1 National Pulmonary Hypertension Unit, Mater Misericordiae University Hospital, D07 KW66 Dublin, Ireland
2 University College Dublin School of Medicine, D04 V1W8 Dublin, Ireland
†These authors contributed equally.
Abstract
Chronic thromboembolic pulmonary hypertension (CTEPH) is a rare, chronic, progressive, and life-limiting condition with a poor five-year survival in untreated patients with advanced disease. Multi-modality imaging techniques are used in making the diagnosis and determining treatment options in CTEPH. Pulmonary endarterectomy (PEA) surgery is the treatment of choice in selected patients, providing excellent long-term survival. Balloon pulmonary angioplasty (BPA) is an established intervention for selected patients who are not suitable for surgery. Riociguat, a soluble guanylate cyclase stimulator, is a vasodilator and indicated in patients with inoperable disease. In this state-of-the-art review, we provide an update in the diagnostic evaluation with a focus on imaging modalities, treatment strategies, and future directions in CTEPH.
Keywords
- chronic thromboembolic pulmonary hypertension
- diagnosis
- imaging modalities
- treatment
- pulmonary endarterectomy
- balloon pulmonary angioplasty
- riociguat
- medical therapy
Pulmonary embolism (PE) is one of the most common causes of cardiovascular mortality (behind coronary artery disease, cardiomyopathy, and stroke) and is a common cause of sudden cardiac death [1]. While the majority of patients have resolution of their symptoms following treatment of their acute PE, a significant proportion of patients have symptoms that persist for more than three months following their PE, including exertional dyspnoea, chest pain, and limitation in functional capacity. This condition is termed post-PE syndrome and encompasses a wide range of conditions, including patients without any obvious residual pulmonary vascular disease [2].
With appropriate treatment of an acute PE, the clot typically resolves. However, incomplete resolution does occur, and residual perfusion defects can be detected in up to 50% of patients who have completed three months of anticoagulation therapy [3]. Patients who have symptoms and evidence of persistent organised clot in the pulmonary circulation without elevated pulmonary arterial pressures (PAP) are classified as having chronic thromboembolic pulmonary disease (CTEPD), which occurs in 6% of cases [4]. Between 2% and 3% of patients have elevated PAP 2–3 years following an acute PE and are diagnosed with chronic thromboembolic pulmonary hypertension (CTEPH) [5, 6, 7, 8]. Under the classification of pulmonary hypertension (PH), CTEPH is within group 4 PH associated with pulmonary artery obstructions and is a rare, chronic, progressive, life-limiting condition [5].
Multi-modality imaging techniques with ventilation/perfusion (V/Q) imaging, computed tomography pulmonary angiogram (CTPA), magnetic resonance pulmonary angiogram, and invasive pulmonary angiogram are used for the diagnosis and treatment selection of patients with CTEPH. The multimodal treatment options in CTEPH include pulmonary endarterectomy (PEA) for technically operable disease, balloon pulmonary angioplasty (BPA) for inoperable disease, and PH targeted medical therapy for predominantly microvascular disease [9].
Despite advancements in treatment options, the diagnosis of CTEPH remains challenging due to nonspecific symptoms and subtle imaging findings, resulting in delayed recognition of the condition. In this review, we provide an update on the diagnostic evaluation with a focus on imaging modalities, treatment strategies, and future directions in CTEPH.
CTEPH is defined as a condition with one or more large mismatched perfusion
defects on V/Q scan, or signs of obstruction on CTPA, with a resting mean
pulmonary artery pressure (mPAP)
CTEPH is classified utilising imaging techniques into proximal and distal disease depending on the distribution of obstructions in the pulmonary vasculature. Proximal CTEPH involves the main, lobar, and segmental pulmonary arteries, while distal CTEPH involves sub-segmental or smaller pulmonary arteries. In addition, CTEPH can also be classified using surgical classification from the University of California San Diego (UCSD) (Fig. 1, Ref. [11]).
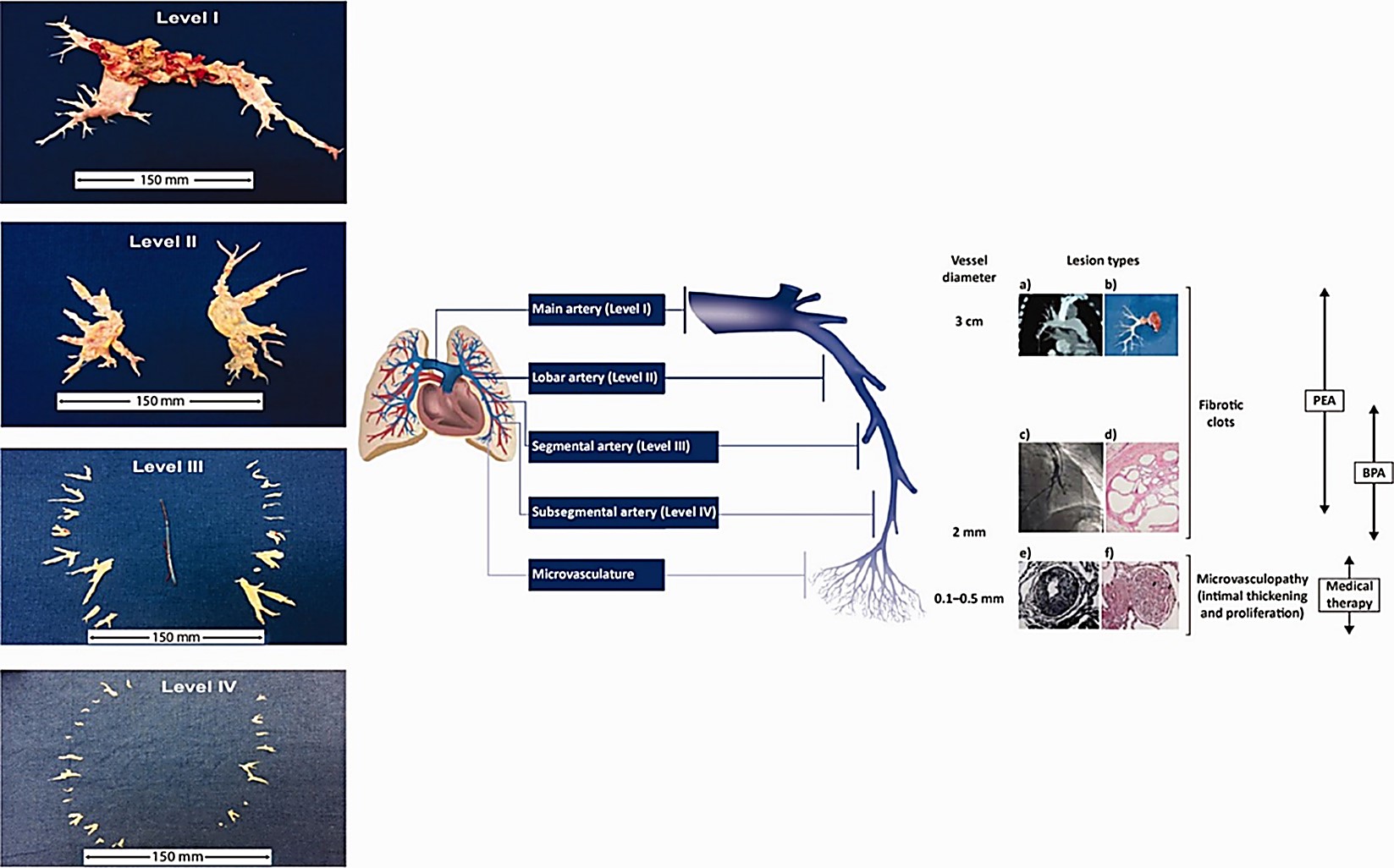 Fig. 1.
Fig. 1.
Diagram demonstrating the University of California San Diego (UCSD) surgical classification and size of material retrieved from PEA. Level 0: No evidence of chronic thromboembolic disease in either lung. Level I: Lesion begins at the level of the main pulmonary arteries. Level II: Lesion begins at the level of the lobar arteries. Level III: Lesion begins at the level of the segmental arteries. Level IV: Lesion begins at the level of the sub-segmental arteries. (a) Computed tomography of a pulmonary artery. (b) Organised thromboembolic material removed at pulmonary endarterectomy. (c) Pulmonary angiogram of segmental and subsegmental pulmonary arteries showing irregular vessel contour and occlusion. (d) Microscopic examination showing a luminal defect with recanalised chronic thrombus (web lesion) and no evidence of vasculopathy in the subsegmental pulmonary artery. (e) Intimal fibromuscular proliferation. (f) Plexiform lesion and vessel occlusion due to vasculopathy and proliferation. PEA, pulmonary endarterectomy; BPA, balloon pulmonary angioplasty. Reprinted with permission from Madani [11], available under the CC BY-NC 4.0 https://creativecommons.org/licenses/by-nc/4.0/.
