1 Obstetrics and Gynecology Unit, Department of Woman, Child and General and Specialized Surgery, University of Campania “Luigi Vanvitelli”, 80138 Naples, Italy
2 Saveetha Dental College and Hospitals, Saveetha Institute of Medical and Technical Sciences (SIMATS), Saveetha University, 600077 Chennai, India
3 Pediatric Unit, Department of Woman, Child and General and Specialized Surgery, University of Campania “Luigi Vanvitelli”, 80138 Naples, Italy
4 Department of Orthodontics, Faculty of Dentistry, Trakya University, 22030 Edirne, Turkey
5 Department of Biomedical, Surgical and Dental Sciences, University of Milan, 20122 Milan, Italy
6 Department of Biomedical and Surgical and Biomedical Sciences, University of Catania, 95123 Catania, Italy
7 Multidisciplinary Department of Medical-Surgical and Dental Specialties, University of Campania “Luigi Vanvitelli”, 80138 Naples, Italy
Abstract
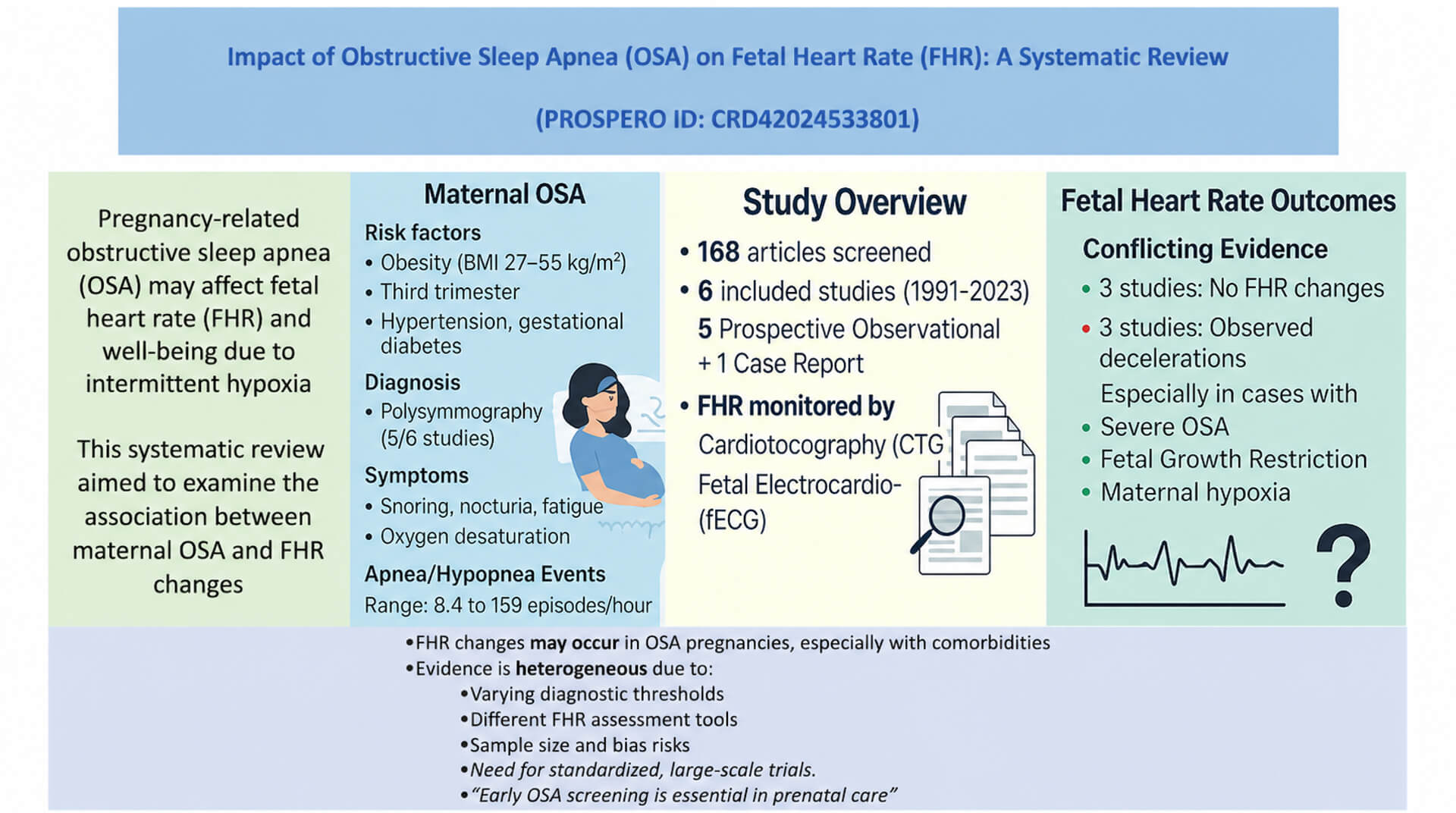
Obstructive sleep apnea (OSA) and intermittent maternal hypoxia during pregnancy have been hypothesised to impact fetal heart rate (FHR). To evidence this possible link, we performed a systematic review of the current evidence about maternal OSA and FHR alteration.
