1 Division of Medicine, University of Nottingham, NG5 1PB Nottingham, UK
2 Department of Respiratory Medicine, Nottingham City Hospital, Nottingham University Hospitals NHS Trust, NG5 1PB Nottingham, UK
Abstract
Randomised controlled trials have shown that early detection of lung cancer by low-dose computed tomography (LDCT) reduces lung cancer and all-cause mortality. This, and a detailed health economics evaluation, led to a recommendation in September 2022 by the UK National Screening Committee (UKNSC) that all four UK nations move towards implementation of a targeted lung cancer screening programme. The National Health Service England (NHSE) Targeted Lung Health Check (TLHC) programme launched in 2019 and has been adopted as the national screening programme in England with national rollout expected to be complete in 2030. Given the complexities of lung cancer screening, a comprehensive protocol and linked quality assurance standard were developed to provide a mechanism to ensure uniform and regulated implementation of a high-quality programme. This review will describe the importance of quality assurance and show how this works in a national programme.
Keywords
- lung cancer
- screening
- quality assurance
Lung cancer is the leading cause of cancer-related deaths in the UK and
worldwide. Two-thirds of people with lung cancer are diagnosed at stage 3 and 4
where there is a low chance of cure, and the five-year survival is
Following a recommendation in September 2022 from the UK National Screening Committee (UKNSC), National Health Service England (NHSE) [4] is implementing a targeted lung cancer screening (LCS) programme which began as a pilot, the “Targeted Lung Health Check” (TLHC) in 2019. National rollout is expected to be completed by 2030 across England. Five years into the programme, over 2.5 million people have been invited from a total eligible population of approximately 7.5 million. There have been over 500,000 participants, and more than 7000 lung cancers found with 76% at stage 1 and 2 [5]. The Department of Health and Social Care [6] estimate that, every year, once fully implemented, the programme will cost £270 million, will deliver almost one million scans and detect lung cancer in 9000 people. In February 2025, the TLHC was renamed the national lung cancer screening programme (LCSP) in England. Remarkably, the stage 1 and 2 lung cancer detection rate in the most socioeconomically deprived quintile for all lung cancers has moved from the bottom to the top [5]. The LCSP may be the first screening programme to demonstrate a reduction in health inequalities. This was achieved with approximately 32% coverage of the invitation to screening assessment and 49% uptake of the offer. Of the 49%, 47.5% met the risk threshold for screening and 91% underwent LDCT [5].
For screening programmes to be effective, they must ensure a favourable balance between benefit and harm. This is achieved through governance and quality assurance (QA). This review will consider the processes and QA standards that are critical to the design, implementation, safety and functioning of the National Health Service (NHS) LCSP in the UK.
Quality standards encompass the entire pathway, from identification of an eligible population to treatment and follow-up. Translating these standards into good screening programmes involves a multi-step approach that aligns established national guidelines, ensures consistency and supports continuous audit and quality improvement to ensure safe, effective and equitable delivery of screening that maximises benefits of screening and mitigates harm.
A clear description of what should occur at each step of the screening pathway will improve consistency across the programme. To ensure a standardised approach, NHSE developed an evidence-based standard protocol and quality assurance framework in 2019; this has since been updated [7, 8]. The latter encompasses 15 quality standards for the LCSP that aim to ensure that the structure and functioning of the pathway supports high quality. An example of the participant pathway in the LCSP is shown in Fig. 1. Each standard clearly outlines the objective, definition and metric, and the local and national assurance and audit process to demonstrate that each standard is being met. They apply to areas including certification, experience and training of workforce, technical infrastructure, imaging protocols and reporting, and onward referral for management of screen-detected findings. Effectiveness standards, which measure performance metrics have been developed but not yet published by NHSE.
