1 Department of Obstetrics and Gynecology, Renmin Hospital, Wuhan University, 430074 Wuhan, Hubei, China
2 Reproductive Medicine Center, Renmin Hospital, Wuhan University, 430074 Wuhan, Hubei, China
†These authors contributed equally.
Academic Editor: Michael H. Dahan
Abstract
Background: To evaluate both the impact of hepatitis B virus (HBV)-DNA copies in women with
HBV infection on the ovarian reserve function and outcomes of in vitro
fertilization (IVF). Methods: We conducted a retrospective study on a
total of 9927 couples undergoing their first IVF cycle. After filtering, 1570
couples (546 HBV-seropositive women and 1024 HBV-seronegative women whose
partners were HBV-seronegative) failed to meet inclusion criteria. According to
the HBV-DNA titers in serum, the HBV-seropositive group was divided into three
groups: DNA-high copy group (n = 139), DNA-low copy group (n = 241), and
DNA-negative group (n = 166). All patients underwent controlled ovarian
hyperstimulation using the long downregulation protocol followed by IVF.
Results: Compared with the HBV-negative group, HBV-positive women with
high DNA copy exhibited lower antral follicle count (AFC) (11.9
Keywords
- HBV-DNA
- Female infertility
- Ovarian function
- In vitro fertilization
Chronic hepatitis B virus (HBV) infection, which can cause hepatic inflammation and severe diseases such as hepatocirrhosis and hepatocellular carcinoma, is a worldwide public health concern [1]. It is estimated that a global prevalence of 3.5% [2], i.e., 2.57 billion people worldwide have been infected with HBV and that roughly 270 million people have been chronically infected, of whom women of reproductive age account for 25.3% [3]. China accounts for some of the highest prevalence of HBV around the world [4, 5]. The main source of transmission for HBV is via familial vertical transmission, including cases of both mother- and father-to-infant vertical transmission, the former being dominant [6, 7, 8]. Newborns from HBsAg positive pregnant women who begin passive-active immunization immediately after birth can significantly reduce vertical transmission, but still have 5%–10% infection with HBV [9, 10].
Hepatitis B virus is a double-stranded DNA virus of the Hepadnaviridae family. Serum HBV-DNA content is the most direct and reliable indicator of viral activity in HBV-infected patients [11, 12]. Apart from hepatocytes, HBV-DNA is also found and expressed in reproductive gonads, such as testis, sperm, seminal plasma, granulosa cells, and oocytes [13]. One study reported that HBV-DNA high-level mothers are more susceptible to mother-to-child transmission [14]. Furthermore, HBV can infect spermatozoa, which in turn affects its quality and function [15]. Recently, as an increasing number of HBV seropositive infertile couples have chosen ART, certain challenges have emerged: could chronic HBV infection affect the ovarian reserve function, egg quality, embryonic development, embryo quality, and pregnancy outcome of in vitro fertilization (IVF) and embryo transfer (ET)?
Given that the cases in our medical center have been routinely and systematically screened for both partners for repertoire virus prior to IVF treatment, we undertook a retrospective study to evaluate the impact of HBV-DNA on ovarian function, ovarian stimulation and outcome of IVF for women with or without infection by HBsAg.
This study included 9927 infertile couples (20–43 years of age) seeking IVF
treatment for the first cycle from October 2014 to August 2019 in the
Reproductive Medical Center of Renmin Hospital of Wuhan University. All couples
were tested for repertoire viruses such as HBV, HCV, HIV and syphilis within 1
year of the ART cycle during routine HBV-DNA inspection in patients with Serum
HBsAg positive. HBV-seropositive patients with DNA titers
Based on the above inclusion and exclusion criteria, we excluded 8357 infertile
couples and screened out 1570 infertile couples for investigation, among whom,
all male partners were HBV-seronegative. According to the criterion whereby HBsAg
is positive or not and the HBV-DNA titers in serum of female partners, the
patients were divided into four groups, HBV-DNA-high copy group (n = 139): serum
HBsAg (+), HBV-DNA
All patients underwent IVF treatment with routine pituitary suppression protocol, as previously described [17]. In brief, patients were subcutaneously injected with gonadotropin-releasing hormone agonist (GnRH-a; Decapeptyl or Diphereline; Ispen, Paris, France) in the mid-luteal phase for at least 14 days. Once the pituitary activity was completely suppressed, the process of ovarian stimulation was commenced with human menopausal gonadotropin (hMG; LIVZON Group, Guangzhou, China) or recombinant follicle-stimulating hormone (FSH; Gonal-f; Merck Serono, Geneva, Switzerland or Puregon; Schering-Plough; Kenilworth, NJ, USA). Human chorionic gonadotropin (HCG; LIVZON Group, Guangzhou, China) was injected when at least three follicles had an average diameter of 18 mm. Then, the oocytes were retrieved after HCG had been injected for 34–36 hours. In the following 16 to 20 hours, IVF was conducted, followed by embryo selection and ET after being cultured in vitro. If there were no available embryos or in the event that patients developed OHSS or showed a risk of OHSS, cycles were canceled.
