1 Department of Reproductive Health, Centre of Postgraduate Medical Education, 01-004, Warsaw, Poland
2 Department of General, Oncologic and Trauma Surgery of Wolski Hospital, Warsaw, 01-211, Poland
3 Collegium Medicum, Cardinal Stefan Wyszynski University, 01-938, Warsaw; Department of Obstetrics and Gynecology, Institute of Mother and Child; 01-211 Warsaw, Poland
Abstract
Endometriosis involvement of the urological tract may lead to significant disabilities. The involvement of the urological tract gives rise to severe complications that burden patient health and quality of life. Two cases of patients with severe urogynecological complications after surgery of deep infiltrating endometriosis were analyzed. Last resort radical surgical treatment did not stop the progression of the disease and failed to prevent further deterioration despite concomitant hormone therapy. Careful clinical consideration is recommended to plan an effective therapeutic strategy and the extent of the surgery for this multidisciplinary and complex disease. The treatment should start and be proceed by a dedicated and experienced team. Radical surgery including a total hysterectomy with a salpingo-oophorectomy could be necessary. Although a radical dissection increases the chances of recovery and long-term remission, it is also associated with an increased risk of serious short-term and long-term postoperative complications. In presented cases, the infiltration of the ureter by endometriosis caused non-symptomatic kidney function loss. The radical surgical treatment of deep infiltrating endometriosis did not stop the progression of a disease and was associated with severe urogynecological complications. Screening for pyelocaliectasis in patients with deep endometriosis could be considered as an important part of preoperative examination and postoperative follow-up.
Keywords
- Deep infiltrating endometriosis
- Gynecological surgery
- Pyelocaliectasis
- Kidney loss
- Urogynecological complications
- Ureteroneocystostomy
Endometriosis is defined as the presence of endometrium-like tissue outside the uterine cavity. Ectopic lesions may cause chronic pain and infertility. Three types of endometriosis are generally recognized: peritoneal, ovarian, and deep infiltrating. The disease affects 6%-15% of women of reproductive age, occasionally occurring during adolescence and postmenopause [1]. Deep infiltrating endometriosis (DIE) infiltrates at least 5 mm below the peritoneal surface. DIE is localized mainly in the sacrouterine ligaments, the rectovaginal septum, the rectum wall, the sigmoid, the urinary bladder, and along the course of the ureters, as well as in the vaginal wall [2, 3]. The type and severity of accompanying symptoms depend mostly on the location of infiltration. Distant localization is relatively rare and presents significant diagnostic challenges. The usual non-genital organs involved are the colon, ureters, and the urinary bladder. The urinary tract is involved in up to 6% of the affected women. Urinary tract endometriosis may be difficult to diagnose since 50% of cases are asymptomatic for a long time, or dysuric symptoms are unobvious for a patient and practitioner unless systematically sought. In cases with urinary bladder involvement, a bladder wall excision is performed. If ureteral involvement occurs conservative ureterolysis or radical segmental resection and ureteroneocystostomy should be carried out. Ureteral endometriosis is considered severe when obstruction of urine flow is present. Severe cases of ureteral endometriosis are more frequent when parametrial infiltration occurs and in patients with low BMI [4]. Urine flow can be obstructed by endometriotic infiltration or less often by fibrotic tissue only. Ureteral involvement in up to 90% of cases is associated with other sited of DIE and affects 10%-14% of DIE cases [5] but severe form is present in 4.6% of DIE cases. Frequency of intrinsic type seems to be underestimated, since in large analysis infiltration of ureteral muscularis was found in 38%-54% of ureteral lesions. In case of intrinsic ureteral endometriosis radical excision, i.e. resection with end to end anastomosis or ureteroneocystostomy, should be performed [6]. The consensus regarding diagnostic and therapeutic protocol is still missing, since most of the studies are uncontrolled, non-randomized and have short follow-up period. The complication rate from 5% to 25% is reported following surgery for ureteral endometriosis depending on the implemented technique [7]. Laparoscopic ureteroneocystostomy performed especially when infiltration is wide and affects the lower portion of ureters is an effective and safe procedure that provides good results [8].
