1 Academic Renal Department, Hull Royal Infirmary, Hull University Teaching Hospitals NHS Trust, HU3 2JZ Hull, UK
2 Department of Medical Sciences, Hull York Medical School, HU6 7RX Hull, UK
Chronic kidney disease (CKD), as defined by the National Institute for Health and Care Excellence (NICE), is an abnormality of kidney structure or function persisting for more than 3 months (typically with an estimated glomerular filtration rate (eGFR) less than 60 mL/min/1.73 m2 and/or albuminuria) [1]. CKD rarely occurs in isolation, as it commonly coexists with cardiovascular disease (CVD), type 2 diabetes mellitus (T2DM), hypertension, and obesity, which share overlapping underlying pathophysiological mechanisms. CKD has an estimated prevalence of 9.1% globally, although some large epidemiological studies report rates as high as 13.4%, with the greatest burden in stage 3 disease and diabetes mellitus (DM) accounting for approximately 30–50% of total cases [2]. Furthermore, it’s estimated that 1 in 3 adults with DM has concurrent CKD [1, 3].
Population data from the National Health and Nutrition Examination Survey suggest that approximately one-quarter of adults have at least one cardiac, kidney, or metabolic condition, defined as CVD, CKD, or T2DM, with the prevalence of overlapping conditions increasing steeply with increasing age. This translates into a disproportionate burden of heart failure, atherosclerotic events, and premature mortality once CKD is established, particularly when diabetes and obesity are also present.
Historically, kidney progression has often been viewed through a kidney-centric lens, with changes in eGFR and albuminuria used as current surrogates of disease trajectory. This approach has underpinned important advances in renin-angiotensin-aldosterone system (RAAS) inhibition and blood pressure control, but it does not fully capture the systemic, multi-organ nature of CKD risk. In practice, the same processes that drive loss of nephron mass (after natural age-related loss) also injure the myocardium, vasculature, and metabolic organs.
The emerging evidence proposes a shift in perspective from slowing eGFR decline in CKD to protecting the entire cardio-kidney-metabolic axis. In this editorial, we focus on how recognising these shared mechanisms and targeting key modifiable drivers, in addition to non-pharmacological interventions, such as lipids, blood pressure, and glycaemic control, can help to slow kidney progression while simultaneously improving cardiovascular and metabolic outcomes. In particular, the advent of sodium-glucose cotransporter 2 inhibitors (SGLT2i), glucagon-like peptide-1 (GLP-1) receptor agonists, non-steroidal mineralocorticoid receptor antagonists (finerenone), and other emerging agents such as endothelin antagonist has shifted the therapeutic goal from simply delaying eGFR decline towards actively preserving kidney function and achieving near-remission, through multimodal combination therapy, and early risk stratification and screening [4]. We also summarize how these advances fit within the Cardiovascular-Kidney-Metabolic (CKM) syndrome framework.
Traditionally, the term Cardiorenal Syndrome (CRS) was introduced to describe the bidirectional interaction between the heart and kidney, originally defined as “moderate or greater kidney dysfunction that exists or develops in people with heart failure” and later refined into five subtypes based on the initiating organ and the acute or chronic nature of the insult [5]. This construct highlighted the haemodynamic crosstalk between the two organs and helped normalise the idea that worsening renal function in heart failure is common and prognostically important. However, CRS remains largely focused on people with established heart failure and pays relatively little attention to upstream metabolic factors such as obesity and insulin resistance that shape who develops heart-kidney dysfunction in the first place.
In parallel, the concept of Metabolic Syndrome (MS) emerged to capture the clustering of central adiposity, hypertension, dyslipidaemia, and dysglycaemia in individuals at high risk of cardiovascular events and T2DM. In clinical practice, MS is typically diagnosed when a person has central (abdominal) obesity plus at least two of the following: impaired glucose regulation or insulin resistance, hypertension, hypertriglyceridaemia, and low high-density lipoprotein (HDL) cholesterol. Some have argued that a more pathophysiologically grounded definition—emphasizing abdominal obesity, insulin resistance, dyslipidaemia and hypertension as interrelated processes rather than discrete laboratory thresholds—better reflects the underlying biology and the concept of risk cluttering that is central to the syndrome.
