1 Department of Infectious Diseases, Beijing Children’s Hospital, Capital Medical University, National Center for Children’s Health, National Center for Children’s Infectious and Allergic Diseases Surveillance, Beijing Research Center for Respiratory Infectious Diseases, Beijing Key Laboratory of Core Technologies for the Prevention and Treatment of Emerging Infectious Diseases in Children, Key Laboratory of Major Diseases in Children, Ministry of Education, 100045 Beijing, China
2 Big Data Center, Beijing Children’s Hospital, Capital Medical University, National Center for Children’s Health, 100045 Beijing, China
Abstract
Invasive fungal infection (IFI) represents a significant global health challenge, particularly in paediatric populations, due to high mortality and severe long-term sequelae. This study aimed to characterize the epidemiological patterns and quantify the disease burden of IFI among hospitalised children in China.
Data were obtained from the face sheets of discharge medical records collected between 2016 and 2022 from 30 tertiary children’s hospitals, aggregated into the FUTang Updating medical REcords (FUTURE) database. Sociodemographic variables, disease spectrum, pathogen distribution, potential risk factors, length of stay (LOS), and overall disease burden among children with IFI were systematically analysed.
A total of 1250 IFI cases were identified, revealing an upward trend in incidence since 2019. The pathogen distribution among the 485 episodes with available microbiological data included Candida (25.36%; 123/485), Aspergillus (23.71%; 115/485), Cryptococcus (22.06%; 107/485), Pneumocystis (21.44%; 104/485), Mucor (4.12%; 20/485), Talaromyces (1.24%; 6/485), Histoplasma (0.82%; 4/485), Blastomyces (0.62%; 3/485), and Sporotrichum (0.62%; 3/485). Disseminated infections accounted for 5.76% (72/1250) of all IFI. Among the 1178 non-disseminated IFI cases, pneumonia (73.34%, 864/1178), central nervous system (CNS) infections (15.53%, 183/1178), and bloodstream infections (8.66%, 102/1178) were the predominant disease types. The most prevalent risk factors were haematological malignancies and myelosuppression. Overall, 181 patients died during their hospitalisation, representing a mortality rate of 14.48%.
The incidence of IFI among hospitalised children in tertiary centres in China has risen since 2019, with Candida, Aspergillus, and Cryptococcus identified as the predominant pathogens. These infections are associated with considerable mortality (14.48%). The findings highlight the urgent need for enhanced surveillance, earlier diagnosis, and targeted therapeutic strategies to reduce morbidity and mortality in this high-risk population.
Keywords
- invasive fungal infection
- epidemiology
- hospitalisation
- children
- Candidiasis
- haematological malignancy
Invasive fungal infection (IFI) constitutes a major global public health challenge, particularly among children [1, 2]. Each year, more than 6.5 million people worldwide are affected by life-threatening fungal diseases [3]. Despite advances in medical technology, the widespread use of broad-spectrum antibiotics, glucocorticoids, organ transplantation, and deep venous cannulation has increased susceptibility to opportunistic fungal infections [4, 5].
Children, especially neonates, are highly vulnerable to IFI because of their immature immune systems, which may result in long-term complications such as neurodevelopmental impairments and organ dysfunction [6]. In 2022, the World Health Organization (WHO) released its first Fungal Priority Pathogens List [7], highlighting the rising importance of IFI. Among these pathogens, Candida species are particularly concerning in paediatric care settings. Candida albicans is the predominant cause of hospital-acquired bloodstream infections in paediatric populations [8, 9]. Additionally, infections caused by Aspergillus and other molds have increased among high-risk groups, including patients with haematological malignancies and those undergoing stem cell transplantation [10]. Mortality rates for invasive mycoses in children remain alarmingly high, at approximately 50%, despite therapeutic advances [11].
The epidemiology of IFI in children is influenced by geographical and socioeconomic disparities, differences in healthcare resources, and varying prevalence of risk factors [12]. Although awareness is growing and targeted studies have been conducted in specific paediatric populations [2, 13], comprehensive, longitudinal national data on paediatric IFI in China, a country with significant regional healthcare inequalities, remain scarce. Therefore, this study retrospectively analysed 1250 cases of hospitalised children diagnosed with IFI across 30 children’s hospitals in 22 provinces of China between 2016 and 2022. The findings aim to provide valuable insights into the epidemiological profile and disease burden of paediatric IFI in China, potentially bridging key knowledge gaps and contributing to the global evidence base. This information could further inform prevention, surveillance, and treatment strategies for these severe infections.
