1 Department of Neurology, Sir Run Run Shaw Hospital, Zhejiang University School of Medicine, 310016 Hangzhou, Zhejiang, China
Abstract
It remains unclear whether vascular characteristics or hemodynamics of the vertebrobasilar artery (VBA) among patients with left- and right-sided hemifacial spasm (HFS) lesions correlate with the risk of HFS occurrence and disease severity. This study aims to investigate the correlation of VBA characteristics with the incidence risk and severity of HFS.
A total of 60 patients with HFS who were admitted to Sir Run Run Shaw Hospital, Zhejiang University School of Medicine, between January and October 2024 were retrospectively enrolled in the HFS group. Another 59 healthy individuals who underwent physical examinations were assigned to the control group. Data on the characteristics of the VBA were collected for both groups, and the correlation between these characteristics and the occurrence risk of HFS was analyzed. In addition, patients in the HFS group were further stratified according to the lesion side (left vs right) and HFS severity grade (mild vs moderate vs severe) for subgroup analysis and comparison of VBA imaging features.
The diameters of the left vertebral artery (VA), right VA, and basilar artery in the HFS group were all found to be larger than those in the control group, and the difference in the diameter of the left VA between the two groups was more significant (p < 0.05). Multivariate logistic regression analysis demonstrated that the diameter of the left VA (odds ratio [OR] = 4.014, 95% confidence interval [CI]: 1.997–8.070), the diameter of the right VA (OR = 3.890, 95% CI: 2.217–6.825), and the diameter of the basilar artery (OR = 2.022, 95% CI: 1.008–4.058) were independent influencing factors for the occurrence of HFS (p < 0.05). There were no statistically significant differences in the diameters of the VA and basilar artery among patients with severe, moderate, and mild HFS (p > 0.05).
The diameters of the left VA, right VA, and basilar artery are significantly correlated with the occurrence of HFS, while the severity of the disease shows no correlation with the diameter of the ipsilateral VA. This suggests that the morphological changes of the VBA system may play an important role in the pathogenesis of HFS.
Keywords
- hemifacial spasm
- basilar artery
- vertebral artery
- disease
Hemifacial spasm (HFS) is a neurological disorder of the brain characterized by involuntary, paroxysmal twitching of the facial muscles, mostly confined to one side, usually starting from the orbicularis oculi muscle and gradually progressing downward to affect even the lower facial muscles, which is more common in middle-aged and elderly people [1]. The prevalence of HFS in the population is about 20/100,000, with a slightly higher prevalence in women than in men. Its diagnosis is mainly based on observation of the characteristic clinical manifestations, such as difficulty opening eyes, facial asymmetry, and twitching noises in the ear, etc. Electrophysiological approach, imaging examinations, and carbamazepine tests are required to facilitate diagnosis for individuals lacking these manifestations. Drugs such as carbamazepine and oxcarbazepine, botulinum toxin injections, and microvascular decompression are the primary modes of treatment for HFS. Vascular compression of the pontine segment of the facial nerve is a major etiological factor in the development of HFS [2], with the vertebrobasilar artery (VBA) being a common implicated factor in clinical practice [3]. However, the VBA is bilaterally asymmetrical, with the left vertebral artery (VA) being the dominant vessel [4]; this dominance is more pronounced in patients with HFS [5]. The bilateral asymmetry of the VBA and the presence of the dominant VBA contribute to the differences in hemodynamics of the bilateral VBA, which, in turn, may cause the VBA to shift or twist to the side with lower blood flow [6]. Fluid dynamics-based analysis has also found that bending of the VBA triggers a pressure difference between the two sides of the vessel wall and increases the wall shear stress, which increases with the angle and degree of bending [7]. A clinical anatomical study demonstrated that the pattern of vessel displacement is consistent with biomechanical characteristics [8]. It is suggested that anatomical variations and hemodynamics differences in the VBA system may contribute to the development of HFS, vascular variations or hemodynamic alterations in the VBA may differ between patients with unilateral and bilateral HFS. However, it is not clear whether vascular variations or hemodynamic changes in the VBA of patients with left- and right-sided HFS are associated with the risk of developing HFS or, in fact, with disease severity. In view of this, 60 patients with HFS were retrospectively included in this study to investigate the correlation of VBA characteristics with the risk of developing HFS and disease severity.