Emphasising insulin resistance, chronic inflammation, and oxidative stress as unifying mechanisms has been useful in focusing attention on lifestyle and metabolic risk factor modification. However, a reliance on categorical cut-offs rather than continuous risk measures may oversimplify cardiometabolic risk and obscure important gradations of vulnerability. Moreover, emerging evidence suggests that additional factors—such as functional hypogonadism in men, which is linked to visceral adiposity, insulin resistance, and adverse cardiovascular risk—may be relevant to the broader metabolic phenotype. Yet within the traditional MS framework, the heart and kidneys are still largely viewed as passive “target organs” injured by metabolic risk, rather than active participants in a dynamic, bidirectional disease network.
Recognising these limitations, the American Heart Association (AHA) recently proposed CKM syndrome as an integrated entity that explicitly encompasses the interdependent pathophysiology of cardiovascular disease, CKD, diabetes, and obesity [6] (Fig. 1). A corresponding staging system, ranging from Stage 0 to Stage 4, was also introduced (Table 1). This staging system emphasises life-course screening and the importance of detecting early metabolic and kidney abnormalities, including albuminuria, in people who may not yet meet traditional criteria for CKD or CVD.
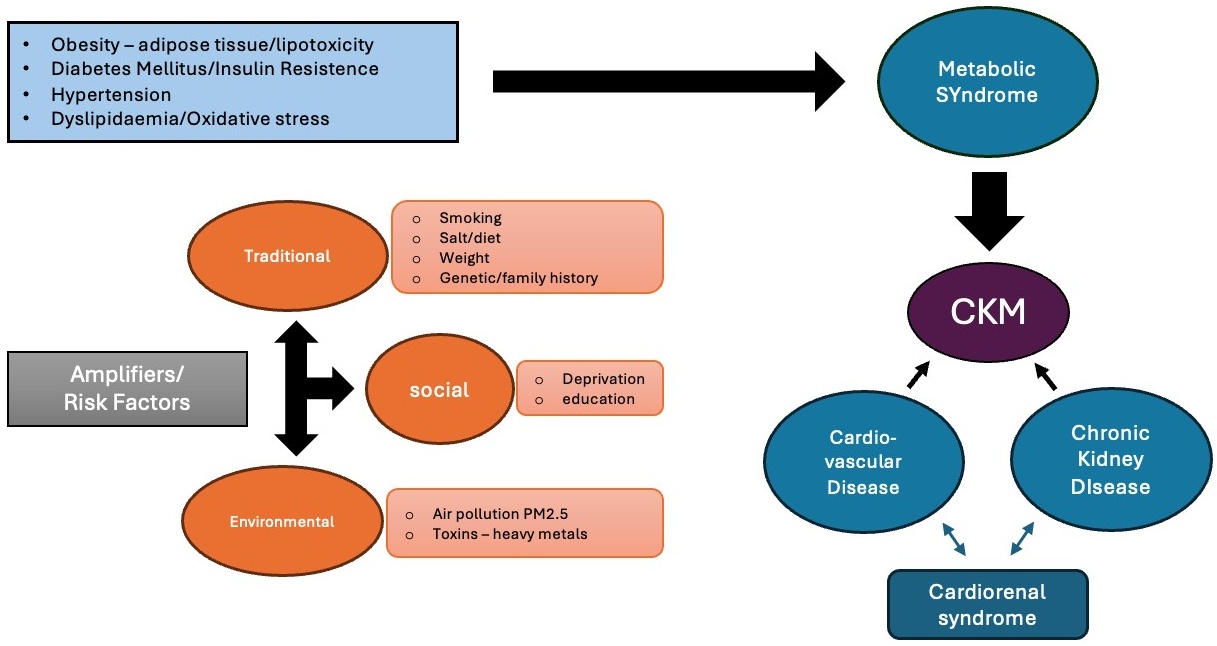 Fig. 1.
