1 Department of Nephrology, Jiangnan University Medical Center (Wuxi No.2 People’s Hospital), 214002 Wuxi, Jiangsu, China
Abstract
There remain significant challenges in assessing the risk of primary nephrotic syndrome (PNS) and concomitant acute kidney injury (AKI) in adult males. AKI is characterized by decreased glomerular filtration rate. This study investigates the levels of serum sex hormones, bone morphogenetic protein-7 (BMP-7), and retinol-binding protein (RBP) and their significance in adult male patients with PNS and concomitant AKI.
This retrospective analysis enrolled 86 adult male PNS patients admitted to Wuxi No.2 People’s Hospital between January 2020 and November 2023 as the PNS group, and 80 healthy adult males as the healthy group. Serum levels of sex hormones—estradiol (E2), testosterone (T), follicle-stimulating hormone (FSH), and luteinizing hormone (LH)—as well as bone morphogenetic protein-7 (BMP-7) and RBP were compared between the two groups. Furthermore, PNS patients were stratified into PNS groups with or without AKI cohorts based on glomerular filtration rate. Additionally, the levels of these parameters were compared between the subgroups.
In the PNS group, the levels of E2 and RBP were 66.67 ± 18.87 pg/mL and 56.65 ± 17.23 mg/L, respectively, both significantly higher than those in the healthy group (p < 0.05). Conversely, FSH and BMP-7 levels were 3.84 ± 1.02 IU/L and 1.83 ± 0.44 ng/mL, respectively, both significantly lower than those in the healthy group (p < 0.05). Within the PNS group, patients with AKI demonstrated significantly higher E2 levels of 69.93 ± 9.95 pg/mL than non-AKI patients (p < 0.05). FSH and BMP-7 levels were substantially lower (3.45 ± 0.96 IU/L and 1.57 ± 0.35 ng/mL) than in non-AKI patients (p < 0.05). However, RBP level was considerably higher (62.23 ± 12.01 mg/L) in those with AKI (p < 0.05). Furthermore, serum creatinine (Scr), blood urea nitrogen (BUN), and cystatin C (Cys C) levels were 167.23 ± 34.43 μmol/L, 11.14 ± 2.23 mmol/L, and 2.02 ± 0.72 mg/L, respectively, all significantly higher in patients with AKI than in non-AKI (p < 0.05). Regression analysis identified E2, BMP-7, and RBP as independent predictors for assessing AKI risk in PNS patients, with corresponding odds ratios (ORs) (95% confidence interval (CI)) values of 1.092 (1.016–1.175), 0.021 (0.002–0.184), and 1.095 (1.032–1.162), respectively (p < 0.05).
Male patients with PNS exhibit abnormal serum sex hormone, BMP-7, and RBP levels, which are associated with AKI. E2, BMP-7, and RBP levels demonstrate significant predictive potential in assessing AKI risk in PNS.
Keywords
- nephrotic syndrome
- males
- gonadal hormones
- bone morphogenetic protein-7
- retinol-binding protein
Primary nephrotic syndrome (PNS) is a clinical condition in which various factors increase glomerular capillary permeability, resulting in significant urinary loss of plasma proteins [1, 2]. PNS incidence is higher in males than in females [3]. Patients with PNS are at risk of acute kidney injury (AKI), characterized by a decline in glomerular filtration rate, which poses a substantial challenge to its clinical management [4].
Currently, clinical diagnosis of PNS-related AKI relies mainly on kidney biopsy and assessment of serum creatinine levels. Although a kidney biopsy provides detailed pathological information, its invasive nature limits its widespread application [5]. On the other hand, the elevation of serum creatinine is often delayed and therefore cannot predict early kidney damage, making it suboptimal as an early warning indicator for PNS and AKI in adult males [6].