The Futang Research Centre of Pediatric Development (FRCPD, http://www.futang.org/ftgk.jhtml), established in 2016, is the first non-profit organisation dedicated to paediatric development research. The centre comprises 47 provincial and municipal medical institutions, creating a nationwide network for children’s health services. Since its inception, FRCPD has systematically collected Face Sheets of Medical Records (FSMRs) from its member hospitals. By 2022, FRCPD had developed an integrated system for the collection and sharing of FSMRs from its member hospitals, thereby improving data utilisation and institutional collaboration. Beijing Children’s Hospital, Capital Medical University, acting as the convening unit, collected FSMRs of paediatric inpatients from 30 tertiary children’s hospitals within the FRCPD network, forming the FUTang Updating medical REcords (FUTURE) database [14]. Data organisation, verification, and quality control were performed by FRCPD personnel.
This was a multicentre, cross-sectional study. Basic medical information for all
paediatric patients (aged 0–18 years) hospitalised with IFI in the FUTURE
database between 2016 and 2022 was retrospectively reviewed. Data were extracted
using the tenth revision of the International Statistical Classification of
Diseases and Related Health Problems, 10th Revision (ICD-10) codes
(https://icd.who.int/browse10/2019/en) for hospitalised children with an IFI
diagnosis. The corresponding ICD-10 codes are listed in Supplementary
Table 1. Extracted variables included sex, age, year of admission, place of
residence, geographic region, length of stay (LOS), primary and secondary diagnoses,
clinical outcomes, and hospitalisation costs. Patients were categorised into six
age groups (days, d; months, m; years, y) corresponding critical stages of immune
system development [15]: 0–28 days,
This retrospective study included basic medical information from the FUTURE
database for all children (
Categorical variables were expressed as frequencies and percentages. LOS and hospital expenditure are reported as medians with interquartile
ranges (IQRs). The Kolmogorov-Smirnov test was applied to assess normality.
Categorical variables were compared using the chi-square test or Fisher’s exact
test, as appropriate. Nonnormally distributed continuous variables were analysed
using the nonparametric Mann-Whitney test for two groups, and the Kruskal-Wallis
