1 The Department of Anesthesiology, The First Affiliated Hospital of Jinan University, Jinan University, 510630 Guangzhou, Guangdong, China
2 The Department of Anesthesiology, Yue Bei People’s Hospital, 512025 Shaoguan, Guangdong, China
Academic Editor: Michael H. Dahan
Abstract
Background: Butorphanol was safely used for obstetric analgesia for
many years. We attempted to determine if it increases the analgesic effect during
patient-controlled epidural labor analgesia (PCEA) and if it affects the
labor-process. Methods: Term pregnant women (N = 60) were randomly
assigned to two groups, to receive either 1 mg intravenously butorphanol or 10 mL
normal saline as placebo at 10 min before intrathecal anesthesia. Pain score,
duration of first and second stage of labor, mode of delivery, umbilical artery
blood gas, Apgar scores and adverse effects were recorded. Result:
Butorphanol shortened the duration of the second stage of labor (t = –2.41,
p = 0.019). Women in butorphanol group showed significantly increased
incidence of pruritus (x
Keywords
- Butorphanol
- Intrathecal anesthesia
- Labor pain
- Patient controlled epidural analgesia
- Visual analogue scale
- Ropivacaine
- Sufentanyl
Labor pain is almost the most serious pain in women’s life. Labor analgesia is extremely necessary and important. Patient controlled epidural analgesia (PCEA) is considered to be an ideal method for labor analgesia. However, there are still some problems in epidural analgesia, such as pruritus [1, 2]. In addition, intrathecal puncture requires the women’s cooperation; strong pain sometimes makes it impossible.
Butorphanol as an opioid analgesic has been safely used for obstetric analgesia for many year [3] and it has good analgesic effect, less risk of pruritus. To the best of our knowledge, no study has compared the role of intravenous butorphanol as an adjunct analgesic agent to the well-established labor analgesia regimes. The combination of a local anaesthetic (ropivacaine) and an opioid (sufentanyl) as PCEA, is a standard procedure for laboring women in many hospitals including ours. It would be of interest to learn if intravenous 1 mg butorphanol could have an analgesia effect for the laboring women, which could have implications for the effect of PCEA and the labor process. The present study was designed to evaluate the efficacy of intravenous infusion of 1 mg of butorphanol as an adjunct, its sparing effect on the delivery process, and the incidence of side effect of epidural analgesia.
