1 Obstetrics and Gynecology Department, Hospital das Clinicas HCFMUSP, Faculdade de Medicina, Universidade de São Paulo, Av. Dr. Enéas de Carvalho Aguiar, 255, 10° andar, 05403-000 Cerqueira Cesar, SP, Brazil
2 Unit of Gynecology and Obstetrics, Department of Women and Children's Health, University of Padua, 35122 Padua, Italy
3 Gynecologic Division, Universidade Federal de São Paulo, Escola Paulista de Medicina, 04023-062 São Paulo, SP, Brazil
4 Department of Public Health, University of Naples Federico II, 80138 Naples, Italy
Academic Editor: Ioannis A. Kalogiannidis
Abstract
Objectives: To provide an updated literature overview about the pathophysiology, natural history, diagnosis and treatment of endometrial polyps (EPs). Findings in Brief: EPs are focal, sessile or pedunculated projections of endometrial lining, resulting from the hyperplastic overgrowths of endometrial glands and stroma around a vascular core. These are the most common intrauterine diseases, affecting up to 34.9% of the women during their lifetime. EPs may vary in size, location, histopathology, natural history and clinical presentation. Importantly, EPs are common in women suffering with infertility (i.e., up to 32% in patients undergoing in-vitro-fertilization) and are not exempt from risk of malignant transformation (occurring in about 3.1% of patients). For these reasons, EPs represent one of the most significant gynaecological problems worldwide. Trans-vaginal ultrasound has fair diagnostic accuracy for EPs, with sensitivity and specificity of 91% and 90%, respectively. Nevertheless, the current gold standard for EPs diagnosis is hysteroscopy, which allows a direct visualization of EPs size and morphology. Moreover, this technique enables safe EPs excision, with a low risk of incomplete tissue removal (about 1.9% and 4.3% in inpatient and outpatient settings, respectively), and variable recurrence rate based on individual risk factors (ranging between 2.5% and 43.6%). Besides hysteroscopy, other management options for EPs can be considered on a case by case basis. Conclusions: Our review may be helpful for updating clinicians’ knowledge on the management of EPs.
Keywords
- endometrial polyps
- etiology
- diagnosis
- infertility
- hystopathology
- hysteroscopy
- natural hystory
- polypectomy
- endometrial cancer
Endometrial polyps (EP) are focal, sessile or pedunculated projections of endometrial lining, resulting from the hyperplastic overgrowths of endometrial glands and stroma around a vascular core [1]. They are mostly benign, however areas of atypical hyperplasia or endometrial carcinoma may be found in 3.8% of postmenopausal patients [2].
The main clinical manifestation of EP is abnormal uterine bleeding (AUB), which is reported in 3.7–65% of patients diagnosed with EP [3]. Otherwise, EP are often incidentally discovered in asymptomatic women at routine ultrasound examination [4]. Importantly, EPs may be associated with infertility and recurrent miscarriage in young women [5].
Aim of the present study was to provide an overview of literature about the pathophysiology, natural history, diagnosis and treatment of EPs.
EP can be sessile or pedunculated, single (80%) or multiple (20%) and may vary in diameter from a few millimeters to centimeters, also presenting varied forms [6]. The most common aspect is a single, bulb-shaped, pedunculated mass equal or less than 1 cm in mean diameter [7] (Fig. 1a–d).