The following databases were analysed from the inception to June 2024: Cochrane Library, PubMed-Medline, EMBASE, Web of Science, and Google Scholar. We included studies with pregnant women affected by OSA, and FHR assessed via cardiotocography or fetal electrocardiography. Studies without polysomnography or home sleep testing, cross-over designs, or non-English publications were excluded.
168 studies were screened, and six were included. Five were prospective observational studies, and one was a case report. Maternal age showed a value from 26.6 ± 7.1 to 34.8 ± 3.3 years, and gestational age ranged from 32.1 ± 3.4 to 39.4 ± 1.0 weeks. The body mass index (BMI) values ranged between 27.5 ± 7.2 and 55 kg/m2. Polysomnography was the primary diagnostic tool for OSA in most studies; one study utilised a home sleep test. Apnea/hypopnea episodes frequency varied from 8.4 to 159 events per hour. Findings about the FHR alterations were discordant: three studies did not report an association, other three observed FHR changes linked to maternal respiratory events.
Our systematic review evidenced a significant variability about the maternal OSA impact on fetal heart rate. The study’s results were contradictory. These inconsistencies also derive from different study designs, sample sizes, and outcomes analysed. Further studies are necessary to evidence the real impact of the OSA on the fetal cardiac response.
PROSPERO (CRD42024533801).
Graphical Abstract
Keywords
- decelerations
- fetal assessment
- fetal heart rate
- heart rate variability
- polysomnography
- pregnancy
- sleep
- sleep apnea
The assessment of fetal growth and well-being remains a central focus of prenatal care [1]. In the last years, studies have investigated maternal breathing disorders and pregnancy compliance [2]. The sleep disorder also impacts fetal growth. One such condition is obstructive sleep apnea (OSA), with recurrent apneic episodes during sleep due to upper airway collapse, with consequently intermittent hypoxia and sleep alteration [3].
Management of temporomandibular joint (TMJ) disorders in patients with OSA typically involves conservative treatment approaches aimed at symptom relief and functional improvement [4]. Among these, oral appliances—particularly mandibular advancement devices—are commonly used and have shown efficacy in addressing both TMJ disorders and OSA-related symptoms [5].
OSA is more commonly associated with maternal complications—such as excessive daytime sleepiness, gestational hypertension, pre-eclampsia, and gestational diabetes [6]. Its role in intermittent fetal hypoxia is of growing concern. Prolonged or repeated hypoxic episodes may impair placental function, ultimately compromising fetal growth and development [3].
Different studies showed an increased prevalence of OSA during pregnancy [7]. Common symptoms of OSA include snoring, nocturia, personality changes, and excessive daytime sleepiness [3]. The symptoms worsen during the last pregnancy period due to the hormonal modification, increased body mass index (BMI), reduced lung volume, and trachea restriction [3]. Women with no previous history of sleep disorders may show symptoms during the third trimester [6]. OSA increased the risk of preterm delivery, gestational hypertension and preeclampsia and gestational diabetes [8].