This study was conducted in adherence to the Declaration of Helsinki, and the
research protocol was approved by the Medical Ethics Committee of Sir Run Run
Shaw Hospital, Zhejiang University School of Medicine (Ethics Approval No.:
2025-1076). This retrospective study used fully anonymized data and posed no risk
to patient confidentiality and safety. Therefore, informed consent was waived in
accordance with the “Ethical Review Measures for Biomedical Research Involving
Human Subjects”. A total of 60 patients with HFS treated at Sir Run Run Shaw
Hospital, Zhejiang University School of Medicine, from January 2024 to October
2024 were retrospectively enrolled. The sample size was calculated using the
formula:
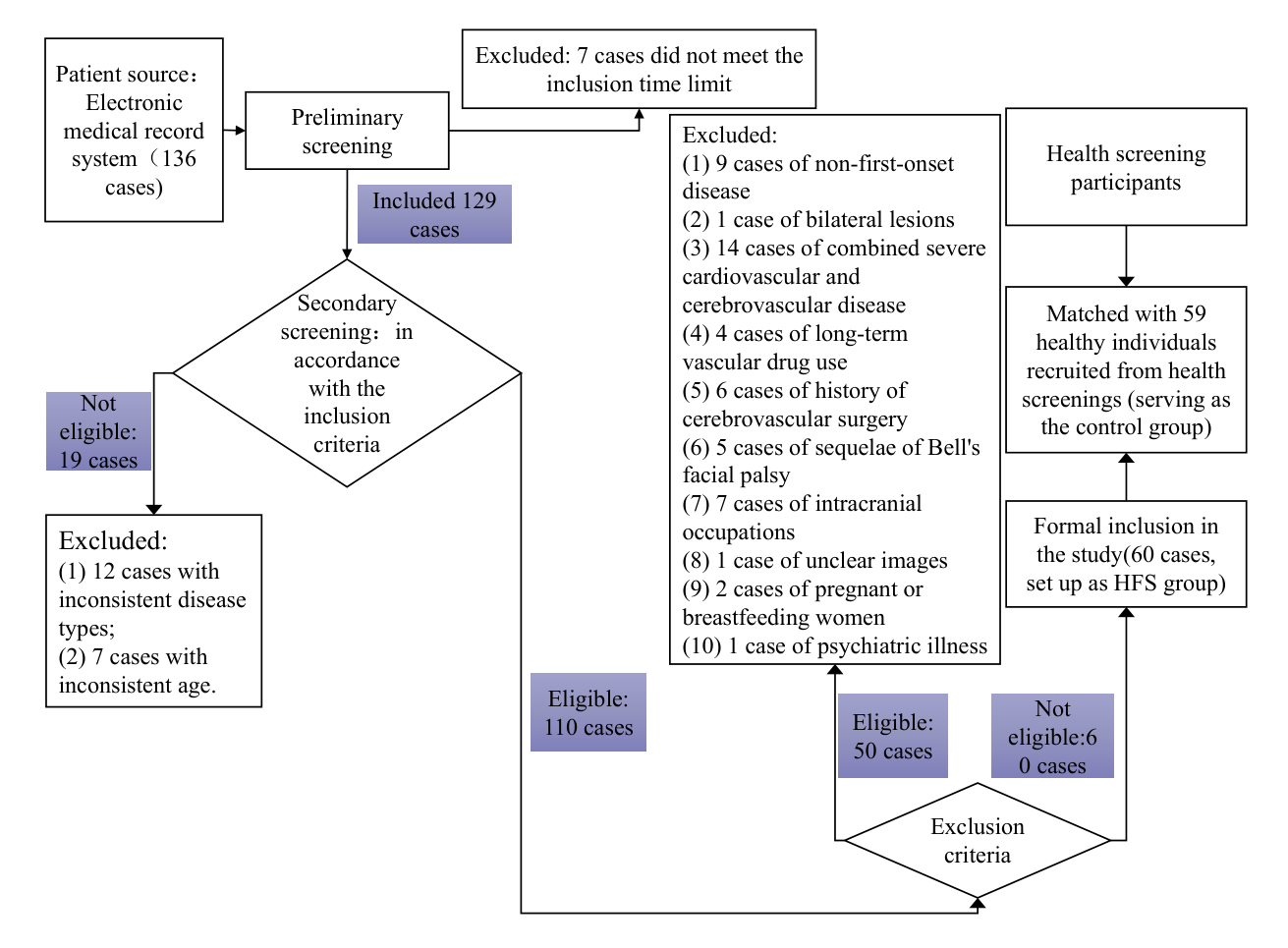 Fig. 1.
