1 Department of Reproductive Endocrinology, Zhejiang University School of Medicine Women's Hospital, 310000 Zhejiang, China
†These authors contributed equally.
Academic Editor: Andrea Tinelli
Abstract
Background: Even though endometriosis is an important risk factor for pelvic inflammatory disease (PID), it is still not clear whether endometriosis influences PID after ultrasound-guided transvaginal oocyte retrieval (TVOR). Therefore, this work was designed to explore whether endometriosis will influence PID after TVOR and study the influencing factors of receiving drainage treatment to improve the outcomes of patients with PID. Methods: A retrospective study was conducted between 2004 and 2017. Data were collected from Women’s Hospital, Zhejiang University School of Medicine, China. This study included 66 patients with acute PID symptoms after TVOR with or without endometriosis. The independent factors predicting drainage treatment were determined using univariate and multivariate logistic regression analyses, and their optimal cut-off points were ascertained using a receiver operating characteristic curve. Results: Among 66 cases, there were 53 women without endometriosis and 13 women with stage III or IV endometriosis. The significantly higher maximum body temperature (p = 0.047), longer days of fever (p = 0.043) and duration of intravenous (IV) antibiotic use (p = 0.001), and more receiving drainage treatment (p = 0.002) were found in the patients with endometriosis. In vitro fertilization (IVF) cycles (odds ratio [OR] = 6.055, 95% confidence interval [CI] = 1.360–26.961, p = 0.018), puncture cyst during TVOR (OR = 60.167, 95% CI = 2.477–1461.619, p = 0.012), and pre-treatment C-reactive protein (CRP; OR = 1.022, 95% CI = 1.003–1.041, p = 0.022) were significant independent risk factors for drainage treatment. The optimal cut-off for IVF cycles and pre-treatment CRP for patients receiving drainage treatment were 2 and 40.3 mg/L, respectively. Conclusions: Patients with endometriosis had more severe PID outcomes after TVOR, requiring more attention during treatment. Early drainage treatment is recommended for patients with pre-treatment CRP levels higher than 40.3 mg/L, puncture cyst during TVOR, and those receiving more than 2 IVF cycles.
Keywords
- in vitro fertilization
- endometriosis
- transvaginal oocyte retrieval
- pelvic inflammatory disease
- drainage treatment
Ultrasound-guided transvaginal oocyte retrieval (TVOR) has been widely used in in vitro fertilization (IVF) because it is relatively easy to learn and is much less invasive than laparoscopic or transabdominal routes. However, this procedure will inevitably breach the abdomen, upper vagina, and endocervix, which may lead to the implantation of microorganisms from the skin, vagina, or cervical tube into the uterus, fallopian tubes, or peritoneal cavity [1]. The incidence of pelvic inflammatory disease (PID) after TVOR has been reported to range from 0.03% to 0.58% [2, 3, 4, 5]. To date, there is no effective method to prevent this undesired complication. Unfortunately, despite adequate treatment, some patients still suffer from significant morbidity, including chronic pelvic pain, infertility, and ectopic pregnancy [6, 7].
A study analyzed the relationship between endometriosis and pelvic abscess in 3215 women and concluded that endometriosis is an important risk factor for PID, with old blood providing a rich culture medium for bacterial proliferation [8]. Several case reports have also described pelvic abscess after TVOR in patients with endometriosis but did not clarify their connection [9, 10, 11]. Furthermore, a 4-year review of infectious processes in women with endometriosis by Villette et al. [12] indicated that some putative complications of assisted reproductive technologies (ART) and endometrioma may not be linked to ART. Rather, these spontaneously occur in endometriosis. Thus, it is still not clear whether endometriosis influences PID after TVOR.
Fouks et al. [13] retrospectively analyzed 15 women with tubo-ovarian abscess (TOA) during IVF cycles and compared the ovarian response to stimulation and the pregnancy rate in two subsequent cycles. It was found that TOA had a detrimental effect on ovarian function after fertility treatment, and the pregnancy rate in the immediate period following TOA was poor. Another study suggested that an increase in pelvic temperature may directly damage the proliferation of embryonic cells as exposure to heat leads to intracellular damage, inhibition of mitosis, inhibition of proliferation, migration, and cell death, resulting in implantation failure or teratogenic effects [14]. During pelvic inflammation, the heightened maternal immune status and excessive secretion of cytokines destroy the delicate balance between cytokines required for embryonic development, which can also lead to failure of embryo development and trophoblast implantation [2]. Therefore, it is necessary to explore whether endometriosis will influence PID after TVOR and study the influencing factors of receiving drainage treatment to improve the outcomes of patients with PID and reduce the impact of inflammation on the follow-up pregnancy outcome through active treatment.