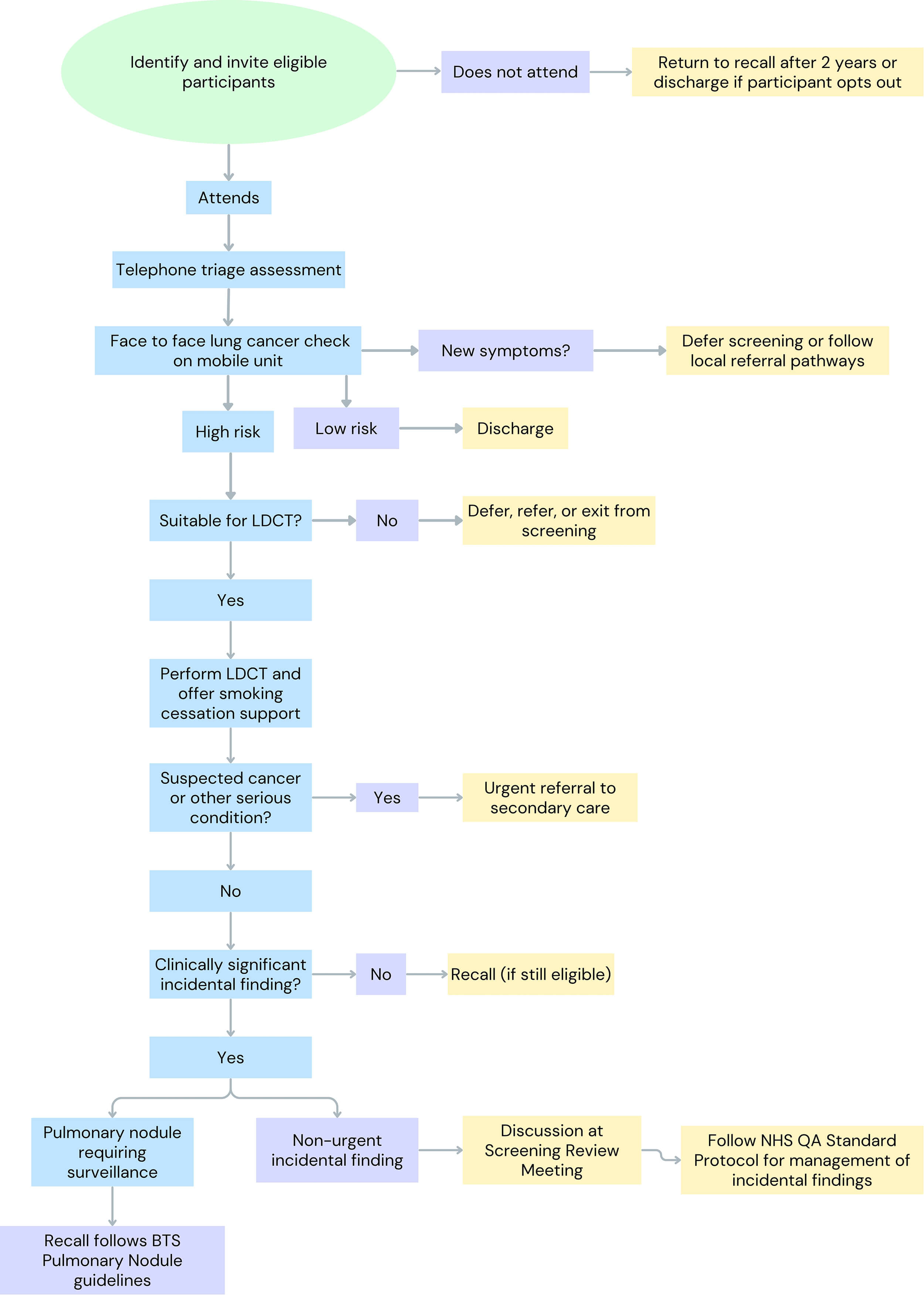 Fig. 1.
Fig. 1.
Example pathway in the National Health Service (NHS) lung cancer screening programme (LCSP). LDCT, low-dose computed tomography; BTS, British Thoracic Society; NHS, National Health Service; QA, quality assurance. Created by Canva (https://www.canva.com/).