Previous studies have shown that estradiol (E2) and follicle-stimulating hormone (FSH) are associated with mesangial cell proliferation, podocyte apoptosis, immune activation, and renal interstitial fibrosis [7, 8]. Similarly, bone morphogenetic protein-7 (BMP-7) exerts anti-fibrotic effects in the renal interstitium, promotes podocyte repair, and has anti-inflammatory properties. However, its expression is negatively correlated with the severity of kidney injury [9, 10]. Retinol-binding protein (RBP), synthesized in the liver, is freely filtered at the glomerulus, with approximately 95% reabsorbed by the proximal tubules, and its expression level is positively correlated with the degree of renal tubular injury [11, 12]. Although the evidence provides partial insights into the pathogenesis of PNS and AKI, systematic investigations into the associations between biomarkers such as E2, FSH, BMP-7, and RBP and PNS in adult males, and their application in assessing AKI risk in this population, remain limited.
Therefore, this study aims to compare the levels of these biomarkers in adult males with PNS and healthy adult males, as well as between PNS with and without AKI. By rigorously evaluating their relationships with AKI complicating PNS, the study seeks to provide more precise and reliable evidence to enhance clinical diagnosis and management.
This study adopted a retrospective analysis design, and the required sample size was calculated using the following formula.
Where n represents the required sample size for each group,
Z1-α/2 is the Z-score corresponding to the significance
level
Using BMP-7 as the core observation indicator, a preliminary sample of 15 adult
males with PNS and 15 healthy males was collected. The mean BMP-7 levels were
2.08
This retrospective study included 86 adult male patients with PNS who received treatment at Wuxi No.2 People’s Hospital, China, between January 2020 and November 2023 as the PNS group.
