1 Departement of Cardiology, The First Affiliated Hospital of Chongqing Medical University, 400016 Chongqing, China
2 Department of Obstetrics and Gynaecology, The Second Affiliated Hospital of Chongqing Medical University, 400060 Chongqing, China
3 Research and Development Institute, Faculty of Medicine, Transilvania University of Brasov, 500484 Brasov, Romania
4 Key Lab of Etiology and Epidemiology, Center for Endemic Disease Control, Chinese Center for Disease Control and Prevention, Harbin Medical University, 150081 Harbin, Heilongjiang, China
Academic Editor: Panagiotis Anagnostis
Abstract
Background: Imbalance of testosterone/estradiol ratio are frequently
reported to be associated with the risk of cardiometabolic diseases; however,
studies have yet to report the testosterone/estradiol ratio and its relationship
to cardiometabolic diseases in women at menopause transition. This study aimed to
explore the association between
testosterone/estradiol ratio with cardiometabolic diseases in women during their
menopausal transition age. Methods: 551 women aged from 45 to 55 years
old were involved in this study. Their baseline information, disease comorbidity,
blood biochemical tests, echocardiography and serum sex hormones were collected.
Women were categorized by tertile distribution of testosterone/estradiol ratio.
We used binary logistic regression model (for odds ratio) and poissoon loglinear
model (for prevalence ratio) to evaluate the association between testosterone/estradiol ratio with cardiometabolic diseases.
Results: The mean age of the study population was 48.6
Keywords
- cardiometabolic diseases
- testosterone/estradiol ratio
- menopause transition
- aging
Cardiometabolic diseases (CMD) are the leading causes of death among women globally [1]. In the past decades, series of campaigns and awareness had helped to ease the impact of CMD on women. Despite these efforts, the overall reduction in the burden of CMD on women has been temporary and stagnant over the last 10 years [2].
The cardio-protective role of estradiol (E2) in women has been a well-established fact [3]. On the other hand, the data regarding the association of testosterone (T) and cardiovascular diseases (CVD) are still conflicting where some showed negative [4, 5], positive [6, 7], while others no association [8, 9]. The testosterone to estradiol (T/E2) ratio may play an important role in the rising cardiometabolic risk throughout the menopausal transition period [10]. A higher T/E2 ratio was reported to be associated with increased risk for CVD, coronary artery diseases (CAD), and heart failure (HF) in older postmenopausal women and hyperlipidemia in CAD [11].
Age and menopause status are the key factors affecting sex hormone level. The previous studies, in their efforts to understand CMD association with T/E2 ratio in women [6, 12, 13], had not considered the effect of age on hormonal level (wider age gap, younger women vs older women). They had included mostly postmenopausal women in favor of premenopausal women [6, 13]. In the quest for establishing an association of CMD risk among women at different ages, change in the balance of T/E2 ratio is more reliable, than T or E2 alone [13]. Currently, there are no available studies that discuss the relationship between T/E2 ratio with CMD in women around menopause transition period [13, 14]. This period generally presents at the age between 45 and 55 years old, and also have been linked with increased risk of CMD [13, 15, 16, 17].
Therefore, we designed this study to generate preliminary findings on the association between T/E2 and CMD to support our hypothesis which states that, “the ratio of T/E2 level in women during their menopausal transition period could be a predictive or diagnostic marker for women who are at risk of CMD.” [14].
This study involved women from general population, residents of Chongqing city, China aged between 45 to 55 years old who visited the First Affiliated Hospital of Chongqing Medical University from May 2018 to August 2021 for their annual medical examination.
We recruited 1147 women. In order to eliminate potential confounder, 596 women were excluded due to their conditions known to heavily impacted on sex hormones level. The exclusion criteria are listed as follow: (1) inaccurate report of their last menstrual cycle date (n = 72); (2) diagnoses of oligomenorrhea [18] (n = 147); (3) pregnancy or had breastfed within the past two years (n = 6); (4) sudden weight loss in the last 6 months prior registration [19] (n = 24); (5) diagnoses of any malignant tumors [20] (n = 46); (6) pituitary or adrenal gland diseases (n = 59); (7) or polycystic ovary syndrome [21] (n = 35); (8) history of hysterectomy or oophorectomy [22] (n = 56); (9) an experience of psychological trauma or emotional distress within past 4 weeks (n = 24); (10) the use of oral contraceptive (OCT) pills or hormone replacement therapy (HRT) in the last 3 months (n = 127). In total 551 women were retained for the study.