There must be accountability for overall quality and performance of the LCSP programme. In the UK, the clinical director of each local site will report data back to NHSE through the LCSP Delivery Group on a quarterly basis [7]. The Screening Quality Assurance Service (SQAS) is responsible for monitoring compliance of the LCSP to national quality assurance standards, supporting services with quality improvement and undertaking quality assurance activities including site visits [9]. SQAS will assess data integrity and regularly audit performance indicators (cancer detection rates, patient outcomes, false positive/negative rates) to ensure areas requiring improvement are addressed promptly and inform policymakers and other stakeholders about the overall effectiveness of the programme. It will work closely with local sites to identify areas for improvement, ranging from eligibility criteria, communication methods, or improving diagnostic accuracy. SQAS also plays a vital role in supporting staff training and development to maintain high standards.
Involvement of stakeholders including healthcare professionals, patient advocates and policymakers will ensure that the quality standards remain relevant and are aligned with the best evidence. Participant feedback is also encouraged in QA efforts to ensure ongoing acceptability of the programme and improve participant experience.
Establishing clear leadership within governance structures will provide mechanisms to support high-quality, effective and safe delivery. The complexity of screening programmes necessitates multiple groups and individuals to work together to provide oversight to the entire screening pathway in administrative, financial and clinical matters.
There is a vast number of different stakeholders involved in the LCSP. Engaging this multidisciplinary group can identify barriers to implementation that need to be addressed in early phases and thus guide programme development. For example, both primary care and secondary care providers in the UK raised valid concerns about increased workload and subsequent strain on resources associated with implementation of the LCSP, particularly regarding management of incidental findings [10]. This highlights the importance of clear processes and sufficient resource allocation to manage onward referrals. Time is allocated locally for screening review meetings (SRMs), where a multidisciplinary team (MDT)—typically comprising the responsible radiologist, responsible clinician, responsible assessor and the LCSP coordinator—follow the LCSP QA standard protocol for management of incidental findings (with some agreed local pathway modifications) [7]. There is no requirement for case finding by primary care as all invitations are managed by a third party. Lesions suspicious for lung cancer or urgent incidental findings are directly referred. Primary care however does support with promotion of the programme and there will inevitably be some administrative burden from adding outcome letters into patient notes.
Key roles and responsibilities of those involved in clinical governance of the LCSP are outlined in Table 1.
| Role | Responsibility |
| UK National Screening Committee (UKNSC) | Evaluates clinical and cost effectiveness. |
| Makes national recommendations. | |
| NHS England (NHSE) | National oversight of LCSP. |
| Develops the standard protocol. | |
| Ensure programmes aligns with national screening policies. | |
| Coordinates funding. | |
| Monitors effectiveness. | |
| NHS Improvement (NHSI) | Supports implementation and drives continuous improvement. |
| Public Health England (PHE) | Provides guidance on public health approaches and engagement strategies, including community outreach. |
| Ensures equitable access to screening. | |
| Integrated Care Boards (previously clinical commissioning groups, CCGs) | Ensure commissioning effective at regional level. |
| Ensure programme is tailored to meet requirements of local population and improving equitable access. | |
| Cancer Alliance Board | Translates recommendations for screening, national cancer plans into national screening programme. |
| Tracks key performance indicators. | |
| Local LCSP Steering Committee | Oversight of the local programme ensuring adherence to the standard protocol. |
| Chaired by clinical director of programme. | |
| Made up of responsible assessors, radiologists and clinicians as well as other stakeholders. | |
| Clinical Director of Programme | Overall clinical oversight and responsibility for local delivery including adherence to the standard protocol and quality assurance protocol. |
| Reports to NHSE through the Cancer Alliance Board. | |
| Ensures data is available for inclusion in national audit. | |
| Responsible Radiologist | Responsible for adherence of radiology team to standard protocol. |
| Responsible Clinician | Responsible for adherence of the clinical team managing screen-detected findings. |
| Responsible Assessor | Responsible for ensuring the correct selection and recruitment processes. |
All individuals involved in the pathway will require specialised training and continuous education in lung cancer detection and screening protocols to maintain diagnostic accuracy. The LCSP standard protocol, QA standard and pan-European standard describe the minimum requirements.