The study was approved by the ethics committee of the First Affiliated Hospital
of Jinan University (Approval No: [2019 LPK No. 020]) and the trial has been
registered at Clinical Trials Registry in China (trial registration number
ChiCTR2100042974). Written informed consent was obtained from all subjects
participating in the trial. All methods we used were performed in accordance with
the Declaration of Helsinki. The study was conducted between June 15, 2019 and
March 13, 2020 at the first affiliated hospital of Jinan University, GuangZhou,
China. After obtaining written informed consent, women requesting analgesia in
the first stage of labor and their cervix being 2 to 4 cm open were enrolled in
this study. Inclusion criteria: American Society of Anesthesiologists physical
status classification (ASA) class I and II, age: 18–35 year-old primipara,
spontaneous onset of labor, body mass index (BMI): 22–32, gestational age:
37–42 weeks, cervix dilation 2–4 cm and a single live fetus in cephalic
presentation. The exclusion criteria were: refusal by parturient, parturient who
had received parenteral opioids in the last four hours, any systemic and local
sepsis, deranged coagulation profile (platelets
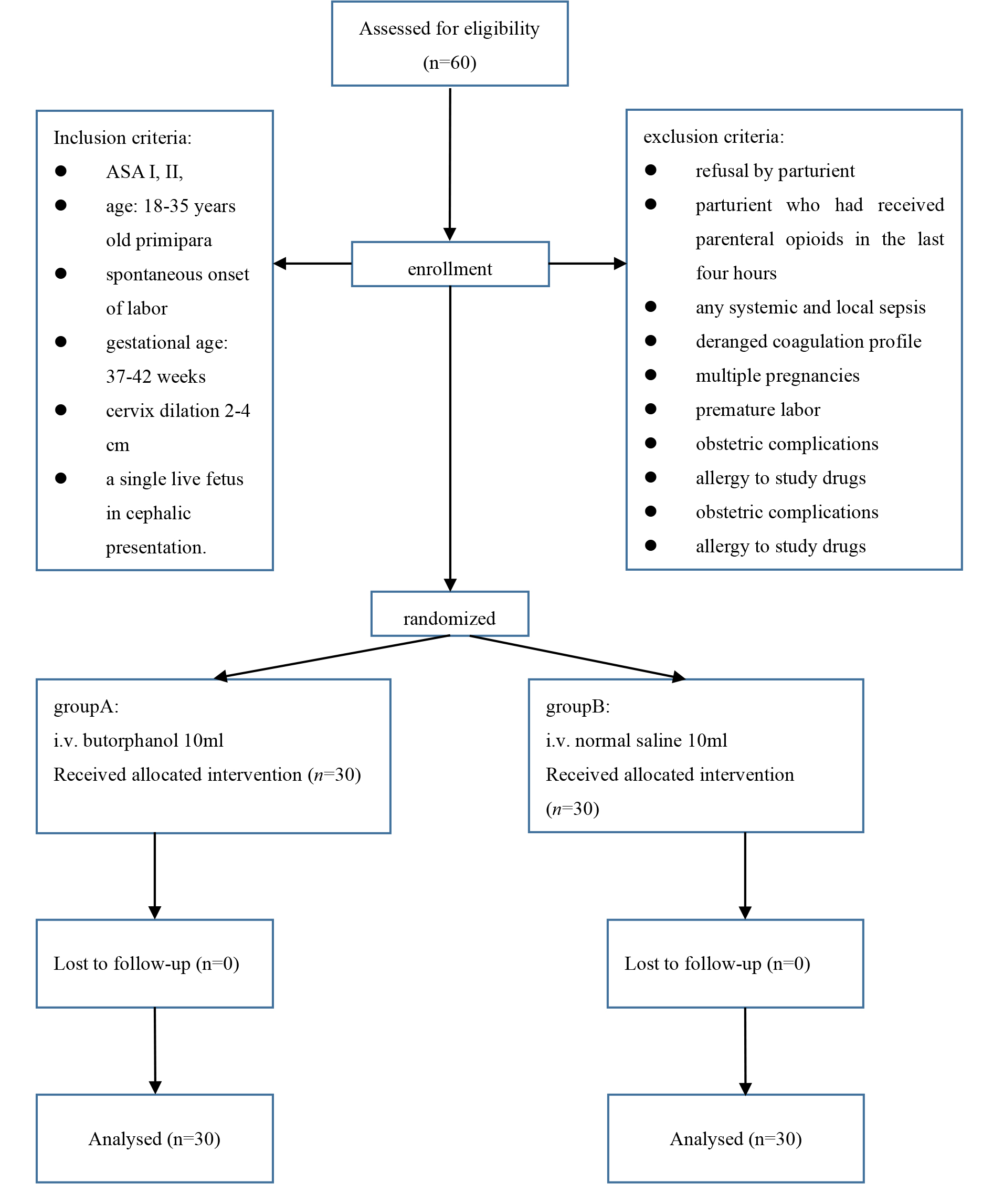 Fig. 1.
Fig. 1.Consort flow diagram. CONSORT indicates consolidated standards of reporting trials.
After the patient was shifted to the clean labor room operation theatre,
intravenous route was accessed and baseline Visual Analogue Scale(VAS) was
recorded. The patient was continuously monitored for heart rate (HR), noninvasive
blood pressure (NIBP) and pulse saturation of oxygen (SpO
Blinding was done by the following means: the drugs were prepared by one investigator who just prepared the drugs for relevant groups which were allocated randomly by using computer generated random numbers. The combined spinal-epidural anesthesia was performed by several well-trained anesthesiologists who don’t know the difference among the two groups. While the assessments were done by another investigator who was blinded to the group and drug received.
