1 Department of Obstetrics & Gynecology, Gifu University Graduate School of Medicine, 1-1 Yanagido, 500-0000 Gifu City, Gifu, Japan
Abstract
The purpose of this study was to investigate the change in uterine artery blood
flow in cases of intrauterine balloon tamponade (IBT). This was a retrospective
cross-sectional study of 132 patients who underwent singleton cesarean section.
IBT was performed in 35 of the 132 patients. There were 21 cases of placental
mediated pregnancy complications (PMPC), which were not included in the IBT
group. Placental positional abnormalities were significantly more common in the
IBT group than in the control group (89% vs. 9%, P
Keywords
- Balloon tamponade
- Uterine artery
- Doppler ultrasound
- Cesarean section
- Placenta previa
In Japan, postpartum hemorrhage (PPH) remains the main cause of maternal deaths, and is responsible for 23% of maternal deaths from direct obstetric causes [1]. Intrauterine balloon tamponade (IBT) is one of the leading methods for managing PPH [2, 3]. The use of IBT can reportedly reduce intra- and postoperative blood loss from placenta previa [4, 5, 6, 7, 8]. Moreover, the use of IBT reduces the need for more invasive procedures, such as radiological uterine artery embolization, compression sutures, and peripartal hysterectomy [9, 10].
Various reports describe bleeding control by filling the balloons, which is called the tamponade test [11, 12]. This test is advantageous in that it is volume-independent and achieves a clinical end-point of hemostasis. IBT produces a tamponade effect by pressure exertion on the myometrial wall, which results in mechanical stimulation of myometrial contraction. Until recently, it has been considered that IBT stops continuous bleeding when the internal myometrial pressure is higher than the systemic arterial blood pressure [13].
The over-inflation of the balloon in the uterus to prevent migration may cause other problems [14]. First, uterine distention is very painful; therefore, hemostasis should be achieved with the minimal volume of uterine distension. Second, there is concern about uterine rupture or uterocervical injury due to excessive expansion. The optimal expansion volume is considered the minimum volume for which the IBT is effective.
Doppler assessment of uterine artery blood flow is frequently used during pregnancy to evaluate placental perfusion. Uterine artery blood flow changes during pregnancy to supply the required blood for fetal development. Past study reported changes in UtA-PI associated with postpartum uterine reconstruction. It was shown that UtA-PI increases progressively during the first eight weeks, regardless of the number of deliveries [15]. UtA-PI increases with postpartum uterine recovery and may be an indicator of poor recovery.
If the hemostatic mechanism of IBT is mechanical compression of the uterine artery, it can be hypothesized that the aforementioned hemostatic effect is objectively quantified by changes in UtA-PI. If this hypothesis is correct, it will not only clarify the mechanism of IBT, but also pose a challenge to the effective management of PPH. The purpose of this study was to examine the utility of IBT as seen from the change in uterine artery blood flow. We believe that our findings would facilitate the understanding of the mechanism of action of IBT.
We conducted a retrospective cross-sectional study of 132 consecutive singleton pregnant women who underwent cesarean delivery at a tertiary medical center from October 2016 to July 2020. Fig. 1 shows the flow diagram of the study. We recruited patients who had undergone elective cesarean section prior to labor onset. We collected data on clinical characteristics, ultrasound and laboratory findings, and perinatal outcomes from the medical records.