The current cases from academic Center of Endometriosis Surgery at the tertiary hospital were described to show the multidisciplinary aspect of the disease and its possible complications. First patient had 4 surgeries before admission to the center. The second patient had initial operation at the center. During therapy both underwent ureteroneocystostomy and developed pyelocaliectasis and subsequent kidney loss despite hormonal treatment (see Figure 1). We would like to depict controversies regarding repeated surgery in the context of the effectiveness and safety of endometriosis treatment [9, 10]. This case report was covered by permission #107/PB-A/2018 of the Centre of Postgraduate Medical Education Bioethics Committee. Both subjects had given informed consents.
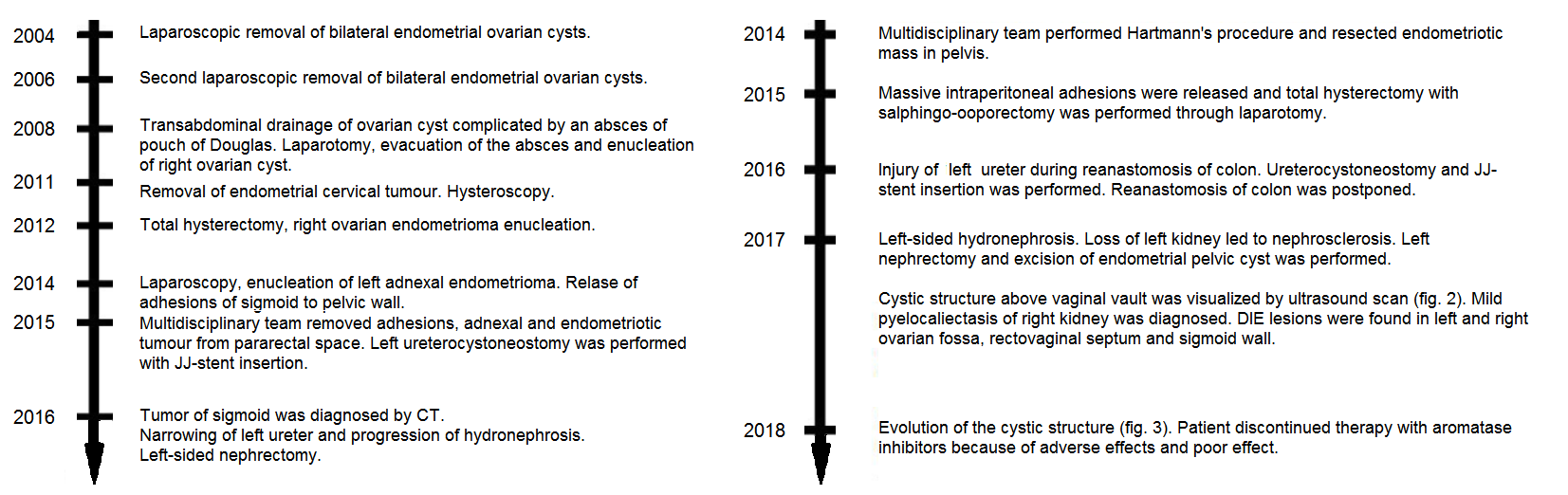 Figure 1.
Figure 1.— Schematic chronological representation of main surgical procedures performed in case #1 (left side) and case #2 (right side). Clinical symptoms of endometriosis progressively worsened despite gradual escalation of surgical interventions.
A 45-year-old woman with a fifteen-year history of endometriosis treatment, primary infertility, ten surgeries completed, presented to the Regional Endometriosis Center of Excellence in 2012. Anamnesis revealed that she had two unsuccessful ovarian hyperstimulations and embryo transfers at the beginning of treatment. Next, severe pain and myomatosis of the uterus were unsuccessfully treated with Gn-RH analogs. In 2004 and 2006, she underwent two laparoscopic removals of bilateral endometrial ovarian cysts. Two years later, a recurrent ovarian cyst was treated by transabdominal drainage complicated by an abscess of the pouch of Douglas. Laparotomy for abscess evacuation and the enucleation of the right ovarian endometrial cyst was required.