Inclusion criteria for patient selection were as follows: (1) age
Additionally, to control unnecessary research costs, 80 of 107 eligible healthy
adult males were enrolled as the healthy group. The inclusion criteria
for control individuals were: (1) age
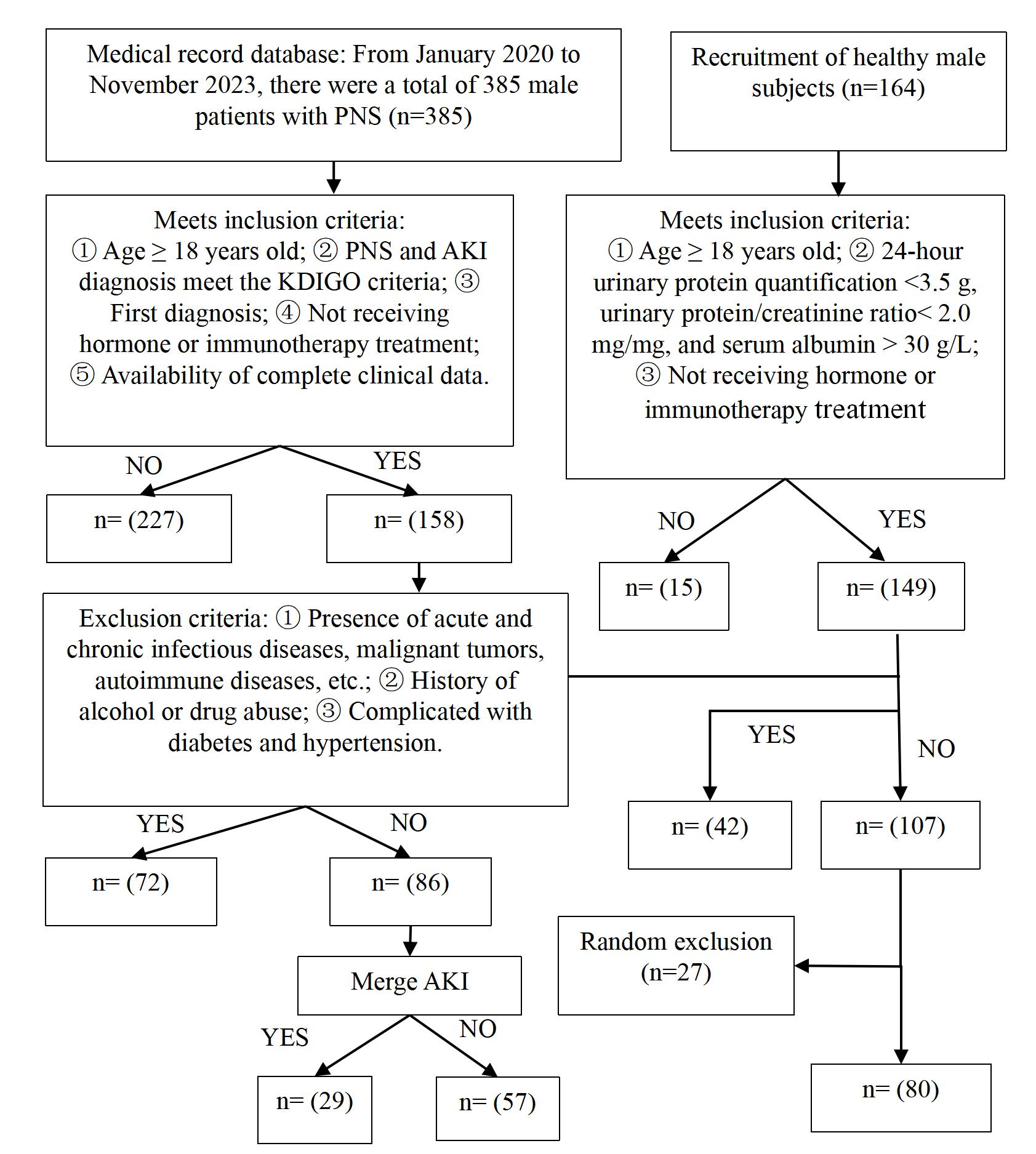 Fig. 1.
Fig. 1.
A flowchart of the study participants’ selection. PNS, primary nephrotic syndrome; AKI, acute kidney injury; KDIGO, Kidney Disease: Improving Global Outcomes.
A 3 mL fasting venous blood sample was collected from each participant and centrifuged at 3000 rpm for 5 minutes to obtain serum. Serum levels of E2, testosterone (T), FSH, luteinizing hormone (LH), and cystatin C (Cys C) were measured using an ADVIA Centaur XP automated chemiluminescence immunoassay system (version 3.2, Siemens Healthcare Diagnostics, Inc., Tarrytown, NY, USA) with the following reagent kits: E2 kit (Beckman Coulter, Inc., Brea, CA, USA; Cat. No. HM2093), T kit (Siemens Healthcare Diagnostics, Inc., Tarrytown, NY, USA; Cat. No. 02789602), FSH kit (Suzhou Changguang Huayi Biomedical Engineering Co., Ltd., Suzhou, China; Cat. No. AE-480), LH kit (Siemens Healthcare Diagnostics, Inc., Tarrytown, NY, USA; Cat. No. LH-110758), and Cys C kit (Siemens Healthcare Diagnostics, Inc., Tarrytown, NY, USA; Cat. No. PH32953).
Furthermore, RBP and BMP-7 levels were determined using an HZ-96A enzyme-linked
immunosorbent assay (ELISA) platform, with assay kits obtained by Siemens
Healthcare Diagnostics Products GmbH (Marburg, Germany; RBP: Cat. No. CB455351;
BMP-7: Cat. No. CHE0182-048). Blood urea nitrogen (BUN) and serum creatinine
(Scr) levels were measured using a DxC 700 AU automated biochemical analyzer
(Beckman Coulter, Inc., Brea, CA, USA) with BUN and Scr reagent kits (Siemens
Healthcare Diagnostics, Inc., Tarrytown, NY, USA; BUN: Cat. No. BC1535; Scr: Cat. No.