Thus, this study retrospectively analyzed the clinical data of patients who required hospitalization due to PID after TVOR. We aimed to investigate the influence of endometriosis on the severity of PID after TVOR and further determine the factors influencing drainage treatment. We conducted this study to provide more evidence for clinical treatment decisions.
This study was approved by the Ethics Committee of the Women’s Hospital, Zhejiang University School of Medicine (approval number: PRO2021-1728). Written informed consent was obtained from all participants. We analyzed the clinical data of patients with acute PID symptoms after TVOR between January 2014 and November 2019. The inclusion criteria in our study were as follows: diagnosis of PID according to the United States Centers for Disease Control and Prevention criteria for PID, receiving hospitalization meets any of the recommended criteria (surgical emergencies cannot be excluded; tubo-ovarian abscess; pregnancy; severe illness, nausea and vomiting, or high fever; unable to follow or tolerate an outpatient oral regimen; or no clinical response to oral antimicrobial therapy) [15], symptoms of PID occurred within 3 months following IVF treatment, and complete clinical data. Patients were excluded if they had an infection before TVOR. A total of 66 patients were eligible for inclusion, which comprised 53 women without endometriosis and 13 women with endometriosis. Among the 13 cases with endometriosis, 5 were diagnosed by ultrasound (the diameter of the cyst was more than 3 cm), and 8 were diagnosed by surgery with stage III or IV endometriosis (r-ASRM classification). We collected the patients’ demographic and clinical data by reviewing their medical records.
According to our department protocol, patients were instructed to perform vaginal scrubbing with povidone-iodine for 2 consecutive days before retrieval. The patients underwent TVOR in the lithotomy position and were covered with sterile surgical sheets. The operator cleansed the entire vulva and vagina with sterile saline while wearing a sterile operating gown and sterile powder-free gloves. The vaginal ultrasonic probe was covered with a sterile plastic sheath. Cysts (including endometriosis or ovarian simple cysts) were not punctured in TVOR unless they interfere with oocyte retrieval. Antibiotic prophylaxis was not used, except when a patient presented with certain risk factors, such as a history of hydrosalpinx, ovarian endometrioma, and PID.
The selection of IV antibiotic therapy was based on the relevant guidelines [15]. IV antibiotic therapy was administered until the patient had been afebrile for at least 48 h (or a minimum of 4 days). Oral antibiotic treatment was continued for 2 weeks. Ultrasound-guided drainage or surgical drainage by laparoscopy or laparotomy was performed in patients whose imaging examination showed visible cysts combined with had positive peritoneal signs or did not improve within 72 h of antibiotic therapy. Following drainage, IV antibiotics were administered for an additional 4 days or until clinical improvement was achieved.
All statistical analyses were performed using the Statistical Package for the
Social Sciences Version 21.0 (SPSS Inc, Chicago, IL, USA). Data are expressed as
frequencies (percentages), means (standard deviation), and median (25th–75th
percentile). The Student’s-test, Chi-square test, and Mann-Whitney U-test were
used whenever appropriate to compare data between the groups. Binary logistic
regression analysis was also performed to determine the significant independent
contribution of specific variables, yielding a p-value of
A total of 66 patients with acute PID symptoms after TVOR were included in this
study. They were divided into two groups, according to the presence or absence of
endometriosis. Table 1 illustrates the comparison of the clinical outcomes
between the two groups. Significant differences were observed in the age (29.66
| Patients without endometriosis (n = 53) | Patients with endometriosis (n = 13) | p | ||
| Age (years) | 29.66 |
32.77 |
0.015 | |
| Gravidity | 0 (0–1) | 0 (0–1) | 0.461 | |