Basic data of patients included: age and BMI of couples; sperm parameters;
duration and cause of infertility; ovarian reserve evaluation (levels of serum
anti-mullerian hormone, menstrual cycle day 3 levels of serum FSH, LH, E
Fertilization rate was defined as the number of confirmed zygotes per the number
of oocytes retrieved in the IVF cycle.
High-grade embryo rate was defined as the number of embryos having
Data were reported as mean
At the outset of this study, we analyzed a total of 9,927 couples (20–43 years of age) who underwent their first IVF cycle in our medical center from October 2014 to August 2019, among whom 596 (6.0%) were HBsAg positive, with the prevalence of HBsAg of each year shown in Fig. 1.
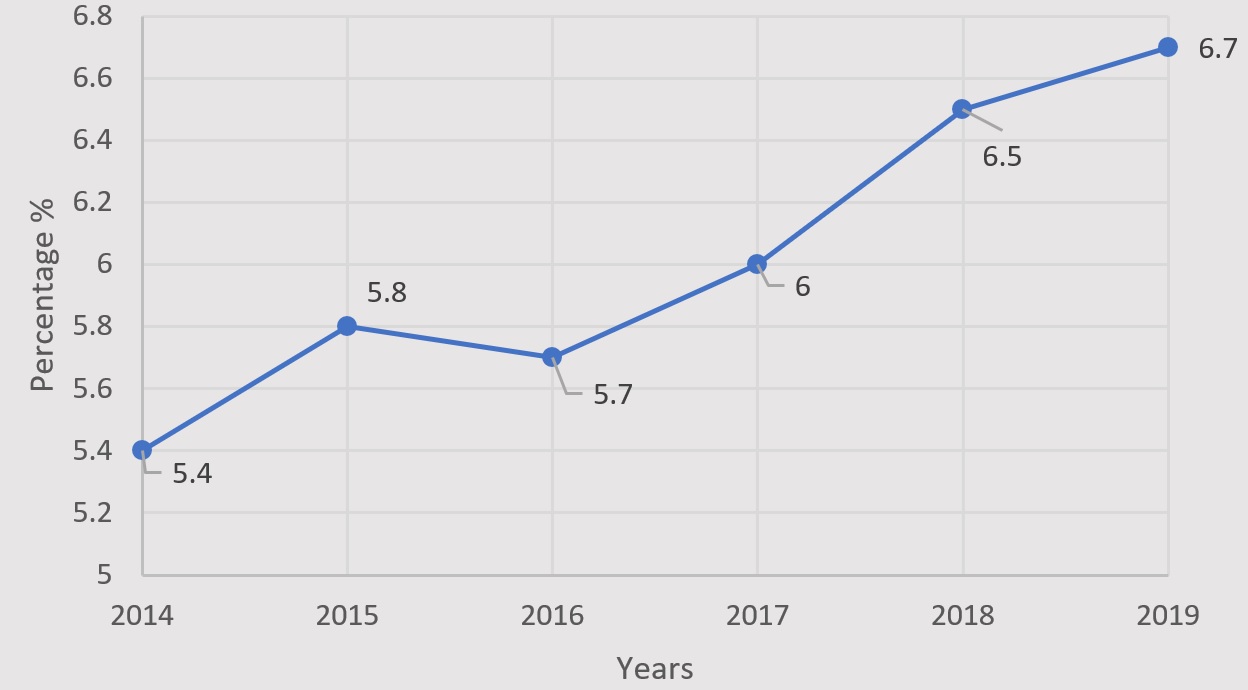 Fig. 1.
Fig. 1.Prevalence of women positive for HBsAg from 2014 to 2019 in our medical center.