ZC100258). Estimated glomerular filtration rate (eGFR) was calculated from serum
Scr using the chronic kidney disease epidemiology collaboration (CKD-EPI) equation [15]. eGFR = 141
Statistical analyses were performed using SPSS version 22.0 (International
Business Machines Corporation, Armonk, NY, USA). Measurement data were evaluated
for normality using the Shapiro-Wilk test and were all confirmed to follow a
normal distribution. These variables were presented as mean
Furthermore, partial Pearson correlation analysis was conducted after adjusting for potential confounding factors, including age, body mass index (BMI), smoking history, and alcohol consumption. Variables with statistically significant
differences in univariate analysis (p
Receiver operating characteristic (ROC) curve analysis was conducted to evaluate
the performance of each parameter in assessing the risk of AKI in PNS patients,
and optimal cutoff values were determined using the Youden index [16]. A
p-value
There were no statistically significant differences in age, body mass index,
smoking history, or alcohol consumption between the PNS and healthy
groups (p
| Group | Number of cases | Age (years) | Body mass index (kg/m2) | Smoking (%) | Drinking (%) |
| PNS group | 86 | 39.93 |
21.82 |
37 (43.02) | 44 (51.16) |
| Healthy group | 80 | 40.45 |
22.04 |
31 (38.75) | 47 (58.75) |
| t/χ2 | –0.350 | –0.678 | 0.313 | 0.963 | |
| p-value | 0.727 | 0.499 | 0.576 | 0.326 |
Note: Measurement data were evaluated for normality using the Shapiro-Wilk test
and were expressed as
E2, FSH, BMP-7, and RBP levels showed statistically significant differences
between the PNS and healthy groups (p
| Group | Number of cases | E2 (pg/mL) | FSH (IU/L) | LH (IU/L) | T (ng/mL) | BMP-7 (ng/mL) | RBP (mg/L) |
| PNS group | 86 | 66.67 |
3.84 |
5.22 |
5.26 |
1.83 |
56.65 |
| Healthy group | 80 | 35.54 |
6.65 |
4.81 |
5.02 |
5.12 |
31.14 |
| t | 13.083 | –16.759 | 1.941 | 1.399 | –27.925 | 11.576 | |
| p-value | 0.054 | 0.164 |
Note: Measurement data were assessed for normality using the Shapiro-Wilk test
and were expressed as
There were no statistically significant differences in serum E2, FSH, LH, and T
levels among PNS patients stratified by age, body mass index, or pathological
type (p
| Clinical characteristic | Number of cases | E2 (pg/mL) | FSH (IU/L) | LH (IU/L) | T (ng/mL) | |
| Age | ||||||
| 43 | 65.82 |
3.86 |
5.18 |
5.30 | ||
| 43 | 67.40 |
3.82 |
5.25 |
5.22 | ||
| t | –0.676 | 0.194 | –0.312 | 0.390 | ||
| p-value | 0.501 | 0.847 | 0.756 | 0.698 | ||
| Body mass index | ||||||
| 48 | 66.45 |
