1 Department of Obstetrics and Gynecology, Maternity and Child Health Hospital Affiliated with Anhui Medical University, Anhui Province Maternity & Child Health Hospital, 230001 Hefei, Anhui, China
Abstract
Aim: Pregnancy is a hypercoagulable state, however it remains unknown
the degree to which pregnancy affects standardly drawn coagulation laboratory
studies. We intended to establish reference intervals for D-dimer and fibrinogen
in the third trimester of pregnancy. Methods: We recruited 1048 healthy
Chinese pregnant women with low-risk pregnancy. Concentrations of D-dimer and
fibrinogen in the third trimester were collected. The 2.5th and 97.5th percentile
reference intervals of D-dimer and fibrinogen in pregnant women with parity = 1,
parity
Keywords
- D-dimer
- Fibrinogen
- Reference interval
- Postpartum hemorrhage
Pregnancy is a unique physiological process for women, which induces increases in coagulation activity and decreases in anticoagulant and fibrinolysis levels. These physiological changes lead to a hypercoagulable state in pregnancy [1], which forms a protective barrier against miscarriage and postpartum hemorrhage (PPH) [2, 3].
PPH causes 27.1% of maternal deaths worldwide [4]. However, the percentage is increased to 34% of maternal deaths in China [5]. PPH has many potential causes, which include uterine atony, placental factors, birth canal trauma and coagulation dysfunction. Although coagulation dysfunction accounts for only about 1.48% of PPH [5], coagulation dysfunction coupled with the early appearance of disseminated intravascular coagulation (DIC) [6], can cause serious adverse pregnancy outcomes, massive obstetric hemorrhage, and pose a threat to the life of pregnant women [7].
The levels of fibrinogen and D-dimer are common indicators to judge the coagulation status. Currently, there is no suitable reference interval of fibrinogen and D-dimer for pregnant women. Herein, we intend to establish a reference interval for blood coagulation function of pregnant women to guide clinical diagnosis and treatment.
This study was performed from Apr. 2019 to Dec. 2019. 1048 low-risk pregnant women in the third trimester were enrolled at the eastern district of Anhui Province Maternity & Child Health Hospital, Hefei, China. Inclusion criteria included singleton pregnant women with natural pregnancy in the third trimester (28 + 0–41 + 6 weeks). Exclusion criteria included (1) medical and surgical diseases (hypertension, diabetes, and abnormal thyroid function), (2) prenatal hemorrhage (placenta previa, abruption of placenta), (3) recent use of anticoagulant or procoagulant drugs, (4) abnormal coagulation function or blood system diseases, (5) a history of abnormal pregnancy (stillbirth).
The gestational week was determined from the last menstrual period of the pregnant woman and the ultrasound examination in the first trimester. If the ultrasound examination differed from the last menstrual period dating by more than 5 days, the gestational week was calculated based on the ultrasound examination. We collected information including maternal age, parity, body mass index (BMI), blood loss, concentrations of D-dimer and fibrinogen in blood. All participants provided informed consent.
PPH is often defined as a blood loss of more than 500 mL within 24 hours after vaginal delivery or 1000 mL after cesarean section. Visual estimation is the most frequently practiced method of determining blood loss during childbirth [8]. The study was checked by obstetrics and transmitted anonymously without experimental intervention, which don’t require the approval of Medical Ethics Committee.
One thousand forty eight participants were divided into two groups as parity = 1
and parity
Data were analyzed using SPSS version 16.0 (IBM, Armonk, NY, USA). Means
As illustrated in the flow chart (Fig. 1) of this study, 1048 low-risk pregnant
women in the third trimester were recruited to investigate the reference
intervals of D-dimer and fibrinogen. The basic information of 1048 women was
listed in Table 1. The average gestational week of pregnant women was 38.78
 Fig. 1.