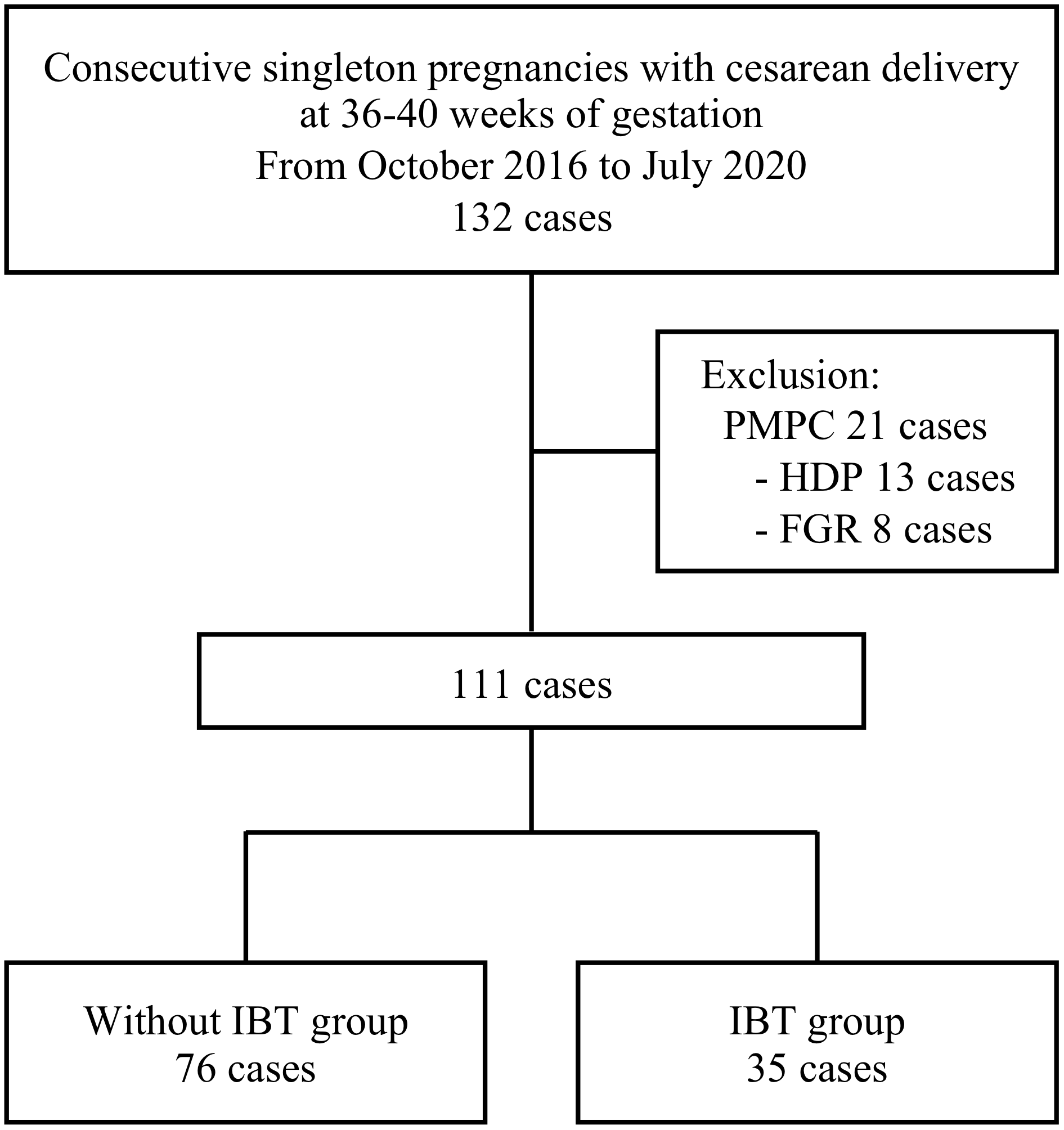 Fig. 1.
Fig. 1.Study participants and inclusion criteria. PMPC, placenta-mediated pregnancy complications; HDP, hypertensive disorders of pregnancy; FGR, fetal growth restriction; IBT, intrauterine balloon tamponade.