This study was approved by the ethics committee of the First Affiliated Hospital of Chongqing Medical University. All the participants were informed of the study content and signed informed consent.
We drew the fasting venous blood samples of the studied group in the morning, 1 to 2 hours after awakening, between 8–9 AM, after approximately 12 hours of overnight fasting, with the subject comfortably seated. The blood samples were centrifuged immediately for 15 min at 4 °C, and the platelet-free serum and plasma were stored at 22 °C to get the most accurate results of hormone levels test. We collected samples from premenopausal women with a normal menstrual cycle from day 19 to 21 of their monthly cycles [23].
The total T, E2, follicle-stimulating hormone (FSH), and luteinizing hormone (LH) were analyzed in the hospital lab using an electrochemiluminescence (ECL) system automatically (COBAS 8000 Series, E602 modular analyzer, Roche Diagnostics International Ltd, CH-6343 Rotkreuz, Switzerland). All the Biochemical samples like high-density lipoprotein cholesterol (HDL-C) and low-density lipoprotein cholesterol (LDL-C), triglycerides (TG), and the total serum cholesterol (TC) were determined by an automated enzymatic procedure (Cobas E602, Roche), using serum samples collected after fasting. The fasting blood glucose was similarly measured. Within a week, we analyzed all samples.
The transition to menopause is characterized by the onset of vasomotor symptoms such as hot flashes, night sweats, and other common menopausal symptoms like sleep disturbances, depression, and anxiety [24].
Menopausal status was defined as FSH that is
above 25 mIU/mL and E2 below 50 pg/mL after permanent cessation of
the menses for 12 consecutive months [25]. T/E2 is the ratio of serum
T (pg/mL) to serum E2 (pg/mL). Obesity was defined as body mass
index (BMI)
HF was diagnosed when there is an increased B-type natriuretic peptide (BNP) or
N-terminal (NT)-prohormone B-type natriuretic peptide (NT-proBNP) profile value
accompanied by clinical manifestations or signs of HF. CAD was diagnosed when
coronary artery stenosis is
CMD refers to risk factors such as elevated blood glucose, abdominal obesity, hypertension (HTN), MetS, hyperlipidemia, and elevated triglycerides, resulting in an interrelated set of conditions that include obesity, HTN, T2DM and CVD [28].
Women were grouped based on the tertiles value of T/E2 ratio, continuous
variables with normal distribution were expressed as the mean
Categorical variables were expressed by a number of cases and percentages, and
the differences between groups were compared by the Pearson