In 2011, recurrent abnormal uterine bleeding led to the diagnosis of a cervical nodule, followed by curettage of uterine cervix and cavity, and excision of the lesion. Histopathology recognized cervical endometriosis and normal endometrial tissue, respectively. Persistent abnormal bleeding was observed and abnormal ultrasound findings were received with a suspicion of a partial uterine septum. The septum was excluded during hysteroscopy.
A few months later, an enlarged immobile uterus was palpated on a gynecological examination. Ultrasound showed a multilocular cyst connected with the cervix, located at the site of the previously treated abscess. Magnetic resonance imaging (MRI) revealed small clots in the uterine cavity. Adenomyosis was excluded, but an 18 × 12 mm endometrial cyst of the right ovary was detected. The left ovary was normal. Next to the cervix, a 63 × 41 × 32 mm solid-cystic nodule filled with dense contents was found. The MRI detected a connection between the cyst and the cervical canal.
In 2012, a diagnostic laparoscopy was performed revealing solid adhesions of the omentum and the abdominal wall, and numerous dense adhesions in the peritoneal cavity. Conversion to laparotomy was performed. The uterus was enlarged by fibroids. The surgery consisted of a total hysterectomy, right ovary endometrioma enucleation, and the release of adhesions. Solid endometriotic nodules were found in the Douglas pouch and the rectovaginal septum. The lesion was connected with the cervix and sacrouterine ligaments. All visible endometriotic changes were removed. Stage IV endometriosis according to the ASRM (American Society of Reproductive Medicine) was recognized. The patient was treated with hormonal continuous oral progestin-only therapy with dienogest and remained asymptomatic for two years.
In 2014, the patient was investigated and surgery was performed due to pelvic pain syndrome. An endometrioma (35 × 30 × 32 mm) of the left adnexa was found on ultrasound. Adhesions of the ileum, the abdominal wall, and sigmoid accretion into the pelvic wall were released by laparoscopy. A multiloculated endometrial cyst of the left ovary was removed. Histopathology confirmed an endometrioma. The pain alleviated for a year with administration of combined oral contraceptives containing ethynylestradiol and dienogest.
In 2015, symptoms returned. An avascular, irregular nodule with a cystic part of the vaginal vault connecting to the rectum was diagnosed on ultrasound. Both-sided mild hydronephrosis was revealed. JJ-stents were inserted to the ureters and a multidisciplinary team (surgeon, urologist, and gynecologist-oncologist) performed a median laparotomy. Intraperitoneal adhesions were released and a bilateral salpingo-oophorectomy was performed, with excision of an endometriotic nodule with o cystic part from the pararectal space. The discoid excision of the upper rectum adjacent to the lesion was carried out. The distal part of the left ureter was removed and a ureteroneocystostomy was performed with JJ-stent insertion. After surgery, the patient was given megestrol acetate with good tolerability and temporal symptoms improvement.
In 2016, she was investigated for changes in bowel movement habits with concomitant anal bleeding. A sigmoid mass was diagnosed by a computer tomography (CT) scan. A sigmoid stricture was confirmed by colonoscopy but the biopsy showed normal mucosa. As endometriosis recurrence was highly suspected, a sigmoidectomy was performed. A pathological examination found an inflammatory nodule with squamous metaplasia, inflammatory infiltration with granulation tissue, and submucosal fibrosis, without histological evidence of endometriosis.
Further follow up revealed a left ureteral stricture and progression of hydronephrosis. Due to hypertension associated with kidney damage, a left-sided nephrectomy was performed. Currently, mild right-sided pyelocaliectasis was observed. The patient does not require antihypertensive treatment and she refused hormonal therapy due to adverse effects and scant endometriosis symptoms.