We recorded general information about the parturient women such as age, height, weight, gestational weeks, haemodynamic parameters of the mother (using multichannel monitor), pain score, and motor block score at pre-set time point (before analgesia, intrathecal puncture, intrathecal injection, 5 min, 10 min, 15 min, 20 min, 1 h, 2 h, 3 h after intrathecal injection, full dilated cervix, 30 minutes and 1 hour after full dilated cervix, newborn delivery). We also recorded the time of delivery, delivery method and adverse effect (These adverse effects such as pruritus, dizziness, somnolence, nausea, and vomiting are evaluated when the analgesic drugs play the strongest analgesic effect-VAS is 0 by asking the mother), the 1-min Apgar score and 5-min Apgar score of the newborn, and the umbilical artery blood gas analysis. After the newborn was delivered and the umbilical cord was cut, a disposable arterial blood collection device (BD®, Roborough, Plymouth, UK) is used to draw 3 mL of umbilical artery blood immediately for measurement of umbilical artery blood gas. VAS was used to assess pain and the score was given by the parturient. The pain levels represented by the scores are as follows: 0~2: no pain 3~4: mild pain 5~6: moderate pain 7~8: severe pain 9~10: insufferable pain. If the parturient fell asleep, the VAS score was counted as 0. The modified Bromage rating was used to analyze the degree of motor block. The neonatal Apgar score was used to assess the physical condition of newborns: 8–10 points indicate normal, 4–7 points indicate mild asphyxia, 0–3 points indicate severe asphyxia.
Statistical analysis: regarding the primary outcome, it was hypothesized that
butorphanol group would have 80% women had lower VAS scale before intrathecal
analgesia than those in placebo group. Therefore, for this hypothesis, about 60
participants required for this research to reach the statistical power of 95%.
Chi-square test was used to analyze the incidence of adverse effects and delivery
method. The general Parametric results like the time of labor, the Apgar score of
the newborn and the umbilical artery blood gas analysis are presented as means
The two groups had similar age, height, weight and maternal hemodynamics fluctuated in the normal range of baseline. There were no significant differences in these general data of the two groups.
The pain scores (the primary outcome) were missing at some time points due to the women’s different delivery time or some women having given birth by cesarean section. Specifically, pain scores were absent in both groups from one hour after the onset of labor analgesia to the time before the delivery. Therefore, in the data analysis, we took repeated measurement ANOVA for the complete data period, and then analyzed the VAS score of each time point by the Independent-Samples t test. Statistics showed that within one hour after intrathecal analgesia, VAS scores of both groups decreased linearly. The results showed that the two experimental groups achieved rapid relief of labor pain after the implementation of intrathecal analgesia. However, the analgesia score of butorphanol group was significantly lower than that of the placebo group from the time of intrathecal puncture to 5 minutes after the onset of intrathecal analgesia, which indicated butorphanol has an advantage in pain relief at the time of intratheacal puncture and the cooperation of pregnant women can be easily obtained. From 10 minutes to 2 hours after the onset of intrathecal anesthesia, the VAS scores of the two groups were low and the mean value of VAS score was close to 0. The analgesic effect of the two groups was good, and the difference was not statistically significant. The VAS score at 3 hours after the onset of intrathecal anesthesia in butorphanol group was significantly lower than that in the placebo group. During the period from cervical full opening to fetal delivery, the pain scores of the two groups increased gradually, and the pain increased, but there was no significant difference between the two groups (Table 1 and Fig. 2).