test for three or more groups. Trends in hospitalisation rates were assessed
using the Cochran–Armitage trend test to evaluate linear changes. All analyses
were conducted with IBM SPSS Statistics for Windows, Version 23.0 (IBM Corp.,
Armonk, NY, USA). Statistical significance was defined as a two-tailed
p-value
A total of 1250 episodes of IFI were identified between January 2016 and
December 2022, based on ICD-10 codes, representing 0.14‰ of all
admissions (1250/9,008,615). The demographic characteristics of patients are
summarised in Table 1. Among these cases, 58.32% (729/1250) occurred in males
and 41.68% (521/1250) in females, yielding a male-to-female ratio of 1.40:1. Age
distribution analysis revealed that children aged 6–12 years accounted for the
highest proportion at 30.08% (376/1250), followed by those aged 3–6 years
(20.64%, 258/1250) and 28 days–1 year (20.00%, 250/1250). Regionally,
Northwest China recorded the highest number of hospitalisations (25.68%,
321/1250), followed by East China (19.20%, 240/1250) and Central China (19.04%,
238/1250). The proportion of IFI-related hospitalisations increased significantly
from 0.09‰ in 2016 to 0.24‰ in 2022
(Cochran–Armitage test,
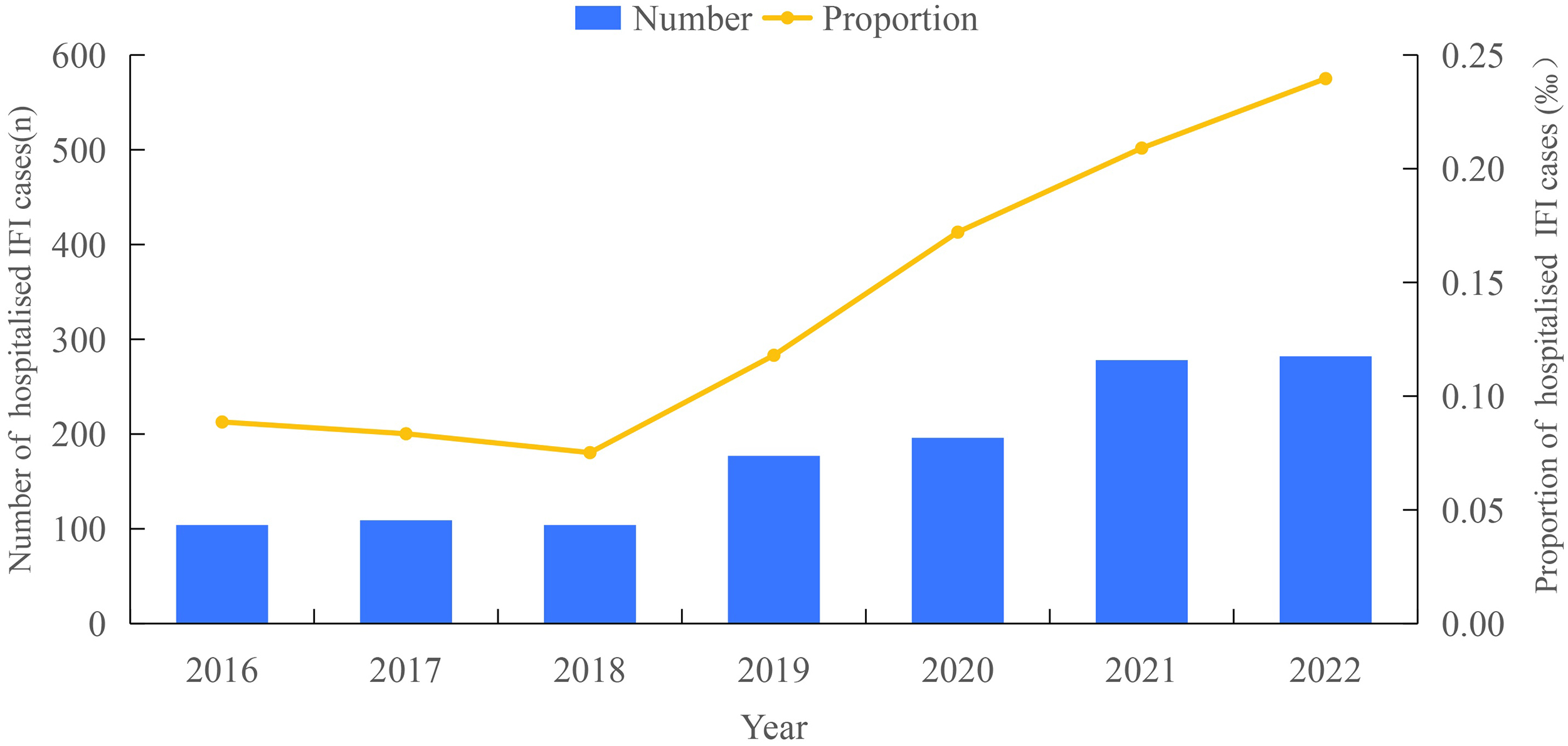 Fig. 1.
Fig. 1.
Annual number proportion of hospitalised children with IFI (2016–2022). Abbreviation: IFI, invasive fungal infection.
| Categories | Hospitalisation | |
| Number of patients | 1250 | |
| Sex, n (%) | ||
| Male | 729 (58.32) | |
| Female | 521 (41.68) | |
| Age group, n (%) | ||
| 0–28 days | 82 (6.56) | |
| 250 (20.00) | ||
| 176 (14.08) | ||
| 258 (20.64) | ||
| 376 (30.08) | ||
| 108 (8.64) | ||
| Region, n (%) | ||
| Northeast China | 25 (2.00) | |
| North China | 213 (17.04) | |
| East China | 240 (19.20) | |
| South China | 91 (7.28) | |
| Central China | 238 (19.04) | |
| Northwest China | 321 (25.68) | |
| Southwest China | 122 (9.76) | |
| Year of admission, n (%) | ||
| 2016 | 104 (8.32) | |
| 2017 | 109 (8.72) | |
| 2018 | 104 (8.32) | |
| 2019 | 177 (14.16) | |
| 2020 | 196 (15.68) | |
| 2021 | 278 (22.24) | |
| 2022 | 282 (22.56) | |
| Place of residence, n (%) | ||
| Urban | 464 (37.12) | |
| Rural | 786 (62.88) | |
| LOS [days, median (IQR)] | 17 (9–29) | |
| Hospitalisation expenditure [USD, median (IQR)] | 4503.36 (1926.15–9330.83) | |
Abbreviations: IQR, interquartile range; LOS, length of stay; USD, United States dollar.