IBT was performed when the intraoperative blood loss exceeded 1000 mL, and the bleeding could not be controlled even after administering uterotonic agents and ligating the bleeding point. Before closing the myometrium incision, we counted the amount of suction bleeding containing amniotic fluid and used to determine IBT indication. Since the final blood loss included suction bleeding during abdominal closure and gauze bleeding count, there were cases in which the total blood loss exceeded 1000 mL even in control group. In all cases, intrauterine balloons were inserted transabdominally during surgery, and removed within 12 hours post-insertion [16]. Following the tamponade test procedure, the balloon was initially expanded with at least 100 mL of distilled water, which was serially increased by 50 mL until bleeding from the drainage tube disappeared. The blood flow (pulsatility index [PI], peak systolic velocity [PSV], and velocity time integral [VTI]) of the ascending branches of the left and right uterine arteries was measured using bedside transvaginal ultrasound. Ultrasound measurements were performed in four stages: immediate preoperative, immediate postoperative (under IBT insertion), 24 h postoperative (after IBT removal), and 5 days postoperative stages. Immediate preoperative UtA-PI was measured within 2 hours before cesarean section. Immediate postoperative UtA-PI was measured in the hospital room after surgery and within an hour after IBT insertion. Oxytocin was administered to all patients in the immediate postoperative period; however, no oxytocin was administered at 24 h post-surgery. In order to eliminate the effects of uterine contraction, all ultrasound assessments were performed in the absence of uterine contractions [17]. After cesareans section, uterine contractions as so-called post labor pains are observed intermittently, so immediate postoperative uterine artery blood flow was measured without uterine contractions. That is, the ultrasound measurement was performed by continuously observing the Doppler waveform for several tens of seconds and confirming that there was no change in the waveform due to uterine contraction.
Doppler measurements were performed transvaginally before and after surgery,
using a Prosound
Factors affecting the immediate preoperative UtA-PI were revealed by multivariate analysis. We compared the uterine artery blood flow at each measurement stage with and without IBT. Thereafter, changes in uterine artery blood flow were compared among the three groups-with balloon volumes 100–199 mL, 200–200 mL, and 300–399 mL.
Statistical analyses were performed using EZR [18], which is a graphical user
interface for R (The R Foundation for Statistical Computing, Vienna, Austria).
Data were expressed as means and standard deviations. Logistic regression
analysis was used for multivariate analysis of factors affecting immediate
postoperative UtA-PI. The Kolmogorov-Smirnov test was used to test the normality
of the distribution. For comparison between two groups, we used the Student’s
t-test (for normal distributions) and Mann-Whitney U test (for
non-normal distributions). The Kruskal-Wallis test with Bonferroni correction was
used to compare the three groups. Statistical significance was set at P
We performed 528 ultrasound scans for 132 women. IBT was performed during surgery in 35 of 132 cases. The balloon volume was less than 200 mL, less than 300 mL, and more than 300 mL in 6, 19, and 10 cases, respectively. Uterine artery embolization was required in one patient in IBT group and two patients in non-IBT group. There were no cases requiring hysterectomy. In this study, no cases required additional analgesics or removal of the balloon due to pain associated with IBT. No uterine rupture or cervical laceration due to IBT was observed.
The background characteristics of the 132 cases are shown in Table 1. Placental
positional abnormalities were significantly more common in the IBT than the
control group (89% vs. 9%, P
| without IBT | IBT | P | |
| (n = 97) | (n = 35) | ||
| primiparity | 39 (40%) | 16 (46%) | 0.690 |
| Placental positional abnormalities | 9 (9%) | 31 (89%) | |
| Placenta mediated pregnancy complications | 21 (22%) | 0 (0%) | 0.001 |
| Hypertensive disorder of pregnancy | 13 (13%) | 0 (0%) | 0.020 |
| Fetal growth restriction | 8 (8%) | 0 (0%) | 0.109 |
| Mother age (years) | 34 |
35 |
0.526 |
| Gestational age at delivery | 37w4d |
37w2d |
0.281 |
| Indications for cesarean section | |||
| Placental positional abnormalities | 9 (9%) | 31 (89%) | |
| Arrest of labor | 10 (10%) | 2 (6%) | 0.515 |
| Repeat cesarean section | 37 (38%) | 1 (3%) | |
| Breech presentation | 13 (13%) | 1 (3%) | 0.112 |
| Non-reassuring fetal status | 5 (5%) | 0 (0%) | |
| Maternal underlying conditions | 23 (24%) | 0 (0%) | |
| Number of cases (%)/mean Maternal underlying conditions include severe hypertensive disorders of pregnancy, severe mental illness, and birth canal obstruction (due to uterine fibroids, ovarian tumors). | |||
Table 2 shows multivariate analysis for immediate preoperative UtA-PI. PMPC was revealed as a relevant factor for UtA-PI. PMPC was excluded to avoid the effect on uterine artery blood flow assessment. Finally, we analyzed 35 and 76 cases in the IBT and control groups, respectively.