Fetal well-being is evaluated through antenatal ultrasound test and cardiotocography (CTG) [9, 10]. Antepartum CTG is a primary method for evaluating fetal cardiac status [11]. This test permits the evaluation of the fetal heart rate (FHR) compensation due to fetal hypoxia [11]. Since CTG is not routinely performed during maternal sleep, episodes of OSA may impact fetal oxygenation and alter FHR variability without detection [9]. The impact of maternal OSA on fetal well-being, particularly in relation to CTG-detected FHR changes, remains under-researched and poorly understood.
We aimed to evidence the maternal OSA impact on the fetal heart rate. This represents a gap in the current research and should improve our understanding of the OSA influence on adverse prenatal outcomes.
The Population, Intervention, Comparison, and Outcomes (PICO) framework was applied to our research:
• Population (P): Third-trimester pregnant woman with a diagnosis of OSA.
• Intervention (I): FHR evaluation performed with CTG or fetal electrocardiogram.
• Comparison (C): Normal pregnancy without OSA diagnosis.
• Outcomes (O): Apnea/hypopnea episodes per hour, and apnea/hypopnea events followed by fetal heart alterations.
Polysomnography or home sleep tests for the OSA diagnosis were necessary to include the studies in our review. Cohort studies, case-control studies, case reports, case series and/or randomised controlled trials were considered eligible.
We excluded: (1) studies without polysomnography or home sleep test diagnosis, (2) cross-over study designs, (3) studies without the PICO framework, (4) no English language, (5) reviews, editorials, letters, posters and conference abstracts.
A systematic review was conducted using the following databases: Cochrane Library (https://www.cochranelibrary.com/advanced-search), PubMed-Medline (https://pubmed.ncbi.nlm.nih.gov/), EMBASE (https://www.embase.com/landing?status=grey), Web of Science (https://mjl.clarivate.com/search-results) and Google Scholar (https://scholar.google.it/). The search covered the period from database inception to June 2024. No geographic restrictions were applied in the selection of studies.
Various Medical Subject Headings (MeSH) were combined to identify studies related to cardiotocography and fetal heart monitoring, including: “decelerations”, “fetal assessment”, “fetal electrocardiogram”, and “heart rate variability”. Obstructive sleep apnea (OSA) was explored using the MeSH terms: “polysomnography”, “pregnancy sleep”, and “sleep apnea”.
We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidelines. A completed PRISMA 2020 checklist is available as Supplementary Table 1. The review protocol was registered PROSPERO ID: CRD42024533801 (https://www.crd.york.ac.uk/PROSPERO/view/CRD42024533801).
Two authors (AP and RM), both holding medical degrees, independently screened the titles and abstracts of all studies identified through the search strategy. Full-text articles of potentially eligible studies were retrieved and assessed for inclusion based on the predefined criteria. To identify additional studies reference lists of included studies and relevant reviews were explored. Discrepancies or uncertainties regarding study inclusion were resolved through discussion with a third reviewer (MLV).
Two reviewers (AP and RM) independently performed data extraction using a Microsoft Excel spreadsheet (version 2501, build 16.0.18429.20132, licensed to Università della Campania “Luigi Vanvitelli”, Italy) specifically designed for individualised data collection. The extracted data included: (1) first author, (2) year of publication, (3) country of origin, (4) patient characteristics (age, gestational age, and BMI), (5) type of sleep test or diagnostic criteria for OSA, and (6) fetal outcomes, including decelerations following apnea/hypopnea events, the presence of sleep-disordered breathing, and the number of apnea/hypopnea episodes per hour. Due to different study designs and outcomes included, we did not perform a quantitative synthesis. As a result, the findings were synthesised descriptively.