To determine the relationship between T/E2 and the parameters of CMD with its components, we calculated odds ratio (OR) and prevalence ratio (PR) and each with its 95% confidence interval (CI). Model 1 adjusted for age. Model 2 further adjusted for Han ethnicity, lifestyle variable: married, never smoke and never alcohol; OCT, HRT use. Model 3 further adjusted for number of live births, menopause staus, CAD family history.
We performed the statistical analysis using the SPSS statistical software
version 27.0.1 (SPSS Inc., Chicago, IL, USA). We also employed the Graph Pad
Prism 9.3.0 (GraphPad Software, Inc., San Diego, CA, USA) for plotting. All the
statistical tests were two-tailed, and the significance level was set at
p
The baseline characteristics of participants were listed in Table 1. The mean
age of the study population was 48.6
| Characteristics | Overall (N = 551) | Tertiles of testosterone to estradiol (T/E2) ratio | p | ||
| T1 |
T2 = 3.9–13.0 | T3 | |||
| n = 183 | n = 198 | n = 170 | |||
| Age, yrs | 48.6 |
46.5 |
48.0 |
51.5 |
|
| Age |
207 (37.6) | 22 (12.0) | 56 (28.3) | 129 (75.9) | |
| Han race, n% | 537 (97.5) | 179 (97.8) | 195 (98.5) | 163 (95.9) | 0.267 |
| Married, n% | 540 (98.0) | 180 (98.4) | 190 (96.0) | 170 (100.0) | 0.020 |
| Chongqing natives, n% | 469 (85.1) | 158 (86.3) | 166 (83.8) | 145 (85.3) | 0.788 |
| Never smoke, n% | 521 (94.6) | 172 (94.0) | 187 (94.4) | 162 (95.3) | 0.861 |
| Never alcohol, n% | 519 (94.2) | 171 (93.4) | 188 (94.9) | 160 (94.1) | 0.820 |
| Never OCT, n% | 523 (94.9) | 171 (93.4) | 189 (95.5) | 163 (95.9) | 0.529 |
| Never HRT, n% | 529 (96.0) | 176 (96.2) | 190 (96.0) | 163 (95.9) | 0.989 |
| Number of parities | 3.0 (2.0, 5.0) | 3.0 (2.0, 5.0) | 3.0 (2.0, 5.0) | 3.0 (2.0, 4.0) | 0.966 |
| Number of livebirths | 1.0 (1.0, 2.0) | 1.0 (1.0, 2.0) | 1.0 (1.0, 2.0) | 1.0 (1.0, 2.0) | 0.157 |
| Number of abortions | 2.0 (1.0, 3.0) | 2.0 (1.0, 3.0) | 2.0 (1.0, 3.0) | 2.0 (1.0, 3.0) | 0.852 |
| Age of menarche, yrs | 14.0 |
13.7 |
14.0 |
14.1 |
0.028 |
| *Days of menstrual period | 4.9 |
5.0 |
4.9 |
4.8 |
0.218 |
| *Days of menstrual circle | 28.1 |
28.0 |
27.9 |
28.5 |
0.055 |
| *Irregular menstrual circle, n% | 28 (5.1) | 15 (8.2) | 7 (3.5) | 6 (3.5) | 0.064 |
| Menopause status, n% | 168 (30.5) | 14 (7.7) | 51 (25.8) | 103 (60.6) | |
| Age of menopause, yrs | 49.0 (46.0, 51.0) | 46.0 (45.0, 50.0) | 47.0 (45.0, 50.0) | 50.0 (47.8, 51.0) | 0.001 |
| Months since menopause | 36.0 (11.0, 72.0) | 6.0 (2.0, 17.3) | 36.0 (11.0, 60.0) | 36.0 (24.0, 78.0) | |
| Menopause |
48 (8.7) | 11 (6.0) | 18 (9.1) | 19 (11.2) | |
| Menopause |
120 (21.8) | 3 (1.6) | 33 (16.7) | 84 (49.4) | |