Administration or nursing staff involved in the pathway must receive training in communication covering the triage process, call quality expectations and control measures, including awareness of red flag symptoms. Training includes consent and ionising radiation regulations. Telephone triage assessments are audited locally on a quarterly basis and fed back nationally by the clinical director of the site.
The British Society of Thoracic Imaging (BSTI) requires radiologists who report
LDCT as part of the LCSP to attend a lung nodule workshop as a minimum. The LCSP standard protocol recommends high volumes of computed tomography (CT) readings (
Specific requirements for CT equipment, software, image acquisition and reporting are described in the LCSP standard protocol [8]. Medical physics audit effective radiation doses locally and feedback through the quarterly QA process to ensure safety.
Tools recommended in screening include computer-aided detection (CAD) of pulmonary nodules and semi-automated measurement of nodule volume. Where volumetric software is updated or changed, processes should be in place to confirm that there is no change in calibration, or the user should be prompted to re-measure previous nodules.
The volume and complexity of data involved in LCS requires a suitable and robust data management system (DMS) that facilitates electronic health record integration, high-quality data collection, data sharing and communication across sites. This is essential to support quality assurance. Nationally specified data items are required in a standardised format across all sites [7]. A successful DMS will also facilitate programme management such as automatic appointment scheduling and recall.
In the LCSP, individual sites collect data from local electronic health records to identify eligible individuals but the approach to information technology is highly variable. Many sites have used basic spreadsheets for data collection which restricts the ability to access real-time data and track programme performance effectively. The NHS LCSP QA standard suggests a minimum mandatory dataset to allow monitoring of key performance indicators [7]. Data from the LCSP are submitted to the National Cancer Registration and Analysis Service (NCRAS), which registers all cancer diagnoses in England. Establishing a single, safe, end-to-end, national DMS with standardised protocols is considered a priority to optimise consistency and standardisation.
All data handling must adhere to information governance and General Data Protection Regulation (GDPR) guidelines [12]. Data sharing agreements must be agreed with data pseudonymised before submission.
Optimal delivery of LCS requires clear eligibility criteria that are targeted to those at highest risk for lung cancer or those with highest predicted screening-derived gain in life years. This approach improves efficiency and cost-effectiveness of the programme by directing resources to those most likely to benefit from early detection and minimises physical and psychological harm in those who do not develop cancer. For example, an analysis of NLST data that 88% of the lung cancer-specific mortality benefit accrued to the 60% of participants at highest risk of lung cancer death [13]. There is inevitably a risk that a proportion of lung cancers will be missed in individuals who do not meet eligibility criteria, but that is the case for all screening programmes.
Eligibility criteria can be stratified by age and cumulative smoking exposure or by using multivariable risk models that incorporate a broader range of risk factors. Age and smoking history alone risk oversimplifying risk prediction by excluding important risk factors, such as smoking intensity, personal history of lung disease, asbestos exposure and family history of lung cancer.
In the UK, these multivariable risk prediction models are already integrated
into eligibility assessment. Specifically, individuals aged 55 to 74 who have
ever smoked and are registered with a General Practice are invited to participate
in the LCSP programme. During the initial assessment, their risk of developing
lung cancer is calculated using two externally validated multivariable models.
Participants that reach a minimum risk threshold of developing lung cancer of
| Liverpool Lung Project version 2 (LLPv2) | Prostate, Lung, Colorectal, and Ovarian 2012 model (PLCOM2012) | |
| Variables | • Age | • Age |
| • Gender | • Education level | |
| • Smoking duration (years) | • Body mass index | |
| • Previous pneumonia/chronic obstructive pulmonary disease (COPD)/emphysema/bronchitis/tuber-culosis (TB) | • COPD/chronic bronchitis/emphysema • Personal history of lung cancer • Family history of lung cancer • Ethnicity • Smoking status • Average number of cigarettes per day • Duration smoked (years) • Years having stopped smoking | |
| • Occupational asbestos exposure | ||
| • Previous history of malignancy | ||
| • Previous family history of lung cancer (relative’s age at onset, i.e., |
||
| Minimum risk threshold |
Adapted from the NHS QA standard protocol [8].