The incidence of CTEPH is estimated to be between 4 and 7 cases per million population per year and could increase to 13 cases per million population per year when a formal outpatient PE clinic is instituted [8]. However, the exact incidence of CTEPH is currently unknown as the condition is underdiagnosed [9].
Pulmonary arterial obstruction followed by aberrant pulmonary vascular remodelling is considered to be the primary contributor to the development of CTEPH. However, the pathogenesis of CTEPH is complex and multifactorial, and factors such as infection, abnormal immune responses, fibrinolysis, genetics, inflammation, and persistent endothelial dysfunction are also believed to contribute (Fig. 2) [9, 12].
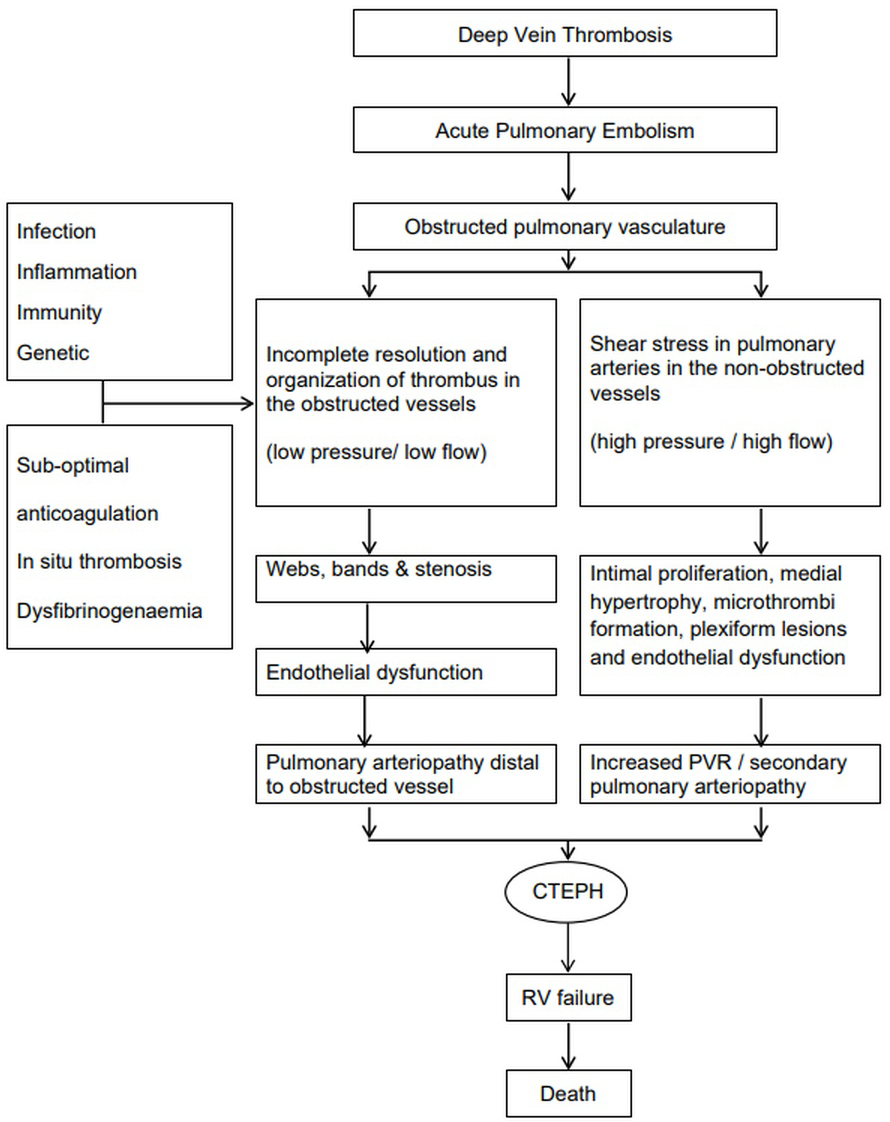 Fig. 2.
Fig. 2.
Proposed pathophysiology of the development of CTEPH. PVR, pulmonary vascular resistance; CTEPH, chronic thromboembolic pulmonary hypertension; RV, right ventricle.
The pathophysiology of CTEPH can be described using a two-compartment model: one involving the obstructed pulmonary arteries, and the other involving the non-obstructed pulmonary arteries. In the majority of patients with acute PE, the thrombus resolves within weeks. However, in a small subset, thrombus resolution is incomplete, leading to persistent pulmonary arterial obstruction. This thrombotic material becomes organised and eventually fibrotic and adheres to the pulmonary arterial vessel wall, forming webs, bands, slits, and stenotic segments. These obstructed regions are characterised by low pressure and flow, resulting in local hypoxia and endothelial dysfunction. In response, hypertrophy of the systemic bronchial arteries and increased bronchopulmonary anastomotic flow contribute to micro-vasculopathy distal to the obstruction [9].
In non-obstructed regions, the pulmonary vasculature is exposed to redistributed blood flow and increased pressure and shear stress. This promotes intimal proliferation, medial hypertrophy, in-situ thrombosis, and in some cases, plexiform lesions. These vascular changes—affecting both obstructed and non-obstructed regions—result in an increase in PVR and the development of CTEPH [9, 13].
Initially, the right ventricle (RV) undergoes adaptive hypertrophy with minimal or no dilatation. With persistent pressure overload, the RV dilates, loses contractile function, and enters a maladaptive phase, ultimately leading to right heart failure and death [14].
There are several risk factors associated with the development of CTEPH, which are presented in Fig. 3.
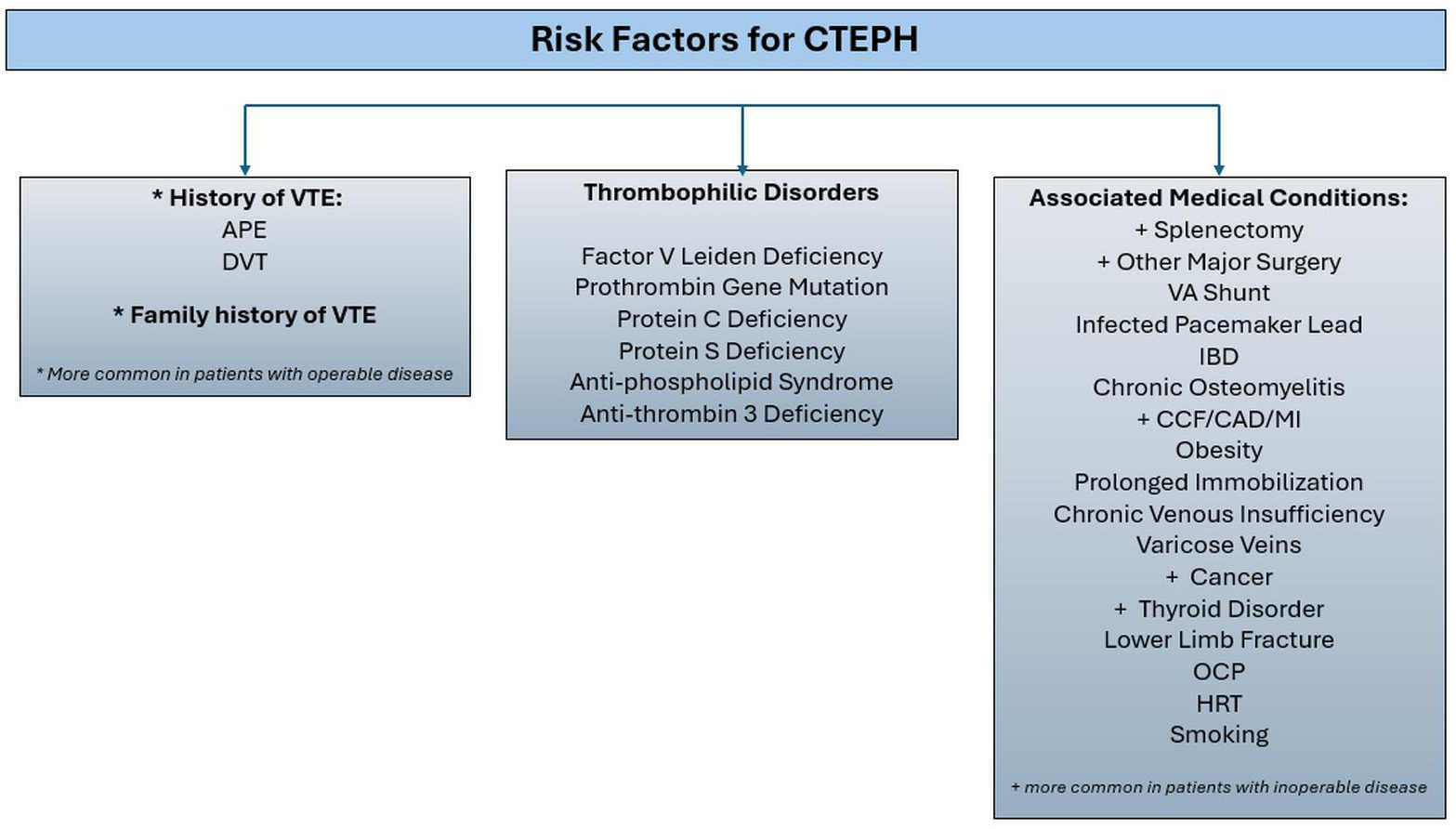 Fig. 3.