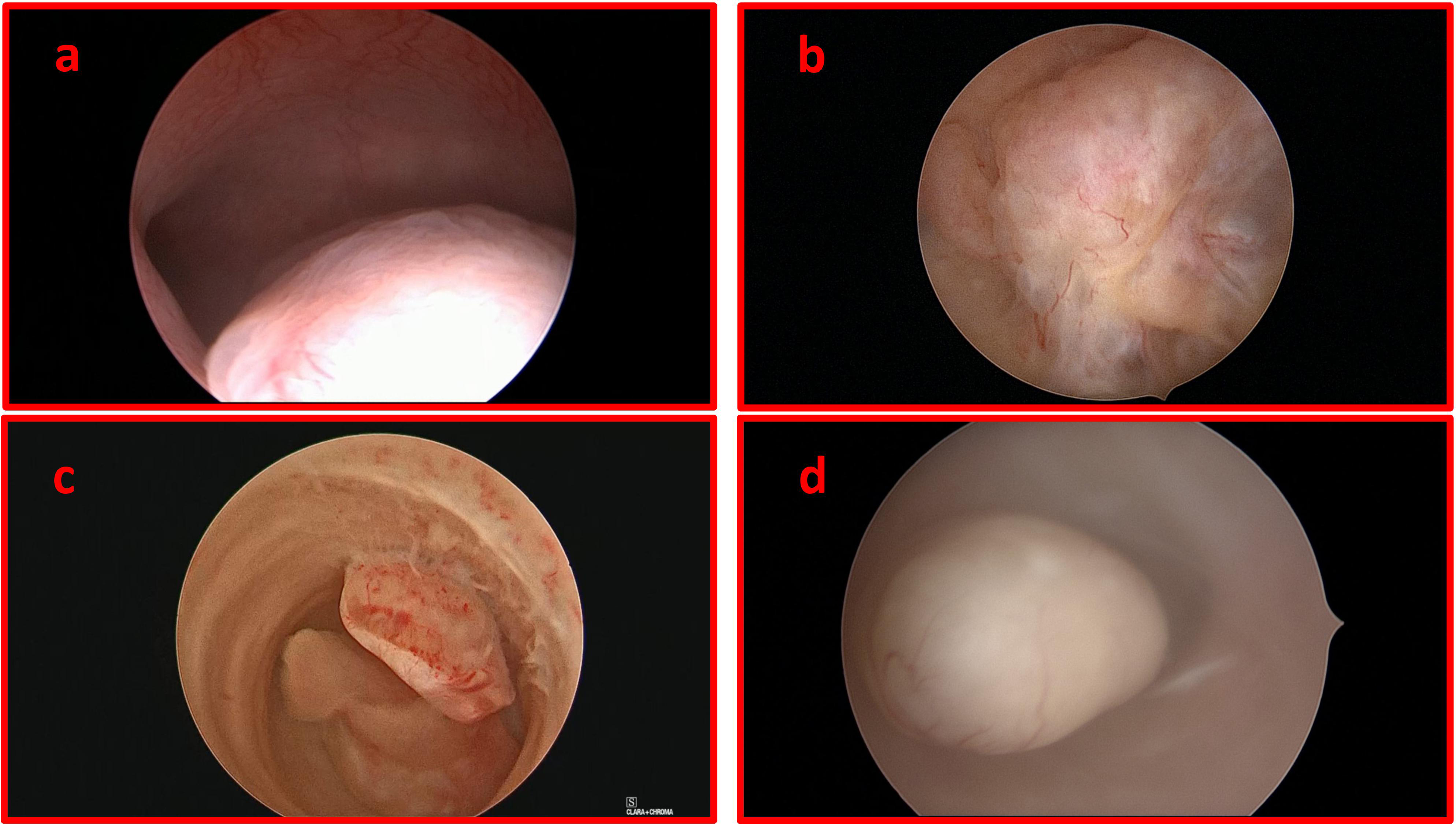 Fig. 1.
Fig. 1.Hysteroscopic pictures of endometrial polyps: functional endometrial polyp in infertile pre-menopausal woman (a); cystic endometrial polyp in a woman undertaking adjuvant tamoxifen therapy for breast cancer (b); atypical endometrial polyps in post-menopausal woman (c); adenomatous endometrial polyp in post-menopausal woman (d).
The majority of EP arise from the uterine fundus (55.8%) and cornual mucosa (29.4%), extending towards the internal orifice of the uterine cervix. Occasionally, they protrude beyond the external cervical orifice (ECO) and can be easily identified at cervical inspection [8].
EP are mainly composed by dense fibrous tissue (stroma) and glands surrounding a vascular core, covered by superficial epithelium. In some cases, smooth muscle tissue may be present. Polyps are currently classified in five categories [9]:
- Hyperplastic polyps: Arise from the basal layer of the endometrium, resulting from the estrogenic stimulus, without opposition of progesterone. They may be associated with endometrial hyperplasia and present atypical areas, especially in postmenopausal women.
- Atrophic polyps: Found in postmenopausal women, resulting from regression of hyperplastic or functional polyps.