Multivariable risk prediction models improve pre-test probability and effectiveness of screening, whilst reducing ethnicity and sex-based inequalities by comparison with age and smoking history alone as used by the US preventive services task force [16].
Robbins et al. [17] have shown that the PLCOM2012 and LLPv2 had only moderate discrimination and were not well calibrated once externally validated in the English primary care population. Several new models have been published. The CanPredict (lung) model for example has shown higher sensitivity than PLCOM2012 and LLPv2 in the UK primary care population and may provide a single-step alternative, further improving efficiency and cost-effectiveness [18]. However, this was designed to be used on primary care data whereas PLCOM2012 and LLPv2 on data directly collected from participants. Further research is required to define the optimal risk model and ensure it is validated for the target population demographics where risk factors may differ and whether these will out-perform the current models recommended in the LCSP standard protocol.
In many countries there is no record of individual smoking habit but developing such a record would have potential benefit in many health interventions, including selection for LCS. Digital health “apps”, such as the NHS Health app, improved data entry forms and alerts may prove helpful.
There is concern that multivariable risk models will select individuals with significant comorbidity. Fitness must be assessed and reassessed during the pathway to ensure participants have a reasonable chance to benefit from early detection of lung cancer and therefore ongoing participation in screening.
Current research is exploring evidence for integration and validation of biomarkers into risk prediction tools to improve early detection and personalise screening intervals [19, 20, 21]. Novel approaches involving machine learning are also in development [22, 23]. Any developments to risk prediction models will need to be validated and integrated into UK primary care software.
LCSPs must be designed to maximise recruitment, participation and adherence of individuals deemed highest risk of lung cancer to improve clinical and cost-effectiveness. Across England, LCSP uptake rates have increased from approximately 40% to nearly 60% (unpublished data from NHSE). National and regional awareness initiatives, supported by charities such as the Roy Castle Lung Cancer Foundation, have aimed to increase public knowledge and engagement.
Several studies and pilot programmes have seen reduced uptake in people who currently smoke and socioeconomically deprived groups, despite them being at highest risk of lung cancer [24, 25]. Older age and female sex were also associated with lower uptake rates [24].
Barriers to participation are driven by a complex interplay of psychological, social, and practical barriers. To ensure LCSPs effectively reduce mortality across high-risk groups, it is imperative to engage underserved groups equitably. Participants in the NLST and NELSON trials were generally younger, better educated, and less likely to be current smokers compared to the broader population eligible for screening or non-responders, which may limit the generalisability of results and potential for equitable benefit [2, 26]. This selection bias underscores the need for targeted strategies to reach and include high-risk individuals who may otherwise be underrepresented in screening initiatives.
Psychological barriers include fatalism, fear of a cancer diagnosis, stigma related to smoking, guilt, misunderstanding and low perceived efficacy of treatment, which also lead to reduced uptake and adherence to screening [27]. Social determinants such as lower health literacy and limited access to healthcare services also contribute to disengagement [28].
Targeted interventions to overcome these challenges include clear and comprehensive communication on potential risks (false positives, unnecessary procedures, overdiagnosis) and benefits are provided in standard letters and participant booklets to those eligible [29]. These are provided at every stage of the pathway so that participants feel adequately informed and supported in a shared decision-making model. Integrating patient navigators who provide “end-to-end” support and can empower individuals to adhere to the programme [30]. Outcome letters from lung health checks or LDCT should be communicated to participants and among healthcare professionals promptly to reduce fear and anxiety associated with LCS and allow timely management of findings [29, 31]. The responsible assessor at each site will audit communication methods to ensure they meet the standard.