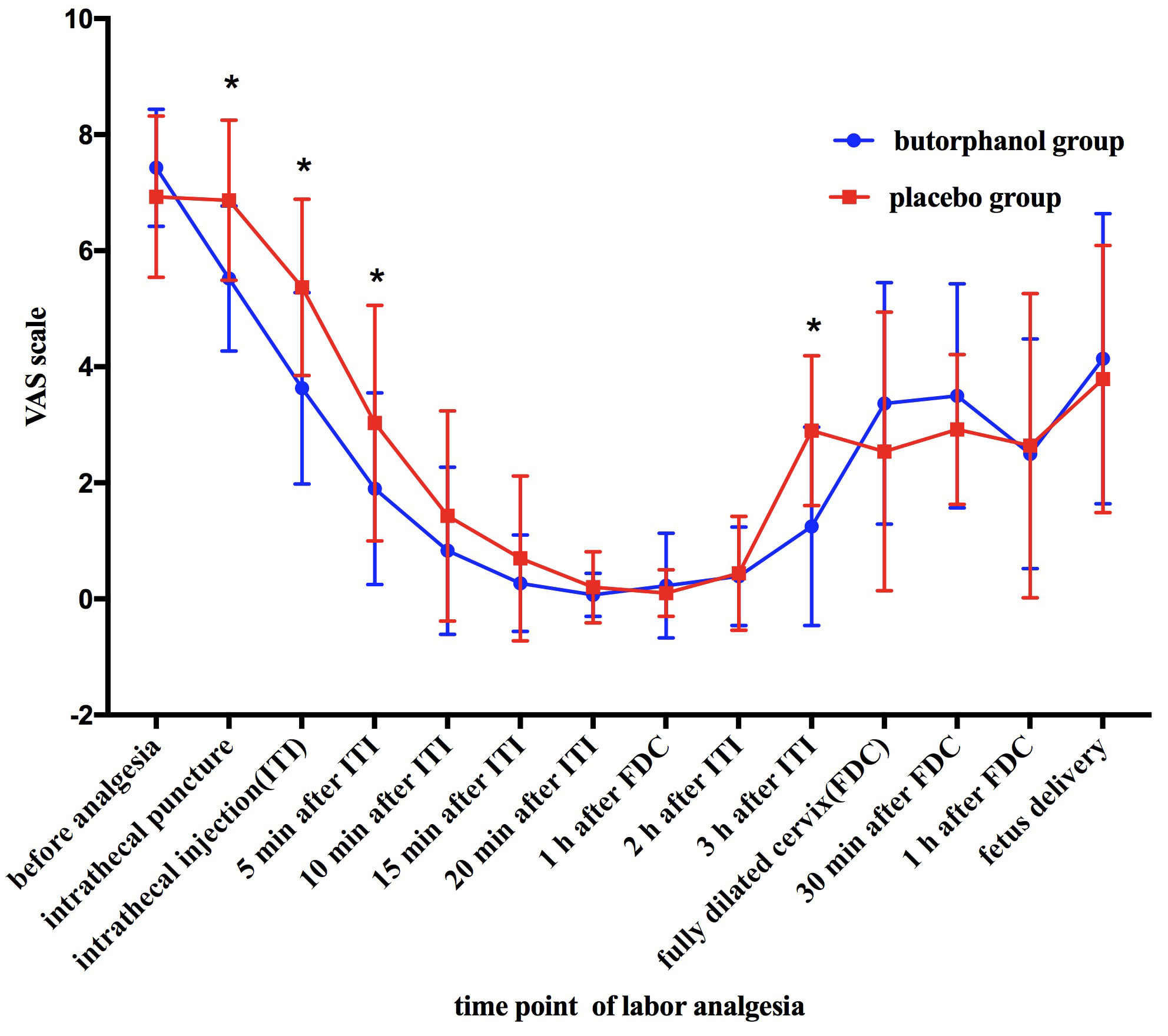 Fig. 2.