| Regression estimate | 95% CI lower | 95% CI upper | P | |
| primiparity | -0.053 | -0.134 | 0.026 | 0.185 |
| Placental positional abnormalities | -0.078 | -0.168 | 0.012 | 0.088 |
| Placenta mediated pregnancy complications | 0.248 | 0.126 | 0.371 | |
| Mother age | -0.001 | -0.009 | 0.007 | 0.813 |
| Gestational age at delivery | -0.023 | -0.051 | 0.004 | 0.097 |
| CI, confidence interval. | ||||
Table 3 shows the perinatal outcomes for each group. Compared to the control
group, patients in the IBT group had significantly lower gestational age at
delivery (37w2d
| without IBT (n = 76) | IBT (n = 35) | P | Power | |
| Gestational Age | 38w0d |
37w2d |
0.783 | |
| Birth weight (g) | 2987 |
2756 |
0.003 | 0.830 |
| Umbilical blood pH | 7.302 |
7.289 |
0.400 | |
| Placental weight (g) | 617 |
575 |
0.119 | |
| Blood loss (mL) | 1058 |
1663 |
0.978 | |
| Pre-surgery Hb (g/dL) | 11.0 |
11.2 |
0.465 | |
| Post-surgery Hb (g/dL) | 10.1 |
9.2 |
0.003 | 0.871 |
| Mean | ||||
Table 4 shows the changes in uterine artery blood flow with and without IBT. In
the IBT group, the immediate pre- and postoperative UtA-PIs were significantly
lower (0.56
| without IBT (n = 76) | IBT (n = 35) | P | Power | |
| Immediately pre-surgery | ||||
| UtA-PI | 0.63 |
0.56 |
0.011 | 0.577 |
| UtA-PSV | 110.6 |
117.0 |
0.393 | |
| UtA-VTI | 59.3 |
62.0 |
0.55 | |
| Immediately post-surgery | ||||
| UtA-PI | 0.96 |
1.37 |
0.986 | |
| UtA-PSV | 106.3 |
92.9 |
0.12 | |
| UtA-VTI | 66.8 |
54.5 |
0.046 | 0.526 |
| Change ratio (immediately post-surgery/immediately pre-surgery) | ||||
| UtA-PI | 1.59 |
2.61 |
0.999 | |
| UtA-PSV | 1.08 |
0.85 |
0.045 | 0.524 |
| UtA-VTI | 1.29 |
1.00 |
0.039 | 0.500 |
| 24 hours post-surgery | ||||
| UtA-PI | 1.05 |
0.93 |
0.073 | |
| UtA-PSV | 89.9 |
101.9 |
0.112 | |
| UtA-VTI | 54.5 |
64.4 |
0.029 | |
| Change ratio (24 hours post-surgery/immediately post-surgery) | ||||
| UtA-PI | 1.13 |
0.78 |
0.999 | |
| UtA-PSV | 0.93 |
1.52 |
0.015 | 0.869 |
| UtA-VTI | 0.93 |
1.97 |
0.912 | |
| 5 days post-surgery | ||||
| UtA-PI | 1.28 |
1.10 |
0.047 | 0.608 |
| UtA-PSV | 87.0 |
95.0 |
0.405 | |
| UtA-VTI | 47.9 |
57.3 |
0.099 | |
| Change ratio (5 days post-surgery/24 hours post-surgery) | ||||
| UtA-PI | 1.31 |
1.27 |
0.837 | |
| UtA-PSV | 1.05 |
1.01 |
0.508 | |
| UtA-VTI | 0.98 |
1.02 |
0.658 | |
| UtA, uterine artery; PI, pulsatility index; PSV, peak systolic velocity; VTI, velocity time integral. | ||||
The immediate postoperative UtA-PIs depending on the balloon expansion volume
were 1.13
In this study, when IBT was performed, the immediate postoperative UtA-PI significantly increased. In addition, UtA-PI decreased to the same level as prior to IBT, after the balloon removal. Uterine artery blood flow velocity was also reduced when IBT was performed.