Fig. 3.
Risk factors for CTEPH. CTEPH, chronic thromboembolic pulmonary hypertension; VTE, venous thromboembolism; APE, acute pulmonary embolus; DVT, deep vein thrombosis; VA, ventriculo-atrial; IBD, inflammatory bowel disease; CCF, congestive cardiac failure; CAD, coronary artery disease; MI, myocardial infarction; OCP, oral contraceptive pill; HRT, hormone replacement therapy.
The history of venous thromboembolism (VTE) is an important risk factor for CTEPH. However, up to 25% of patients with CTEPH do not have any documented history of VTE, which underscores the need to evaluate additional underlying mechanisms for the development of CTEPH [15].
In patients with CTEPH, routine screening for thrombophilia disorders is not
routinely recommended except in the case of antiphospholipid syndrome (APLS),
which carries diagnostic and therapeutic relevance. The highest-risk subset is
triple-positive APLS, defined by positivity for lupus anticoagulant,
anti-cardiolipin antibodies (Immunoglobulin G [IgG] and Immunoglobulin M [IgM])
and anti-
Other important risk factors for CTEPH include prior splenectomy, permanent intravascular devices (pacemaker leads and long-term central lines), inflammatory conditions (inflammatory bowel disease and osteomyelitis), malignancy and thyroid replacement hormone. These conditions may contribute to a prothrombotic state or impair thrombus resolution and should be specifically considered during clinical evaluation [9, 10, 13].
Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) infection was initially speculated to be associated with acute PE and the development of CTEPH. However, a recent study from the UK national registry data showed that CTEPH remained a rare complication after Coronavirus Disease 2019 (COVID-19). In this study, only 1.2% of the patients with confirmed CTEPH had a prior history of COVID-19-related PE [17]. This is in keeping with national registry data from France [18]. These findings suggest that COVID-19 is not a risk factor for the development of CTEPH.
CTEPH should be actively considered as a possible diagnosis in any patient with unexplained dyspnoea and identifiable risk factors.
Patients with CTEPH present with symptoms similar to those of patients with other forms of PH. The symptoms are usually non-specific in nature, with dyspnoea and fatigue initially, then subsequently oedema and other features of RV dysfunction and failure in the more advanced stages [10]. Psychological distress is also common [19]. Due to the nonspecific nature of their symptoms, the diagnosis is often delayed. Recent CTEPH registries have demonstrated a time to diagnosis from symptom onset of 10–15 months [15, 20]. Delays in diagnosis are associated with worse pulmonary haemodynamics and may result in an increase in all-cause mortality [5]. Therefore, it is important to consider CTEPH specifically in patients with a history of acute PE, a new diagnosis of PH, or in patients with unexplained dyspnoea [21].
Recent European expert consensus recommends clinical assessment at three months after acute PE to screen for CTEPH in a structured and standardised follow-up clinic [22].
V/Q imaging remains the diagnostic test of choice to screen for CTEPH and is recommended for all patients who are undergoing workup of newly diagnosed PH. Two main types of V/Q imaging are used: Planar V/Q scan, which provides 2D images and V/Q single photon emission computed tomography (SPECT), which generates 3D perfusion images and imaging of the lungs and mediastinal anatomy. Although either imaging technique is suitable, V/Q SPECT is more sensitive and is the preferred imaging modality [23]. Persistent mismatched perfusion defects are the key finding in CTEPH. In patients with normal lung parenchyma on radiographic imaging and a negative V/Q scan, CTEPH can be ruled out with a negative predictive value of 98% [10]. However, there are some limitations to V/Q imaging; the extent of the perfusion defects does not correlate with disease severity, specifically in proximal disease. Additionally, V/Q imaging does not differentiate CTEPH from acute PE and other causes of vascular obstruction, such as pulmonary veno-occlusive disease or pulmonary capillary haemangiomatosis [10, 23]. V/Q imaging does not require breath-holding, making it more suitable for patients with respiratory distress. In addition, V/Q imaging avoids the use of contrast agents and is therefore safe for patients with renal impairment or contrast allergy [24].
CTPA is the most readily available and widely used imaging modality to visualise the pulmonary vasculature in suspected CTEPH. CTPA identifies direct vascular features of CTEPH, including eccentric thrombus, webs, bands, calcified thrombus and pulmonary artery narrowing. Indirect vascular signs include main pulmonary artery dilatation and bronchial artery enlargement in advanced cases. Parenchymal findings may include a mosaic attenuation pattern: cardiac features suggestive of PH include right heart chamber dilatation, increased RV/left ventricle (LV) ratio, interventricular septal flattening, and hepatic venous reflux of contrast suggestive of tricuspid regurgitation [24]. Fig. 4 (Ref. [24]) shows radiological features of CTEPH on CTPA.
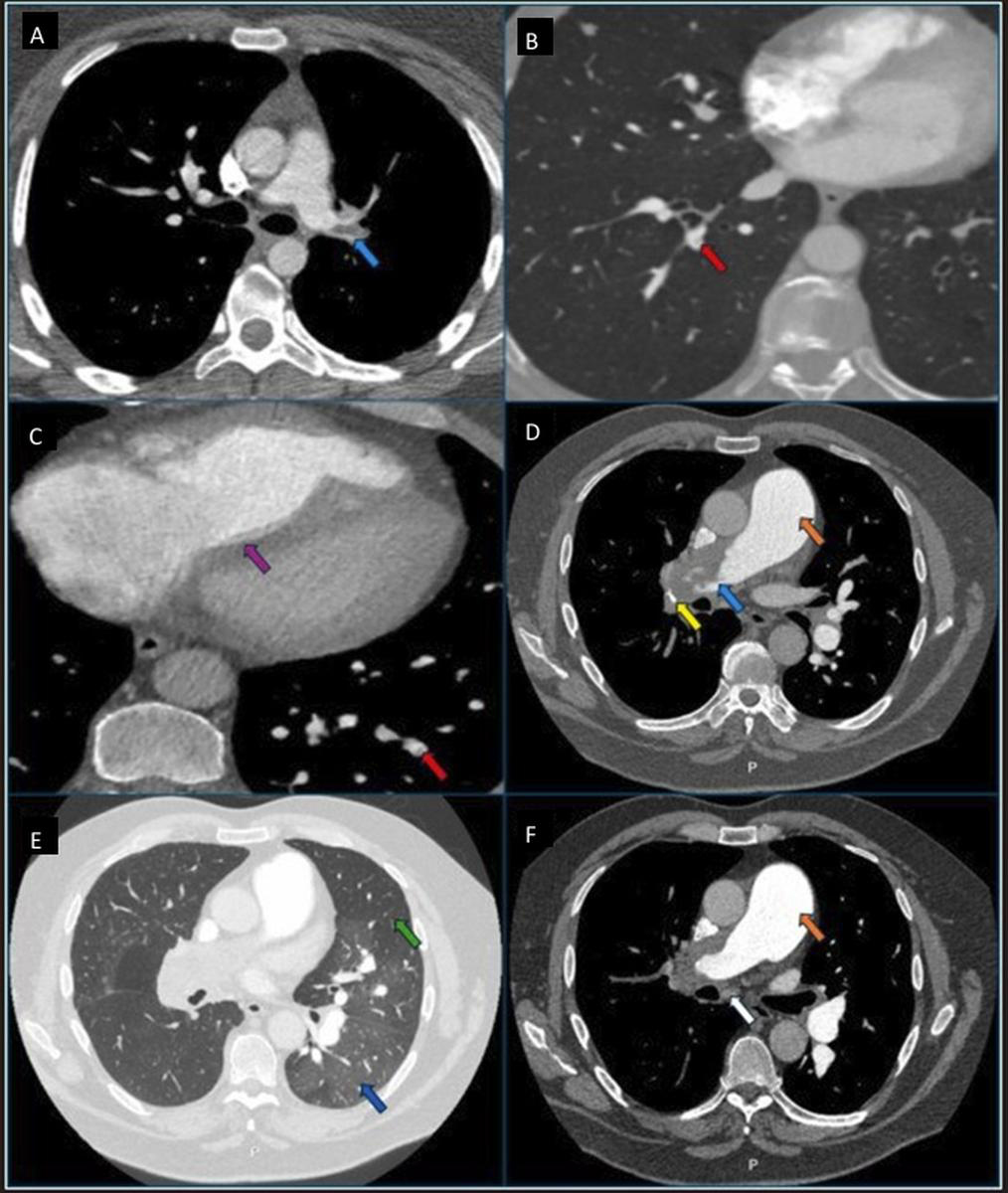 Fig. 4.