Fig. 1.
The process of patient inclusion in this study. HFS, hemifacial spasm.
Inclusion criteria of this study include: (1) meeting the diagnostic criteria of
HFS stipulated in the “Chinese Expert Consensus on Facial Muscle Spasm Diagnosis
and Treatment” [10]; (2) aged
Fifty-nine healthy controls were selected from individuals attending health
screening in the same hospital and included in the control group. Inclusion
criteria for healthy controls include: (1) people who underwent routine physical
examination; and (2) aged
Data including age, gender, BMI, disease duration, past history (hypertension [11], diabetes mellitus [12], hyperlipidemia [13]), mean arterial pressure (MAP), total cholesterol, fasting blood glucose, HFS lesion side, and HFS severity grades of all subjects were collected. The severity of HFS was evaluated using a classification system established by Sampaio [14]. This system classifies spasm intensity into five grades: (1) grade 0: no spasm; (2) grade I: spasm induced by external stimulation; (3) grade II: mild facial muscle spasm; (4) grade III: obvious facial muscle spasm with mild dysfunction; (5) grade IV: severe spasm with dysfunction. For analysis purposes in this study, spasm severity was further categorized into three levels: grades 0 and I as mild, grades II and grade III as moderate, and grade IV as severe.
A 1.5T magnetic resonance machine (SIGNA Voyager type, General Electric,
Chicago, IL, USA) and the accompanying 8-channel head phase-array control coil
were utilized for a conventional head scan magnetic resonance angiography (MRA)
scan to obtain characterization data of VBA. Routine scans included axial
T1-weighted imaging (T1WI) scans with a repetition time (TR) of 1780 ms and an
echo time (TE) of 9 ms; axial T2WI fluid-attenuated inversion recovery (FLAIR)
scans with a TR of 8000 ms and a TE of 92 ms; axial diffusion-weighted imaging
(DWI) scans with a TR of 6061 ms and a TE of 75 ms; and sagittal T2WI scans with
a TR 6014 ms, TE 105 ms. MRA was performed by three-dimensional time-of-flight
(3D-TOF) scanning with a TR of 23 ms, a TE of 6.8 ms, and a field of view of 24
cm
Images were reconstructed by using maximum-intensity projection (MIP), and individual image data were independently evaluated by two senior radiologists using Functool software (version 4.5.5, General Electric Healthcare, Waukesha, WI, USA) on oblique coronary MIP images aligned parallel to the basilar arteries. The coefficient of concordance for the measurements performed by the two senior radiologists was 0.832. The diameters of VA were measured at 3 mm intervals with the VA confluence as the origin for three consecutive measurements, and the average of these measurements was taken as the final measurement of the diameters of the right and left VA. Using phase-contrast magnetic resonance angiography (PC-MRA) technique, the center of the vascular cross-section of the basilar artery (proximal/middle/distal) and bilateral vertebral arteries (segment V4) was selected as the region of interest (ROI), and the mean flow velocity (MFV) was measured at three consecutive levels, and the resulting flow velocity data were averaged separately as the final measurement.