Based on inclusion and exclusion criteria, we excluded 8357 infertile couples
and screened out 1570 infertile couples, among whom 546 women were HBsAg
positive with 139 women categorized as high DNA copy and 241 women as low DNA
copy. The demographic data of the subjects are shown in Table 1. Regarding the
indexes of age, BMI of couples, the duration and cause of infertility, and the
basal semen parameters, there were no significant differences between the
HBV-positive and HBV-negative groups (p
| Characteristics | HBV-positive groups | HBV-negative group | |||
| DNA-high copy | DNA-low copy | DNA-negative | (n = 1024) | ||
| (n = 139) | (n = 241) | (n = 166) | |||
| Female age (years) | 31.2 |
31.2 |
31.3 |
30.9 | |
| Male age (years) | 35.1 |
35.0 |
34.8 |
34.2 | |
| Female BMI (kg/m |
21.9 |
21.9 |
21.8 |
21.8 | |
| Male BMI (kg/m |
23.3 |
23.6 |
23.4 |
23.3 | |
| Duration of infertility (years) | 4.5 |
4.4 |
4.3 |
4.20 | |
| Cause of infertility | |||||
| Tubal blockage | 114 (82.0%) | 192 (79.7%) | 134 (80.7%) | 811 (79.2%) | |
| Male factor | 40 (28.8%) | 73 (30.3%) | 47 (28.3%) | 267 (26.1%) | |
| Others | 6 (4.3%) | 12 (5.0%) | 8 (4.8%) | 41 (4.0%) | |
| Basal semen parameters | |||||
| Volume (mL) | 1.89 |
1.81 |
1.83 |
1.85 | |
| Concentration (× 10 |
76.24 |
75.24 |
76.01 |
75.35 | |
| Progressive motility (a + b) (%) | 30.4 |
30.9 |
30.7 |
29.98 | |
| Normal morphology (%) | 14.58 |
14.31 |
13.9 |
14.01 | |
| Note: Values are presented as mean | |||||
The data on ovarian reserve function are shown in Table 2. The AFC refers to
follicles with a diameter of 2–9 mm visible in the ovary under vaginal
ultrasound. It can reflect the number of original follicles remaining in the
follicle pool well and is a direct reflection of ovarian reserve function [25, 26]. AFC
| Characteristics | HBV-positive groups | HBV-negative group | ||
| DNA-high copy | DNA-low copy | DNA-negative | (n = 1024) | |
| (n = 139) | (n = 241) | (n = 166) | ||
| FSH level (IU/L) | 7.6 |
7.4 |
7.1 |
7.3 |
| LH level (IU/L) | 4.7 |
4.7 |
4.6 |
5.0 |
| E |
48.9 |
49.4 |
48.5 |
45.6 |
| No. of AFC | 11.9 |
12.8 |
13.1 |
13.3 |
| FSH |
8 (5.8%) | 13 (5.3%) | 8 (4.8%) | 53 (5.2%) |
| FSH/LH |
16 (11.5%) | 25 (10.4%) | 16 (9.7%) | 123 (12.0%) |
| AFC |
11 (7.9%)** | 12 (5.0%) | 6 (3.6%) | 32 (3.1%) |
| AMH |
12 (8.6%)* | 17 (7.1%) | 9 (5.4%) | 44 (4.3%) |
| Note: Values are presented as mean *p **p | ||||
The data on COH and embryology are summarized in Table 3. We found that fewer
oocytes were retrieved in the DNA-high copy group than in the HBV-seronegative
group (9.2
| Characteristics | HBV-positive groups | HBV-negative group | ||
| DNA-high copy | DNA-low copy | DNA-negative | (n = 1024) | |
| (n = 139) | (n = 241) | (n = 166) | ||
| Duration of Gn stimulation (d) | 11.0 |
10.9 |
11.1 |
10.5 |
| Total dose of Gn used (IU) | 2287.9 |
2295.38 |
2310.36 |
2340.48 |
| Endometrial thickness (mm) on the day of HCG injection | 1.1 |
1.0 |
1.1 |
1.0 |
| E |
4512.6 |
4463.54 |
4481.02 |
4532.23 |
| No. of oocytes retrieved | 9.2 |
12.6 |
13.0 |
13.1 |
| No. of embryos transferred in cycles by ET | 2.03 |
2.05 |
2.01 |
2.07 |
| Cycles with rescue ICSI performed | 11 (7.9%) | 11 (7.1%) | 11 (6.6%) | 72 (7.0%) |
| Fertilization rate | 771/1087 (70.9%)** | 1571/2168 (72.5%)* | 1065/1432 (74.4%) | 5997/7985 (75.1%) |
| High-grade embryo rate | 385/748 (51.5%)** | 825/1534 (53.8%)* | 579/1042 (55.6%) | 3364/5913 (56.9%) |