| Parity | 0 (0–0) | 0 (0–0) | 0.986 | |
| BMI (kg/m |
21.36 |
21.44 |
0.902 | |
| History of abdominal surgery | 24 (45.3%) | 10 (76.9%) | 0.410 | |
| AMH (ng/mL) | 3.56 (1.52–6.51) | 1.22 (0.79–3.40) | 0.078 | |
| IVF cycles | 1 (1–2) | 1 (1–3) | 0.604 | |
| Puncture cyst during TVOR | 3 (5.7%) | 5 (38.5%) | 0.006 | |
| Number of collected oocytes | 16 (10–21) | 4 (3–17) | 0.032 | |
| Time interval from TVOR to the onset of Symptoms(days) | 1 (0–3) | 10 (1–21) | 0.004 | |
| Symptoms | ||||
| only pelvic pain | 9 (17.0%) | 2 (15.4%) | 1.000 | |
| only fever | 2 (3.8%) | 1 (7.7%) | 0.488 | |
| pelvic pain and fever | 42 (79.2%) | 9 (69.2%) | 0.471 | |
| pelvic pain, fever and metrorrhagia | 0 (0.0%) | 1 (7.7%) | 0.197 | |
| pre-treatment |
12.8 (10.7–17.5) | 12.4 (11.3–15.7) | 0.796 | |
| pre-treatment |
10.7 (8.4–14.4) | 10.1 (8.9–13.7) | 0.961 | |
| pre-treatment |
26.9 (9.5–63.2) | 76.6 (32.4–152.4) | 0.008 | |
| pre-treatment |
0.06 (0.04–0.16) | 0.48 (0.05–1.26) | 0.102 | |
| Microbiological cultures | 1.000 | |||
| negative | 25 | 7 | ||
| positive | 18 | 4 | ||
| Maximum body temperature ( |
38.15 |
38.60 |
0.047 | |
| Days of fever (days) | 2 (1–4) | 3 (2–7) | 0.043 | |
| Duration of IV antibiotics use (days) | 6 (4–7) | 14 (7–22) | 0.001 | |
| Receiving drainage treatment | 2 (3.8%) | 5 (38.5%) | 0.002 | |
| Ultrasound-guided drainage | 1 | 1 | 1.000 | |
| Surgical drainage | 1 | 2 | 1.000 | |
| Ultrasound-guided and surgical drainage | 0 | 2 | 1.000 | |
| Hospitalization period (days) | 6 (6–9) | 10 (6–20) | 0.019 | |
BMI, body mass index; AMH, anti-Mullerian hormone; IVF, in vitro fertilization; TVOR, transvaginal oocyte retrieval; WBC, white blood cells; N, neutrophil; CRP, C-reactive protein; PCT, procalcitonin. Notes: The “pre-treatment” refers to the time before antibiotics were administered. | ||||
Table 2 shows the univariate and multivariate logistic regression analyses of the independent predictors of drainage treatment. Univariate analysis showed that age, stage III or IV endometriosis, IVF cycles, puncture cyst during TVOR, time interval from TVOR to the onset of symptoms, and pre-treatment CRP were significantly correlated with receiving drainage treatment.
| Univariate analysis | Multivariate analysis | |||
| OR (95% CI) | p | OR (95% CI) | p | |
| Age | 1.424 (1.088–1.863) | 0.01 | 1.321 (0.852–2.048) | 0.213 |
| Gravidity | 1.223 (0.447–3.348) | 0.695 | - | - |
| Parity | - | 0.999 | - | - |
| BMI | 0.789 (0.538–1.155) | 0.223 | - | - |
| History of abdominal surgery | 2.586 (0.464–14.406) | 0.278 | - | - |
| AMH | 0.180 (0.026–1.248) | 0.083 | - | - |
| Stage Ⅲ or Ⅳ endometriosis | 15.937 (2.631–96.538) | 0.003 | 3.167 (0.164–61.265) | 0.446 |
| IVF cycles | 3.915 (1.422–10.779) | 0.008 | 6.055 (1.360–26.961) | 0.018 |
| Puncture cyst during TVOR | 18.333 (3.005–111.869) | 0.002 | 60.167 (2.477–1461.619) | 0.012 |
| Number of collected oocytes | 0.480 (0.214–1.076) | 0.075 | - | - |
| Time interval from TVOR to the Onset of symptoms(days) | 1.116 (1.014–1.227) | 0.024 | 1.102 (0.948–1.28) | 0.206 |
| pre-treatment WBC | 0.909 (0.746–1.107) | 0.341 | - | - |
| pre-treatment N | 0.933 (0.768–1.133) | 0.481 | - | - |
| pre-treatment CRP | 1.018 (1.005–1.031) | 0.007 | 1.022 (1.003–1.041) | 0.022 |
| pre-treatment PCT | 0.984 (0.893–1.085) | 0.75 | - | - |
| Microbiological cultures | 3.333 (0.554–20.068) | 0.189 | - | - |
| Univariate and multivariate logistic regression analysis. OR, odds ratio; CI, confidence interval; BMI, body mass index; AMH, anti-Müllerian hormone; IVF, in vitro fertilization; TVOR, transvaginal oocyte retrieval; WBC, white blood cells; N, neutrophil; CRP, C-reactive protein; PCT, procalcitonin. | ||||
Multivariate analysis by backward elimination of nonsignificant associated factors in the univariate analysis showed that IVF cycles (odds ratio [OR] = 6.055, 95% confidence interval [CI] = 1.360–26.961, p = 0.018), puncture cyst during TVOR (OR = 60.167, 95% CI = 2.477–1461.619, p = 0.012), and pre-treatment CRP level (OR = 1.022, 95% CI = 1.003–1.041, p = 0.022) were significant independent risk factors for drainage treatment.