Fig. 4.
Chronic thromboembolic pulmonary disease (CTEPD) features on computed tomography pulmonary angiogram (CTPA). (A) CTEPD without pulmonary hypertension (PH) patient with extensive left main pulmonary artery chronic thrombi with intimal irregularity and obtuse angle (light blue arrow). Note the non-dilated main pulmonary artery. (B) CTEPD without PH patient with pulmonary artery web/band (red arrow). Note the normal right-to-left ventricle ratio. (C) CTEPH patient with dilated right heart chambers with flattening of the interventricular septum (purple arrow) and pulmonary artery web/band (red arrow). (D) CTEPH patient with calcification (yellow arrow) in extensive occluding proximal chronic thrombi, irregular intimal contour with obtuse angle (light blue arrow) and dilated main pulmonary artery (orange arrow). (E) CTEPH patient with mosaicism with areas of hypo-attenuation and vascular pruning in the region of vascular obstruction (green arrow) and normal/hyper-attenuation in the region without vascular obstruction (dark blue arrow). (F) CTEPH patient with dilated bronchial artery (white arrow) and dilated main pulmonary artery (orange arrow). Reprinted with permission from Ghani et al. [24], available under the CC BY-NC-ND 4.0 https://creativecommons.org/licenses/by-nc-nd/4.0/.
CTPA’s main advantages are its high-definition imaging, short scan duration, and ability to detect both vascular obstruction and secondary cardiac changes. The main limitation includes the requirement of iodinated contrast, making it unsuitable for some patients with severe renal impairment or contrast allergy, and the inability to provide direct lung perfusion assessment. In addition, subtle or distal chronic thrombi may be missed without expert interpretation, with a specificity of 25% in diagnosing sub-segmental lesions in some studies [25].
Despite these limitations, advances in computed tomography (CT) technology and radiologist expertise have significantly improved its diagnostic accuracy, with reported sensitivity of 88%–98% and specificity of 89%–96% [24, 25].
Magnetic resonance imaging (MRI) is increasingly used in the evaluation of suspected CTEPH and offers diagnostic performance comparable to CT in selected centres. It can delineate the pulmonary vasculature and allows for perfusion mapping of the lungs without the need for ionising radiation. MRI also provides detailed information on cardiac morphology and function, including right ventricular ejection fraction, ventricular mass index, and pulmonary artery pulsatility index. Its main advantages are the absence of radiation and its ability to assess both anatomy and function in a single study. Limitations of MRI include inferior image sharpness compared to CT, longer scan duration, reduced availability, and higher cost. In addition, it remains less accurate than CTPA in visualising distal pulmonary arterial anatomy. MRI may be considered in patients where radiation exposure is a concern or when a comprehensive cardiopulmonary assessment is required in a single modality [10, 24, 26].
Non-invasive multi-modality imaging techniques in CTEPH are presented in Fig. 5.
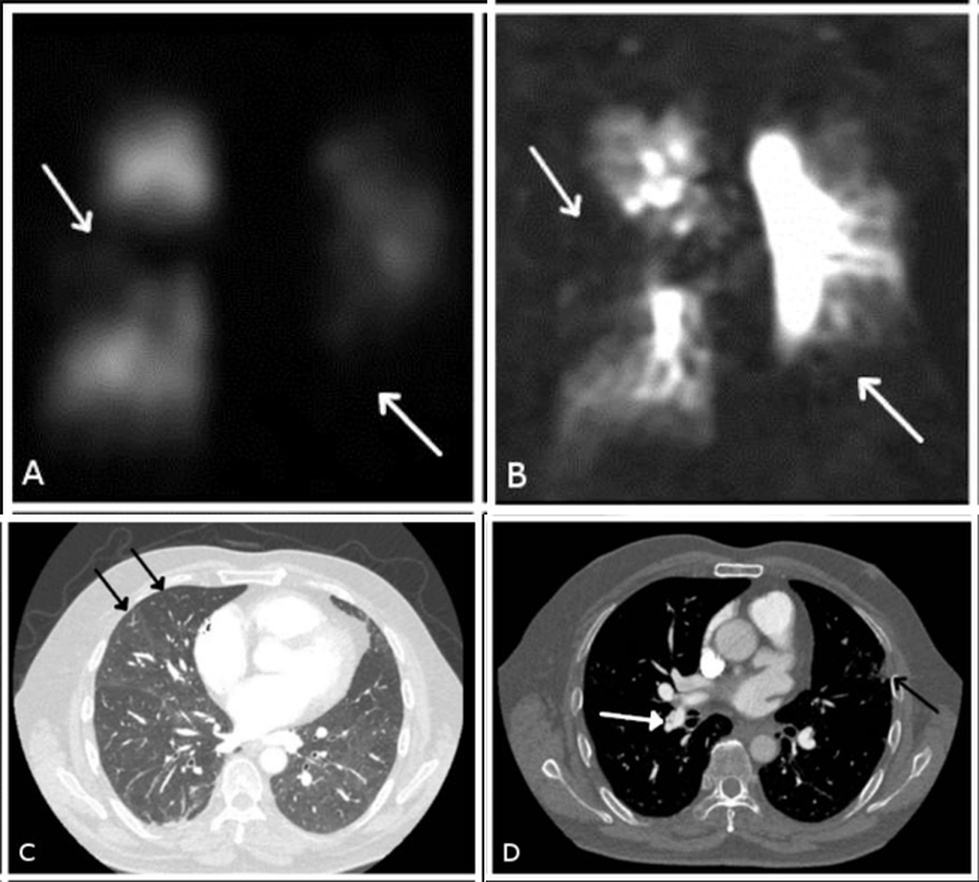 Fig. 5.
Fig. 5.
Non-invasive multi-modality imaging in chronic thromboembolic disease. Bilateral segmental perfusion defects (white arrows in (A) and (B)) demonstrated on (A) planar perfusion scintigraphy, (B) 3D dynamic contrast-enhanced perfusion magnetic resonance imaging (MRI) and (C) and (D) CTPA with the typical findings of a central filling defect (web) (white arrow in (D)), mosaic perfusion pattern (black arrows in (C)) and subpleural scarring from infarction (black arrow in (D)).
Standard pulmonary angiography involves advancing a pigtail catheter via a sheath placed in the right internal jugular vein or right common femoral vein into the pulmonary arteries, followed by iodinated contrast injection and fluoroscopic imaging during breath hold on deep inspiration. Digital subtraction angiography (DSA) is not a separate procedure but a software-based technique that improves the quality of the images by subtracting non-vascular structures. Both standard pulmonary angiography and DSA images can be performed in the radiology suite or cardiac catheter lab. DSA is helpful for the diagnosis and treatment selection of patients with CTEPH, whereas standard pulmonary angiography can be used during real-time BPA procedures [27].
Although pulmonary angiography was historically considered the gold standard for assessing operability, its routine use has declined with the advent of high-resolution CTPA. In current practice, conventional angiography is generally reserved for two key indications: (1) when CTPA is of inadequate quality or non-diagnostic despite repeat imaging, and (2) for detailed anatomical mapping prior to BPA, particularly in patients not undergoing surgery [10, 28].
When used for BPA planning, DSA plays an important role in assessing the appearance of vascular lesions. Five common lesion types have been described: type A (ring-like stenosis), type B (webs or bands), type C (subtotal occlusion), type D (total occlusion), and type E (tortuous vessels). Following BPA, types A and B lesions have better treatment outcomes and lower complication rates compared to types C, D, and E lesions [28, 29].