| Dysmenorrhea, n% | 119 (21.6) | 48 (26.2) | 42 (21.2) | 29 (17.1) | 0.111 |
| Hypermenorrhea, n% | 193 (35.0) | 70 (38.3) | 63 (31.8) | 60 (35.3) | 0.420 |
| CAD family history, n% | 96 (17.4) | 32 (17.5) | 31 (15.7) | 33 (19.4) | 0.638 |
| Heart rate, bpm | 85.4 |
84.8 |
86.5 |
84.8 |
0.293 |
| SBP, mmHg | 125.5 |
121.7 |
125.7 |
129.4 |
|
| DBP, mmHg | 77.5 |
74.2 |
78.1 |
80.3 |
|
| Weight, kg | 58.9 |
57.9 |
59.5 |
59.4 |
0.083 |
| Height, cm | 157.1 |
157.3 |
157.1 |
157.0 |
0.875 |
| BMI, kg/m |
23.9 |
23.4 |
24.1 |
24.1 |
0.045 |
| Hemoglobin, g/L | 118.3 |
117.5 |
117.7 |
119.9 |
0.150 |
| Albumin, g/L | 40.3 |
40.1 |
40.4 |
40.3 |
0.862 |
| Serum creatinine, mmol/L | 59.1 |
58.2 |
59.0 |
60.1 |
0.531 |
| Serum calcium, mmol/L | 2.3 |
2.3 |
2.3 |
2.3 |
0.007 |
| eGFR, mL/(min × 1.73 cm |
105.2 |
108.6 |
106.2 |
100.5 |
|
| Total cholesterol, mmol/L | 4.5 |
4.3 |
4.6 |
4.6 |
0.359 |
| Triglyceride, mmol/L | 1.1 (0.9, 1.7) | 1.1 (0.8, 1.9) | 1.2 (1.0, 1.6) | 1.2 (0.9, 1.9) | 0.766 |
| LDL-C, mmol/L | 2.8 |
2.6 |
2.9 |
2.9 |
0.118 |
| HDL-C, mmol/L | 1.4 |
1.4 |
1.4 |
1.3 |
0.991 |
| HDL-C/LDL-C | 0.5 |
0.6 |
0.5 |
0.5 |
0.659 |
| Fasting glucose, mmol/L | 5.4 |
5.3 |
5.4 |
5.6 |
0.074 |
| LVDD, mm | 45.9 |
45.9 |
46.0 |
45.8 |
0.898 |
| RV, mm | 18.4 |
18.3 |
18.2 |
18.5 |
0.646 |
| IVS, mm | 9.6 |
9.4 |
9.5 |
9.9 |
|
| LVEF, % | 67.4 |
67.7 |
67.4 |
67.1 |
0.598 |
| *Postmenopausal women data from previous periods. OCT, oral contraceptive therapy; HRT, hormone replacement therapy; CAD, coronary artery disease; SBP, systolic blood pressure; DBP, diastolic blood pressure; BMI, body mass index; eGFR, estimated glomerular filtration rate; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; LVDD, left ventricular diastolic diameter; RV, Right ventricle size; IVS, interventricular septum; LVEF, left ventricular ejection fraction. | |||||
However, women in the three different T/E2 value groups had some similarities such as alcohol consumption, smoking, OCT and HRT, parities and live births, menstruations, CAD family history, heart rate, serum creatinine, fasting glucose and lipid level, heart size and its left ventricular ejection fraction (LVEF).
The overall distribution of T, E2 and T/E2 ratios were presented in the supplementary materials (see Supplementary Figs. 1,2). When grouped by age segments, serum T levels had a slight decreasing trend as age increased, while serum E2 levels decreased inversely proportional to age, resulting in T/E2 ratio to be positively correlated with age (Fig. 1).
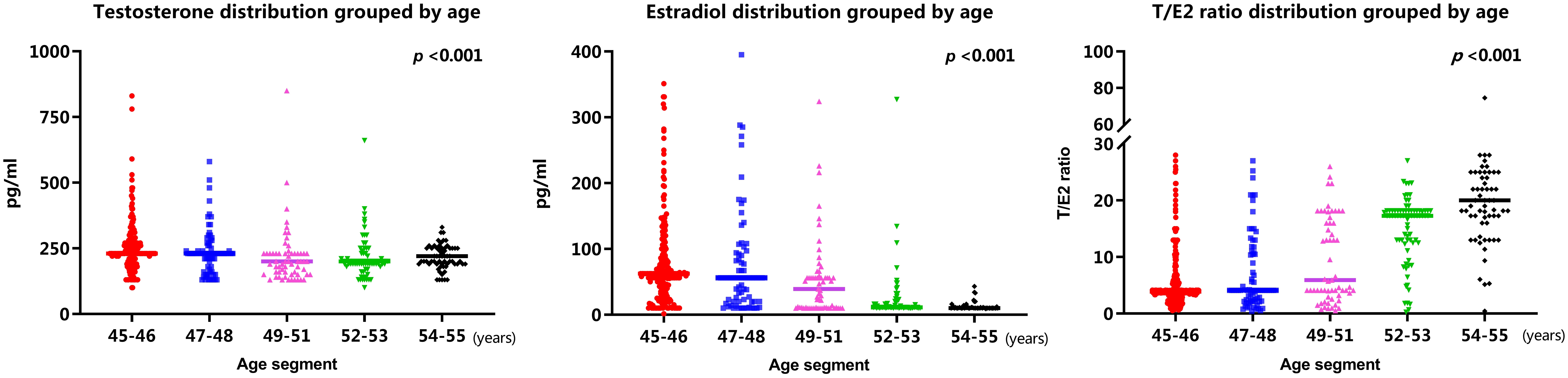 Fig. 1.
Fig. 1.The distribution of testosterone and estradiol levels, and T/E2 ratio grouped by five age segments.