The ROC curves comparing the performance of the independent factors and their joint predictors (p = 1 / (1 + e-Z), Z = –8.072 + 1.801 × IVF cycles + 4.097 × puncture cyst during TVOR + 0.022 × pre-treatment CRP) for predicting patients receiving drainage treatment are shown in Fig. 1. Table 3 shows that the AUC was 0.722 (95% CI = 0.470–0.973) for IVF cycles, 0.752 (95% CI = 0.521–0.983) for puncture cysts during TVOR, and 0.877 (95% CI = 0.778–0.975) for pre-treatment CRP, which were all lower than that of the joint predictor (AUC = 0.944, 95% CI = 0.865–1.000). The optimal cut-off of IVF cycles and pre-treatment CRP for predicting patients receiving drainage treatment were 2 times and 40.3 mg/L, respectively.
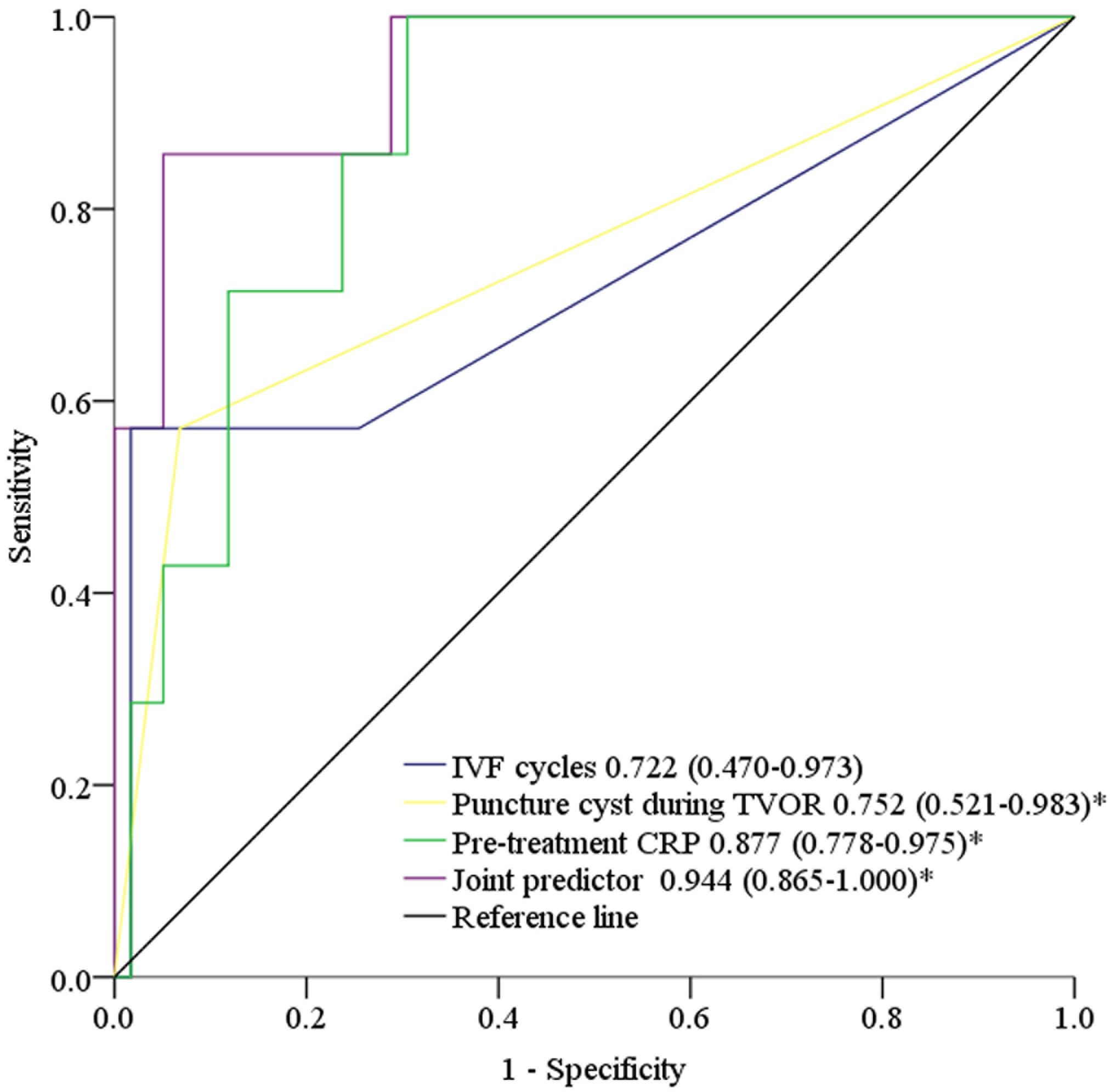 Fig. 1.