A 42-year-old woman presented to the Center in 2013 with a diagnosis of endometriosis. Her history included two vaginal deliveries and two spontaneous miscarriages. At the initial presentation, she demonstrated chronic pelvic pain, abnormal vaginal bleeding, dyschezia, and dysuria that lasted two months. She underwent dienogest treatment for two months with no effect and had an unsuccessful attempt of vaginal removal of rectovaginal nodule a district hospital outside from the Centre. Histology form initial surgery did not prove endometriosis in a structure described as an inflammatory mass. Ultrasound revealed the limited mobility of the uterus and of the left ovary. The remaining genital organs appeared normal and there was no evidence of pyelocaliectasis. CT scans showed extensive pelvic lesions. A pelvic bimanual examination revealed a slightly flattened right and posterior vaginal fornix, the uterine body attached to the right side through a wide endometrial infiltration containing the right ovary, rectum, and right pelvic wall, and obliteration of the pouch of Douglas. The right vaginal fornix was occluded, without significant resistance on the left side. The vesicouterine recess was normal. A rectal examination revealed a 9 cm long extensive infiltration of the large intestine. The mass was located on the right side. Following colonoscopy that showed no infiltration of mucosa, the patient was scheduled for surgery.
In October 2014, a multidisciplinary team (gynecologist-oncologists and a general surgeon) performed a laparoscopy that was converted to a laparotomy due to a solid endometriotic infiltration affecting the rectovaginal septum with partial obliteration of the colon spreading up to distal sigmoid. The discrepancy between rectal examination and intraoperative picture was a result of rectovaginal space nodule, which pulled down the rectosigmoid flexure down into Okabayashi space. When the adhesion was released the nodule was located on the rectosigmoid flexure. The endometriotic mass with a part of the vaginal wall and the upper rectum with a distal part of the sigmoid involved in the endometriotic infiltration was excised. Blurred anatomy, tissue injury, long procedure and a need for vaginal wall opening in the opinion of general surgeon precluded safe anastomosis and resulted in decision on postponing anastomosis by a Hartmann’s procedure. Histopathology revealed a sigmoid DIE involving all layers of the intestine, which was removed with a margin of normal tissue. Oral progestin-only therapy with dienogest was recommended.
Six months later, the patient presented a full-featured recurrence of symptoms, with diffused pelvic pain and hemorrhagic menstruation. At the gynecologic examination, the uterus had limited mobility, without evidence of infiltration within the vesicouterine recess. A large, painful nodule was found in the projection of the right cardinal ligament. A rectal examination found a pathological infiltration involving the rectal stump. After the comprehensive discussion of treatment options with the patient, it was decided to perform a total hysterectomy with gonads.
In July 2015, massive intraperitoneal adhesions were released through laparotomy, and a hysterectomy with bilateral salpingo-oophorectomy was performed. Histopathology showed an endometrial cyst of the right ovary, extensive adhesions of the right oviduct, the left ovary with follicular cysts, a fragment of the normal left oviduct, and a uterine with adenomyosis. The patient received low dose hormonal replacement therapy afterwards.
A year later, she was admitted for the restoration of gastrointestinal tract continuity. Attempts to dissect and release the ureter from adhesion to the rectal stump caused distal left ureter damage. Left side ureteroneocystostomy and bilateral JJ-stent insertion was carried out. The reanastomosis of the colon was postponed.
Subsequently, the patient developed left-sided hydronephrosis. The progressive loss of left kidney function led to a nephrectomy due to nephrosclerosis in June 2017. Simultaneously, an endometrial pelvic cyst was removed and intraperitoneal adhesions were released during surgery. The continuity of the gastrointestinal tract was not restored due to recurrent endometriosis within the rectal stump.
In November 2017, the pain persisted and a cystic structure mimicking ovarian endometrioma (34 × 44 × 35 mm) above the vaginal vault was visualized by ultrasound (Figure 2). The right JJ-stent was found near the nodule. Mild pyelocaliectasis (12 mm) of the right kidney had developed. Other DIE lesions were found in the area of the left and right ovarian fossa, the rectovaginal septum, and the sigmoid wall. Additional painless points of resistances were palpable in the pelvis at the gynecological examination - probably scars and adhesions. The 1-year evolution of the cystic structure is shown in Figure 3.