Fig. 1.A Flow chart of this study.
| Means |
Range | |
| Gestational week | 38.78 |
28–41 |
| Age (years) | 28.58 |
15–43 |
| Parity | 1.49 |
1–4 |
| Maternal BMI (kg/m |
26.69 |
17.91–37.17 |
| D-dimer (mg/L) | 1.83 |
0.34–7.8 |
| Fibrinogen (g/L) | 4.28 |
1.36–7.71 |
| Abbreviations: SD, standard deviation; BMI, body mass index. | ||
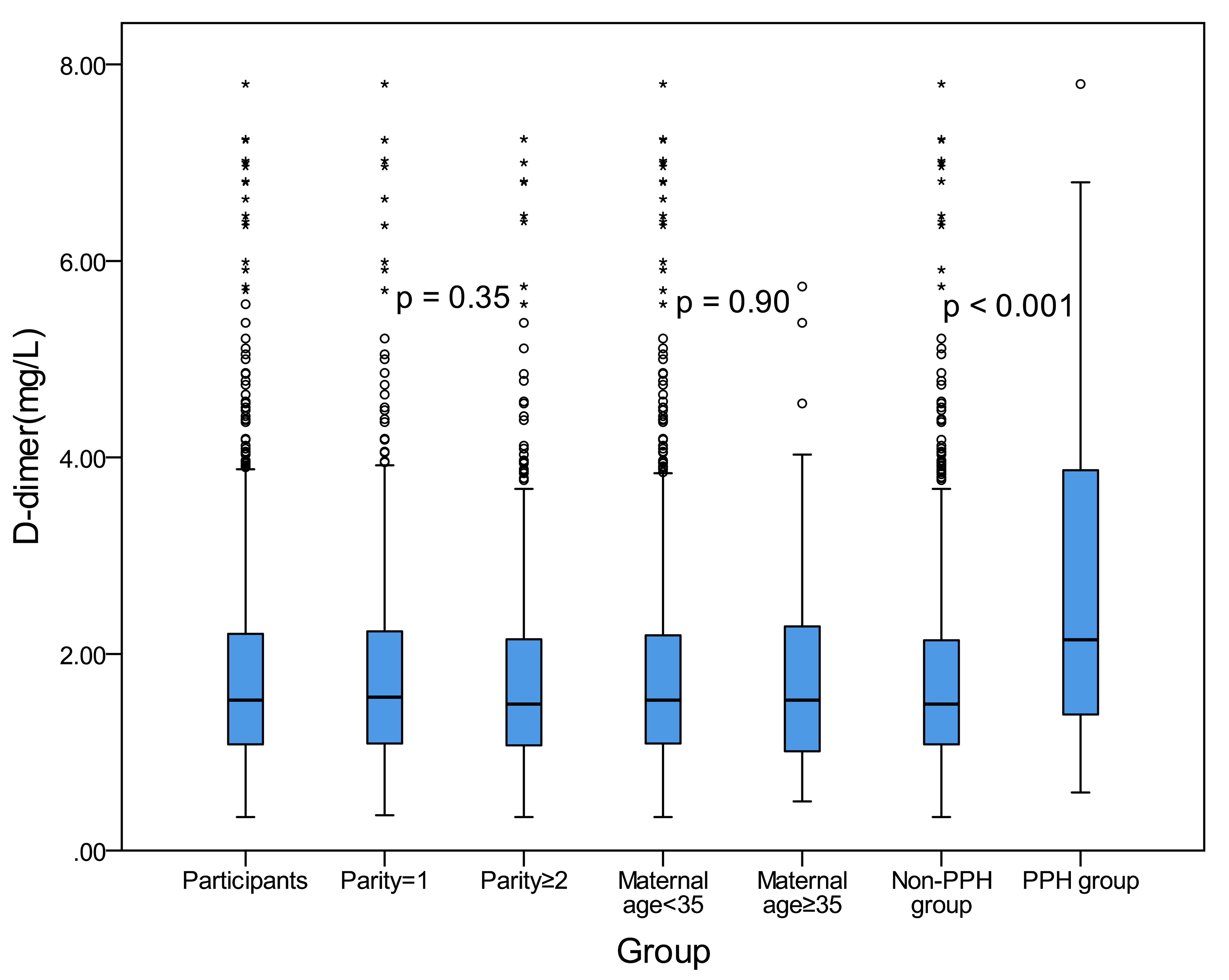
We analyzed blood levels of D-dimer for pregnant women in each group. As can be