Women with higher level of T/E2 ratio were likely to have obesity, HTN, T2DM,
MetS, CVD and CMD all p
| Diseases | Overall (N = 551) | Tertiles of T/E2 ratio | p | ||
| T/E2 |
T/E2 = 3.9–13.0 | T/E2 | |||
| n = 183 | n = 198 | n = 170 | |||
| Obesity | 48 (8.7%) | 8 (4.4%) | 18 (9.1%) | 22 (12.9%) | 0.017 |
| Hypertension | 164 (29.8%) | 35 (19.1%) | 70 (35.4%) | 59 (34.7%) | 0.001 |
| T2DM | 44 (8.0%) | 8 (4.4%) | 16 (8.1%) | 20 (11.8%) | 0.038 |
| Hyperlipidemia | 21 (3.8%) | 9 (4.9%) | 6 (3.0%) | 6 (3.0%) | 0.613 |
| MetS | 74 (13.4%) | 18 (9.8%) | 24 (12.1%) | 32 (18.8%) | 0.037 |
| CVD | 53 (9.6%) | 11 (6.0%) | 14 (7.1%) | 28 (16.5%) | 0.001 |
| CAD | 48 (8.7%) | 10 (5.5%) | 13 (6.6%) | 25 (14.7%) | 0.004 |
| HF | 6 (1.1%) | 1 (0.5%) | 1 (0.5%) | 4 (2.4%) | 0.161 |
| CMD | 246 (44.6%) | 54 (29.5%) | 99 (50.0%) | 93 (54.7%) | |
| T2DM, type 2 diabetes mellitus; T/E2 ratio, testosterone/estradiol ratio; MetS, metabolic syndrome; CVD, cardiovascular diseases; CAD, coronary artery diseases; HF, heart failure; CMD, cardiometabolic diseases. | |||||
As displayed in Fig. 2, when grouped by tertiles, after increasing adjustments
in binary logistics regressions, women with T/E2
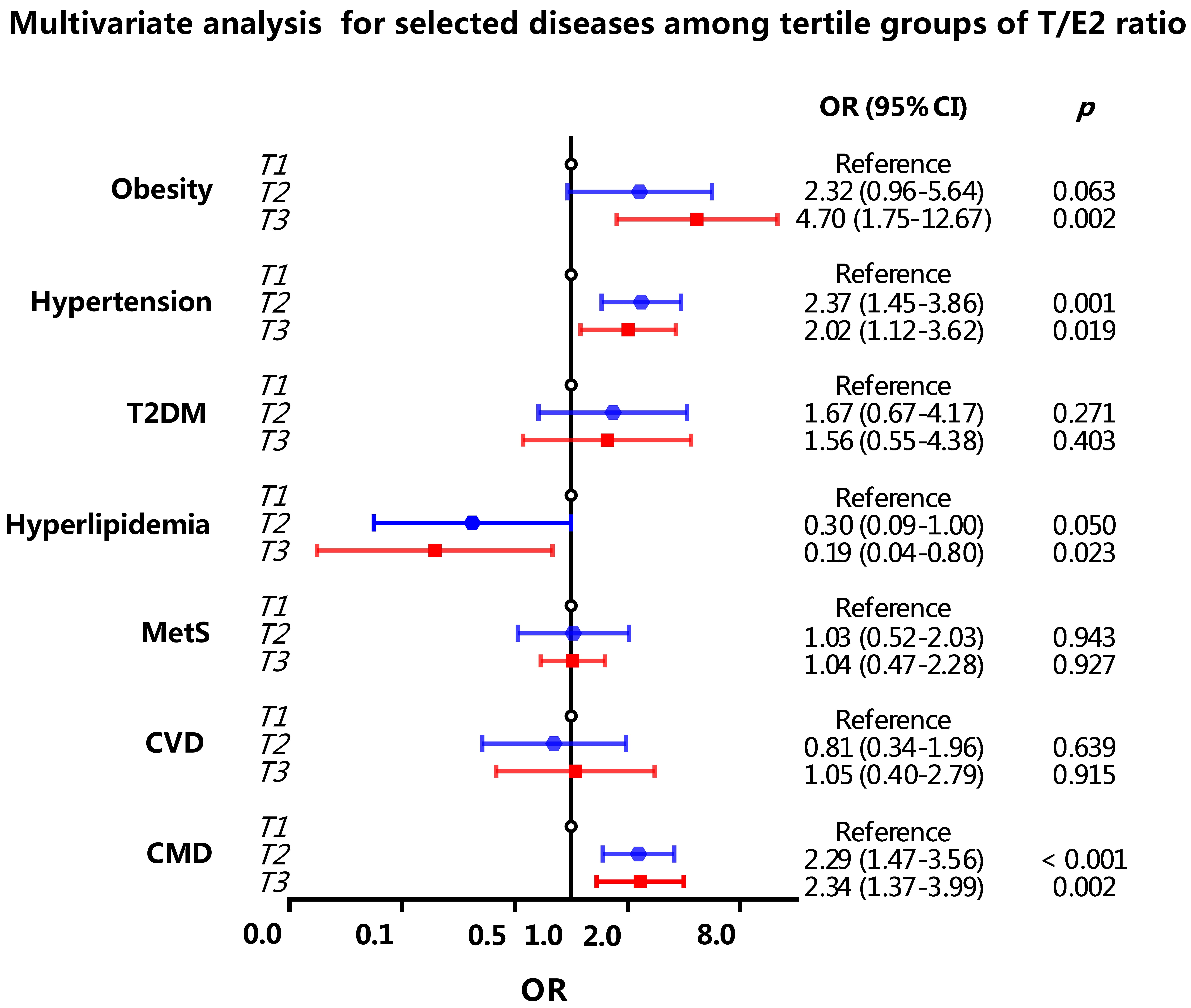 Fig. 2.