Fig. 2.Pain score of parturient at different time points during labor
analgesia. *p
| Time point | Group A (n = 30) | Group B (n = 30) | p-value |
| (butorphanol group) | (placebo group) | (*p | |
| Before analgesia | 7.43 |
6.93 |
0.116 |
| Intrathecal puncture | 5.52 |
6.87 |
0.000* |
| Intrathecal injection (ITI) | 3.63 |
5.37 |
0.000* |
| 5 min after ITI | 1.90 |
3.03 |
0.021* |
| 10 min after ITI | 0.83 |
1.43 |
0.162 |
| 15 min after ITI | 0.27 |
0.70 |
0.155 |
| 20 min after ITI | 0.07 |
0.20 |
0.310 |
| 1 h after ITI | 0.23 |
0.10 |
0.461 |
| 2 h after ITI | 0.39 |
0.44 |
0.857 |
| 3 h after ITI | 1.25 |
2.90 |
0.021* |
| Fully dilated cervix (FDC) | 3.37 |
2.54 |
0.163 |
| 30 min after FDC | 3.50 |
2.92 |
0.349 |
| 1 h after FDC | 2.50 |
2.64 |
0.906 |
| Fetus delivery | 4.14 |
3.79 |
0.583 |
| Data are shown as mean Abbreviations: VAS, visual analogue scale; ITI, intrathecal injection; FDC, fully dilated cervix. | |||
After the implementation of intrathecal labor analgesia, we observed that the Bromage score of the two groups was always zero, indicating that both of our analgesia program had no effect on lower limb movement. During the study, one patient in the butorphanol group was transferred to cesarean section because of sever fetal heart rate deceleration, while two cases in the placebo group were converted to cesarean section, one case was suspected of placental abruption due to bloody amniotic fluid, and the other was fetal heart rate deceleration. All these three women were converted to cesarean section one hour after intrathecal analgesia. So the delivery mode of the two groups included normal delivery, forceps assisted delivery and cesarean section, and No significant difference in the delivery mode between the two groups.
Analyzing the time duration of the first and the second stage of labor between the two groups, there was no significant difference in the length of the first stage of labor between the two groups, while the duration of the second stage of labor in the butorphanol group was significantly shorter than those in the placebo group, suggesting that butorphanol might help shorten the second stage of labor (Table 2). Apgar score at 1 and 5 minutes showed that infants of the two groups had no asphyxia, with no statistical significance. Umbilical artery blood gas analysis showed that the pH value, lactic acid and blood glucose of the two experimental groups were in the normal range in both groups, without significant difference (Table 3). Therefore, butorphanol had no effects of the mode of delivery, Apgar score, umbilical artery blood gas.
| Labor process | Group A (n = 30) | Group B (n = 30) | p-value |
| (butorphanol group) | (placebo group) | (*p | |
| First stage of labor (h) | 8.94 |
9.12 |
0.855 |
| Second stage of labor (h) | 0.85 |
1.22 |
0.019* |
| Data are shown as mean | |||
| Blood Gas value | Group A (n = 30) | Group B (n = 30) | p-value |
| (butorphanol group) | (placebo group) | ||
| pH value | 7.23 |
7.21 |
0.329 |
| Lactic acid | 4.29 |
4.78 |
0.201 |
| Blood sugar | 4.92 |
5.05 |
0.762 |
| Data are shown as mean | |||
However, butorphanol might have caused dizziness and somnolence. It reduced the incidence of pruritus caused by sufentanil. In butorphanol group, there was one case of nausea and one case of vomiting, but no nausea and vomiting was found in the placebo group: incidence of nausea and vomiting between the two groups was not statistically significant. Two experimental groups caused some maternal urine retention and fever, the difference between the two groups was not statistically significant (Table 4).