The proportions of IFI hospitalisations were analysed relative to total
hospitalisations across different patient age groups. As shown in Fig. 2A,
children aged
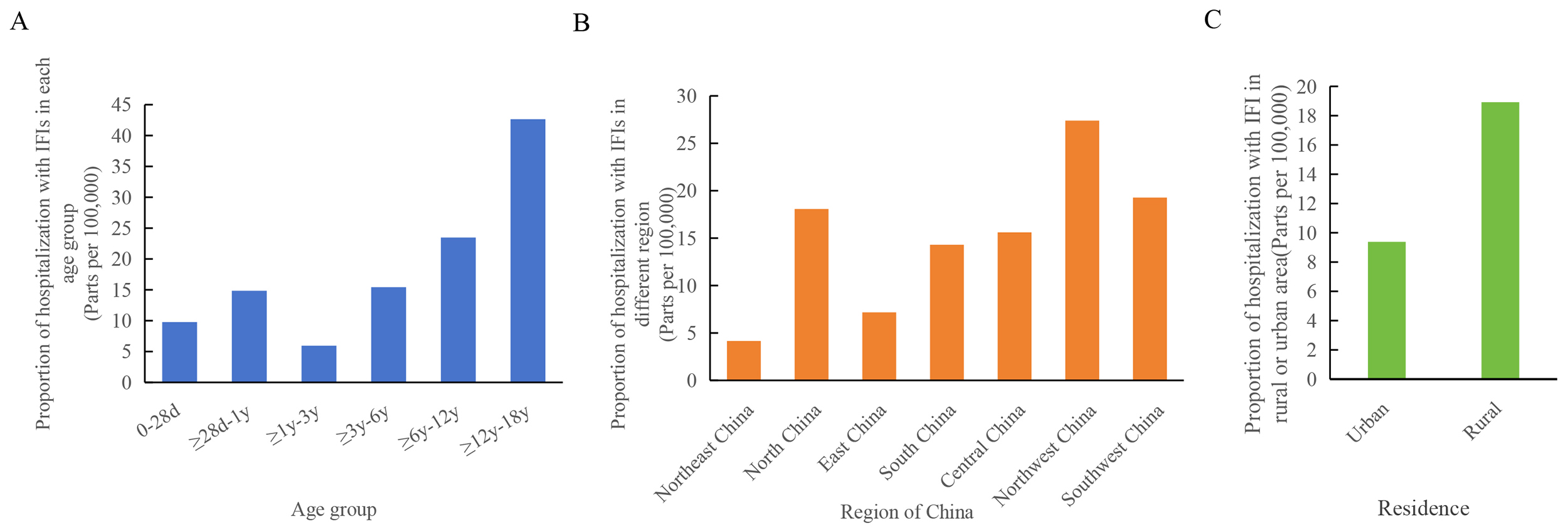 Fig. 2.
Fig. 2.
Distribution of hospitalised children with IFI by category: (A) age group, (B) region, and (C) place of residence. Abbreviations: d, days; y, year(s).
Among the 1250 patients enrolled in this study, 5.76% (72/1250) had disseminated IFI, whereas 94.24% (1178/1250) presented with non-disseminated IFI (Fig. 3A). Within the non-disseminated group, pneumonia, central nervous system (CNS) infections, and bloodstream infections were predominant disease types, representing 73.34% (864/1178), 15.53% (183/1178), and 8.66% (102/1178), respectively. Less frequent infections included nasosinusitis (1.44%, 17/1178), endocarditis (0.51%, 6/1178), arthritis (0.34%, 4/1178), keratitis (0.08%, 1/1178), and peritonitis (0.08%, 1/1178) (Fig. 3B).