3.80 |
5.29 |
5.30 | ||
| 38 | 66.91 |
3.88 |
5.14 |
5.20 | ||
| t | –0.212 | –0.378 | 0.694 | 0.427 | ||
| p-value | 0.832 | 0.706 | 0.490 | 0.670 | ||
| Pathological type | ||||||
| Membranous nephropathy | 58 | 66.31 |
3.79 |
5.26 |
5.35 | |
| Other | 28 | 67.34 |
3.93 |
5.15 |
5.09 | |
| t | –0.417 | –0.650 | 0.509 | 1.091 | ||
| p-value | 0.678 | 0.518 | 0.612 | 0.278 | ||
| Acute kidney injury | ||||||
| Yes | 29 | 69.93 |
3.45 |
5.26 |
5.28 | |
| No | 57 | 64.82 |
4.06 |
5.20 |
5.25 | |
| t | 2.244 | –2.747 | 0.257 | 0.136 | ||
| p-value | 0.027 | 0.007 | 0.798 | 0.892 | ||
Note: In the “Pathological type” category, “other” includes pathological
types such as minimal change disease and focal segmental glomerulosclerosis,
excluding membranous nephropathy. Measurement data were evaluated for normality
using the Shapiro-Wilk test and were expressed as
There was no significant difference in serum BMP-7 and RBP levels among PNS
patients when stratified by age, body mass index, and pathological subtype
(p
| Clinical characteristics | Number of cases | BMP-7 (ng/mL) | RBP (mg/L) | |
| Age | ||||
| 43 | 1.85 |
57.03 | ||
| 43 | 1.81 |
56.32 | ||
| t | 0.416 | 0.272 | ||
| p-value | 0.678 | 0.786 | ||
| Body mass index | ||||
| 48 | 1.84 |
56.15 | ||
| 38 | 1.82 |
57.20 | ||
| t | 0.290 | –0.401 | ||
| p-value | 0.772 | 0.690 | ||
| Pathological type | ||||
| Membranous nephropathy | 58 | 1.81 |
55.34 | |
| Other | 28 | 1.86 |
59.08 | |
| t | –0.531 | –1.513 | ||
| p-value | 0.597 | 0.134 | ||
| Acute kidney injury | ||||
| Yes | 29 | 1.57 |
62.23 | |
| No | 57 | 1.98 |
53.48 | |
| t | –5.001 | 3.228 | ||
| p-value | 0.002 | |||
Note: Measurement data were evaluated for normality using the Shapiro-Wilk test
and were expressed as
Within the PNS group, patients with AKI had higher Scr, BUN, eGFR and Cys C levels
than those without AKI (p
| Acute kidney injury | Number of cases | Scr (µmol/L) | BUN (mmol/L) | Cys C (mg/L) | eGFR (mL/min/1.73 m2) |
| Yes | 29 | 167.23 |
11.14 |
2.02 |
52.75 |
| No | 57 | 89.92 |
5.76 |
0.89 |
87.54 |
| t | 15.510 | 15.427 | 10.940 | –6.826 | |
| p-value |
Note: Measurement data were assessed for normality using the Shapiro-Wilk test
and were expressed as
After adjusting for potential confounding factors, including age, BMI, smoking
history, and alcohol consumption, partial Pearson correlation analysis revealed that
BMP-7 was negatively correlated with Scr, BUN, and Cys C, and positively
correlated with eGFR (p
| Scr | BUN | Cys C | eGFR | |
| E2 | 0.270a | 0.213a | 0.294a | –0.252a |
| FSH | –0.084 | –0.138 | –0.154 | 0.042 |