There are various reports on factors that affect UtA-PI [15, 19]. In our study, multivariate analysis revealed PMPC as a factor affecting immediate preoperative UtA-PI. By excluding PMPC, we closely evaluated the effect of IBT on uterine artery blood flow. Immediate preoperative UtA-PI was significantly lower in IBT group. We speculated this was because there were many cases of placental positional abnormalities in IBT group. As shown in Table 2, using multivariate analysis for preoperative UtA-PI, placental positional abnormalities was extracted as a factor that independently affects UtA-PI. In cases of placental positional abnormalities, it is speculated that development of blood flow in placental bed around the lower uterine segment, which is close to the main trunk of uterine artery, reduces uterine artery vascular resistance. Immediate postoperative UtA-PI increased significantly with IBT. On the other hand, 24 h postoperative UtA-PI tended to be lower in IBT group, although there was no significant, and 5 days postoperative UtA-PI was significantly lower in IBT group. From the above, we think UtA-PI is originally lowered in case of placental positional abnormalities, and it is emphasized UtA-PI is increased by IBT performed.
A previous study reported that intrauterine pressure does not change even when
the expansion volume of IBT exceeds 100 mL [20], and it is better to avoid
unnecessary balloon inflation if the bleeding is reduced [21]. In our study,
increased UtA-PI was observed even with relatively small expansion volumes
(100–199 mL). Although there was no statistically significant change, UtA-PI and
flow velocity tended to increase and decrease, respectively, as the balloon
volume increased. It is speculated that the direct compression effect on the
uterine artery increases depending on the expansion volume, even if the
intrauterine pressure does not increase. In clinical practice, hemostasis is
often achieved with a small balloon volume (volume
In cases of placental positional abnormalities, which made up the majority of IBT group, bleeding from the placental bed increased significantly. in these cases, hemostasis is likely to be achieved by direct compression of the bleeding point by IBT. In cases without placental positional abnormalities, intraoperative uterine atony was the main cause of increased bleeding. It is considered that direct compression of the uterine artery by IBT was effective in achieving hemostasis even in these situations. One patient in IBT group who required uterine artery embolization was not associated with placenta positional abnormalities. Due to the marked uterine atony and manifestation of abnormal coagulation, we stepped up the hemostasis treatment. Two patients underwent UAE in control group. One case was PMPC (severe preeclampsia), and sudden massive bleeding was observed 3 hours after surgery. In this case, UAE was selected over IBT because of concerns about the manifestation of abnormal coagulation. The other case was PPH on 3 days after surgery, which pointed out a vascular image suggesting pseudoaneurysm and intermittent extravasation at the bottom of uterus. In all cases, UAE showed good hemostasis and no hysterectomy was required.
This retrospective study was limited by the fact that the IBT group contained many placental positional abnormalities, and there was only a small number of cases of IBT. Moreover, in this study, it is difficult to show that the higher UtA-PI, the smaller the amount of bleeding; therefore, future studies will need to assess the aforementioned relationship with and without IBT. However, it is clear that IBT reduces postoperative blood loss in patients with placental positional abnormality, and the changes in uterine artery blood flow are consistent with the IBT performed. We recommended performing UtA-PI measurement in prospective, extensive studies to determine the effect of IBT and the volume of balloon expansion in more detail.
IBT during cesarean section increases UtA-PI. And UtA-PI tends to increase as the balloon volume increases. We believe that our study findings will help to clarify the effect of IBT on uterine blood flow.
RS designed and analyzed the data. RS, TS, and KM performed the operation and collected the data. All authors approved the final version. KM supervised the study.
The study was conducted in accordance with the Declaration of Helsinki, and the protocol for this study was approved by the ethics committee of Gifu University Hospital, Gifu, Japan (approval number: 2019-165).
We gratefully acknowledge the work of past and present members of my department, and my precious family for the understanding they showed for my work.
This research received no external funding.
The authors declare no conflict of interest.