Data were analyzed using SPSS 26.0 software (IBM Corp., Armonk, NY, USA). Normal
distribution was assessed using the Shapiro-Wilk test. Measurements that
conformed to the normal distribution were expressed as mean
There were no statistically significant differences in age, gender, BMI,
past history (hypertension, diabetes mellitus, hyperlipidemia), mean
arterial pressure, total cholesterol, and fasting blood glucose between the HFS
and control groups (p
| Items | HFS group (n = 60) | Control group (n = 59) | t/ |
p | |
| Age (year), |
54.15 |
58.85 |
1.846 | 0.067 | |
| Gender, n (%) | 0.072 | 0.788 | |||
| Male | 27 (45.00) | 28 (47.46) | |||
| Female | 33 (55.00) | 31 (52.54) | |||
| BMI (kg/m2), |
25.94 |
26.75 |
1.420 | 0.159 | |
| Disease duration (years), |
4.40 (1.32, 5.97) | / | |||
| Past history, n (%) | |||||
| Hypertension | 25 (41.67) | 26 (44.07) | 0.070 | 0.791 | |
| Diabetes mellitus | 17 (28.33) | 9 (15.25) | 2.908 | 0.084 | |
| Hyperlipidemia | 6 (10.00) | 11 (18.64) | 1.815 | 0.178 | |
| MAP (mmHg), |
108.44 |
111.78 |
1.415 | 0.160 | |
| Total cholesterol (mmol/L), |
3.58 |
3.63 |
0.321 | 0.749 | |
| FBG (mmol/L), |
4.83 |
4.47 |
1.340 | 0.183 | |
| HFS lesion side, n (%) | |||||
| Left side | 33 (55.00) | / | |||
| Right side | 27 (45.00) | / | |||
| HFS severity grades, n (%) | |||||
| Mild | 11 (18.33) | / | |||
| Moderate | 33 (55.00) | / | |||
| Severe | 16 (26.67) | / | |||
Notes: p
Abbreviations: BMI, body mass index; HFS, hemifacial spasm; MAP, mean arterial pressure; FBG, fasting blood glucose.
The diameters of the left and right VA and the basilar artery were all greater
in the HFS group than in the control group (p
| Items | HFS group (n = 60) | Control group (n = 59) | t | p |
| Diameter of left VA (mm) | 4.38 |
3.30 |
6.174 | |
| Blood flow velocity of left VA (cm/s) | 35.44 |
37.19 |
1.884 | 0.062 |
| Diameter of right VA (mm) | 3.91 |
3.30 |
3.422 | 0.001 |
| Blood flow velocity of right VA (cm/s) | 34.94 |
36.07 |
1.190 | 0.237 |
| Diameter of basilar artery (mm) | 4.31 |
3.84 |
3.013 | 0.003 |
| Blood flow velocity of basilar artery (cm/s) | 34.45 |
36.31 |
1.962 | 0.052 |
Abbreviation: VA, vertebral artery.
Multivariate logistic regression analysis was performed using HFS occurrence
as the dependent variable and the factors showing significant differences between
the two groups in Tables 1,2 as the independent variables. The results showed
that diameter of the left VA (odds ratio [OR] = 4.014, 95% confidence interval
[CI]: 1.997–8.070), diameter of the right VA (OR = 3.890, 95% CI:
2.217–6.825), and diameter of the basilar artery (OR = 2.022, 95% CI:
1.008–4.058) are independent predictors of HFS occurrence (p
| Variables | Standard error | Wald | p | VIF | OR | 95% CI | ||
| Lower limit | Upper limit | |||||||
| Diameter of left VA | 1.390 | 0.356 | 15.218 | 1.104 | 4.014 | 1.997 | 8.070 | |
| Diameter of right VA | 1.358 | 0.287 | 22.423 | 1.059 | 3.890 | 2.217 | 6.825 | |
| Diameter of basilar artery | 0.704 | 0.355 | 3.926 | 0.048 | 1.046 | 2.022 | 1.008 | 4.058 |
| Constant | –12.801 | 2.449 | 27.311 | |||||
Abbreviations: CI, confidence interval; OR, odds ratio; VBA, vertebrobasilar artery; VIF, variance inflation factor.