| Cycles without ET for no viable embryos | 6 (4.3%)* | 9 (3.7%)* | 4 (2.4%) | 14 (1.4%) |
| Cycles without ET for danger of OHSS | 13 (8.6%) | 21 (8.7%) | 14 (8.4%) | 87 (8.5%) |
| Implantation rate | 67/214 (31.3%)* | 131/401 (32.7%)* | 107/283 (37.8%) | 750/1948 (38.5%) |
| Clinical pregnancy rate/cycle with OR | 56/139 (40.3%)* | 102/241 (42.3%)* | 80/166 (48.2%) | 508/1024 (49.6%) |
| Clinical pregnancy rate/cycle with ET | 56/123 (45.5%)** | 102/209 (48.8%)* | 80/148 (54.1%) | 508/894 (56.8%) |
| Early abortion rate | 11/56 (19.6%)** | 16/102 (15.7%)** | 8/80 (10.0%) | 36/508 (7.1%) |
| Note: Values are presented as mean *p **p | ||||
The duration of Gn stimulation, total dose of Gn used, endometrial thickness on
the day of HCG injection, No. of embryos transferred per ET cycle, cycles with
rescue ICSI performed as well as the cycles without ET for the danger of OHSS
were not significantly different among the HBV-seropositive and seropositive
groups (p
Furthermore, compared with the HBV-negative group, the implantation rate and clinical pregnancy rates were significantly lower in both DNA-high copy and DNA-low copy groups (implantation rate: 31.3% vs 38.5%, p = 0.039 and 32.7% vs 38.5%, p = 0.028; clinical rate: 40.3% vs 49.6%, p = 0.039 and 42.3% vs 49.6%, p = 0.042 per cycle with OR and 45.5% vs 56.8%, p = 0.018 and 48.8% vs 56.8%, p = 0.036 per cycle with ET), and the early abortion rate was significantly higher in both DNA-high copy and DNA-low copy groups (19.6% vs 7.1%; p = 0.001 and 15.7% vs 7.1%; p = 0.005).
China is a highly endemic area for HBV, with an overall prevalence of 5.2% among women of reproductive age [18]. However, the prevalence in this study was slightly higher at 5.4–6.7%, possibly on account of geographical variation. The number of seropositive infertile women in our medical center has gradually risen over the past six years, as shown in Fig. 1. Recently, laboratory tests for hepatitis B cases and clinical diagnosis have become increasingly standardized, thereby reducing the rate of missed reports. With improvements in economic conditions, more infertile women, especially in the surrounding countryside which is often at a heightened risk of hepatitis B, have embraced ART. This may further explain the higher prevalence during the study period.
In this study, 546 infertile patients infected with HBV were collected; the positive rate of HBV-DNA was 69.60% out of the total 1570 cases, among which the low copy of HBV-DNA was the majority (44.14%). This suggests that the viral replication in HBV-infected women of childbearing age is predominantly associated with low replication, that adults have a certain capacity of immune clearance of HBV, while a significant proportion of women are still highly active (DNA-high copy group: 25.46%).
Among several studies [19, 20, 21, 22, 23] that have addressed the reproductive performance of couples infected with HBV, results are discordant. Some studies [19, 20] showed significantly lower implantation in couples conflicting for HBV infection, while others reported a higher implantation rate in couples of HBV infection than the control individuals [23]. However, none of them took into account the state of HBV-DNA copies. Our study was grouped on the copy amount of HBV-DNA, designed to evaluate the varying degrees of copy amount of HBV-DNA infection on ovarian function, ovarian stimulation and outcome of IVF for women.