seen in Fig. 2, the median blood D-dimer levels were 1.53 mmol/L. The median
level of D-dimer was 1.56 mmol/L in parity = 1 group, and 1.49 mmol/L in parity
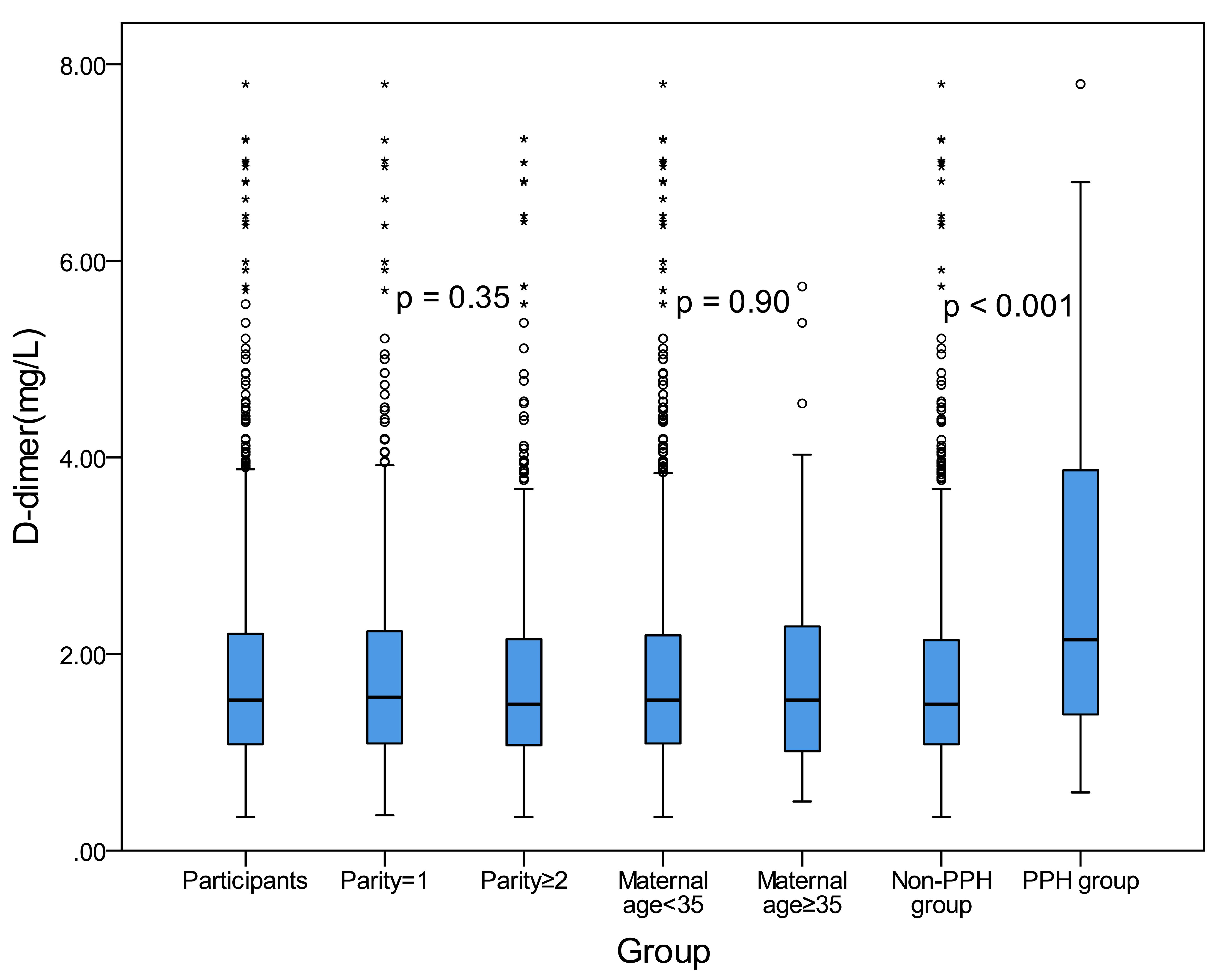 Fig. 2.
Fig. 2.The D-dimer concentration distribution in each group. The upper edge and the lower edge of the box indicate the 75th percentile and 25th percentile, respectively. The horizontal line in the middle of each box represents the median value. The p value was listed.