Numerous practical barriers to screening have also been described including travel (physical distance, access), employment, costs of screening, comorbidity and other commitments [32, 33]. To overcome these barriers, the NHS LCSP offers a hybrid approach with some centralised screening units, as well as decentralised units to enhance population coverage. Mobile CT scanners for LCS are a common delivery method in England that can provide a service in convenient locations such as shopping centre and supermarket car parks.
Feedback should be encouraged from programme non-attendees to improve uptake in those eligible. Engaging patients and public, particularly in underserved communities, is vital to improving methods that improve equitable access to screening for all eligible individuals and avoid disparities in care.
Careful consideration is required to ensure there is sufficient workforce capacity to deliver LCS. Primary roles involved in the screening programme are vast, including but not limited to community health workers, primary care physicians and nurses, smoking cessation advisers, respiratory physicians, radiologists, and patient navigators.
National LCS will lead to increase in resource demands across all involved in the screening pathway. The Royal College of Radiologists [34] reported a 30% shortage of clinical radiology consultants in the UK, which may rise to 40% by 2028 without immediate action. Such shortages threaten the scalability and sustainability of national screening programmes. Implementation of artificial intelligence solutions and standardised reporting platforms may mitigate some burden by reducing radiologist workload, alongside robust validation and oversight mechanisms. Respiratory physician demand is substantial given involvement in screening review and investigation of screen-detected findings. This can be helped through streamlining administrative staff support, including automated scheduling and follow-up systems, to reduce time burden on clinicians. Standardised communication and guideline-driven management will also allow for efficient triage and consistency in management decisions. Table 3 (Ref. [7]) shows suggested management of incidental findings recommended in the LCSP QA standard protocol. With earlier detection of lung cancer, surgical demand will also increase. In the US, it is estimated that there will be 37% more lung cancer surgeries in 2015–2040 compared to no screening [35]. The design of any programme must consider workforce capacity limitations to maintain clinical effectiveness and optimise deliverability and national consideration to investment in workforce training and retention.
| Finding | Reporting recommendation | Suggested action if baseline or new |
| Pulmonary | ||
| Emphysema | Classify as: | Inform participants with moderate to severe radiological emphysema about the findings and recommend they seek advice from primary care if they have symptoms. |
| Mild ( |
Smoking cessation for all current smokers | |
| Moderate (25–50%) | ||
| Severe ( |
||
| Bronchiectasis | Classify as: | No action if mild/borderline |
Mild (airways are 1.5–2 Moderate (2–3 |
If moderate/severe, refer for specialist review if chronic cough or recurrent LRTI documented at health check | |
| Severe ( |
Notify individual and GP regarding standard bronchiectasis/infection prophylaxis management and give option of referral | |
| Bronchial wall thickening | Do not report | None |
| Respiratory bronchiolitis-associated interstitial lung disease (RB-ILD) | Do not report | None |
| Interstitial lung abnormalities (ILAs) | Report all ILA and recommend: | |
| If |
Consider referral if | |
| Consolidation | Classify as: | Refer to lung cancer service if cancer possible |
| - Likely inflammatory | Interval CT at 6 weeks or 3 months if appears inflammatory | |
| - Possibly malignant | Assess for clinical infection and prescribe antibiotics as required | |
| Tuberculosis (TB) | Indicate if likely to be TB and include differential diagnosis | Refer into TB service |
| Pleural effusions/thickening | Report size and evidence of malignant features | Discuss at screening review meeting or refer lung cancer service if suspicious appearances |
| Extrapulmonary | ||
| Mediastinal mass | Report size, position and whether cystic | Cystic lesions do not require further investigation |
| Review at lung cancer MDT or screening review MDT to consider management options including surveillance as part of screening programme or work-up with positron emission tomography (PET)/CT/magnetic resonance imaging (MRI) | ||