| LH | 0.038 | 0.100 | 0.016 | 0.115 |
| T | –0.056 | –0.025 | 0.003 | –0.047 |
| BMP-7 | –0.328a | –0.289a | –0.265a | 0.319a |
| RBP | 0.264a | 0.313a | 0.254a | –0.196a |
Note: Pearson correlation analysis was performed with age, BMI, smoking history,
and alcohol consumption as control variables; a, the correlation analysis
showed p
The four indicators that demonstrated statistically significant differences
between PNS patients with and without AKI in the univariate analysis (E2, FSH,
BMP-7, and RBP) were included in further modeling. Multicollinearity analysis
showed that these four indicators had VIF values
Multivariable logistic regression analysis revealed that E2, BMP-7, and RBP levels were
independently associated with the risk of AKI in PNS patients (p
| SE | Wald | p-value | OR (95% CI) | ||
| E2 | 0.088 | 0.037 | 5.722 | 0.017 | 1.092 (1.016–1.175) |
| FSH | –0.672 | 0.368 | 3.330 | 0.068 | 0.511 (0.248–1.051) |
| BMP-7 | –3.843 | 1.098 | 12.260 | 0.021 (0.002–0.184) | |
| RBP | 0.091 | 0.030 | 8.907 | 0.003 | 1.095 (1.032–1.162) |
| Constant | –1.450 | 3.954 | 0.134 | 0.714 |
Note: Multivariate logistic regression analysis was used; A p
Receiver operating characteristic (ROC) curve analysis showed that the areas
under the curve (AUC, 95% CI) for E2, BMP-7, and RBP in assessing AKI risk were
0.829 (0.733–0.926), 0.900 (0.833–0.966), and 0.725 (0.609–0.841),
respectively (p
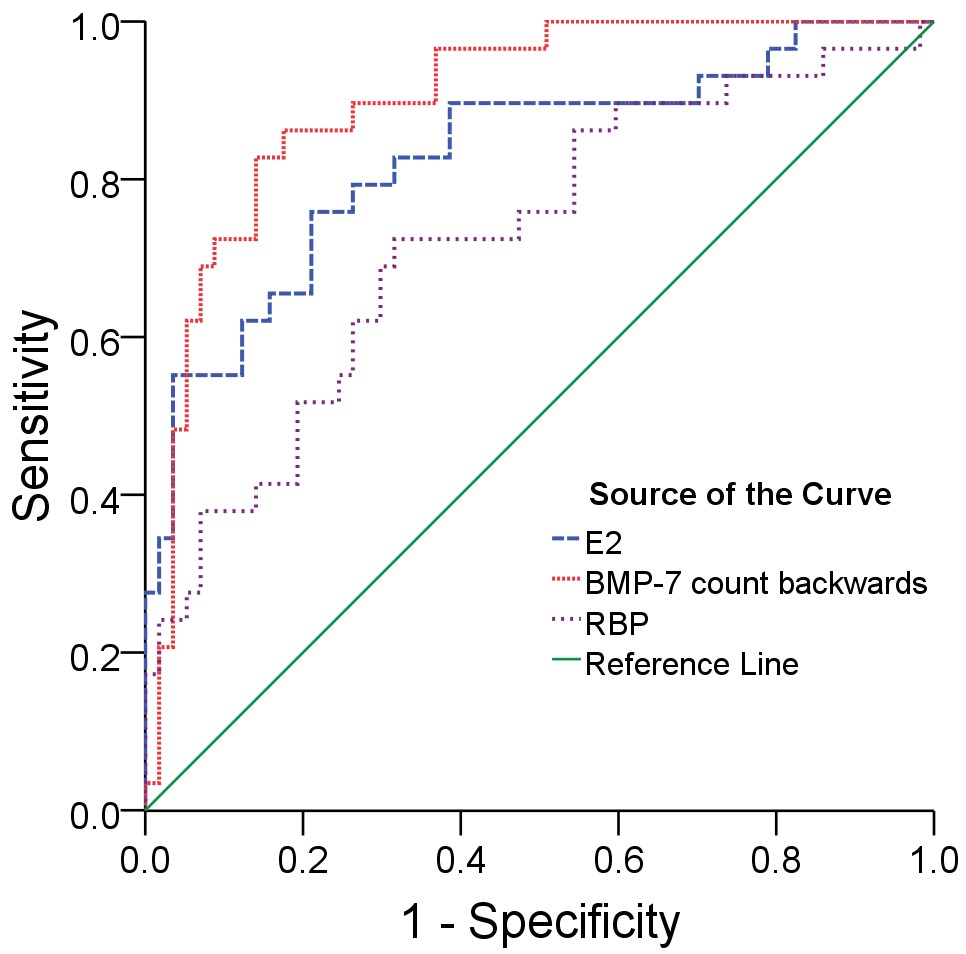 Fig. 2.
Fig. 2.
ROC curve parameters of E2, BMP-7, and RBP in the diagnosis of AKI. Note: The BMP-7 count backwards calculation method is 1/BMP-7.
| Indicator | Areas under the curve (95% CI) | p-value | Cutoff value | Sensitivity | Specificity |
| E2 | 0.829 (0.733–0.926) | 68.67 pg/mL | 0.759 | 0.782 | |
| BMP-7 | 0.900 (0.833–0.966) | 1.73 ng/mL | 0.828 | 0.861 | |
| RBP | 0.725 (0.609–0.841) | 59.59 mg/L | 0.725 | 0.685 |
Note: Cutoff values were determined by maximizing the Youden index. A p
PNS is an immune-inflammatory kidney disease in adults, often complicated by infections, which can reduce glomerular filtration rate or impair tubular reabsorption, thereby affecting life [17]. In this study, we analyzed serum sex hormone levels in adult males with PNS and found significantly higher E2 levels and substantially lower FSH levels than in the healthy group. These findings indicate a pronounced disruption of sex hormone homeostasis in PNS patients, characterized by elevated E2 and reduced FSH levels. This imbalance has been reported to have a profound impact on kidney disease on the sex hormone secretion system [18].