Advantages of pulmonary angiography include higher imaging clarity, dynamic real-time imaging, and the ability to measure pulmonary haemodynamics at the same time. It provides direct visualisation and accurate lesion classification. Disadvantages include radiation exposure, its invasive nature that comes with associated procedural risks, and limitations caused by motion artefact if the patient is unable to breath-hold efficiently. There is a requirement for iodinated contrast, and there are risks to patients with severe renal impairment and contrast allergy. As a result, DSA use is generally limited to specific scenarios rather than routine diagnostic work-up [24, 25].
In recent years, several advanced imaging techniques have been developed to improve the diagnosis and treatment selection in CTEPH. These technologies offer a more detailed visualisation of the pulmonary vasculature and may better support treatment planning, particularly in centres performing BPA. Some of these modalities are limited to expert PH centres but are expected to be more widely available in the future.
Dual-energy CT (DECT) is an imaging modality that produces CTPA images and lung perfusion imaging at the same time. This is useful for both diagnosis and follow-up. DECT has a sensitivity of 97%–100% and a specificity of 86%–92% which is preserved for segmental disease with a sensitivity of 85% and a specificity of 99%. The main limitations of DECT are that it has limited availability and the requirement for specific expertise in the interpretation of the pulmonary vascular imaging [24, 25].
Area detector CT is a newer imaging modality that uses a wide detector range (usually 256–320 rows), similar to the DECT scan. It produces both CTPA imaging and reconstructs lung ventilation and perfusion imaging. One key advantage is that it provides a higher resolution of the distal pulmonary arteries, including sub-segmental branches, making it useful for BPA planning. In addition, it can also provide full cardiac CT information, including heart size and function, valve assessment, and coronary artery and myocardial perfusion. The main limitations are that it involves a higher radiation dose, requires increased patient cooperation, and is only available in a very few expert PH centres, so there is limited clinical experience in its use [5, 30, 31].
Cone beam CT, also known as C-arm CT, is an advanced form of pulmonary angiography performed using a C-arm system that rotates 180–360 degrees around the patient while contrast is administered through a catheter into the pulmonary artery. It generates 2D images over a few seconds and reconstructs them into a high-resolution 3D image of the pulmonary arteries. The technique provides better views of the distal segmental and sub-segmental pulmonary arteries, making it useful for BPA planning. Limitations include the need for invasive access, use of contrast, operator expertise, and it is mainly available in very few high-volume BPA centres [24, 25].
Three-dimensional reconstruction can be created from standard CTPA images using specialised software. This provides a detailed visualisation of the distal pulmonary arteries, making it useful in BPA planning without the need for an extra scan or contrast. Limitations include the need for specialised software, time for post-processing, and image quality that depends on operator technique [32].
Despite the availability of multiple imaging modalities, including emerging techniques, the subtlety of the diagnostic signs, limited specialist training, and restricted access outside expert PH centres often lead to missed or delayed recognition of CTEPH. Even with standard tools like CTPA and V/Q scanning, a lack of familiarity amongst radiologists remains a key contributor to delayed referral to expert PH centres.
Transthoracic echocardiography (TTE) is an essential non-invasive imaging modality recommended in the initial evaluation of suspected PH. It not only serves as a screening tool but also provides important information on RV function and associated cardiac co-morbidities [10].
Features suggestive of PH include elevated tricuspid regurgitation velocity (TRV
In addition, TTE provides assessment of LV function, left atrial size, and left-sided valvular abnormalities, which is useful in excluding group 2 PH and contributing to comprehensive risk stratification [10].
RHC is mandatory, not only to confirm the diagnosis of precapillary PH but also to plan treatment. Post-capillary disease, from left heart comorbidities, can increase operative risk. PVR is helpful in assessing overall prognosis and calculating the risk associated with PEA and BPA interventions [9, 33].
In the diagnostic workup of CTEPH, initial screening with V/Q scanning or other perfusion imaging is crucial for identifying perfusion defects. If abnormalities are detected, TTE is used to screen for the presence of PH. Patients with suspected CTEPH are then referred to a specialised PH/CTEPH centre for comprehensive evaluation, which includes CTPA and DSA if required. The final diagnosis of CTEPH is confirmed via RHC, after which a multidisciplinary team (MDT) determines the most suitable treatment strategy for each patient [9]. The European Society of Cardiology (ESC) and European Respiratory Society (ERS) diagnostic algorithm for the workup of CTEPH is presented in Fig. 6 (Ref. [10]).
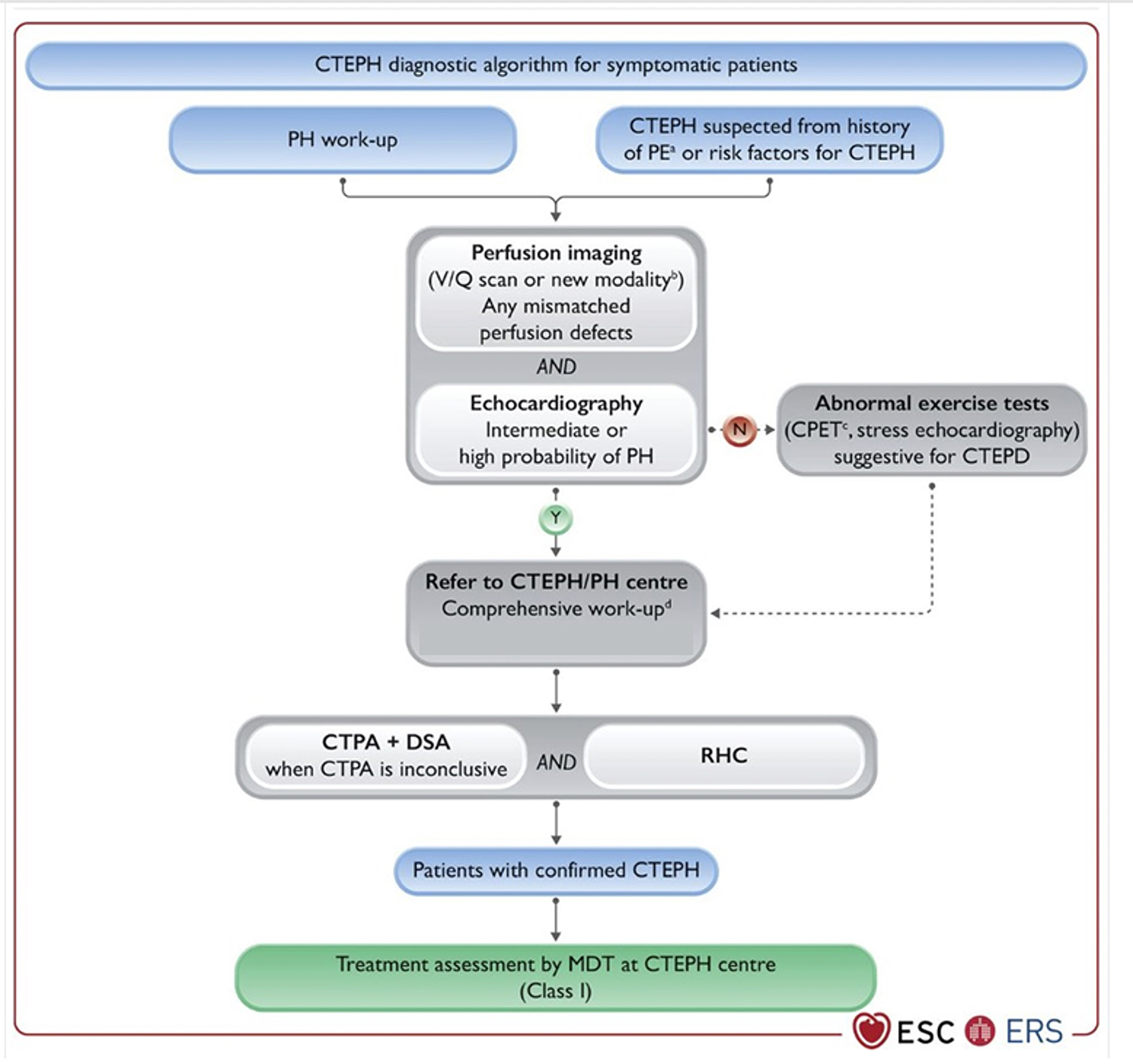 Fig. 6.
Fig. 6.