- Functional polyps: Glandular changes similar to those found in the rest of the endometrium. They respond to the hormonal stimuli of the menstrual cycle.
- Adenomatous polyps: Displaying the presence of smooth and fibrous muscle tissue in varying proportions. The “atypical” forms are characterized by the concomitant presence of structural atypia, and may be associated with malignant transformation.
- Pseudopolyps: Small sessile lesions, generally smaller than 1 centimeter, similar to the rest of the endometrium; detected only in the secretory phase of the menstrual cycle and may disappear after the menstrual flow.
The actual prevalence of EP in the general population is difficult to be determined. Current knowledge is main limited by the lack of studies focusing on asymptomatic patients. However, it is estimated that EP may affect a proportion of women from 7.8% to 34.9%, depending on individual risk factors and population studied [7]. The prevalence of EP is higher in postmenopausal (11.8%) than premenopausal women (5.8%), whilst is even superior in women affected by infertility (up to 32% in women candidates to in-vitro-fertilization) [7, 8, 9, 10].
Many risk factors have been correlated with the development of EP, including: family history, age, diabetes, hypertension, obesity, hyperestrogenism and Tamoxifen therapy [11].
- Genetic and hereditary factors: Chromosomal abnormalities, such as alterations in chromosomes 6 and 20, may play a role on the development of EP [12].
- Age: Regarding age, prevalence of EP seems to increase with age during reproductive years, but it is unclear whether it continues to increase or decrease after menopause [13].
- Diabetes: Diabetes mellitus can act as a risk factor for EPs. However, it is still not clear whether or not this condition primarily influences growth or the malignant degeneration of EP [14].
- Hypertension: Systemic hypertension, especially associated with obesity, seems to be an important factor that may be correlated with the development of EP. Furthermore, advancing age is related to the increase in systolic pressure and the prevalence of hypertension [14].
- Obesity, hyperestrogenism and tamoxifen: Exposure to relative or absolute excess of estrogens is supposed to increase the risk of developing EP. Indeed, obesity, early menarche and late menopause (all conditions with higher exposure to estrogens) are associated with higher prevalence of EP. Interestingly, tamoxifen users have been also showed to be at higher risk of developing EP (besides endometrial cancer). It is probably due to the lack of anti-proliferative activity of progesterone hormone on endometrial mucosa [15].
Monoclonal origin of EPs was first demonstrated by Jovanovic et al. [16] on hysterectomy specimens from 22 women in 1996. However, the exact cause of EP is still unknown. Due to their histopathological heterogeneity, a multifactorial origin has been hypothesized. In particular, genetic, inflammatory, hormonal and iatrogenic factors are supposed to play some role in their origin [12].
- Genetic factors: The importance of genetics in the pathogenesis of EP is emphasized by the models of Lynch or Cowden syndrome [17]. These two genetic syndromes are associated with higher risk for EP, as well as for endometrial cancer [18].
Abnormalities in chromosomes 6 and 12, such as chromosomal rearrangements (translocation), have been described in women with EP, resulting in altered endometrial pattern with the appearance of polypoid structures [19]. Cytogenetic rearrangements in the family of high mobility chromosomal proteins (HMGs) appear to be involved in the development of EP [20]. Moreover, the higher prevalence of polyps in postmenopausal women has been associated with an higher expression of the p63 protein in EP and the adjoining endometrium [21]. Finally, identified the presence of the p53 protein as a biological activity marker of endometrial polyps in postmenopausal patients [22]. Some authors suggested that p53 overexpression in the polyps may be associated with the increased risk for endometrial cancer development. Known as the “guardian of the genome”, p53 exerts a tumor suppressor activity by downregulating genes governing proliferation and up regulating genes involved in preventing growth. It is considered essential for preventing inappropriate cell proliferation and in maintaining genomic integrity following genotoxic stress [23].
Inflammation: High levels of matrix metalloproteinase (MMPs), proteins involved in mechanisms of angiogenesis, migration, proliferation and cell death have been described in endometrial fluid of women with various uterine pathologies, including also EP (in addition to leiomyomas and adenomyosis). Recent studies and review showed increased risk of EP in women suffering from chronic endometritis [24]. This data supports a potential inflammatory origin for some kinds of EP [25].