The risk of bias in the included studies was assessed by two independent authors, adopting a modified Newcastle-Ottawa Quality Assessment Scale [12]. The assessment covered five domains: (1) study design and sample representativeness, (2) sampling technique, (3) description of the fetal heart rate evaluation method, (4) quality of population description, and (5) completeness of outcome data (Table 1). Disagreements between reviewers were resolved by consultation with a third reviewer (MLV).
| (1) Study design and sample representativeness: |
| 1 point: Study design involved a control group, sample size was greater than or equal to 50 participants. |
| 0 points: Uncontrolled study, sample size less than 50 participants. |
| (2) Sampling technique: |
| 1 point: Patients recruited consecutively. |
| 0 points: Potential convenience sampling or unspecified sampling technique. |
| (3) Description of the FHR evaluation technique: |
| 1 point: The authors provided a comprehensive description of the equipment, setting, and adopted technique. |
| 0 points: The study did not report adequate information on the fetal evaluation technique. |
| (4) Quality of population description: |
| 1 point: The study reported a clear description of the population (e.g., age, BMI, apnea/hypopnea events |
| were followed by decelerations, etc.) with proper measures of dispersion (e.g., mean, standard deviation). |
| 0 points: The study did not report a clear description of the population, incompletely reported descriptive |
| statistics, or did not report measures of dispersion. |
| (5) Incomplete outcome data: |
| 1 point: The study reported complete data on the procedures and/or fetal outcomes. |
| 0 points: Selective data reporting cannot be excluded. |
The individual components listed above are summed to generate a total modified Newcastle-Ottawa risk of bias score for each study. Total scores range from 0 to 5.
For the total score grouping, studies were judged to be of low risk of bias
(
168 studies were identified after the screening. After removing duplicates and excluding studies that did not meet the inclusion criteria, six articles were deemed eligible for inclusion (Fig. 1) [13, 14, 15, 16, 17, 18].
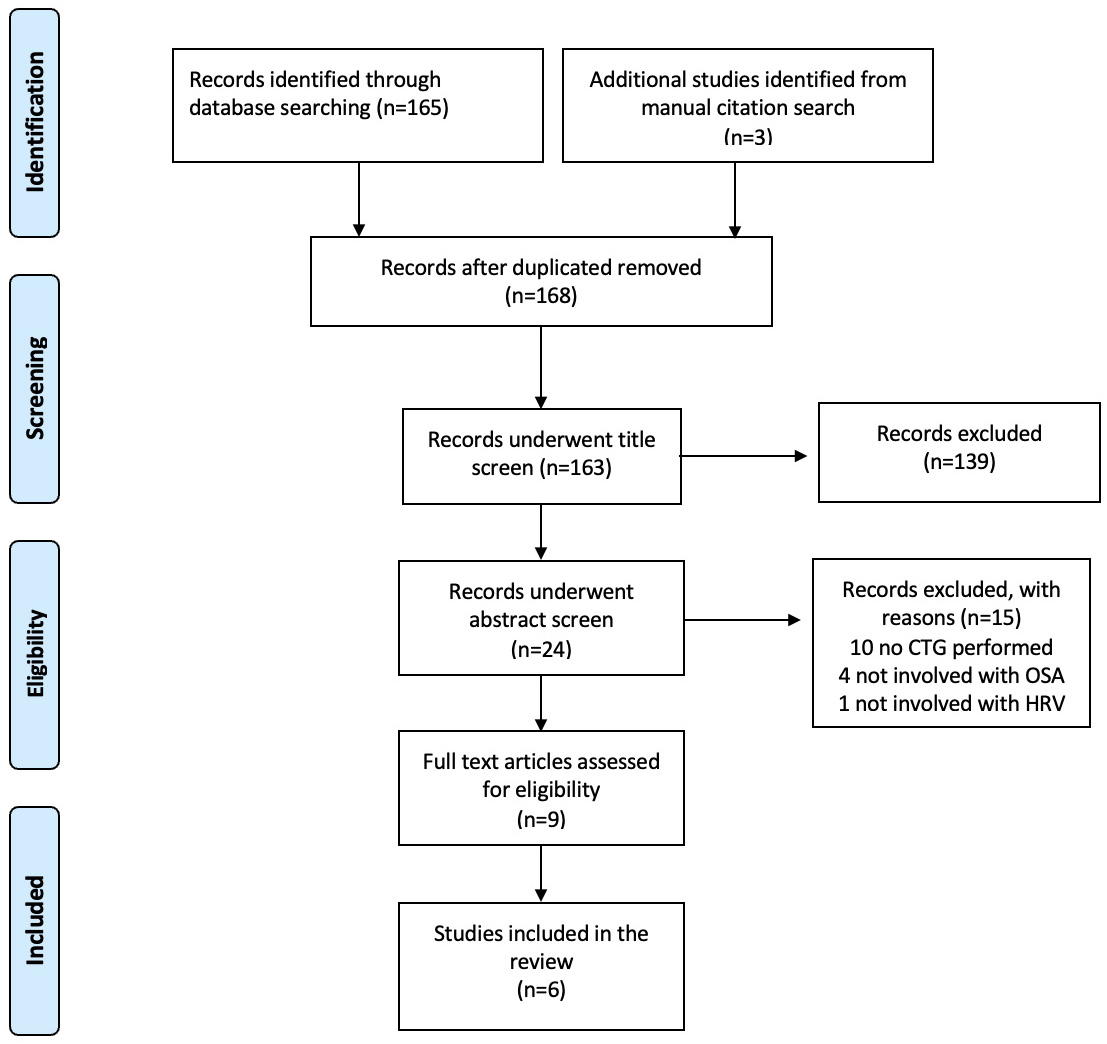 Fig. 1.