Fig. 1.The ROC curves compared the performance of IVF cycles, puncture
cyst during TVOR, pre-treatment CRP and the joint predictor in the prediction of
patients receiving drainage treatment. *Significant differences (p
| AUC | 95% CI | Cut-off | Sens (%) | Spec (%) | YI | p | |
| IVF cycles | 0.722 | 0.470–0.973 | 2 | 57.1 | 98.3 | 0.555 | 0.057 |
| Puncture cyst during TVOR | 0.752 | 0.521–0.983 | - | 57.1 | 93.2 | 0.504 | 0.030 |
| Pre-treatment CRP | 0.877 | 0.778–0.975 | 40.3 | 100 | 69.5 | 0.695 | 0.001 |
| Joint predictor | 0.944 | 0.865–1.000 | 0.192 | 85.7 | 94.9 | 0.806 | |
| Receiver–operating characteristic (ROC) curves. IVF, in vitro fertilization; TVOR, transvaginal oocyte retrieval; CRP, C-reactive protein; AUC, area under the curve; CI, confidence interval; YI, Youden index. | |||||||
The effect of endometriosis on PID after TVOR has not been conclusively determined. Analyzing 66 cases of patients with PID after TVOR, the present study showed that patients with endometriosis had more severe outcomes, such as higher maximum body temperature, longer days of fever, longer duration of IV antibiotic use, and more cases of drainage treatment. Moreover, IVF cycles, puncture cyst during TVOR, and pre-treatment CRP were found to be significant independent risk factors for predicting patients receiving drainage treatment. ROC curve analysis also confirmed that the joint predictor of IVF cycles, puncture cyst during TVOR, and pre-treatment CRP is useful for the prediction of patients who are likely to undergo drainage treatment.
TVOR is the most used method for obtaining oocytes during IVF cycles. Several observational studies assessing the incidence of complications associated with this procedure indicated that it is safe, with very low rates of serious adverse events [16]. Although PID after TVOR is rare, the risk should not be underestimated, which may be life-threatening. Several mechanisms for the etiology of PID after TVOR have been proposed, such as (1) inoculation of vaginal microorganisms into the ovary or peritoneal cavity during needle puncture through a nonsterile vaginal wall, (2) reactivation of latent PID, or (3) direct contiguous organ injury [17]. Bacterial contamination from the transvaginal route is the most common cause because the typical vaginal flora has been found in microbiological cultures from abscesses caused by TVOR [18]. The distribution of microorganisms in our statistical data showed the same phenomenon. It has been reported that the hospitalization rate of patients with pelvic pain was 0.03%–0.7%. It is difficult to collect objective data about pelvic pain since it is a symptom that is experienced differently by each patient, and these cases may be underreported [16, 19]. In this study, 11 out of 66 patients were hospitalized because of simple abdominal pain, another three were hospitalized because of fever, and the remaining cases were complicated by other symptoms. We also found that the time interval from TVOR to the onset of symptoms can be as long as 65 days. Other studies have reported that the time interval may range from 4–120 days [9, 12, 20, 21], highlighting its variability. It is undeniable that we estimated that the diagnosis of PID by retrospective investigation was not reliable enough. In particular, mild cases may have been underreported. However, through statistical analysis, we can also understand the relevant results of the patients who developed PID after TVOR and were hospitalized for IV antibiotic treatment to provide more evidence for clinical treatment decisions. As pelvic infection becomes clinically evident within a few hours to several days following TVOR, it is also recommended to extend the follow-up of the patient to detect the changes in the patient’s condition early.