- Hormonal factors: The role of sexual hormones (estrogen, progesterone) in the pathogenesis of EP is currently subject of debate. Some Authors speculate that a greater expression of aromatase [26] and sexual hormone receptors may be responsible for the stretching of the endometrial glands, stromal tissue and spiral arteries that confer their usual morphology [27].
- Iatrogenic factors: The impact of hormone replacement therapy on the development of EP is controversial [7], whilst the use of levonorgestrel-releasing intrauterine device seems to be protective [28].
EP may regress, persist, enlarge or undergo malignant transformation. The spontaneous regression rates of EP are mainly dependent on their size. In particular, the bigger is the polyp, the lower is the chance of spontaneous regression. At this regard, a recent study found a regression rate of 26.7% after 1 year follow-up for EP with a mean diameter of 10.7 mm. Differently, the regression rate of EP with mean diameter of 15.1 mm was considerably lower (mean difference, 4.4; 95% confidence interval (CI), 0.2–8.6; p = 5.04) [29].
Corroborating these findings, a prospective study showed a tendency of polyps bigger than 1 cm to persist and cause AUB, compared to polyps smaller than 1 cm [30]. The phenomenon of EP regression may be associated with one or more episodes of menorrhagia and mild/moderate pelvic pain [31].
Although the majority of EP are benign, a small proportion (ranging from 0% to 12.9%) may be subject to transformation in pre-malignant and malignant lesions [32]. Nevertheless, solid evidence on this topic is lacking [33]. First, it is hard to establish if pre-malignant and malignant lesions originate from the polyp itself or from adjacent endometrium (involving EP in a later time). Second, published studies evaluated the correlation between EP and cancer, rather than the biological evolution of EP [34].
A prospective controlled study, including 248 women with abnormal uterine
bleeding (AUB) with and without endometrial polyps, detected a higher frequency
of endometrial hyperplasia in women with polyps. However, in this study
population, no difference in malignancy was observed between the groups [32].
Ferrazzi et al. [35] in a multicenter study including 1922 women,
compared the presence of malignant and premalignant lesions in EP of
postmenopausal patients with and without AUB. The prevalence of atypical
hyperplastic polyps was 1.2% in asymptomatic women and 2.2% in symptomatic
patients (p
In a recent systematic review including 9266 women, the pooled prevalence of endometrial cancer confined to EP was 3.1%. The rates were lower for premenopausal women (1.12%) than post-menopausal ones (4.93%) [36]. Moreover, if considering only postmenopausal patients (2586 women), the presence of premalignant and malignant lesions was 1.8% and 2.3%, respectively [37].
When EP are diagnosed, the risk factors for endometrial cancer are advanced age
(
In support of the first hypothesis, a retrospective study on 1467 patients with EP found a prevalence of 8.5% of endometrial cancer (n = 125, of which 10.4% confined to EP and 89.6% included EP and the adjacent endometrium. Insightfully, the rate of endometrial cancer in women with EP was lower in comparison to those affected by uterine fibroids (11.7%), leading authors to the conclusion that EP are not precursor of endometrial cancer, but only common incidental findings in women affected by endometrial cancer [47].
The majority of EP are asymptomatic, incidentally discovered at routine ultrasound examination or during the diagnostic workup of infertile couples. When symptomatic, they are associated with AUB, where the intensity of vaginal bleeding is independent from the number, size and location of EP [48]. When large, EP may externalize through the uterine external cervical orifice (ECO) and can be easily identified during routine gynecological examination [49].
- Abnormal uterine bleeding: During menacme, the presence of intermenstrual bleeding is the most frequent complaint. EP are found in 10% to 40% of women suffering from premenopausal bleeding. In the postmenopausal period uterine bleeding with or without hormone replacement therapy use, may occur [13].
- Infertility: EP are found in about 15–25% of infertile women. Although a causal relationship between EP and infertility is not well established, some local factors found in women with EP may exert a negative effect on sperm migration and endometrial receptivity [9, 50]. In particular, different studies showed that EP are associated with increased concentrations of inflammatory cytokines and Glycodelin (glycoprotein secreted by the endometrium that inhibits the function of natural killer cells), as well as with the a reduced expression of some mediators of implantation such as tumor necrosis factor (TNFa) and Insulin-like Growth Factor Binding Protein 1 (IGFBP-1) [51]. Accordingly, a recent systematic review demonstrated that hysteroscopic polipectomy prior to ARTs can improve clinical pregnancy rate in women suffering from infertility [5].