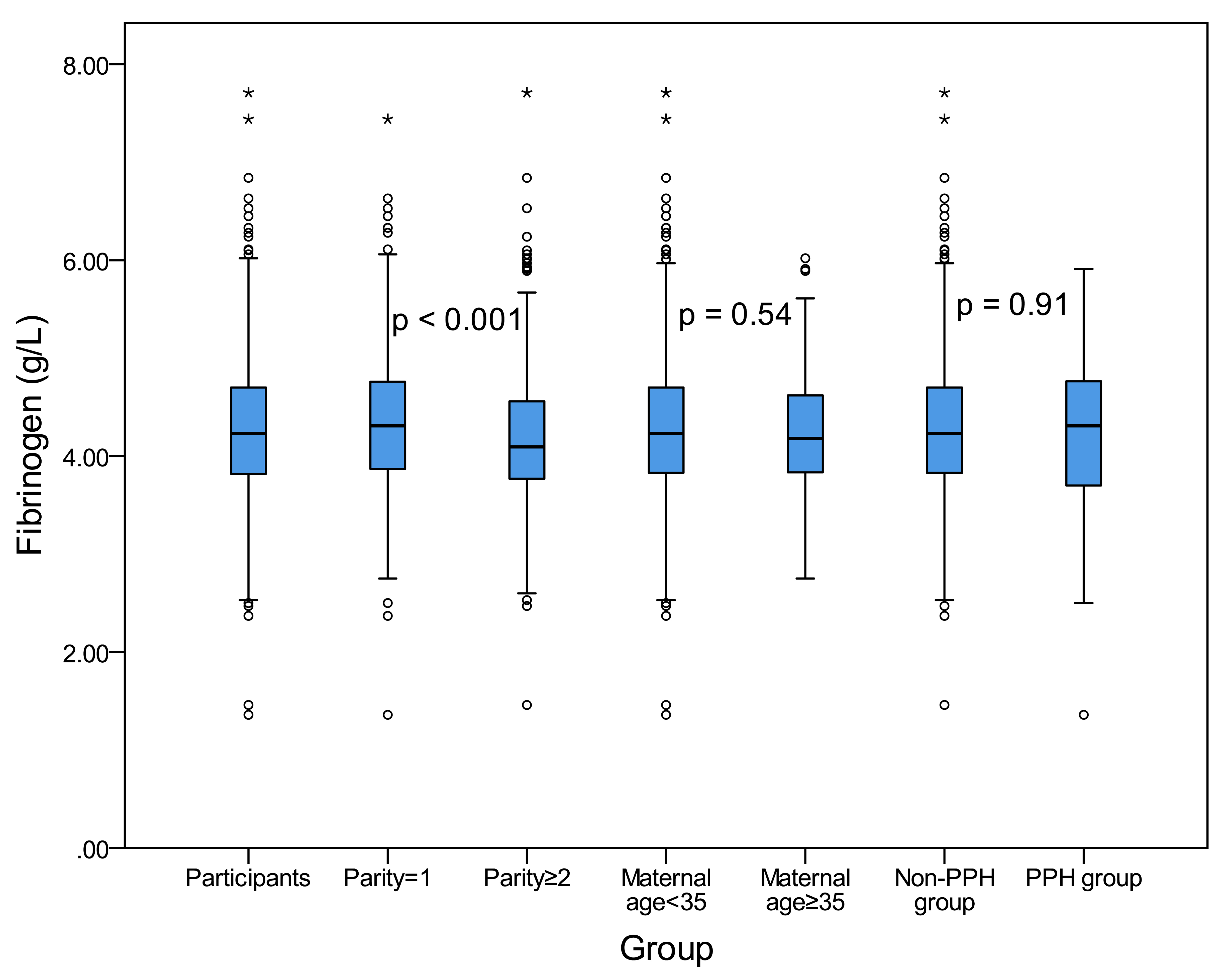
Next, blood levels of fibrinogen for pregnant women in each group were studied.