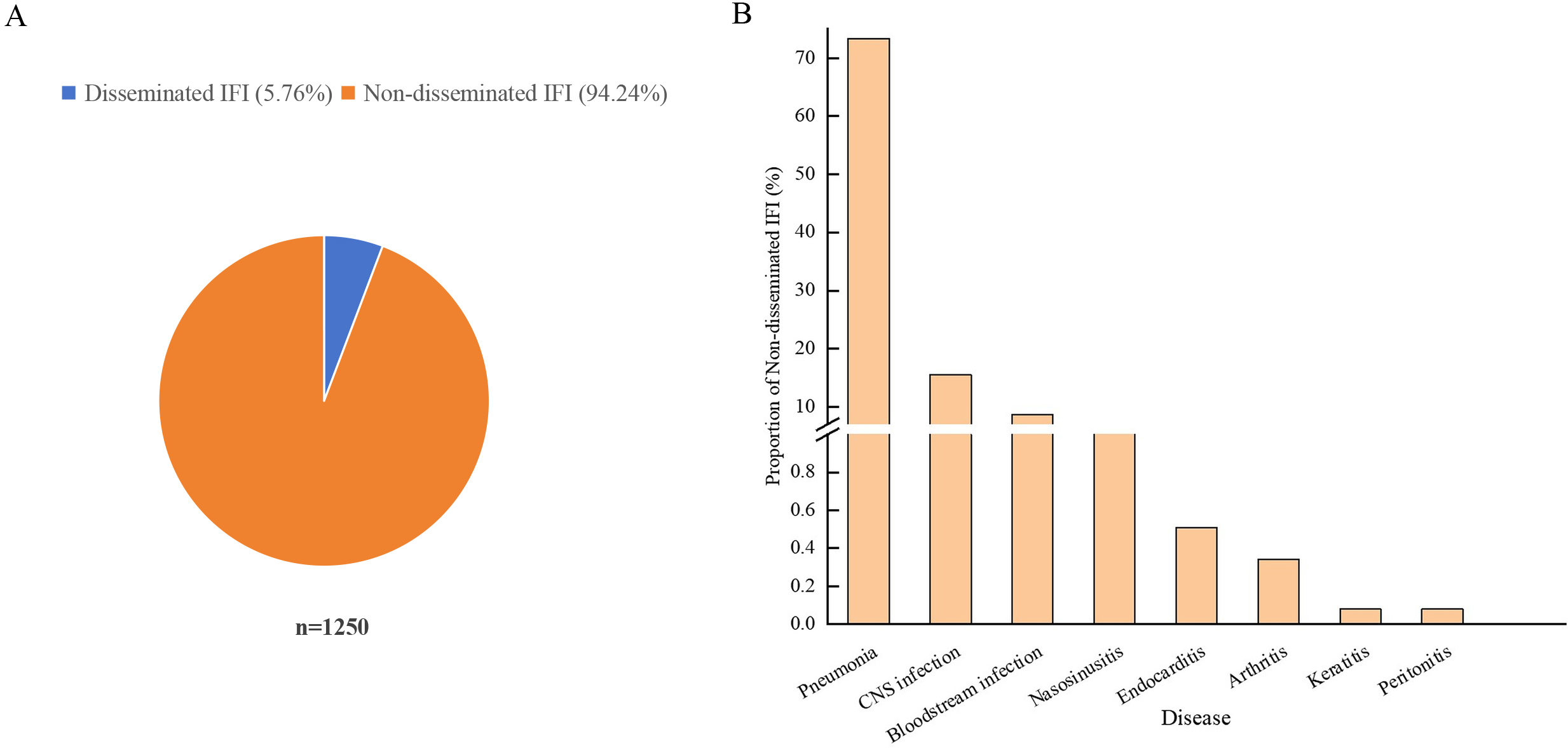 Fig. 3.
Fig. 3.
Clinical spectrum of IFI in hospitalised children. (A) Proportion of disseminated versus non-disseminated infections. (B) Proportion of non-disseminated infections by type, including pneumonia, CNS infections, bloodstream infections, nasosinusitis, endocarditis, arthritis, keratitis, and peritonitis. Abbreviation: CNS, central nervous system.
Only patients with pathogen information documented in the FSMRs were included in
this analysis, resulting in 485 episodes (38.8%) with available microbiological
data. The fungal pathogens were: Candida (25.36%, 123/485), Aspergillus (23.71%, 115/485), Cryptococcus (22.06%, 107/485), Pneumocystis (21.44%, 104/485), Mucor (4.12%, 20/485), Talaromyces (1.24%, 6/485), Histoplasma (0.82%, 4/485), Blastomyces (0.62%, 3/485), and Sporotrichum (0.62%, 3/485) (Fig. 4).