In HBV-infected patients, serum HBV-DNA assay is the most direct and reliable
indicator of viral activity, particularly useful in assessing the risk of disease
progression and candidacy for antiviral therapy and to distinguish active
hepatitis B from the inactive carrier state [24]. We found that compared with the
HBV-negative group, among HBV-infected infertile women, high copy status of
HBV-DNA generated fewer AFCs, a lower number of retrieved oocytes, larger
proportion of AFC
In women, AMH is secreted by pre-sinus follicles and small sinusoidal follicle
granule cells. Compared with other existing hormone tests, AMH fluctuates
slightly during the menstrual cycle and is less susceptible to gonadotropins
[28]. Therefore, AMH is a sensitive and accurate indicator of
ovarian reserve function [29]. AMH
Several studies have provided evidence of HBV infection in the ovaries and even in oocytes [32]. Moreover, HBV-DNA positive rates in oocytes and embryos are positively correlated with serum HBV-DNA levels [33, 34]. Studies have also shown that HBV carrying can cause local immune or inflammatory reactions in the destruction of related functions in local tissues through the deposition of antigen-antibody complexes [35, 36, 37]. HBV infection can increase the proportion of sperm cell necrosis and apoptosis, destroy the integrity of the sperm membrane, cause the loss of sperm mitochondrial membrane, and reduce the function of sperm [20]. Therefore, HBV-DNA may damage ovarian tissue and oocytes, cause changes in ovarian reserve functions such as AMH and AFC, and affect the quality of eggs and embryos. Accordingly, we postulate that when a large amount of HBV-DNA is present in the blood, it can have negative effect on ovarian reserve function.
In this study, we observed a lower fertilization rate, lower high-grade embryo rate and more cycles without ET for no available embryos in both infertile women with high and low copies of HBV-DNA. Fertilization is the process of the egg and sperm fusing into a zygote. HBV-DNA sequences may pass through the zona and oolemma and integrate with oocyte genome, and then replicate and exist in the ovum at different stages [38, 39, 40, 41]. It may be an important mechanism for HBV vertical transmission that when oocytes are fertilized with normal spermatozoa, these sperm transmit HBV-DNA sequences to the embryo [39]. We speculate that when oocyte quality is thus damaged, the process of fertilization is affected in women with both high and low copies of HBV-DNA.
We observed lower implantation and clinical pregnancy rates in both DNA-high and DNA-low groups along with a higher early abortion rate in the above-mentioned two groups. This is consistent with the observations by Cui et al. [42] who showed that among 21,004 pregnant women in a prospective cohort study, the proportion of abortions in HBsAg carriers was significantly higher than in the control. Embryo is transferred in the last step of the IVF cycle and will subsequently develop into a new individual. The success rate is associated with endometrial receptivity as well as the quality of embryo. A separate study reported that integration and replication of HBV genes can cause chromosomal mutations in early embryonic cells while viral protein products can interfere with embryo metabolism and development [43]. As previously described, HBV-DNA can be detected in the female reproductive tract system and might have an impact on the embryo tissue and endometrial local immunity, and for HBV-seropositive women with impaired oocyte binding, these may be responsible (at least in part) for poor pregnancy results. In addition, we did not find differences among three HBV-positive groups, potentially attributable to insufficient sample sizes. As the sample size and research indicators of this study are still limited, large sample clinical research and related basic research are necessary to further confirm the specific underlying mechanism of this research.
Our study shows that HBV-DNA in HBV-seropositive women may interfere with ovarian reserve function and ultimately affect pregnancy outcome. For women of childbearing age with HBV infection, especially with high copy status of HBV-DNA, it is recommended that treatment and childbearing occur as early as possible in order to reduce the impact of HBV infection on ovarian reserve function. Prior to assisted pregnancy treatment, this population should be fully informed about possible outcomes to avoid medical risks. With enhanced prediction of ovarian reserve function, embryo quality, fertilization rate and pregnancy rate, improved consultation can be provided to HBV-seropositive infertile women.
AFC, antral follicle count; AMH, anti-mullerian hormone; ART, assisted
reproductive technology; BMI, body mass index; COH, controlled ovarian
hyperstimulation; E
LL and HL wrote the paper and analyzed the data; FS and JY proposed the idea and help revise the manuscript; WL collected the data and provided some suggestion for drafting the manuscript.
The institutional review board of the Renmin Hospital of Wuhan University approved the ethics of this research (20190522002). Informed consent was obtained from all patients before data collection.
The authors wish to thank all peer reviewers for their opinions and suggestions.
This research received no external funding.
The authors declare no conflict of interest.