Fig. 1.
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2009 Flow Diagram summarising the study selection process and inclusion criteria. CTG, cardiotocography; OSA, obstructive sleep apnea; HRV, heart rate variability.
The key characteristics of the included studies are summarised in Table 2 (Ref. [13, 14, 15, 16, 17, 18]). One study was a case report [13]. The remaining five studies were prospective observational studies [14, 15, 16, 17, 18]. Of these, four studies were from North America [13, 15, 16, 18], one from Turkey [14] and one from Australia [17].
| Author, year, questionnaire | Study characteristics | Country | Patient characteristics | Diagnosis | ||
| Age mean (SD), y | GA mean (SD), wk | BMI mean (SD), kg/m2 | Sleep test/criteria | |||
| [18] | Prospective observational study | USA | Not specified | 36.2 |
Polysomnography | |
| [16] | Prospective observational study | USA | 32.0 |
34.6 |
37.1 |
Home sleep test |
| [13] | Case report | Canada | 32 | 36 | 55 | Polysomnography |
| [14] | Prospective observational study | Turkey | 34.8 |
37.5 |
Polysomnography | |
| [17] | Prospective observational study | Australia | FGR: 31.6 |
32.1 |
30.6 |
Polysomnography |
| C: 31.6 |
39.4 |
31.5 | ||||
| [15] | Prospective observational study | USA | 26.6 |
32.3 |
27.5 |
Polysomnography |
Abbreviations: GA, gestational age; FGR, fetal growth restriction; C, control group; SD, standard deviation.
Among the included studies, five demonstrated a low risk of bias in at least three of the evaluated domains [14, 15, 16, 17, 18], while one study was classified as having a high risk of bias [13]. A detailed description of the risk of bias assessment across all domains for each study is presented in Table 3 (Ref. [13, 14, 15, 16, 17, 18]).
| Author, year | Study design and sample representativeness | Sampling technique | Description of the fetal heart rate evaluation technique | Quality of population description | Incomplete outcome data | Total score |
| [18] | - | 1 | 1 | 1 | 1 | 4 |
| [16] | - | 1 | 1 | 1 | 1 | 4 |
| [13] | - | - | 1 | - | - | 1 |
| [14] | 1 | 1 | 1 | 1 | - | 4 |
| [17] | - | 1 | 1 | 1 | 1 | 4 |
| [15] | - | 1 | 1 | 1 | - | 3 |
Table 2 summarises patient characteristics and the type of sleep test used in
each of the included studies. The studies analysed were published between 1991
and 2023. The maternal age showed a mean of 26.6
OSA was diagnosed using polysomnography in five studies [13, 14, 15, 17, 18], and one study adopted the home sleep test [16].