Table 2 shows the age-specific distribution of confirmed pathogens
among IFI cases across different age groups. Candida was predominant in
children
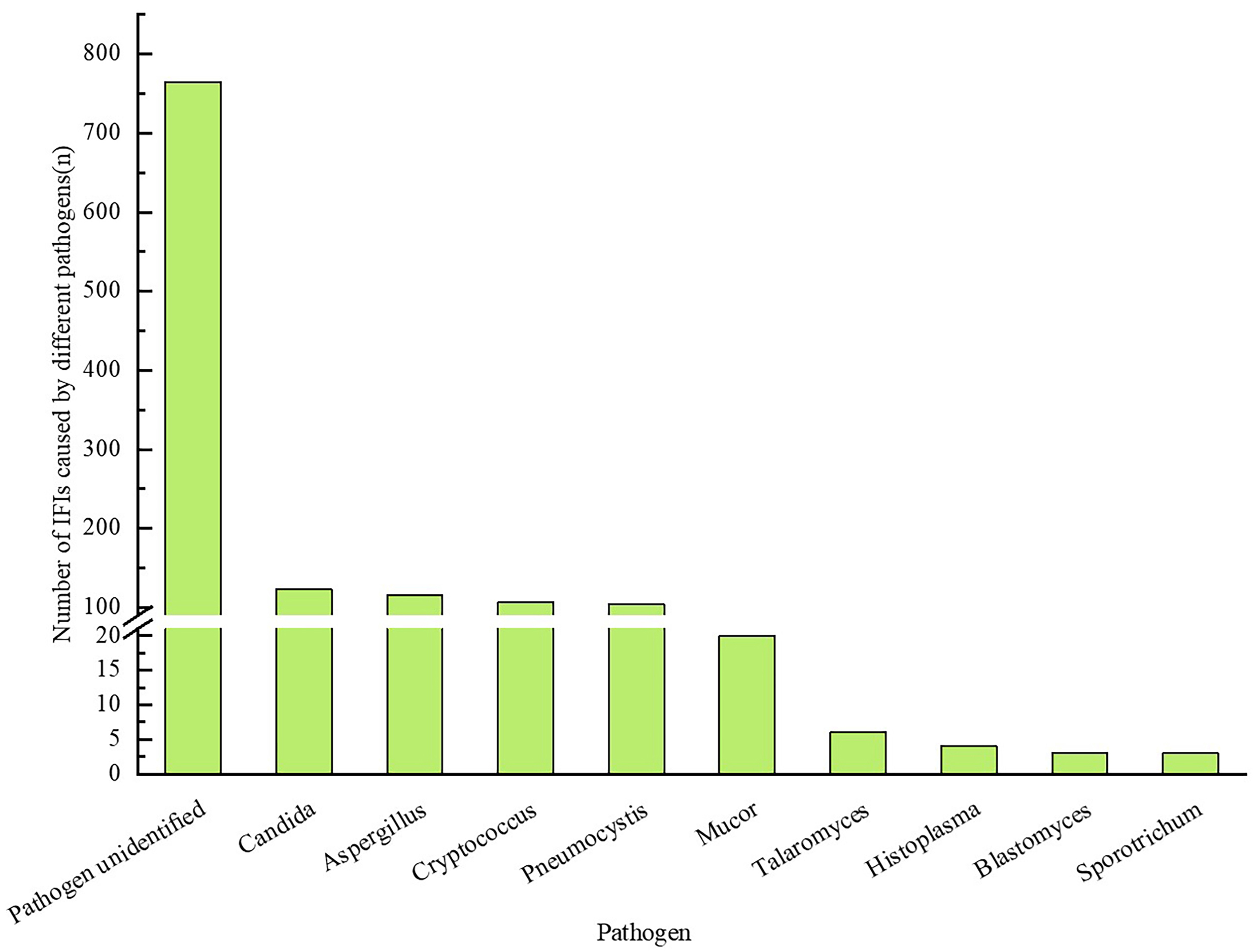 Fig. 4.
Fig. 4.
