populations based on the POSEIDON criteria.
HMG, human menopausal gonadotropin; IVF, in vitro fertilization; POSEIDON, Patient-Oriented Strategy Encompassing IndividualizeD Oocyte Number; POR, poor ovarian response; COS, controlled ovarian stimulation; FSH, follicle stimulating hormone; HMG, human menopausal gonadotropin; LH, luteinizing hormone; ICSI, intracytoplasmic sperm injection.
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Concept and design: XL. Acquisition, analysis, and interpretation of data: XL, JQ, SL, YB and XZ. Drafting of the manuscript: JQ and SL. Critical revisions of the manuscript for important intellectual content: XL. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was conducted in accordance with the Declaration of Helsinki for Medical Research involving Human Subjects and was approved by the Ethics Committee of West China Second Hospital, Sichuan University (ethical permit number: 2021029). The study was registered at Chinese Clinical Trial Registry (https://www.chictr.org.cn), registration number: ChiCTR2100043040. Informed consent was obtained from all participants prior to participation in the study. All data were deidentified to protect patient privacy.
Not applicable.
This work was supported by Key R & D projects of Sichuan Provincial Department of Science and Technology under Grant 2021YFS0243.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.