As shown in Fig. 3, the median fibrinogen level was 4.23 mmol/L. The median
fibrinogen level was 4.31 mmol/L in parity = 1 and 4.10 mmol/L in parity
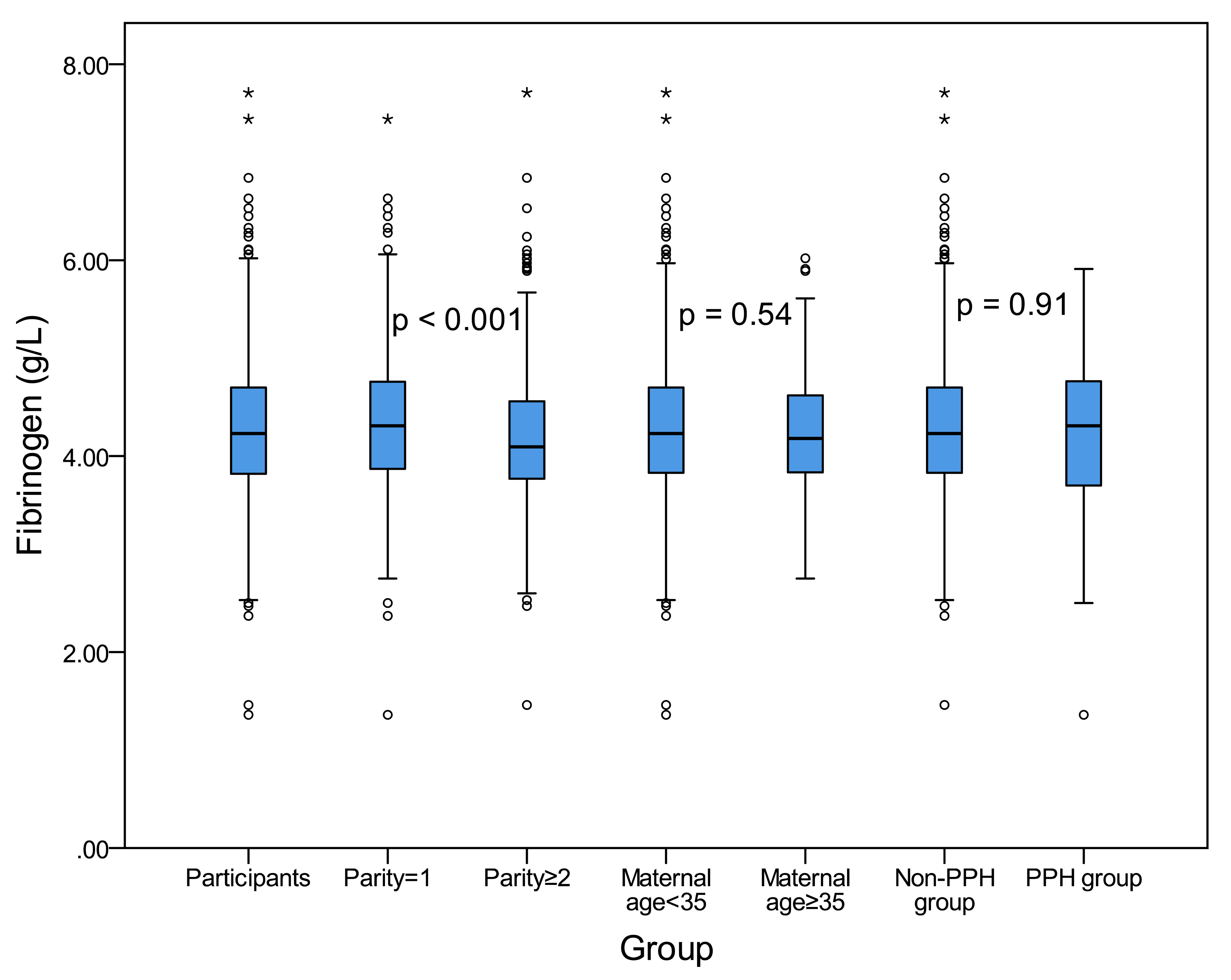 Fig. 3.
Fig. 3.The fibrinogen concentration distribution in each group. The upper edge and the lower edge of the box indicate the 75th percentile and 25th percentile, respectively. The horizontal line in the middle of each box represents the median value. The p value was listed.