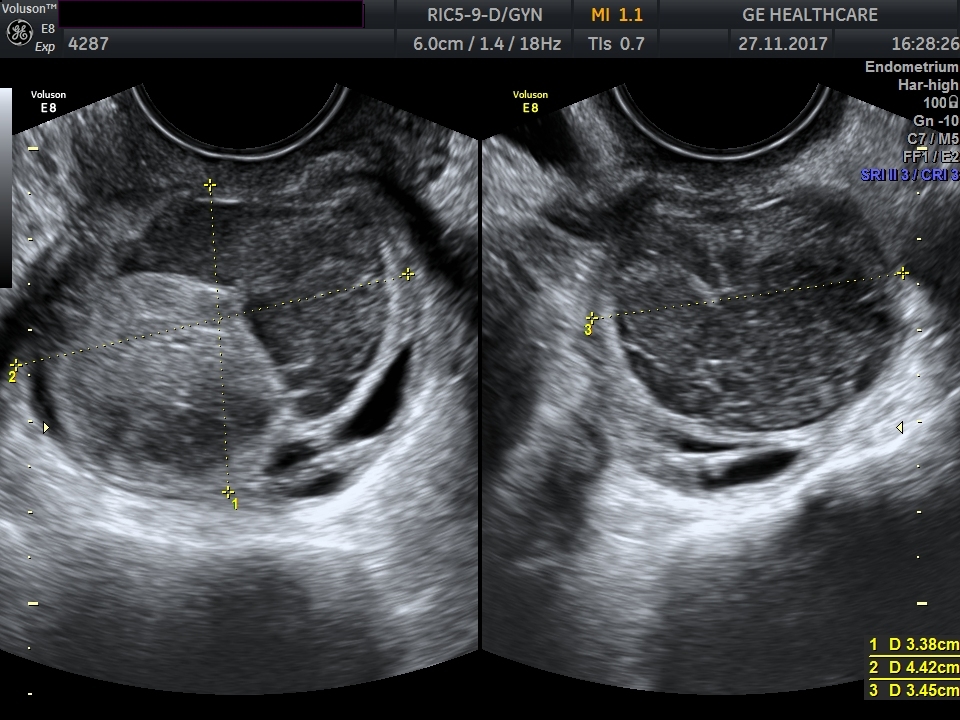 Figure 2.
Figure 2.— Transvaginal ultrasound scan of a cystic structure (presumably an endometriotic cystic nodule) above the vaginal vault of patient case #2 (as seen on November 27th, 2017). The cyst imitates common ovarian endometrioma, however, at the time of the US-scan the patient was after a bilateral adnexectomy. The size of the cystic structure is 34 × 44 × 35 mm.