Diagnostic algorithm for the workup of CTEPH. CTEPH, chronic thromboembolic pulmonary hypertension; PH, pulmonary hypertension; PE, pulmonary embolus; V/Q, ventilation/perfusion; CPET, cardiopulmonary exercise testing; CTEPD, chronic thromboembolic pulmonary disease; CTPA, computed tomography pulmonary angiogram; DSA, digital subtraction angiography; RHC, right heart catheterisation; MDT, multidisciplinary team; N, no; Y, yes. a CTEPH suspected from the history of PE, including elevated systemic pulmonary arterial pressure and signs of CTEPH on CTPA performed at the time of the acute PE. b Alternative imaging techniques. c Typical pattern seen low PETCO2 (PETCO2, end tidal CO2), high VE/VCO2 (VE, minute ventilation; VCO2, volume of exhaled carbon dioxide), low VO2/HR (VO2, volume of O2; HR, heart rate), and low peak O2. d Comprehensive assessment after 3 months of effective anticoagulation or sooner in the deteriorating patient. Reprinted with permission from Humbert et al. [10], available under the CC BY-NC 4.0 https://creativecommons.org/licenses/by-nc/4.0/.
General management of CTEPH consists of lifelong anticoagulant therapy. Oral vitamin K antagonists (VKA) have historically been the preferred anticoagulant, although many patients are now treated with direct oral anticoagulants (DOACs). International guidelines recommend VKA in patients with APLS, especially those who have a triple-positive antibody profile [10]. Bunclark and colleagues [34] demonstrated that in patients with CTEPH, DOACs were associated with higher rates of recurrent thrombosis compared to warfarin, despite similar bleeding rates. Another prospective observational multinational registry showed no difference in mortality between patients receiving VKA or DOAC [15].
Patients are supported with diuretics and oxygen therapy when required. General preventive measures such as vaccinations, regular guided exercise, and counselling regarding the risk of pregnancy in women of childbearing age are recommended [9, 10, 35].
Patients with CTEPH should be evaluated in an expert centre with current guidelines recommending a multidisciplinary approach to decision-making. This MDT should consist of experienced pulmonary vascular radiologists, PEA surgeons, BPA interventionists, PH physicians, CTEPH nurse specialists, and administrative staff. The role of the CTEPH MDT is to confirm the diagnosis, assess the suitability for surgical operability or BPA intervention, determine the indication for medical therapy or, in some cases, consider multimodal therapy. This decision is primarily based on the patient’s case history, disease distribution and pulmonary haemodynamics. In addition, the MDT can advise on follow-up post-intervention and initiate further therapies as clinically indicated [9, 29]. This approach is routinely implemented in expert PEA centres.
Surgical resection of occlusive chronic thromboembolic material is the recommended treatment for CTEPH. CTEPH is potentially curable with PEA, and treatment improves symptoms, quality of life, and prognosis [33, 36, 37]. PEA is a major surgical procedure, and the method is based on data from Jamieson and colleagues at UCSD. It involves a median sternotomy with cardiopulmonary bypass, enabling gradual cooling to 20 degrees Celsius. Deep hypothermic circulatory arrest (DHCA) is then initiated to allow dissection of the pulmonary artery. DHCA is limited to twenty-minute intervals, and typically only one interval is required per side. The usual approach is to make an incision along the right main pulmonary artery extending into the right lower lobe. From there, chronic thromboembolic material is resected to the sub-segmental level. On completion of the right PEA, the procedure is repeated on the left side. The overall hospital stay is typically 7–10 days post-procedure [33, 36, 38, 39].
Suitability for undergoing PEA depends on various factors. Surgically accessible pulmonary arterial occlusions are proximal and typically involve the main, lobar, segmental and proximal sub-segmental pulmonary arteries. Haemodynamic measurements taken at the time of RHC help to determine suitability for surgery. Historically, patients with higher PVR preoperatively have had poorer outcomes, and this was once a contraindication to surgery. However, with extra-corporeal membrane oxygenation (ECMO) support, the operative mortality in these cases has declined to less than 5%. Other patient factors, including advanced age and the presence of multiple comorbidities, are perceived clinical risks to operability but are not absolute contraindications. There are no strict criteria for determining operable candidates, and ultimately, patient selection depends on the experience of the unit [5, 9, 40].
A UK-based national registry identified predictors of short and long-term mortality following PEA. 90-day mortality was associated primarily with haemodynamic burden, increased age, reduced six-minute walk distance (6MWD), reduced cardiac index, elevated mPAP, and increased PVR. In contrast, five-year mortality was associated with comorbidity burden, including increased left atrial area, history of tobacco smoking, older age, ischaemic heart disease, and reduced 6MWD [41]. In addition, extremes of body mass index pre-operatively have been identified as a predictor of mortality following surgery [42].
In expert centres, PEA significantly reduces pulmonary artery pressures in 70% of cases with a postoperative mortality of 2% [9]. Long-term outcomes are excellent with survival of 87%–96% at 3 years and 79% at 10 years [15, 20, 43, 44, 45, 46]. The Assessing the Spectrum of Pulmonary hypertension Identified at a Referral centre (ASPIRE) registry has shown that patients undergoing PEA surgery and those eligible for but declining surgery due to patient choice have an estimated 5-year survival of 83% and 55% respectively [10]. This suggests that surgery improves survival showing the importance of surgical counselling in the management of patients with technically operable disease.
Patients who have mild CTEPH (mPAP 21–24) may not benefit from intervention. The UK national registry demonstrated that these patients who were not on PH therapy and did not receive any intervention had no evidence of disease progression on non-invasive testing and had an overall mortality of 9% at 3 years follow-up [47]. In this group of patients with technically operable disease, PEA is only reserved for patients who have severe exertional symptoms and poor quality of life.
In selected patients with significant residual PH post-PEA and worsening symptoms despite PH therapy, repeat PEA may be considered. Data from UCSD support its use in expert centres with similar post-operative mortality and higher complication rates as compared to patients undergoing PEA for the first time [48].
There is a strong association between centre volume and PEA outcomes. Heuts and colleagues [49] observed a non-linear relationship, with around 40 surgeries per year being required to reduce postoperative mortality below 5%. Centres performing over 100 surgeries annually had postoperative mortality rates of 2.9%. This reinforces the importance of centralising care in high-volume, experienced institutions. Expert PEA centres are able to perform more distal endarterectomies with fewer complications and lower mortality compared to less experienced centres [50].
As per the recent consensus statement, expert PEA centres are defined as centres
that perform greater than 50 PEA surgeries per annum with a postoperative
mortality of less than 3% and the ability to operate at segmental or
sub-segmental levels. Non-expert PEA centres are defined as centres that perform
PEA is the treatment of choice for operable disease, offering excellent outcomes. The procedure has evolved from treating proximal disease to relatively distal disease in select high-volume expert centres with excellent outcomes.
BPA is a treatment option for patients deemed to have inoperable disease or for patients who have residual PH post-PEA. BPA is a percutaneous intervention which involves the treatment of pulmonary vascular obstructions with balloon dilatation. Target lesions are identified using selective pulmonary angiography. A guidewire is advanced into the diseased areas, and a balloon is advanced over the guidewire. The balloon is then inflated at low pressures, and this process is then repeated to target other diseased vessels. The current BPA technique requires multiple staged procedures with several diseased segments targeted during each session, usually confined to an individual lung. The current technique was pioneered in Japan and has been adopted by multiple expert centres across Europe and North America. Three-year survival rates are reported as 92%–95% [9, 15, 28, 51].
The balloon pulmonary angioplasty versus riociguat for the treatment of inoperable chronic thromboembolic pulmonary hypertension (RACE) trial was a phase 3 open-label randomised controlled trial (RCT) comparing BPA to riociguat in non-operable patients with CTEPH. Improvements in pulmonary haemodynamics were more pronounced in the BPA group, with a reduction of PVR to 40% of baseline as compared to 67% of baseline in the riociguat group at 26 weeks. Patients who underwent BPA had a higher rate of treatment-related adverse events (42% in the BPA group vs. 9% in the riociguat group), of which lung injury was found to be the most common complication (35% in the BPA group vs. 4% in the riociguat group) [52]. These results are in keeping with contemporary BPA clinical trials in Japan [53]. The RACE ancillary study followed up patients for a further 26 weeks and showed that patients who were pretreated with riociguat before undergoing BPA showed similar improvement of pulmonary haemodynamics but reduced treatment-related adverse events compared to patients who did not receive riociguat before BPA (14% riociguat and BPA group vs. 42% in BPA only group) [52]. A post hoc analysis of the RACE trial further highlighted that BPA led to significantly greater improvements in RV function compared to riociguat, reinforcing the physiological advantage of BPA in non-operable disease [54].