Table 4 (Ref. [13, 14, 15, 16, 17, 18]) presents findings related to fetal compromise and sleep-disordered breathing events in pregnant women. Two studies assessed fetal compromise using a fetal electrocardiogram device (Monica AN24 and Monica datakey (DK), Monica Healthcare Ltd., Buckinghamshire, UK) [16, 18], and four studies applied cardiotocography (CTG) [13, 14, 15, 17].
| First author | Fetal compromise evaluation | Patients with sleep-disordered breathing events, number | Episodes of apnea/hypopnea per hour | Apnea/hypopnea events were followed by decelerations |
| [18] | fetal electrocardiogram (Monica AN24) | 82 | 8.7 | 45/2936 (1.5%) |
| [16] | fetal electrocardiogram (Monica DK) | 23 | 8.4 |
* |
| [13] | CTG | 1 | 159 | 0 |
| [14] | CTG | 4 | 13.5 |
** |
| [17] | CTG | 23 | NR | *** |
| [15] | CTG | 20 | 12.2 | 0 |
* A total of 37 late decelerations were observed in n = 18 women. Fifty percent (n = 9) of these women met the criteria for sleep-disordered breathing. Overall, 84% of late decelerations were temporally associated with a maternal respiratory event.
** 3/4 women with obstructive sleep apnea (OSA) had fetal heart decelerations accompanying maternal desaturation.
*** 20.2% of fetal heart rate (FHR) events had one or more apnoeas or hypopnoeas in the 5 minutes prior to the event (deceleration 19%, prolonged deceleration 17%, prolonged period of reduced variability 26%).
Abbreviations: CTG, cardiotocography; NR, not reported; DK, datakey.
The sleep-disordered breathing events ranged from 1 to 82 events, with different apnea/hypopnea episodes per hour among the studies [13, 14, 15, 17, 18]. DiPietro et al. [18] reported an average of 8.7 apnea/hypopnea episodes per hour, with only 1.5% of these events followed by fetal heart rate decelerations.
This finding is consistent with the results of Pitts et al. [16], who
reported an average of 8.4
In the 1991 case report by Charbonneau et al. [13], CTG revealed fetal
heart rate variability, with a reported 159 apnea/hypopnea episodes per hour, yet
no decelerations were observed. Sahin et al. [14] reported an average of
13.5
Skrzypek et al. [17] performed CTG monitoring on 23 pregnant women and found that 20.2% of fetal heart rate alterations were preceded by one or more apneic or hypopneic events. Finally, Olivarez et al. [15] applied CTG to 100 patients, reporting an average of 12.2 apnea/hypopnea episodes per hour, with no detected fetal abnormalities.
Our systematic review included six studies published between 1991 and 2023 that analysed the effects of maternal obstructive sleep apnea (OSA) on fetal heart rate (FHR), assessed using cardiotocography (CTG) and fetal electrocardiography (fECG) [13, 14, 15, 16, 17, 18, 19]. Except for Pitts et al. [16], which diagnosed the maternal sleep disorders with the home sleep test, all the studies diagnosed sleep disorders with polysomnography [13, 14, 15, 17, 18].
Half of the studies included in our review did not provide evidence of an impact of maternal OSA on fetal heart rate [13, 15, 18]. Three studies evidenced the association between maternal OSA and fetal heart distress, despite the limited sample [14, 16, 17]. Skrzypek et al. [17] investigated OSA in pregnant women with preterm fetal growth restriction (FGR) comorbidity. The results indicated that fetuses with this condition have more acute FHR events compared to the controls [17]. Moreover, FGR is a fetal condition that predisposes to hypoxia and adversely affects fetal outcomes [20, 21].
Olivarez et al. [15] and DiPietro et al. [18] did not evidence a direct correlation between OSA and fetal abnormalities. The predictive value of the Berlin screening questionnaire during pregnancy was explored by Olivarez et al. [15]. They included 100 pregnant women with gestational ages ranging from 26 to 39 weeks, and the authors concluded that the mechanism by which OSA may affect the fetus during maternal apneic episodes remains unclear [15]. DiPietro et al. [18] included 84 obese pregnant women at term of gestation. They observed only a modest compensatory increase in fetal heart rate variability in response to apneic and hypopneic episodes, without any pathological fetal heart response [18].