The findings of this study are consistent with previous research reporting
altered sex hormone levels in patients with nephrotic syndrome [19]. E2, through
its interaction with estrogen receptors, regulates the activity of multiple organ
systems. In the context of PNS, elevated E2 levels may represent a feedback
protective mechanism, reversing renal fibrosis and preserving kidney function by
downregulating the expression of mesangial cell adhesion molecules and inhibiting
the activity of transforming growth factor-
Additionally, the onset of PNS can disrupt the regulation of the hypothalamic-pituitary-gonadal axis, leading to decreased FSH secretion [22]. Current evidence suggests that PNS may also alter LH and T levels by affecting gonadal axis function [23]. In contrast, the present study did not find statistically significant differences in LH and T levels between adult males with PNS and healthy controls. This result may reflect the impact of PNS on specific components of the gonadal axis, suggesting that its effects on LH and T are relatively limited [24].
This study revealed that RBP levels in adult male PNS patients were higher than in healthy controls, whereas BMP-7 levels were significantly lower. Under physiological conditions, this low-molecular-weight protein is mainly reabsorbed by proximal tubular epithelial cells; elevated RBP levels indicate impaired glomerular filtration and tubular reabsorption and may contribute to the onset of primary nephrotic syndrome [25]. Previous research has shown that increased RBP is a predictor of disrupted renal filtration and reabsorption, and the present study further supports its role as a sensitive indicator of tubular function in PNS patients [26].
BMP-7 is predominantly expressed in podocytes, distal renal tubules, and collecting ducts [27]. Reduced BMP-7 expression promotes epithelial-to-mesenchymal transition in tubular epithelial cells and exacerbates podocyte injury, thereby accelerating renal fibrosis, a key pathogenic mechanism in PNS. The decreased BMP-7 levels observed in this study are consistent with these pathological characteristics and suggest that changes in BMP-7 may serve as an important diagnostic biomarker for PNS.
In male patients with PNS complicated by AKI, E2 levels were significantly higher, and FSH levels were substantially lower than in those without AKI. One underlying mechanism may be that E2 enhances immune defence by activating lymphocytes and promoting immunoglobulin production against exogenous pathogens; however, an excessive immune activation may exacerbate renal inflammation, trigger glomerulonephritis and related lesions, and thereby promote the onset of AKI in PNS [28]. Although E2 exerts renoprotective effects, such as inhibiting mesangial cell adhesion molecule expression and alleviating fibrosis, these beneficial effects may be disrupted in PNS patients who develop AKI, resulting in aggravated kidney injury [29].
Furthermore, decreased FSH levels may be a key contributor to the development of AKI. FSH plays a crucial role in maintaining testicular function and sex hormone balance, and its reduction may impair androgen synthesis and secretion, thereby disrupting normal regulation of cellular proliferation and apoptosis [30]. This disruption can diminish anti-fibrotic capability and weaken renal self-repair mechanisms, further accelerating AKI progression.
Our study findings found that BMP-7 levels were significantly decreased in adult male PNS patients complicated by AKI. This reduction is directly associated with tubular epithelial-mesenchymal transition (EMT) and podocyte injury, indicating impairment of the kidney’s intrinsic protective barrier, loss of functional homeostasis, accelerated renal fibrosis, and exacerbated PNS pathology [31]. Concurrently, RBP levels were markedly elevated in these patients, reflecting compromised reabsorptive capacity of injured renal tubules and supporting the potential of RBP as a sensitive early warning biomarker for AKI in this cohort.
Additionally, we observed that compared with PNS patients without AKI, those with concurrent AKI exhibited significantly higher serum Scr, BUN, and Cys C levels, along with a substantially lower eGFR. These observations confirm significant renal dysfunction in PNS patients who develop AKI. The elevations in Scr, BUN, and Cys C directly reflect reduced renal capability to remove metabolic wastes, while the significant decline in eGFR largely reflects AKI-induced reductions in renal blood flow, dysfunction of the glomerular filtration membrane, and potential tubular obstruction, collectively leading to a sharp decrease in nephron function. Notably, correlation analysis revealed that BMP-7 levels were negatively associated with Scr, BUN, and Cys C, whereas E2 and RBP levels were positively linked to these renal function parameters. A decrease in BMP-7 indicates impaired renal protective mechanisms, facilitating the accumulation of metabolic waste products such as Scr, BUN, and Cys C. In contrast, elevated RBP levels directly reflect diminished renal excretory function, showing a trend consistent with the accumulation of metabolic waste products [32, 33]. The positive correlation between E2 levels and markers of renal dysfunction observed in this study may reflect an inadequate compensatory response to renal injury, wherein the protective mechanisms of E2 become overwhelmed in the setting of severe tubular damage and oxidative stress—a phenomenon described as hormonal resistance in other inflammatory conditions [34, 35].