Number of IFI cases stratified.
| Category | Total, n | Candida, n (%) | Aspergillus, n (%) | Cryptococcus, n (%) | Pneumocystis, n (%) | Mucor, n (%) | Others, n (%) | ||
| Year | |||||||||
| 2016 | 60 | 23 (38.33) | 7 (11.67) | 19 (31.67) | 5 (8.33) | 1 (1.67) | 5 (8.33) | ||
| 2017 | 55 | 22 (40.00) | 7 (12.73) | 17 (30.91) | 5 (9.09) | 2 (3.64) | 2 (3.64) | ||
| 2018 | 32 | 6 (18.75) | 9 (28.12) | 5 (15.62) | 7 (21.88) | 3 (9.38) | 2 (6.25) | ||
| 2019 | 78 | 19 (24.36) | 23 (29.49) | 20 (25.64) | 12 (15.38) | 1 (1.28) | 3 (3.85) | ||
| 2020 | 66 | 13 (19.70) | 16 (24.24) | 16 (24.24) | 16 (24.24) | 2 (3.03) | 3 (4.54) | ||
| 2021 | 94 | 28 (29.79) | 28 (29.79) | 10 (10.64) | 25 (26.60) | 2 (2.13) | 1 (1.06) | ||
| 2022 | 100 | 12 (12.00) | 25 (25.00) | 20 (20.00) | 34 (34.00) | 9 (9.00) | 0 (0.00) | ||
| Age | |||||||||
| 0–28 days | 22 | 21 (95.45) | 1 (4.54) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | ||
| 123 | 61 (49.59) | 20 (16.26) | 3 (2.44) | 32 (26.02) | 0 (0.00) | 7 (5.69) | |||
| 64 | 17 (26.56) | 20 (31.25) | 16 (25.00) | 4 (6.25) | 3 (4.69) | 4 (6.25) | |||
| 82 | 11 (13.41) | 21 (25.61) | 28 (34.15) | 17 (20.73) | 1 (1.22) | 4 (4.88) | |||
| 137 | 10 (7.30) | 39 (28.47) | 40 (29.20) | 36 (26.28) | 11 (8.03) | 1 (0.73) | |||
| 57 | 3 (5.26) | 14 (24.56) | 20 (35.09) | 15 (26.32) | 5 (8.77) | 0 (0.00) | |||
| Disease type | |||||||||
| Disseminated IFI | 68 | 31 (45.59) | 4 (5.88) | 25 (36.76) | 0 (0.00) | 4 (5.88) | 4 (5.88) | ||
| Non-disseminated IFI | |||||||||
| Pneumonia | 282 | 34 (12.06) | 109 (38.65) | 7 (2.48) | 104 (36.88) | 16 (5.67) | 12 (4.26) | ||
| CNS infection | 111 | 35 (31.53) | 2 (1.80) | 74 (66.67) | 0 (0.00) | 0 (0.00) | 0 (0.00) | ||
| Bloodstream infection | 23 | 22 (95.65) | 0 (0.00) | 1 (4.35) | 0 (0.00) | 0 (0.00) | 0 (0.00) | ||
| Nasosinusitis | 0 | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | ||
| Endocarditis | 0 | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | ||
| Arthritis | 1 | 1 (100) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | ||
| Keratitis | 0 | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | ||
| Peritonitis | 0 | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | ||
For disseminated IFI, Candida and Cryptococcus accounted for 45.59% (31/68) and 36.76% (25/68), respectively. In bloodstream infections, Candida was identified in 95.65% (22/23), while in CNS infections, Cryptococcus and Candida accounted for 66.67% (74/111) and 31.53% (35/111), respectively. In pneumonia cases, Aspergillus and pneumocystis were predominant, representing 38.65% (109/282) and 36.88% (104/282), respectively.
Among hospitalised patients diagnosed with IFI, 84.00% (1050/1250) exhibited identifiable risk factors for fungal infections. The predominant risk factor was haematological malignancy, affecting 36.80% (460/1250) of children, followed by myelosuppression in 18.08% (226/1250). Immune deficiency, prematurity, and organ transplantation were observed in 8.72% (109/1250), 7.20% (90/1250), and 4.4% (55/1250) of the patients, respectively. Among those with immunodeficiency disorders, organ transplant recipients, and patients with haematological malignancies, Pneumocystis was the predominant pathogen, followed by Candida and Aspergillus. In premature infants and surgical cases, Candida predominated, while Mucor was the leading pathogen in patients with diabetes (Supplementary Table 2).