At present, endometriosis is a common indication for IVF and embryo transfer in patients who have failed surgery and/or drug treatment [22]. The sensitivity and specificity of transvaginal ultrasound have been reported to be 93% and 96%, respectively [23, 24]. Although there is no pathological diagnosis when using ultrasound, the increase in the sensitivity and specificity of ultrasound diagnosis and the findings of current research that surgical treatment does not improve the ovarian response to gonadotropin [25, 26, 27, 28] have changed the viewpoint of whether endometriosis should be operated on before ART. Some studies have proposed that endometriosis is an important risk factor for PID [8, 10, 29], though some have concluded otherwise [12, 30]. In this study, we compared the outcomes of PID after TVOR in patients with or without endometriosis. We found that patients with endometriosis had more severe outcomes, such as higher maximum body temperature, longer days of fever, longer duration of IV antibiotic use, and more cases of drainage treatment. Although the relationship between endometriosis and infection remains controversial, the treatment of patients with PID after TVOR should be properly monitored, especially those with endometriosis, whose condition may be more serious.
The treatment of PID varies depending on the clinical situation. The success rate of drug treatment alone ranges from 34%–87.5% [13, 31]. Ultrasound-guided drainage of pelvic abscesses is an alternative to surgical procedures. However, despite repeated aspiration under ultrasound guidance, 6.6% of residual abscesses still required further surgery in a study involving 302 women who underwent 449 aspirations [32]. In our study, 7 patients (10.6%) could not be cured by simple ultrasound- or surgery-guided drainage. CRP is generally believed to reflect the severity of infection. Further analysis of our study showed that pre-treatment CRP higher than 40.3 mg/L was one of the significant independent risk factors for patients receiving drainage therapy. It is well known that multiple IVF cycles with TVOR and puncturing of cysts increase the number of pelvic operations and easily lead to the onset of pelvic inflammation. Meanwhile, puncture cysts during TVOR and receiving more than 2 IVF cycles were also identified as independent risk factors in our study. The existence of these independent risk factors makes it difficult to control PID with drugs alone. It is worth mentioning that we also found that the AUC of the joint predictor of these risk factors was 0.944, which indicates its high clinical predictive potential. In general, in patients with PID after TVOR combined with the comprehensive evaluation of IVF cycle, puncture cyst during TVOR, and pre-treatment CRP, early drainage treatment may be a better treatment for these patients.
Although every effort has been made to match endometriosis and control subjects, there were still several limitations to our study. The main limitation of this study was its retrospective nature and the small sample size of patients with endometriosis. Furthermore, due to the different pain experiences of each patient, mild PID can easily be ignored, leading to the underestimation of PID. Meanwhile, the data were collected from a single center. Therefore, a multicenter and large sample setting is needed to exclude possible single-center biases. However, most of the relevant data were obtained from medical records, making our conclusion credible.
Our data indicated that pelvic infection becomes clinically evident within a few hours to several days after TVOR. Therefore, following T healthcare providers should be vigilant against PID after TVOR. Patients with endometriosis had more severe outcomes, such as higher maximum body temperature, longer days of fever, longer duration of IV antibiotic use, and more cases of drainage treatment. Proper attention should be provided to the treatment of patients with PID after TVOR, especially those with endometriosis, whose condition may be more serious and may thus affect the outcome of fertility. Meanwhile, pre-treatment CRP higher than 40.3 mg/L, puncture cyst during TVOR, and receiving more than 2 IVF cycles could be used as the combined predictor for recognizing patients with a high risk of undergoing drainage treatment, helping clinicians identify this indication and take timely measures.
WZ and RZC have contributed equally to this work and share first authorship. WZ and RZC designed the study. FL, XYY and RZC performed the statistical analysis. HYL, XJC and HJG acquisited data. HYL and YMZ revised the paper. All authors read and approved the final manuscript.
This retrospective study was approved by the institutional ethics committee of Women’s Hospital, Zhejiang University School of Medicine, China (approval number: PRO2021-1728). All patients provided written informed consent.
We thank all the members of our department for their support and valuable suggestions.
This work was supported by the National key R&D Program of China (2018YFC1004900).
The authors declare no conflict of interest.