The diameter of the left VA in the right-sided HFS group was lower than that in
the left-sided HFS group; the diameter of the right VA in the right-sided HFS
group was higher than that in the left-sided HFS group; and the diameter of the
basilar artery in the right-sided HFS group was slightly higher than that in the
left-sided HFS group. However, none of these differences were found to be of
statistical significance between the two HFS subgroups (p
| Items | Right-sided HFS group (n = 27) | Left-sided HFS group (n = 33) | t | p |
| Diameter of left VA | 4.20 |
4.38 |
0.975 | 0.334 |
| Diameter of right VA | 4.00 |
3.81 |
0.831 | 0.410 |
| Diameter of basilar artery | 4.33 |
4.29 |
0.170 | 0.865 |
There were no statistically significant differences in diameter of VA or basilar
artery between patients with mild, moderate, and severe grades of HFS (p
| Items | Diameter of left VA | Diameter of right VA | Diameter of basilar artery |
| Mild (n = 11) | 4.09 |
3.75 |
4.25 |
| Moderate (n = 33) | 4.32 |
4.00 |
4.36 |
| Severe (n = 16) | 4.63 |
4.35 |
4.40 |
| F | 1.654 | 2.177 | 0.694 |
| p | 0.200 | 0.123 | 0.504 |
Asymmetry between left and right VAs is a common anatomical variation, with the left-sided dominance observed more frequently. This dominance of the VBA is particularly prominent in HFS patients [15], suggesting that the high-risk population for HFS could be identified by analyzing VBA features. The study also found that the left VA is often more dominant. The proportion of left-sided dominance of the VAs in patients with left-sided HFS is as high as 96.2%, whereas the proportion of right-sided dominance of the arteries in patients with right-sided HFS is 52.4% [16]. This indicates that there may be significant differences in VBA characteristics between patients with left-sided and those with right-sided HFS. Based on this hypothesis, the present study assessed the VBA characteristics in patients with HFS and in healthy individuals, in order to analyze the relationship of these characteristics with the risk of HFS occurrence. Further analysis was conducted to identify differences in VBA characteristics between right-sided HFS and left-sided HFS patients, and examine differences in these characteristics according to different HFS severity grades. In addition, to reduce the influence of age, gender and the correlation of blood pressure, blood glucose and lipid levels on the risk of developing HFS in patients and their condition, 59 cases of healthy controls were included in the analysis, with their age, gender, BMI, hyperlipidemia, mean arterial pressure, total cholesterol level, and fasting glucose levels all matched to the corresponding variables in the HFS group.
Our results showed that the diameter of the left and right VA, as well as the diameter of the basilar artery, were all independent influencing factors for HFS occurrence. There were no statistically significant differences in VBA diameter between patients with HFS on different sides or affected by HFS at different severity levels.
The influence of VBA on HFS occurrence is possibly reflected mainly in vascular compression and nerve excitation. Anatomically, asymmetry of the VBA, the presence of a dominant VA and the tortuosity of blood vessels increase the risk of vascular compression at the pontine exit of the facial nerve. It has been found that a larger diameter of blood vessel is associated with a greater risk for compression [17]. From a hemodynamic point of view, pressure differences and changes in wall shear stress caused by blood flow promote vascular displacement and deformation. The arterial diameter may indicate that the greater the vascular deformation, the higher the risk of compressing the pontine exit of the facial nerve and the greater the risk of HFS occurrence [18]. This study also found that blood flow velocity in the left and right vertebral and basilar arteries was lower in the HFS group than in the control group, though this difference was not statistically significant. This indicates that HFS patients experienced reduced blood flow velocities in the left and right vertebral and basilar arteries; however, the lack of significant differences compared to healthy controls may be attributed to the fact that most included patients had only mild to moderate levels of HFS. A study by Li et al. [19] reported that the dominant VA was associated with HFS and was more prevalent on the left. These findings indirectly support the conclusion of this study that the diameter of the left VA, the diameter of the right VA, and the diameter of the basilar artery are independent influencing factors for the occurrence of HFS.