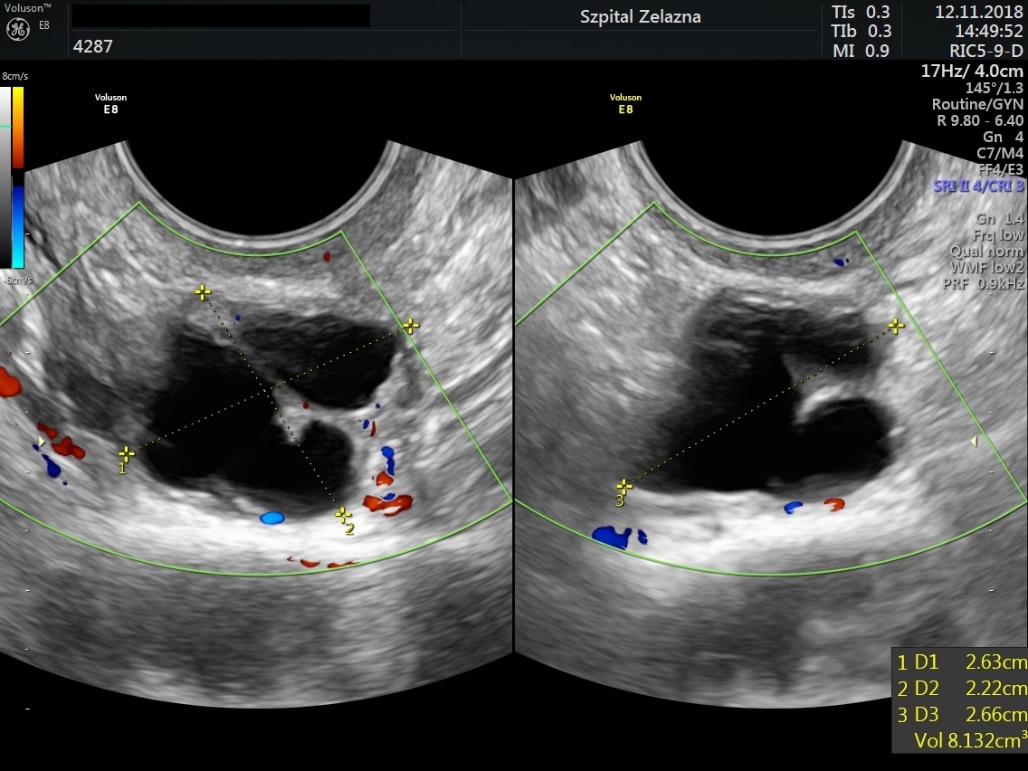 Figure 3.
Figure 3.— Transvaginal ultrasound scan showing the evolution of a cystic structure above the vagina vault of patient #2 in year 1 follow-up (as seen on November 12th, 2018). The evolution of the lesion is not characteristic of a common endometrioma. The size of the cystic structure is 26 × 22 × 27 mm.
Since pelvic pain syndrome persisted, therapy with aromatase inhibitors was introduced. The patient discontinued the treatment because of adverse effects and little therapeutic effect. Currently, the patient is receiving nonsteroid analgesia and weak opioids for breakthrough pains combined with antidepressive and antiepileptic drugs.
Symptoms of endometriosis are chronic pelvic pain, abdominal pain, abnormal vaginal bleeding, dyschezia, dysuria, dyspareunia, anal bleeding, abnormal bowel movements or infertility. In some cases, there are no symptoms and endometriosis may be revealed accidentally by ultrasound examination or laparoscopy. Evaluation of intestinal and ureteral endometriosis is necessary when deep lesions are palpable. The progress of the disease also destructs other organs, causing disabilities. About 20% of patients require recurrent surgery due to symptoms persistence or recurrence. Ureter infiltration is extremely dangerous because it may lead to stenosis and secondary asymptomatic loss of kidney function. DIE management may be initiated by medical therapy, while non-symptomatic patients should be carefully followed-up. Indications for surgery include ineffective pharmacological treatment, adverse effects, and the occurrence of hydronephrosis. The recommended surgical approach is laparoscopy with radical excision of endometriotic infiltrating tissues [11]. Damage of ureter with concomitant hydronephrosis may be painless, therefore follow up of DIE cases that includes US-scan of kidneys may be indicated. In cases of DIE suspicion, careful ultrasound examination or MRI are recommended [9, 10]. On the other hand, many general surgical teams consider colonography performed by CT and MRI are comparable methods to diagnose bowel masses. CT colonography is cheaper and much more available in our setting, moreover, local radiologists are well acquainted with CT assessment. Surgery is advised in instances were intestinal endometriosis is refractory to medical treatment or hormone therapy is contraindicated. A surgical approach largely depends on the location, the number and the extent of the lesions, as well as the infiltration of extra-genital organs [9]. The excision of DIE should be considered as surgery that reduces pain, improves bowel function, reduces diarrhea, constipation, anal bleeding associated with endometriosis, and improves patients' quality of life. Nevertheless, radical surgery is associated with a significant rate of complications. According to the ESHRE Guidelines, surgery of DIE is threatened by a 2.1% rate of intra operative complications (such as bleeding resulting in conversion to open surgery and ureteral or small bowel damage), and up to 13.9% of postoperative complications (such as rectovaginal, ureteral or vesicovaginal fistula, pelvic abscess, bleeding, ureteral stenosis, vaginal apex necrosis or stenosis of a protective ileostomy) [12, 13]. There is currently insufficient evidence to clearly confirm whether and to what extent these procedures are appropriate. More research and follow-up are necessary in patients with radical, optimal or sub-optimal surgery. It seems that careful consideration needs to be given in order to balance between the chance of recovery or long-term remission and the increased risk of serious complications [14, 15]. If hormonal and conservative surgical treatment are unsuccessful, a total hysterectomy with bilateral adnexectomy is recommended, but the decrease in sex hormone levels is associated with increased morbidity as well as general and local symptoms of estrogen deficiency [16].