- Physical Examination: During the gynecological examination, only the prolapsed EP can be identified as a globular, friable, pedunculated and externalized structure through the external cervical orifice. However, this finding may correspond, more commonly, to a polyp of cervical origin [52].
- Transvaginal Ultrasound (TUS): EP are often diagnosed by TUS. It is a good cost-effective method, with mean sensitivity of 91%, 90% specificity, 86% positive predictive value (PPV) and 90% negative predictive value (NPV) [7]. In ultrasound image, polyps typically appear as hyperechogenic, regularly contoured lesions within the uterine cavity and along the endometrial line. Cystic spaces may be associated, corresponding to dilated glands with proteinaceous content. These findings, however, are not specific and may be confused with other uterine pathologies, such as submucosal leiomyomas, endometrial hyperplasia and endometrial cancer [45].
Recent studies investigated the role of Power Doppler for the differential diagnosis between endometrial polyps, hyperplasia and cancer. Nevertheless, demonstrated low specificity based on parameters of vascular resistance index, pulsatility and polyp size. The power doppler, however, showed better sensitivity and specificity in the differentiation of the unique vascular pattern present in the typical polyps, compared to the multiple diffuse vessels present in the endometrial hyperplastic and neoplastic lesions. Thus, power doppler may help in the suspicion of malignancy, but may not distinguish between others pathologies, such as endometrial hyperplasia and submucosal myomas. So, there are not enough data in the literature that support the use of Doppler or power doppler ultrasonography instead of histopathological analysis of endometrial polyps after surgical removal [45].
- Hysterosonography: Ultrasound performed with saline infusion allows a better accuracy for the detection of EP through distension of the uterine cavity, being a safe, well tolerated and minimally invasive diagnostic method [7]. Compared to conventional TUS, it allows the detection of minor lesions and a better characterization of the location and size. Compared to hysteroscopy, hysterosonography has demonstrated similar sensitivity and specificity, with the advantage of allowing the concomitant evaluation of other pelvic structures. However, it does not allow the definitive anatomopathological diagnosis or therapeutic possibility [7].
- Diagnostic hysteroscopy: Diagnostic hysteroscopy is considered the gold standard method for the diagnosis of EP, with sensitivity and specificity described of 90% and 93%, respectively; PPV of 96% and VPN of 93% [7]. By offering the direct visualization of endometrial mucosa, hysteroscopy allows a better characterization of EP and the possibility of “see and treat” in outpatient regimen [53]. Office polipectomy has demonstrated high effectiveness in the management of EP, as it allows reaching a definitive (hystopathological) diagnosis in a single procedure step on. When compared to traditional resectoscopic surgery, office hysteroscopy is cheaper, more safe and does not require hospital stay in and analgesia [54].
During the endoscopic evaluation, the following parameters related to EP should be described [9]:
- Number;
- Size: estimate size from the comparative analysis with the Grasping 5-French forceps, which with open jaws has a diameter of 6 mm;
- Location and relationship with the tubal ostium;
- Texture: Soft, friable, dense and semi-myomatous (ademiomatous polyps);
- Characteristics of the implantation base: sessile or pediculated;
- Superficial vasculature: the presence of abundant and tortuous vessels, is the most relevant hysteroscopic finding in the suspicion of a malignant lesion;
- Coexistence of other pathologies: Myomas, adenomyosis, Mullerian abnormalities;
- Blind biopsy or curettage: This approach is currently discouraged for the management of EP due to low sensitivity 10%, low PPV 66% and NPV 33%, when compared to hysteroscopy. As a blinded procedure, it may not retrieve adequate samples from EP and may miss malignant cells at the base of EP. Moreover, histopathological examination may be limited by the fragmentation of the tissue obtained [55].
The differential diagnosis of endometrial polyps should be made with other uterine pathologies, such as intracavitary leiomyomas, endometrial hyperplasia or endometrial cancer [45].