However, some studies suggest that primary HFS is caused by vascular compression, where offending blood vessels increase the excitability of the facial nerve through mechanical irritation. This increased excitability facilitates transmission between nerve branches, thereby exacerbating the patient’s symptoms [20]. An increased VA diameter is associated with greater vascular compression of the facet nerve from the cerebral pontine segments due to mechanical factors. A study by Xia et al. [21] also found that mechanical compression of the offending blood vessels can lead to the overexpression of Nav1.8 in the sodium ion channel. Nav1.8 overexpression affects the resting potential of neurons and increases the excitability of the facial nerve nucleus. Excessive excitation and ‘cross-excitation’ between axons can cause more severe HFS symptoms. Additionally, in unilateral lesions, the contralateral dominant vessel often exhibits anatomical variations with increased diameter. However, our analysis of VBA characteristics in patients with HFS at different sides and severity levels did not reveal significant differences in VBA diameter between the subgroups. Although the specific mechanism remains unclear, we speculate that the lack of significant results may be attributed to the relatively small sample size of patients included in this study. Furthermore, the study did not adequately control for, or perform stratified analysis of, confounding factors such as vascular atherosclerosis. This may have obscured relevant vascular changes in HFS patients, leading to the lack of statistically significant differences between groups.
The limitations of this study are as follows: Firstly, owing to its retrospective nature, this study is subject to the inherent biases commonly associated with retrospective analyses. Secondly, the small sample size used and the variations in VBA characteristics in patients across different regions could limit extrapolation of the research results. Differences in the site of compression in the exit zone of the facial nerve were not thoroughly examined, limiting the study’s generalizability and mechanistic insight. Thirdly, VBA characteristics may vary depending on the site of vascular compression along the facial nerve’s root exit zone and the sides affected; however, this study did not perform a correlation analysis to investigate these differences. Therefore, further research is warranted in the future to explore this aspect in greater depth.
In summary, the diameters of the left and right VA and the basilar artery are independent influencing factors for HFS occurrence, with greater diameters of these vessels linked to improved predictive value for HFS. However, vessel diameter does not appear to correlate with disease severity.
• The relationship of VBA characteristics with the risk of HFS occurrence and disease severity remains unclear.
• The diameter of the left, right VA, and the basilar artery are associated with the risk of HFS occurrence, but not with HFS severity classification.
• Morphological alterations in the VBA may play a key role in the pathogenesis of HFS, but do not directly influence HFS clinical grading.
All data included in this study are available from the corresponding author upon reasonable request.
XH and JS designed the study. XH and JS conducted the study. XH provided assistance and guidance during the experiment. JS analysed the data. JS drafted the manuscript. Both authors contributed to important editorial changes to the manuscript. Both authors read and approved the final manuscript. Both authors participated fully in the work and agreed to take responsibility for all aspects of it.
This study was conducted in adherence to the Declaration of Helsinki, and the research protocol was approved by the Medical Ethics Committee of Sir Run Run Shaw Hospital, Zhejiang University School of Medicine (Ethics Approval No.: 2025-1076). This retrospective study used fully anonymized data and posed no risk to patient confidentiality and safety. Therefore, informed consent was waived in accordance with the “Ethical Review Measures for Biomedical Research Involving Human Subjects”.
We would like to express our sincere gratitude to our supervisor for designing this study, and to the Department of Neurology, Sir Run Run Shaw Hospital, Zhejiang University School of Medicine, and its doctors for their valuable support and assistance.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