| Coronary artery calcification (CAC) | Report (based on visual estimation of most affected artery) as: | Clinical assessment of cardiovascular risk if moderate or severe CAC |
| - None | Primary prevention measures (if not already in place) | |
- Mild - Moderate - Severe |
Local agreement as to whether assessment included in lung health check appointment | |
| Aortic valve disease | Report if moderate or severe calcification involving 2 or 3 cusps | Primary care to refer for echocardiogram if moderate or severe and not known to have aortic valve disease |
| Aortic aneurysms | Thoracic: | As per reporting recommendation |
| Report dilation of |
||
| Abdominal: | ||
| 3–5 cm: to refer | ||
| Breast nodules | Specify site and size | Refer for breast MDT |
| Liver lesions | Classify as malignant, indeterminant, benign or incompletely imaged/unable to evaluate | Refer malignant lesions to appropriate local cancer pathway |
| Indeterminate lesions can be reviewed at screening review meeting | ||
| Renal lesions | Classify as malignant, indeterminant, benign or incompletely imaged/unable to evaluate | Refer malignant lesions to appropriate local cancer pathway |
| Indeterminate lesions can be reviewed at screening review meeting | ||
| Bone abnormalities | GP to refer for bone density evaluation for |
Recommend bone risk assessment and protection |
| Thyroid abnormalities | Report only if local lymphadenopathy and/or punctate calcification | Refer to thyroid MDT |
| Adrenal lesions | Report size and attenuation | |
| 1–4 cm and | ||
Adapted from NHS LCSP quality assurance standards [7]. HU, hounsfield units, SRM, Screening Review Meeting; ILD, interstitial lung disease; GP, general practitioner; CT, computed tomography; MDT, multidisciplinary team; LRTI, lower respiratory tract infection.
Radiologists that report LDCT for the NHS LCSP use a LDCT reporting proforma to ensure standardisation. In the UK, clinical management of pulmonary nodules should follow the British Thoracic Society (BTS) 2015 pulmonary nodule guidelines [36] and will not be addressed in this review. An update to these guidelines is expected in 2025–2026. Many nodule services in the UK run as “virtual” nodule clinics, with standardised information and communication.
Over two-thirds of individuals screened with LDCT for lung cancer have “incidental findings” [37]. The majority are coronary artery calcification and/or emphysema. Management of these findings risks physical harm from unnecessary, potentially invasive, investigations and psychological harm such as fear and anxiety. There is a concern that incidental findings will place a significant burden on the NHS. This may be mitigated by provision of clear and robust guidelines and protocols to standardise management and reduce the need for onward referrals. The NHS LCSP quality assurance standard outlines management of the most common incidental findings [7].
Decisions on management should be made by a multidisciplinary team, including a radiologist, respiratory physician, nurse and coordinator, as this improves adherence to guidelines [38]. Patient preference should be considered, where appropriate. Evidence shows that an MDT approach to management reduces the number of findings requiring further action. Adherence to national guidelines, e.g., BTS, National Institute for Health and Care Excellence (NICE) and LCSP will help minimise cumulative radiation exposure by determining optimal screening intervals while maintaining best practice.
The overall clinical and cost-effectiveness of LCS can be improved by integration of evidence-based smoking cessation interventions. Evidence from trials and pilot programmes show that up to 55% of participants identify as current smokers, vs 15–20% in the general population aged 55–80 years. These studies have further shown that people who smoke are more likely to be from lower socioeconomic status groups and worry about stigma associated with their smoking status [39]. The UK Lung Cancer Screening (UKLS) trial demonstrated higher smoking cessation rates in screened individuals, particularly those receiving an abnormal result [40]. LCS therefore presents a “teachable moment”, where people who smoke may be more receptive to smoking cessation advice and support [41]. Individuals who are eligible for LCS and who smoke often have multiple smoking-related diseases such as chronic obstructive pulmonary disease (COPD) and ischaemic heart disease [42]. Smoking cessation interventions can therefore maximise reduction in morbidity and mortality from lung cancer and smoking-related comorbidities in this high-risk group [42, 43].