Fig. 1.
Metabolic risk factors contribute to the development of CKM syndrome. Traditional (smoking, diet, weight, family history), societal (deprivation, education), and environmental (air pollution, toxins) amplifiers further increase susceptibility. Within this framework, CVD and CKD are integral components that contribute to CKM, with cardiorenal syndrome highlighting the bidirectional interaction between the heart and kidney. CVD, cardiovascular disease; CKD, chronic kidney disease; CKM, Cardiovascular-Kidney-Metabolic; PM2.5, particulate matter 2.5. Microsoft 365 (Microsoft Corporation, 2024, Redmond, WA, USA).
| Stage | Label | Definition |
| 0 | No risk factors | No known CVD, kidney disease, or metabolic disease/risk factors. |
| 1 | Excess/dysfunctional adipose tissue | Excess body fat and/or unhealthy fat distribution and/or prediabetes/impaired glucose tolerance*. |
| 2 | Metabolic risk factors and CKD | Metabolic risk factors (e.g., diabetes, hypertension, hypertriglyceridaemia [ |
| 3 | Subclinical CVD in CKM | Early (asymptomatic) CVD in people with metabolic risk factors/CKD or high predicted CVD risk. |
| 4 | Clinical CVD in CKM (4a/4b)** | Symptomatic/clinical CVD in people with excess adiposity and/or metabolic risk factors and/or kidney disease. |
*BMI
**Stage 4a: no kidney failure & Stage 4b: kidney failure present.
CVD, cardiovascular disease; CKD, chronic kidney disease; CKM, Cardiovascular-Kidney-Metabolic; BMI, body mass index; HbA1c, hemoglobin A1c.
At the centre of CKM is excess, dysfunctional adipose tissue, especially visceral fat, driving insulin resistance, dyslipidaemia, and ectopic lipid deposition in the liver, heart, and kidney. This promotes chronic low-grade inflammation and oxidative stress, leading to endothelial dysfunction, microvascular damage, and progressive fibrosis in both myocardium and kidney parenchyma. In parallel, activation of the RAAS and sympathetic activation, amplified by obesity, hypertension, and nephron loss, causes vasoconstriction, sodium retention, and further inflammatory and oxidative injury [7, 8].
Disordered lipid metabolism is a key modifiable driver of CKM-related kidney progression, with accumulating evidence linking dyslipidaemia to worsening CKD. Its impact on cardiovascular outcomes is substantially altered as kidney function declines [7]. People with CKD typically develop atherogenic dyslipidaemia—high triglycerides, low HDL, and small, dense low-density lipoprotein (LDL)—that accelerates atherosclerosis and microvascular injury. Increasing attention has focused on triglyceride-rich remnant lipoproteins, including chylomicron remnants and very-low-density lipoprotein (VLDL) remnants. These particles are particularly atherogenic because they readily penetrate the arterial wall and deliver a high cholesterol load per particle to the intima, while also inducing endothelial dysfunction and inflammation. Consequently, remnant lipoproteins are strongly associated with coronary artery disease, ischaemic stroke, and residual cardiovascular risk beyond LDL. This may explain why cardiovascular disease, rather than kidney failure, is the leading cause of death in CKD [9]. Statins remain a cornerstone therapy in non-dialysis CKD and transplant populations, substantially reducing major vascular events and mortality even though their effect on eGFR decline is modest in advanced disease [9]. Beyond statins, Proprotein Convertase Subtilisin/Kexin Type 9 (PCSK9) inhibitors, such as Alirocumab and Evolocumab, provide substantial LDL reduction with proven cardiovascular benefit in high-risk groups [10, 11]. Lipoprotein(a) and triglyceride-rich lipoproteins are emerging therapeutic targets, with multiple phase 3 trials in progress [12]. At the severe end of CKM spectrum, fish oil has shown potential for reducing cardiovascular events in people on haemodialysis [13]. Within CKM, lipid-lowering should therefore be viewed as part of broader vascular protection, with potential benefits for both cardiovascular and renal outcomes.