In the presented cases, the disease progressed despite radical treatment. Interestingly endometriosis infiltration caused a need of ureteroneocystostomy performance and further loss of kidney on left side in both cases. Left-sided localization of ureteral endometriosis is more common [5] and can be related to asymmetrical development of DIE lesion caused by the unidirectional circulation of peritoneal fluid and anatomical differences between right and left hemipelvis [2]. Although radical excision of ureteral lesions is required for complete treatment [6], it not warrants long time remission of the disease. Described cases, as the prevailing majority of ureteral endometriosis cases, had multiple localization of deep lesions [5] and were burdened with many surgical procedures that can destructs normal anatomy. Performing repeated surgeries significantly decrease the effectiveness of treatment and increases the rate of complication. Poor local conditions and high risk of dehiscence may enforce the decision of performing non-anatomic anastomosis, like in Hartmann procedure, and a need of subsequent surgeries.
Our experiences agree, with the opinion that there is a high risk of recurrence following conservative treatment [17]. In addition, the effectiveness of surgery and recurrence rate depends not only on surgical excellence, but also on patient characteristics, biological aggressiveness of the disease and compliance to pharmacological therapy. Postoperative therapy with continuous or cyclic combined or progestin-only pill is a proven method of prevention of endometriosis recurrence [18], and was applied in both cases. The effect of postoperative treatment depends largely on length of treatment and patients’ compliance, that can be limited by side effects or individual patient decisions. Unfortunately, despite radical dissection being performed by an experienced surgical team and prescription of hormonal therapy, treatment can be unsuccessful and endometriosis can still progress further [19, 20].
Comprehensive diagnosis should be done before surgery to allow preparation of multidisciplinary team for one-step surgery. Since involvement of ureters can be asymptomatic for a long time in majority of patients or signs could be overlooked unless systematically sought, careful questioning, physical examination and renal ultrasonography may be helpful before operation [6]. Ureteral endometriosis is not presumed in the majority of patients before the surgery [21], therefore, ureters should be carefully evaluated in all surgeries performed for DIE, even when pyelocaliectasis was not found before [5]. Moreover, the surgery should be well tailored to needs of patients and reserved for cases that can benefit from invasive treatment and performed in well prepared team to avoid repeated surgery. Current therapeutic spectrum does not always fit well to all clinical circumstances and could be related to severe complications, therefore, novel, less invasive, safer and more effective management strategies are urgently sought for better personalization of treatment.
DFT, RM: project development, data collection, management data analysis, manuscript writing and editing, AK, AR, MR: manuscript writing and editing, MS, BPS: data collection, performance of surgery, manuscript writing and editing.
We would like to thank Elżbieta Marchlewska, Dorota Sys and Kinga Kalita for their technical assistance. Funding support from NCN (2011/03/N/NZ5/05899) is greatly appreciated (R.M.). The authors acknowledge the institutional support from the Center of Postgraduate Medical Education.
The authors declare no conflict of interest.