The LCSP standard protocol recommends that where possible, smoking cessation services are offered on-site and involve an opt-out referral to local smoking cessation services, and treatment, e.g., nicotine replacement therapy [8]. The Yorkshire Enhanced Stop Smoking study (YESS) showed that for services available on the mobile screening unit, provided on an opt-out basis, 89% of eligible people saw a smoking cessation practitioner and 75% agreed to ongoing support. Confirmed quit rates at 1 year were around 30% with this approach [44]. There is strong evidence for intensive intervention with multiple counselling sessions and provision of pharmacotherapy [45].
In areas with local authority budget constraints, smoking cessation interventions may be less comprehensive, with poorer access to pharmacotherapy and follow-up which will reduce the overall effectiveness of LCSP in smoking cessation. The LCSP will inevitably identify more and more smokers and without sufficient and sustained funding, the services will have increasing difficulty to meet the demand. Provision will therefore be based on various local factors, such as availability, available workforce and training and costs [46].
The figures relating to the English lung cancer screening programme used in this review reflect the most up-to-date published data. However, unpublished data show that the programme has now detected over 10,000 cases and participation rates are increasing. As with any ongoing programme, further analysis will be dependent on reporting according to defined performance standards. The Rapid Cancer Registration dataset has provided almost real-time information about cancer but this fact also means that validation of data is less assured. This is described in detail on the Rapid Cancer Registration Data (RCRD) website. Nevertheless, the large scale means that trend analyses are likely to be accurate, and are useful to examine possible population impacts of the programme.
Like other screening programmes, LCS has to be organised in such a way as to maximise the clinical and cost effectiveness whilst ensuring high levels of equitable coverage of invitations and participation. The English model, developed from the TLHC (now the English national LCSP) demonstrates that this is achievable. The key to success is the use of a single, nationally mandated, standardised protocol supported by quality assurance standards and an effective governance and implementation structure—ensuring consistency across all sites. Compared to the United States, where implementation has been largely decentralised and uptake remains variable, the UK benefits from its integrated health system and ability to standardise delivery. Most other European countries are pursuing evaluations, smaller pilots and research studies. Those that currently have national programmes (Croatia and Poland) are much smaller that the UK programme which stands out for rapidly scaling up a population-level programme with embedded quality assurance. This centralised, protocol-driven structure is key to its success and may serve as a model for other nations seeking to balance effectiveness, equity and operational feasibility. The English programme has demonstrated that this approach works at a national level with impacts seen on early-stage lung cancer detection and a likely reduction in health inequalities.
• Quality assurance ensures lung cancer screening programmes are delivered consistently across sites, following evidence-based standardized protocols, for eligibility criteria, scanning procedures, and reporting methods.
• Strong clinical governance structures oversee the delivery of lung cancer screening programmes, with leadership responsible for setting quality standards, ensuring accountability, and supporting continuous quality improvement.
• QA involves auditing the accuracy and consistency of CT interpretation, radiologist performance, image quality, and use of standardised reporting frameworks.
• All personnel involved must be trained to national standards, with ongoing competency assessments to maintain high-quality delivery.
• Robust systems are necessary to track key performance indicators, enabling benchmarking and identification of areas for service improvement.
• QA ensures that services are accessible, safe and improve equity by identifying and addressing barriers to access so that underserved and high-risk populations are effectively reached.
Not applicable.
AB: Conceptualisation of the review; Critical interpretation of the literature; Writing—original draft preparation. ELOD: Conceptualisation of the review; Critical interpretation of the literature. DRB: Conceptualisation of the review; Critical interpretation of the literature. All authors contributed to the important editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
AB declares no conflicts of interest. ELOD declares receipt of grants from National Institute for Health and Care Research (NIHR), Horizon Europe, Cancer Research UK (CRUK) and Roy Castle Lung Cancer Foundation. DRB declares honoraria for speaking from Astra Zeneca, Roche and Merck Sharp Dohme (MSD) and receipt of grants to his institution from NIHR, Horizon Europe, Small Business Research Initiative (SBRI), Innovate UK and CRUK. Fig. 1 was created using Canva. The authors have no financial or personal relationship with Canva, and the use of this tool does not imply any endorsement.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