Hypertensive nephropathy is currently the second leading cause of end-stage kidney disease (ESKD) after diabetes-related kidney disease. Hypertension is a strong and independent risk factor for CKD onset and progression, affecting up to 90% of people with CKD, and remains the leading modifiable contributor to premature mortality [14]. It is also a defining feature of CKM, emerging early as a metabolic risk factor (CKM stage 1–2) [6]. Hypertension in CKD is characteristically salt-sensitive, driven by tubular sodium retention, nephron loss, extracellular volume expansion, arterial stiffness, and maladaptive RAAS activation, often in the absence of overt oedema. Established hypertension perpetuates oxidative stress and relative kidney hypoxia, creating a self-reinforcing cycle of kidney injury and hypertension. Effective blood pressure control is crucial to slowing eGFR decline, delaying progression to ESKD, and reducing cardiovascular events.
Renin-angiotensin system inhibitors remain the first-line therapy in CKD with
hypertension and albuminuria (urinary albumin-to-creatinine ratio (uACR)
Mineralocorticoid receptor overactivation is now recognised as a key driver of cardiac and kidney fibrosis and chronic inflammation in CKD. Finerenone, a non-steroidal selective mineralocorticoid receptor antagonist (MRA), provides incremental benefit when added to ACEi/ARB therapy, reducing albuminuria, slowing CKD progression, and lowering heart-failure and atherosclerotic events in people with T2DM and CKD, with a more favourable safety profile than steroidal MRAs [16]. Emerging data suggest potential kidney-protective effects in non-diabetic CKD, although definitive outcome trials are ongoing [16]. Notably, the CONFIDENCE trial demonstrated greater uACR reduction with combined finerenone and SGLT2i compared with finerenone alone [17]. Aldosterone synthase inhibition has also shown promise, with phase 2 EASi-KIDNEY data demonstrating approximately 40% uACR reduction, irrespective of background SGLT2i use [8].
Endothelin receptor antagonists represent another emerging strategy for attenuating albuminuria and slowing eGFR decline in both diabetic and non-diabetic CKD. Recent randomised trials have demonstrated clinically meaningful efficacy with an acceptable safety profile, particularly when combined with SGLT2i therapy [18].
Chronic hyperglycaemia, obesity, and insulin resistance sit at the core of CKM and cause kidney injury long before eGFR begins to fall. Excess adiposity and insulin resistance increase tubular glucose and sodium reabsorption via upregulated sodium-glucose cotransporter-2 (SGLT2), which reduces sodium delivery to the macula densa, blunts tubuloglomerular feedback, and leads to glomerular hyperfiltration and glomerulomegaly. This “metabolic kidney” phenotype is already present in CKM stages 1 and 2. Over time, the combination of high flow, high pressure, oxidative stress, and pro-fibrotic signalling makes the kidney structurally vulnerable even when routine measures of kidney function still appear normal.
Therapeutically, this has driven a shift from glucose control alone to metabolic and organ protection. SGLT2 inhibitors are arguably the single biggest advance in delaying CKD progression in the past decade. Across multiple large trials, they consistently reduce composite kidney endpoints and slow eGFR decline in diabetic and non-diabetic CKD, with benefits largely independent of additional hemoglobin A1c (HbA1c) lowering [19].
Glucagon-like peptide-1 (GLP-1) receptor agonists provide complementary cardiovascular benefit, clinically meaningful weight loss, and anti-inflammatory effects, with consistent secondary evidence for reduced albuminuria and slower eGFR decline in T2DM and CKD [20]. These kidneys and cardiorenal benefits appear broadly consistent across semaglutide trials, where effects were most pronounced when combined with an SGLT2 inhibitor [20]. Similar benefits have been reported with oral semaglutide (SOUL) and the dual long-acting gastric inhibitory polypeptide (GIP)/GLP-1 receptor agonist tirzepatide (SUMMIT) [21, 22]. More recently, the triple GIP/GLP-1/glucagon receptor-agonist Retatrutide has demonstrated substantial weight loss and improved glycaemic control in T2DM, with a safety profile similar to established incretin-based therapies, although CKD-specific outcome data are not yet available [23].