Fig. 2.Odds ratio between T/E2 ratio with CMD and its components.
Groups were the tertiles of T/E2 ratio, and were set with T1
In detail, after multivariate analysis, the OR (95% CI)
across the tertile groups for obesity were: 1.0 (reference), 2.32 (0.96–5.64),
4.70 (1.75–12.67) (p
In comparison, PR (95% CI) for obesity were: 1.0 (reference), 2.16
(0.93–5.03), 4.01 (1.59–10.11) (p
As for hyperlipidemia, multivariate regression analysis indicated that women
with T/E2
This study objective was to evaluate the relationship between T/E2 ratio with cardiometabolic risk of Chinese women at menopause transition age (generally between 45 to 55 years old). Our results revealed that (1) despite the short enrollment age range, both T and E2 level varied differently through age segments, this had resulted to significant differences in T/E2 among age groups; and that (2) women with high T/E2 ratio had a higher prevalence risk of CMD such as obesity and HTN.
Erratic changes in E2 levels in women happened long before menopause [18]. And when they are near menopause, E2, and progesterone level decrease but FSH increases [29]. Women may be exposed to higher estrogenic activity before the drastic decline, because of early drop-in ovary activity in anticipation of menopause transition period [30]. This hormonal alteration is generally translated to early menopausal symptoms such as infrequent menstruation (oligomenorrhea), night sweating, sleep disturbance and hot flashes (vasomotor symptoms) [31].
Following early sign of menopause comes the amenorrhea periods, which is the period during which women ovarian function slowly decline along with the decrease level of E2. As women progress through this time, they completely loss the ovarian function and the permanent cessation of menses at which after 12 months they are considered postmenopausal. Consequently, women permanently remain in low hypoestrogenic state as the E2 and progesterone level also remained low after menopause. Studies on T in women have shown that there is either increase or stable T level during menopause [32]. However, more evidence suggest that the decline is due to age instead of menopause [33]. Most importantly, owing to the steep decline of E2 at menopause transition, T/E2 increases in post-menopausal women, which may involve endothelial and cardiovascular health [34].
Data regarding T and its relationship with CMD are mixed. While Lambrinoudaki et al. [35] have claimed that T is positively correlated with CVD-related risk in postmenopausal women, Mudali and colleagues pointed to its deleterious effects on lipid profiles [36]. The latter consequently lead to the occurrence of atherosclerosis formation, specifically in carotid arteries [37], therefore, cardiometabolic related risk factors [38] such as HTN, obesity, and insulin resistance in postmenopausal women escalate [39]. It is important to consider that studies have been contradictory despite the potential pathological linkage between CVD events and T levels.
In addition, a cohort study claimed that higher testosterone levels were associated with an increased risk of CAD in a study with 30 years follow-up [7], which contradicted the finding from the Laughlin and colleagues [40], that lower T levels were associated with increased CVD events in post-menopausal women. Furthermore, another cohort study in Germany involving 2129 middle-aged women [8] and the Women’s Health Study [41] have both concluded a null association between T with CVD incidents.
The protective effects of estrogens on cardio-metabolic physiology works through several mechanisms. One of these mechanisms is its through increasing the synthesis of nitric oxide, which in turn causes vasodilation [31]. Another mechanism involves its regulatory effect on serum lipids which further decreases the risk of plaque formation; thus, ensuring a normal blood flow.