A large single-centre study from Japan demonstrated that patients with both technically operable and inoperable disease achieved comparable 10-year survival following BPA (84.3% vs. 90.1%) [55]. This was in keeping with the results of the national Japanese registry data (5-year survival: 95.6%; BPA group vs. 95.4%; PEA group) [56]. In addition, a French single-centre study demonstrated comparable improvement in pulmonary haemodynamics six months after completion of all BPA sessions in patients with proximal and distal CTEPH (proximal CTEPH: PVR decreased from 6.5 to 4.0 WU; distal CTEPH: PVR decreased from 7.6 to 3.8 WU) [57]. These findings suggest that BPA may provide durable long-term outcomes even in patients with disease distributions that would now be considered operable in expert surgical centres, supporting BPA’s expanding role in CTEPH management.
In a Japanese single-centre registry, approximately 65% of patients achieved a
mPAP
Based on the recent international consensus statements, the aim of BPA is to reduce the mPAP to below 30 mmHg, improve resting saturations above 95% and dilate all segments and sub-segments of the pulmonary artery that are technically possible [9, 28].
Expert BPA centres are defined as doing 100 BPA interventions per annum with
less than 1% mortality and have ECMO support. Non-expert BPA centres are defined
as doing
BPA is an established alternative treatment option for patients who are unable to undergo PEA due to distal disease distribution. In select high-volume expert centres, it can also be performed in patients with technically operable disease, achieving good clinical outcomes.
Multiple clinical trials have been performed to assess the efficacy of PH therapies in CTEPH. These trials have been performed in patients who have inoperable disease or those who have persistent PH post-PEA (Table 1, Ref. [60, 61, 62, 63, 64, 65]). To date, the most robust evidence for medical therapy is for riociguat, a soluble guanylate cyclase stimulator. The evidence for riociguat was demonstrated in the trials riociguat for the treatment of chronic thromboembolic pulmonary hypertension (CHEST-1) and its long-term extension, riociguat for the treatment of chronic thromboembolic pulmonary hypertension: a long-term extension study (CHEST-2). CHEST-1 was a phase 3 randomised double-blind placebo-controlled trial evaluating riociguat versus placebo. Riociguat met the primary end point of improvement in 6MWD, with an improvement of 39 m compared to –6 m in the placebo group [60]. CHEST-2 was an open-label extension of CHEST-1 and demonstrated sustained efficacy of riociguat and a favourable benefit-risk profile at 2 years [66].
| Trial | Study drug | Trial phase | Patients | Primary endpoint | Treatment effect on 6MWD (m) | Treatment effect on PVR (%), mean | Reference |
| BENEFiT | Bosentan | Phase 3 | 157 | Change in PVR as a percentage of baseline and 6MWD from baseline to week 16 | +2 | –24% | [61] |
| CHEST-1 | Riociguat | Phase 3 | 261 | Change in 6MWD from baseline to week 16 | +46 | –246 dynꞏsꞏcm–5 | [60] |
| MERIT-1 | Macitentan | Phase 2 | 80 | Resting PVR as a percentage of baseline to week 16 | +34 | –19% | [62] |
| CTREPH | Treprostinil | Phase 3 | 105 | Change in 6MWD from baseline to week 24 | +40.7 (high dose versus low dose) | Mean change from baseline: high dose –214 dynꞏsꞏcm–5, low-dose +73 dynꞏsꞏcm–5 | [63] |
| JapicCTI | Selexipag | Phase 2 | 78 | Change in PVR from baseline to week 20 | +2 | –93.5 dynꞏsꞏcm–5 | [64] |
| SELECT* | Selexipag | Phase 3 | 91 | Percent of baseline PVR to week 20 | - | - | [65] |
*Discontinued early due to futility.
6MWD, six-minute walk distance; BENEFiT, Bosentan Effects in iNopErable Forms of chronIc thromboembolic pulmonary hypertension; CHEST-1, riociguat for the treatment of chronic thromboembolic pulmonary hypertension; CTREPH, subcutaneous treprostinil for the treatment of severe non-operable chronic thromboembolic pulmonary hypertension; JapicCTI, Selexipag for chronic thromboembolic pulmonary hypertension in Japanese Patients - A Double-Blind, Randomised, Placebo-Controlled, Multicentre Phase II Study; MERIT-1, Macitentan for the treatment of inoperable chronic thromboembolic pulmonary hypertension: results from the multicentre, phase 2, randomised, double-blind, placebo-controlled study; PVR, pulmonary vascular resistance; SELECT, Efficacy and safety of selexipag in patients with inoperable or persistent/recurrent CTEPH (SELECT randomised trial).
Subcutaneous treprostinil is the only other PH targeted therapy to have shown improvements in 6MWD in CTEPH in any phase 3 randomised controlled trial. Subcutaneous treprostinil for the treatment of severe non-operable chronic thromboembolic pulmonary hypertension (CTREPH) trial was a multi-centre double-blind controlled phase 3 clinical trial involving 105 patients with inoperable disease or persistent PH post PEA surgery, evaluating the efficacy of continuous subcutaneous treprostinil, a stable prostacyclin analogue in CTEPH. Patients were randomised to high-dose versus a low-dose treprostinil (30 ng/kg/min vs. 3 ng/kg/min). At 24 weeks, patients receiving the continuous high-dose subcutaneous treprostinil had an improvement of 41 m in their 6MWD compared to patients receiving lowdose treprostinil [63]. The open-label extension study of CTREPH showed sustained clinical benefit and tolerability of long-term subcutaneous treprostinil in severe inoperable CTEPH, with many patients receiving additional BPA or PH therapy. No new safety concerns occurred over 24 months [67]. In Macitentan for the treatment of inoperable chronic thromboembolic pulmonary hypertension: results from the multicentre, phase 2, double-blind, placebo-controlled study (MERIT-1), Macitentan, an oral endothelin receptor antagonist, was shown to significantly reduce PVR in patients with inoperable CTEPH [62]. The long-term safety and efficacy of Macitentan in inoperable chronic thromboembolic pulmonary hypertension: results from MERIT and its open-label extension (MERIT-2), the open-label extension and crossover study following MERIT-1, demonstrated that patients who had received macitentan from the start maintained their walk distance at approximately 388 m over 6 and 36 months, while those who crossed over from placebo improved by +10 m at 6 months and +23 m at 36 months with good tolerability. Additionally, in a progressive disease like inoperable CTEPH, the lack of functional decline over three years and a 23 m improvement despite delayed initiation highlights the practical value of ongoing medical therapy in this condition [68]. While not approved for the treatment of CTEPH, these findings make a strong case for the use of macitentan for inoperable disease. Combining an endothelin receptor antagonist with other pulmonary vasodilator therapies targeting the nitric oxide and or prostacyclin signalling pathways is likely to offer synergistic benefit in severe cases, given the shared pathophysiology with pulmonary arterial hypertension (PAH).
Riociguat is currently the only medication licensed for the treatment of CTEPH, as recommended by current international guidelines. Other pulmonary vasodilator therapies may be used off-label, on compassionate grounds, for symptomatic benefit.
The ESC/ERS three-strata risk stratification model for PAH has demonstrated differences in long-term mortality among medically managed CTEPH patients. The ASPIRE registry demonstrated a better 5-year survival of 91% in low-risk CTEPH cases compared to 59% and 29% in intermediate and high-risk cases, respectively, in keeping with the results of the Comparative, Prospective Registry of Newly Initiated Therapies for Pulmonary Hypertension (COMPERA) registry, suggesting shared prognostic features and overlapping pathophysiology with PAH [37].
Hybrid therapy offers new possibilities in CTEPH management by combining surgery, intervention, and medical therapy in tailored sequences. One approach, referred to as sequential hybrid therapy, involves performing BPA after PEA to treat residual distal lesions not accessible surgically. In high-risk patients with severe haemodynamic compromise, combined hybrid therapy may be used where BPA is performed pre-operatively to improve stability and reduce surgical risk, often alongside PH-targeted medical pre-treatment. BPA is also used as a rescue strategy in life-threatening situations, such as following unsuccessful PEA or in patients with CTEPH who develop acute PE. In both scenarios, ECMO support is often required. These strategies are typically applied in expert centres based on MDT evaluation [69, 70].
The treatment algorithm for patients with CTEPH is presented in Fig. 7 (Ref. [9]).