The most appropriate management of EP should be established on the basis of a thorough evaluation of patient’s history (i.e., desire for pregnancy), associated symptoms, risk factors for malignancy, and technical qualification of the physician. In view of such factors, clinicians may opt for conservative non-surgical management, conservative surgery or radical surgery [13].
- Expectant management: “Wait and see approach” can be considered in
patients in premenopausal age, asymptomatic, affected by small EP (
- Hormonal therapy: The use of medical management in patients with EP has a limited role in the literature. Wada-Hiraike et al. [56] demonstrated that intermittent administration of oral contraceptives (OC) for 2–5 months (median 3 months) led to higher regression rate of sessile polyps compared to pedunculated polyps (76% vs. 44%), suggesting that sessile polyps may be more sensitive to OC treatment than pedunculated polyps [56]. The observed regression of EPs could be due to several factors including pro-apoptotic activity of OC and anti-inflammatory effects of progesterone, resulting in progressive flattening of endometrial protrusions [28, 56, 57].
The use of some types of hormonal therapies, especially progestogen with high antiestrogenic activity, may have a preventive role for the formation of polyps [28]. Gardner et al. [57] demonstrated the prevention of EP in women using levonorgestrel intrauterine system (LNG-IUS).
- Hysteroscopic polipectomy: It is a conservative surgical approach which an effective and safe therapeutic method, promoting symptomatic relief for 75 to 100% of patients [37].
Can be performed in an outpatient setting or surgical center. Outpatient polypectomy is associated with lower intraoperative risks and greater cost-effectiveness. On the other hand, polypectomy in the operating room is associated with a considerably higher risk of uterine perforation and penetration compared to office hysteroscopy [58].
(a) Hysteroscopic polypectomy in office setting
Small polyps (less than 0.5 cm) can be easily removed using only 5-Fr mechanical instruments, such as scissors or grasping forceps [59]. In addition, the use of energy, like laser or 5-Fr bipolar electrode, allows precise cuts in less time compared to mechanical instruments [60].
The recent advent of the 15-Fr bipolar office resectoscope made it possible to treat multiple polyps and EP larger than 3 centimeters in a shorter period of time [57]. Smaller tissue removal device (TRD), namely “hysteroscopic morcellators”, can also be a valuable option to minimize pain and reduce procedure time, since the combination of tissue cutting and tissue aspiration minimizes the need for several re-insertions of the material through the cervix. TRD use a single-use rigid metal inner tube with cutting edges rotating and/or reciprocating with an outer tube of variable diameter (9 to 4 mm), avoiding thermic damage to removed tissue.
Additionally, the learning curve is short and made simpler by the availability of computer simulators which allow clinicians to gain confidence with the tool [61].
(b) Hysteroscopic polypectomy in the operating room
Currently, there are no precise limits on polyp characteristics to define whether polypectomy should be performed in an outpatient or surgical setting. Patients reluctant to outpatient treatment or with extensive pathologies may be favored with the procedure in a surgical setting [4].
According to the endometrial pathology and the patient’s characteristics (parity, difficulty in cervical dilation, cervical stenosis), the appropriate size of the resectoscope (15 Fr, 22 Fr, 26 Fr or 27 Fr) and the type of energy applied (monopolar or bipolar) must be defined [56]. Several studies have shown that the use of TRD allows a reduction in surgical time and a shorter learning curve when compared to the standard resectoscope [62, 63].
The recurrence rate after complete excision of the lesion is present in approximately 2.5% to 43.6% of cases. Previous polypectomies, multiple polyps and endometriosis are factors associated with a higher risk of recurrence [64, 65].
- Hysterectomy: It is a radical therapeutic option that guarantees no recurrence of polypoid lesions. Nevertheless, it is burdened by high costs, high rate of complications and morbidity. Therefore, it is not currently considered as a first-line therapy of EP [13].
A suggested algorithm for the management of EP is summarized in Fig. 2.