Beside these main effects, estrogens further contribute to endothelial vasodilation through regulating regulate specific inflammatory markers and cytokines [42]. Conversely, literature has shown that decreased E2 levels could increase the risk of CAD in women [7]. When studying the menopausal transition, it was observed that women with higher premenopausal E2 levels had lower risk of developing carotid plaque after menopause, compared to those that had a lower premenopausal E2 level [43]. This evidences that the decrease of E2 may associate with increased risks of CMD.
In contrast, other studies argued that high exposure to estrogen in premenopausal women, such as in pregnancy [13], is associated with disadvantageous metabolic profiles, including high blood pressure or hyperglycemia, which led to an increased risk of HTN and T2DM later in life [44]. High estrogenic exposure was also reported to be associated with the risk of CAD and stroke among older postmenopausal women [45].
While the aforementioned studies revealed that high E2 exerted possible benefits on the cardiometabolic health, other data suggested its detrimental effects. However, these studies converge with the “timing hypothesis”, in which estradiol has an undesirable vascular effect in older women, while it has neutral or positive effects in younger women [46].
During the menopausal transitioning, the increasing T/E2 ratio coincides with an increased incidence of CVD, T2DM, and other metabolic risk factors including HTN, dyslipidemia, impaired glucose metabolism, and obesity [43]. The dysregulated serum T levels association with aging and/or the diminishing cardio-protective E2 levels [47] could be the reasons for the increased risk of CMD [11].
In the present study, we found an association between a higher T/E2 ratio and increased incidence of CMD. We previously assumed that the imbalance in the T/E2 ratio among these women was a result of decreased E2 as they experience a swift decline of E2 during the menopausal transition age compared to the gradual decline of T level. Therefore, we evaluated the association of T/E2 ratio with CMD-related risk factors. The results indicated that higher T/E2 ratio was associated with CMD and its components such as obesity and HTN. This suggest that a balance level of T/E2 ratio may play a reliable key role in CMD-related risk in women around the menopause transition period.
Our study is intended to provide preliminary data on T/E2 ratio association with CMD in women transitioning into menopause.
Treatment with an appropriate of T/E2 ratio appeared to attenuate injury to the endothelium and also reduced inflammation in cell and animal models [48]. This finding suggested that a combined therapy of T and E2 based on their ratio would prove to be more efficient compared to the exclusive replacement therapy of T or E2.
Based on our results, we suggest further prospective studies assessing women transitioning into menopause with intended purpose of proving the importance of the T/E2 ratio diagnosis in women transitioning into menopause.
Though our study has many strengths, such as the inclusion of only women transiting to menopause, between the ages of 45 and 55 years, articulated exclusion criteria, and well-diagnosed comorbidities, we observed few limitations.
This is a single-center cross-sectional study with a small sample size due to strict exclusion criteria for enrollment and regional confinement of southwestern Chinese women. Therefore, we recommend a large-scale multicenter cohort study across race/ethnicity among women within the age bracket of 45–55 years in the future.
We also acknowledge that we did not use fertility monitors for to appropriately account menstrual cycle phase among premenopausal women. While obesity is a quantifiable measure emphasizing the stratification parameters associated to the BMI and waist circumference, adiposity refers to the distribution of body fat. In our study, we did not calculate the amount of visceral fat presented. However we found that BMI was statistically significant with T/E2 ratio.
Our study indicates that a higher T/E2 ratio is associated with a high prevalence of CMD in women at menopause transition age. From our findings, we suggest further prospective research to confirm the sensitivity of the T/E2 ratio in predicting the possibility of CMD in women around their menopause transition period.
The dataset used and analyzed in this study is available from the corresponding author [GL] upon reasonable request.
FBA—Investigation, Data collection, Visualization, Writing - Original Draft, Conceptualization; YX—Investigation, Data collection; BH, BRS—Supervision, Resources; AC, SCO—Writing - Review & Editing; AS—Visualization; GL—Writing - Review & Editing, Methodology, Software, Formal analysis, Project administration, Funding acquisition; SL—Project administration, Supervision. All authors read and approved the final manuscript.
This study was approved by the ethics committee of the First Affiliated Hospital of Chongqing Medical University, No. 2020-23. All the participants were informed of the study content and signed informed consent.
Not applicable.
This study is supported by the cultivation fund of the First Affiliated Hospital of Chongqing Medical University, No. PYJJ2019-25.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/j.ceog4912260.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