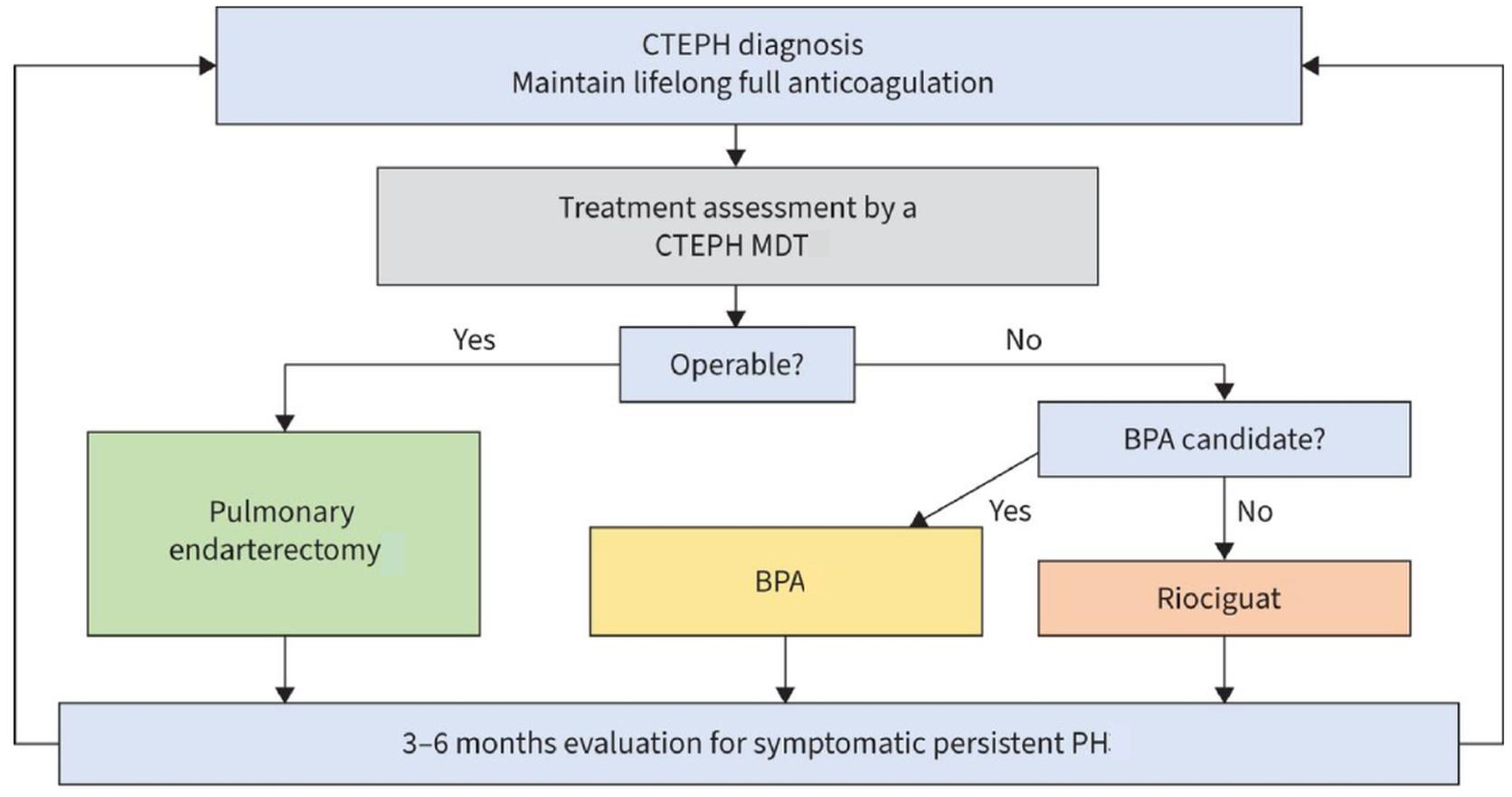 Fig. 7.
Fig. 7.
Management algorithm for treatment of patients with CTEPH. CTEPH, chronic thromboembolic pulmonary hypertension; MDT, multidisciplinary team; PH, pulmonary hypertension; BPA, balloon pulmonary angioplasty. Reprinted from Kim et al. [9], available under the CC BY-NC 4.0 https://creativecommons.org/licenses/by-nc/4.0/.
Questions still remain unanswered about management in CTEPH, where PEA and BPA are both feasible. PEA remains the treatment of choice in patients with technically operable disease. Operability is not clearly defined and is based on the expertise of the surgical centre. PEA has been increasingly performed by high-volume centres on patients with relatively distal disease, with similar outcomes as compared to patients with more proximal obstructions. Balloon pulmonary angioplasty versus pulmonary endarterectomy in patients with chronic thromboembolic pulmonary hypertension: a non-inferiority randomised trial (GO-CTEPH) is a non-inferiority study comparing BPA with standard of care PEA in patients who are eligible for both procedures (GO-CTEPH; clinicaltrials.gov identifier NCT05110066, https://clinicaltrials.gov/study/NCT05110066).
The Initial Dual Oral Combination Therapy Versus Standard-of-care Initial Oral Monotherapy Prior to Balloon Pulmonary Angioplasty in Patients With Inoperable Chronic Thromboembolic Pulmonary Hypertension (IMPACT CTEPH) RCT is assessing if there is a benefit of dual oral combination therapy with macitentan and riociguat prior to undergoing BPA (IMPACT-CTEPH; clinicaltrials.gov identifier NCT04780932, https://www.clinicaltrials.gov/study/NCT04780932). This strategy is currently used in some expert BPA centres based on anecdotal evidence. If the study meets its primary endpoint and there is a significant improvement in pulmonary haemodynamics, this could further reduce the risks of pulmonary vascular injury during the BPA procedure and improve overall outcomes in this patient group.
Sequential use of surgery, BPA, and medical therapy is an area of emerging interest, but prospective studies are needed to guide patient selection.
In parallel with these clinical trials, technological innovations in imaging and artificial intelligence (AI) are likely to reshape the diagnostic and interventional landscape in CTEPH. Emerging imaging modalities such as DECT, Area Detector CT, cone beam CT, and three-dimensional reconstructions are already enhancing anatomical resolution and interventional planning, particularly for BPA. In the future, emerging AI-driven tools—including automated pulmonary arterial segmentation, machine-learning–derived CT scoring systems, and combined models incorporating perfusion imaging and thrombotic burden—may enable more accurate prediction of haemodynamic compromise and individualised treatment selection. While such approaches have shown promise in PAH, their application in CTEPH remains investigational. No validated AI-based scoring systems or diagnostic algorithms currently exist in CTEPH; however, their development could mark a paradigm shift in diagnosis, operability assessment, and treatment planning for both PEA and BPA [24].
In patients with CTEPH, PEA remains the treatment of choice where feasible, as supported by current guidelines, with excellent outcomes even in distal disease at expert centres. However, it is invasive and resource-intensive.
BPA offers a less complex alternative and is increasingly applied to distal disease. Future advances, including improved imaging technologies, pre-treatment targeted medical therapy, and increasing operator experience, may further enhance BPA planning and safety. Real-world data from Japan and worldwide suggest that BPA may achieve outcomes comparable to PEA in selected cases. The early uptake in Japan may also reflect a cultural preference for less invasive treatments, contributing to broader acceptance of BPA in that setting. Wider adoption in Europe is ongoing, and further expansion in the indication for BPA will depend on results from ongoing RCTs.
Much like coronary artery disease, where bypass surgery and percutaneous coronary intervention are used in complement, PEA and BPA, with or without adjunct medical therapy, may ultimately coexist with selection based on patient profile, disease distribution, and the refinement of available diagnostic and therapeutic tools.
• Structured follow-up at three months after an acute PE improves early recognition and timely referral of patients with suspected CTEPH.
• Multi-modality imaging techniques are required for the diagnosis and determination of treatment planning in CTEPH.
• Pulmonary endarterectomy (PEA) surgery is the treatment of choice in patients with technically operable disease.
• Balloon pulmonary angioplasty (BPA) offers a valuable alternative in patients with CTEPH who are unable to undergo PEA surgery.
• The soluble guanylate cyclase stimulator, riociguat, is the only currently approved oral medical treatment option in CTEPH.
• Advances in the pulmonary vascular imaging techniques are expected to further improve patient selection, procedural planning and outcomes in patients with CTEPH.
Not applicable.
CF, SRQ and SG conceived the idea. CF and SRQ performed the literature search. CF, SRQ and SG analysed the data. CF, BM, SRQ, and SG interpreted the data. CF, SRQ and SG drafted the manuscript. All authors contributed to the important editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All images are anonymised and used under institutional ethical approval for retrospective research use (REC 06/Q2308/8). The research was conducted in strict accordance with the ethical principles outlined in the Declaration of Helsinki.
Not applicable
The work was supported by a grant from the Pulmonary Hypertension Association, Ireland (PHA2025-01).
The authors declare no conflicts of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.