The case report by Charbonneau et al. [13] described a 32-year-old pregnant woman at 36 weeks of gestation, affected by severe obesity (155 kg), gestational diabetes, and fetal growth restriction. Despite an apnea–hypopnea index of 159 events per hour and maternal oxygen desaturation, CTG monitoring showed normal fetal heart rate reactivity in this patient [13].
Sahin et al. [14] analysed 35 term pregnant women, among whom only four were diagnosed with OSA. In these four cases, a strong correlation (75%) was observed between fetal heart rate decelerations and maternal oxygen desaturation [14]. Furthermore, neonates of women affected by OSA showed lower Apgar scores and birth weights [14]. Similarly, Pitts et al. [16] reported a strong correlation between maternal respiratory events and the occurrence of late and prolonged fetal heart rate decelerations. In their cohort of 40 women, 17 were diagnosed with sleep-disordered breathing during the third trimester; among them, 37 late decelerations were recorded, 84% of which were associated with maternal respiratory events [16].
Other studies evidenced the relation between the maternal hypoxic episodes and the fetal growth restriction, preterm delivery, and other adverse perinatal outcomes [22, 23, 24]. Apneic phase is associated with maternal desaturation, with an impact on the fetal circulation and fetal heart rate [25]. This association is not well recognised in pregnant populations. Fetal and placental circulatory systems may be well adapted, allowing the fetus to tolerate brief episodes of maternal desaturation without adverse effects [26, 27, 28].
Other risk factors—such as elevated body mass index (BMI), gestational hypertension, and gestational diabetes—may worsen fetal outcomes, predisposing affected individuals to increased susceptibility to the effects of maternal sleep disorders [29, 30, 31]. According to DiPietro et al. [18], maternal apneic episodes may cause fetal compensatory mechanisms.
Based on our systematic review, the studies included showed contradictory findings. This difference may originate from different study designs and different outcomes. Even studies that employed overnight polysomnography with continuous fetal ECG monitoring during maternal respiratory events reported contrasting findings [16, 18]. The presence of comorbid conditions may directly influence fetal heart rate patterns and confound the study’s results [13, 17].
Obesity remains the most significant confounding factor, as the majority of patients with maternal sleep disorders associated with fetal heart abnormalities were also affected by severe obesity [32]. Most studies were conducted in obese pregnant women, which represent an independent risk factor [13, 14, 16, 17, 18].
Other limitations are related to the geographic variability among the study settings [33]. Main studies were conducted in high-resource settings, North America and Australia [13, 15, 16, 17, 18], and could not represent the low-resource setting. The high-resource study predominance limits our generalizability [33, 34, 35].
Biases were evaluated with the modified Newcastle-Ottawa scale, to a rigorous systematic review.
OSA represents a possible risk factor for the fetal well-being and fetal heart modification. The current evidence are not unanimous about the OSA impact on the fetal heart modification. Despite these unclear findings, early diagnosis and management of maternal sleep disorders remain a significant point of prenatal care. Further standardized studies are needed to understand the complete relationship between OSA and fetal heart adaptation.
• We conducted a systematic review of the literature on the influence of maternal OSA on FHR.
• Six studies were included in our analysis, with three showing no significant effect on FHR and the other three demonstrating fetal distress in OSA pregnant women, characterised by FHR decelerations and changes.
• These discrepancies could be related to differences in study settings, sample size, diagnostic criteria for OSA, and methods of fetal monitoring.
• Despite those limitations, for the first time, this review has focused on the possible relationship between maternal OSA and FHR, which is significant for maternal sleep disorder management during pregnancy in order to optimize prenatal care and minimize the impacts of associated risks.
All the data of this study are included in this article.
Conceptualization, MLV, MMM and GM; methodology, MDB, RM and MC; formal analysis, AP; investigation, MLV; data curation, AP and RM; writing—original draft preparation, AP and RM; writing—review and editing, MDB and GM; visualization, HU and MMM; supervision, MDB and GM; All authors contributed to the important editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
GM was serving as one of the Editorial Board Members of this journal. We declare that GM had no involvement in the review of this article and has no access to information regarding its review. Full responsibility for the editorial process for this article was delegated to John Alcolado. Other authors declared no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/BJHM53217.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