Regression analysis revealed that E2, BMP-7, and RBP were significantly associated with the onset of AKI in patients with PNS. Elevated E2 and RBP levels, along with reduced BMP-7 levels, were all linked to an increased risk of AKI in PNS patients. In contrast, FSH, LH, and T showed no statistically significant associations with PNS complicated by AKI, suggesting that E2, BMP-7, and RBP may serve as key indicators for assessing AKI risk in PNS patients, whereas FSH, LH, and T may have comparatively weaker associations. Similarly, ROC curve analysis further demonstrated that the AUC (95% CI) for E2, BMP-7, and RBP in diagnosing PNS complicated by AKI were 0.829 (0.733–0.926), 0.900 (0.833–0.966), and 0.725 (0.609–0.841), respectively. These findings indicate that all three markers have high potential for evaluating AKI risk in PNS and may offer new insights into understanding the mechanisms of acute kidney injury in adult male patients with PNS.
However, as this was a retrospective study, longitudinal time-series data and interventional evidence were not available to explore in depth the causal relationships and underlying mechanisms linking serum sex hormones, BMP-7, and RBP with the onset and progression of PNS, as well as with PNS complicated by AKI. Our findings can therefore only confirm that these parameters differ between male PNS patients with AKI, those without AKI, and healthy males, and that E2, BMP-7, and RBP are associated with the onset of AKI in PNS. Moreover, although the sample size was formally determined, the estimation was based solely on BMP-7 as the core parameter, which may have resulted in insufficient statistical power for other indicators. Furthermore, all participants were obtained from a single hospital, potentially limiting the generalizability of these results to broader patient populations in other regions. Future studies should include larger sample sizes and adopt a multicenter, prospective observational study design and interventional trials to further elucidate the relationships between serum sex hormones, BMP-7, and RBP, and the development and progression of PNS and its complications with AKI.
In conclusion, adult male PNS patients demonstrate significantly abnormal levels of serum sex hormones, BMP-7, and RBP, and these changes are closely related to the occurrence of AKI. E2, BMP-7, and RBP serve as reference indicators for assessing AKI risk in patients with PNS. In clinical practice, managing these biomarkers and implementing early intervention approaches based on their levels may reduce the risk of AKI in patients with PNS.
• Compared with healthy adult males, those with PNS exhibited elevated levels of E2 and RBP but lower levels of FSH and BMP-7.
• Compared with PNS adult male patients without AKI, those with AKI had higher E2 and RBP levels and lower BMP-7 and FSH levels.
• E2, BMP-7, and RBP are independent predictors of AKI in adult male PNS patients. Combining these parameters can enhance risk assessment.
• Future studies should expand the sample size and examine whether the correlations of E2, BMP-7, and RBP with disease progression and AKI in adult male patients with PNS in different regions and ethnic groups.
• We recommend routine monitoring of E2, BMP-7, and RBP levels in these patients to enable timely assessment of AKI risk.
PNS, primary nephrotic syndrome; AKI, acute kidney injury; E2, estradiol; FSH, follicle-stimulating hormone; BMP-7, bone morphogenetic protein-7; RBP, retinol-binding protein; T, testosterone; LH, luteinizing hormone; Cys C, cystatin C; BUN, blood urea nitrogen; Scr, serum creatinine; eGFR, estimated glomerular filtration rate.
The data used to support the findings of this study are available from the corresponding author upon request.
YZ and LZ designed the research study and wrote the first draft. YZ and LZ performed the research. YZ and LZ analyzed the data. Both authors contributed to the important editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was approved by the Ethics Review Committee of Wuxi No.2 People’s Hospital (Approval No.: 2020-Y-7) and was conducted in accordance with the principles of the Declaration of Helsinki. All patients participated voluntarily and provided written informed consent.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