Finally, the 2.5th and 97.5th percentile reference intervals of D-dimer and fibrinogen for pregnant women in the third trimester for each group were established and listed in Table 2.
| Case | D-dimer (mg/L) | Fibrinogen (g/L) | |||
| Median | Reference interval | Median | Reference interval | ||
| P2.5th and P97.5th | P2.5th and P97.5th | ||||
| All | 1048 | 1.53 | 0.63–5.05 | 4.23 | 3.02–5.92 |
| Parity = 1 | 578 | 1.56 | 0.62–5.47 | 4.31 | 3.03–5.91 |
| Parity |
470 | 1.49 | 0.62–4.80 | 4.10 | 2.93–5.94 |
| Age |
961 | 1.53 | 0.63–5.05 | 4.23 | 3.02–5.92 |
| Age |
87 | 1.53 | 0.59–5.21 | 4.18 | 3.07–5.91 |
| Non-PPH | 976 | 1.49 | 0.62–4.39 | 4.23 | 3.04–5.93 |
| PPH | 72 | 2.15 | 0.62–7.8 | 4.31 | 2.30–5.83 |
D-dimer is an important molecular marker of the physical hypercoagulable state and hyperfibrinolysis [9]. The reference range of D-dimer in healthy non-pregnant population is 0–0.5 mg/L. The high D-dimer levels found in our study population demonstrate the hypercoagulable state and secondary hyperfibrinolysis that occur during pregnancy. For pregnant women, the level of D-dimer is positively correlated with the gestational week, and increases gradually during normal pregnancy, reaching a peak in the third trimester [10, 11, 12]. In our study population, 99.33% of pregnant women in the third trimester had D-dimer levels exceeding the cut-off point (0.5 mg/L). Kline et al. [11] reported that the proportion of pregnant women whose D-dimer exceeded the 0.5 mg/L threshold was 50%, 77% and 100% in the first, second and third trimester, respectively [3, 11].
Fibrinogen is the precursor of fibrin. In the final stage of coagulation, soluble fibrinogen becomes insoluble fibrin, which promotes blood coagulation. As a classic coagulation parameter index, it is of great significance for judging the body’s hypercoagulable state or bleeding tendency. In order to match the reduction of fibrin primordial rationality during pregnancy, the fibrinogen level reached to twice higher than that of non-pregnant women [13].
The risk of venous thromboembolism (VTE) in pregnant women is 4–5 times higher than that of non-pregnant women [14]. However, the diagnosis of VTE is difficult and expensive. A negative D-dimer result can exclude the diagnosis of VTE without further testing, reducing unnecessary radiological examinations [15, 16]. However, the existing D-dimer level may not be a reliable predictor of VTE because there is a physiologic increase of D-dimer levels during pregnancy. If we continue to use the known reference range, false positive results in pregnant women without VTE will be caused.
Previous studies showed that the levels of D-dimer and fibrinogen in pregnant
women were higher than that of non-pregnant, but did not include reference
ranges. Some obstetricians recommend re-establishing their reference intervals,
especially in the third trimester. According to the recommendations of the
Association for Clinical and Laboratory Standards, Gutiérrez et al.
[3] calculated the reference interval of D-dimer to be 169–1202
In this study, the reference intervals of D-dimer and fibrinogen in the third trimester for Chinese low-risk pregnant women were established. We found that the concentration of D-dimer in the PPH group was significantly higher than that of the non-PPH group, but there were only 72 samples in the PPH group, and none of them developed severe organ failure or died. Therefore, the reference interval for pregnant women with high PPH risk can still refer to the whole.
Our research also has some limitations. First, this study was a cross-sectional study and cannot eliminate individual differences. It should be set as a longitudinal study. Second, this study did not count the plasma D-dimer and fibrinogen levels of pregnant women after delivery, because in the early stage of PPH, low level of fibrinogen will aggravate PPH [18]. Third, this study only established the reference interval for D-dimer and fibrinogen in the third trimester of pregnancy, and did not establish the reference interval for D-dimer and fibrinogen in the first and second trimester.
In summary, reference intervals for D-dimer and fibrinogen during the third
trimester in Chinese low-risk pregnant women were established. As the
concentration of D-dimer and fibrinogen in the blood during pregnancy differ from
the normal population, the current reference intervals can’t satisfy the needs of
pregnant women, particularly in the third trimester. Herein, we provided
reference intervals of D-dimer and fibrinogen in pregnant women with parity =1,
parity
WS and HL designed the study. WS, HL, YZ and ZL performed the research. HL provided help and advice on the ELISA experiments. WS analyzed the data and wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
The study did not involve experimental intervention. The related data were checked routinely and transmitted anonymously by obstetrics. The approval of the Medical Ethics Committee was not required.
We would like to express our gratitude to all those who helped us during the writing of this manuscript. Thanks to all the peer reviewers for their opinions and suggestions.
This research received no external funding.
The authors declare no conflict of interest.