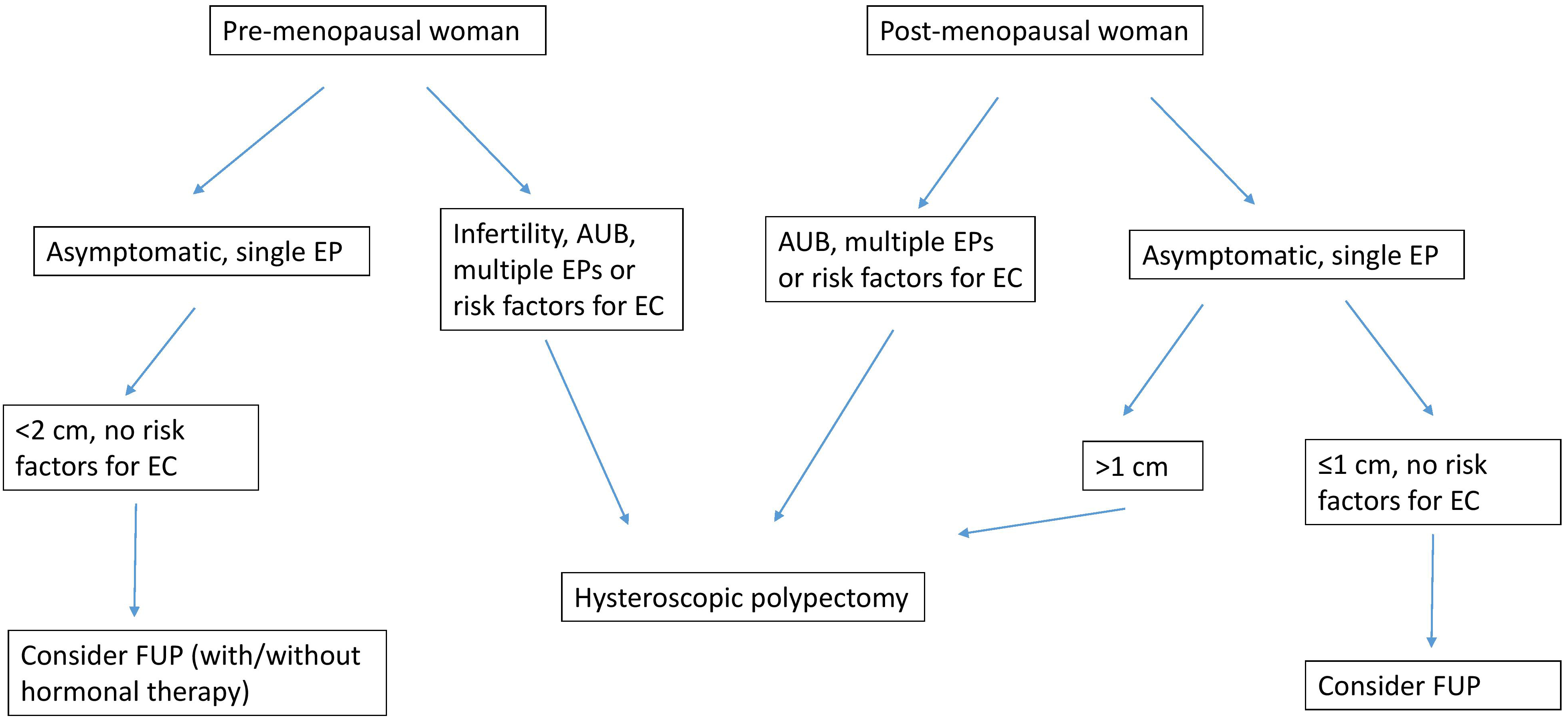 Fig. 2.
Fig. 2.Management algorithm based on authors’ experience for women with endometrial polyps. Polyp diagnosis performed by using trans-vaginal ultrasound examination with/without diagnostic hysteroscopy; EP, endometrial polyp; AUB, abnormal uterine bleeding; FUP, follow-up; EC, endometrial cancer. Risk factors for endometrial cancer, obesity, diabetes mellitus, hypertension, tamoxifen therapy, additional findings at ultrasound and/or diagnostic hysteroscopy.
MDCV–Study conceptualization and writing; AV—Study writing and revision; MCR—Study writing; LCAN—Study editing; AG—Study editing; ADSS—Study coordination and writing.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest. AV is serving as one of the Editorial Board members/Guest editors of this journal. We declare that AV had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to IAK.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