Beyond incretin pathways, emerging mechanistic data implicate upregulation of renal tubular cannabinoid-1 (CB1) receptors and inducible nitric oxide synthase in obesity related CKD. In preclinical models, CB1 receptor antagonism attenuates kidney inflammation, fibrosis, oxidative stress, and structural injury, while improving metabolic parameters, highlighting a potential future therapeutic target [24].
The emerging evidence supports layered, multi-pathway therapy as the default
strategy for people with high-risk CKM, particularly those with CKD, where agents
targeting complementary pathways can achieve synergistic benefit. GLP-1 receptor
agonists primarily improve metabolic control, weight, and inflammation; SGLT2
inhibitors confer haemodynamic (blood pressure and intraglomerular pressure) and
metabolic benefits, including reductions in intraglomerular pressure; and
finerenone attenuates haemodynamic stress, fibrosis, and inflammation through
mineralocorticoid receptor blockade [3]. Modelling studies and network
meta-analyses consistently demonstrate that combining these classes yields
greater reductions in major cardiovascular events, CKD progression, and all-cause
mortality than monotherapy or dual regimens, translating into meaningful gains in
event-free survival for people with T2DM and albuminuria [25]. In this
context, modest improvements in eGFR slope or uACR should no longer be considered
sufficient when rational combination therapy can deliver substantially greater
kidney protection and, in some patients, near-remission trajectories -
particularly in those with non-severe albuminuria (uACR
Current NICE guidance recommends screening for serum creatinine and uACR in adults with diabetes, hypertension, cardiovascular disease, or previous acute kidney injury (AKI) [26], while KDIGO adopts a broader risk-based approach, extending screening to multisystem and genitourinary disorders [3]. AHA CKM framework further broadens eligibility by defining CKM stages (1–2) on the basis of metabolic risk, hypertension, and cardiovascular disease, with routine assessment of kidney function and albuminuria embedded within risk evaluation [27]. Building on this, the CKM Predicting Risk of Cardiovascular Disease EVENTs (PREVENT) score incorporates eGFR, and, where available uACR, alongside traditional cardiovascular risk factors to predict total cardiovascular disease, including heart failure [28]. Use of risk tools dependent on eGFR and uACR is therefore likely to promote earlier and more widespread testing, enabling more accurate CKD stratification and timely initiation of intensified, multi-pathway therapy. Importantly, accumulating evidence supports the cost-effectiveness of CKD screening in high-risk groups and the general population from around age 55 years of age, particularly when linked to improved uptake of SGLT2 inhibitors, which remains suboptimal in routine practice [29].
Closing the implementation gap in cardiorenal-metabolic medicine is now the key challenge. By adopting a CKM framework, health systems can expand CKD screening, normalise earlier and combined use of kidney–heart–protective therapies, and embed collaborative care models that translate strong trial evidence into real-world benefit. In primary care, this could include automated prompts for eGFR and uACR testing in people with CKM risk factors, integration of CKM-adapted risk scores (such as PREVENT) into electronic health records, and nurse- or pharmacist- led reviews to optimise RAASi, SGLT2i, glucagon-like peptide-1 receptor agonist (GLP-1RA), and finerenone use.
• CKD, CVD, and type 2 diabetes form a unified CKM syndrome driven by shared pathophysiology.
• CKM staging highlights early disease (including albuminuria), life-course risk, and roles of adiposity and oxidative stress.
• Layered therapy (RAASi, SGLT2i, GLP-1RA, MRAs) enables greater albuminuria reduction and disease modification.
• CKM supports earlier, risk-based CKD screening (eGFR, uACR) in high-risk populations.
Not applicable.
Conceptualisation: RMD, SS, RD, SB; literature review and drafting: RMD, SS, RD. All authors contributed to the important editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
SS has received honoraria from Pharmacosmos. SB has received honoraria from Vifor CSL, MEDICE, Bayer, Boehringer Ingelheim, Pharmacosmos, GSK and Astellas for lectures. RMD and RD declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