| Adverse effects | Group A (n = 30) | Group B (n = 30) | p-value |
| (butorphanol group) | (placebo group) | (*p | |
| Pruritus n (%) | 1 (3.33%) | 17 (56.67%) | 0.000* |
| Dizziness n (%) | 9 (30.00%) | 0 (0%) | 0.004* |
| Somnolence n (%) | 20 (66.67%) | 1 (3.33%) | 0.000* |
| Nausea n (%) | 1 (3.33%) | 0 (0%) | 1.000 |
| Vomitting n (%) | 1 (3.33%) | 0 (0%) | 1.000 |
| Urinary retention n (%) | 10 (33.33%) | 13 (43.33%) | 0.427 |
| Fever n (%) | 2 (6.67%) | 4 (13.33) | 0.667 |
| *means p | |||
Painless childbirth greatly improves the delivery experience of women. The Patient-controlled epidural analgesia (PCEA) is an extremely effective and popular treatment for labor pain, and it’s the only method providing complete analgesia without maternal or fetal sedation [6, 7, 8]. The PCEA is superior to continuous epidural infusion (CEI) in terms of analgesia satisfaction [9].
In many previous studies, epidural analgesia was started when the cervix was 2–4 cm open [10] and we also chose this. However, many women felt some difficulties in taking the suitable posture for epidural needle insertion because of the insufferable pain. Our study found that intravenous administration of 1 mg butorphanol 10 minutes before intrathecal puncture significantly reduced maternal pain, especially in the period of intrathecal puncture to 5th minute after the onset of labor analgesia. Intravenous butorphanol can quickly produce analgesic effect, it makes easier to get the cooperation of the pregnant women.
In our experiments, we found that butorphanol improved the pruritus caused by epidural analgesia, while the incidence of pruritus in our placebo group was more than 50%. SEC. [11] found that the incidence of pruritus was even as high as 95%, which may be related to the dose of sufentanil [12]. Kappa-agonists have been shown to inhibit pruritus in both animals and human beings [13]. Many previous studies also have pointed out butorphanol can be used for treatment of pruritus [11, 13, 14], but it will increase the incidence of dizziness and somnolence [15]. The occurrence of somnolence may also be related to the fatigue that occurred the day before delivery due to the pain of uterine contraction and failure to rest well. What’s more, Opioids have mild sedative effects [11, 16]. We found that the second stage of labor was significantly shorter for women in butorphanol group than those in the placebo group. So, we believe that dizziness and somnolence allow a woman to have a full rest in the process of labor, and good rest finally benefit for shortening the second stage of labor.
In our study, the 1 and 5 minute Apgar scores were normal for both groups, indicating that both the butorphanol and epidural analgesia did not affect the Apgar score. However, Apgar score does not always reflect the homeostasis of neonatal internal environment, so we also measured pH value, lactic acid value, and blood glucose value of neonatal umbilical arterial blood. According to our study, there was no statistical difference in the pH value of neonatal umbilical arterial blood gas between the two groups, and the pH values of the two groups fluctuated in the normal range. The umbilical artery blood gas analysis showed the both groups have normal lactic acid value and blood glucose: the difference was not statistically significant. Taken together, we believe that butorphanol is a safe adjunct to patient-controlled epidural labor analgesia and has no adverse effects on neonates.
The present data showed that butorphanol is a safe and effective adjunct for labor analgesia. Intravenous administration of 1 mg butorphanol before the implementation of intrathecal labor analgesia can relieve maternal pain and improve the comfort experience for intrathecal puncture. It may help to shorten the second stage of labor. Intravenous analgesics before the implementation of intrathecal analgesia according to the needs and conditions of pregnant women may be an option for labor analgesia.
YLL, TZ and FYN designed the research study. TZ, FYN, MXW and ZJL performed the research. YLL, TZ and FYN analyzed the data. YLL and TZ wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
This study has been approved by the ethics committees of the first affiliated hospital of Jinan University (Approval No: [2019 LPK No. 020]). Informed consent was obtained from all participants.
Thanks to the midwives Lin Jinrong, Feng Caizhen, Chen Danliang and the obstetricians of the First Affiliated Hospital of Jinan University for their help to this study. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
This research received no external funding.
The authors declare no conflict of interest.