This study assessed LOS and hospitalisation costs in paediatric patients with
IFI (Table 3). Significant differences were observed based on age, region,
disease, and pathogen (p
| Category | LOS [days, Median (IQR)] | H/U | p-value | Expenditure [USD, Median (IQR)] | H/U | p-value | |
| Age group | 45.4392 | 27.7998 | |||||
| 0–28 days | 32 (16–49) | 7828.02 (3932.51–13,113.69) | |||||
| 18 (10–35) | 4003.26 (1822.39–8808.02) | ||||||
| 16 (10–28) | 4011.02 (2372.01–6367.87) | ||||||
| 17 (9–28) | 4099.94 (1853.97–8122.16) | ||||||
| 14 (8–22) | 4268.01 (1971.15–7868.83) | ||||||
| 16 (8–24) | 5607.21 (2733.97–10,731.06) | ||||||
| Region | 22.3903 | 0.0010 | 20.0842 | 0.0027 | |||
| Northeast China | 14 (7–36) | 4427.93 (1294.05–13,304.77) | |||||
| North China | 18 (10–31) | 5963.46 (2760.36–11,281.56) | |||||
| East China | 16 (8–29) | 4541.68 (1829.32–9647.31) | |||||
| South China | 11 (6–21) | 3661.27 (1508.86–6561.97) | |||||
| Central China | 16 (8–33) | 3949.74 (1760.14–8801.70) | |||||
| Northwest China | 19 (11–28) | 4526.09 (2743.17–6934.69) | |||||
| Southwest China | 15 (8–27) | 3530.44 (1816.36–7869.42) | |||||
| Residence | 0.0061 | 0.9378 | 0.7641 | 0.3821 | |||
| Rural | 17 (9–29) | 4296.45 (2088.57–8908.03) | |||||
| Urban | 16 (9–33) | 4431.53 (2263.19–8908.03) | |||||
| Disease type | 108.1920 | 60.7752 | |||||
| Disseminated IFI | 25 (7–44) | 5942.60 (1635.03–12,761.15) | |||||
| Pneumonia | 15 (9–23) | 3947.78 (2092.17–6939.30) | |||||
| CNS infection | 28 (7–58) | 6711.32 (1745.63–13,867.10) | |||||
| Bloodstream infection | 32 (18–45) | 7774.66 (4287.36–11,902.03) | |||||
| Nasosinusitis | 8 (4–9.5) | 2259.63 (1476.30–3704.76) | |||||
| Endocarditis | 20 (6–34) | 4791.32 (2500.21–22,032.34) | |||||
| Arthritis | 35 (29–46) | 8162.46 (4349.41–12,758.40) | |||||
| Keratitis | 6 (6–6) | 581.36 (581.36–581.36) | |||||
| Peritonitis | 34 (34–34) | 6712.83 (6712.83–6712.83) | |||||
| Pathogen | 22.7164 | 0.0069 | 20.8394 | 0.0134 | |||
| Candida | 22 (8–43) | 6262.34 (1755.02–12,426.40) | |||||
| Aspergillus | 13 (8–21) | 4087.72 (2375.70–7997.47) | |||||
| Cryptococcus | 22 (6–23) | 4252.98 (1614.97–12,119.31) | |||||
| Pneumocystis | 15 (10–20) | 4925.80 (2732.27–10,662.48) | |||||
| Mucor | 19 (8–40) | 7259.04 (2066.23–15,740.04) | |||||
| Talaromyces | 19 (7–36) | 9686.49 (1496.05–15,604.36) | |||||
| Histoplasma | 23 (11–43) | 1589.24 (389.29–7231.84) | |||||
| Blastomyces | 8 (7–9) | 1198.37 (859.28–1358.10) | |||||
| Sporotrichum | 18 (8–27) | 4021.45 (3644.92–13,520.24) | |||||
Among the discharged patients, 181 (14.48%) died, all of whom were included in
the analysis. Mortality varied significantly across age groups (Table 4).
Children aged
| Categories | Deaths, n (%) | Survivors, n (%) | p-value | ||
| Sex (n, %) | 0.821 | 0.365 | |||
| Male | 100 (13.72) | 629 (86.28) | |||
| Female | 81 (15.55) | 440 (84.45) | |||
| Age group (n, %) | 25.15 | ˂0.0001 | |||
| 0–28 days | 7 (8.54) | 75 (91.46) | |||
| 49 (19.60) | 201 (80.40) | ||||
| 30 (17.05) | 146 (82.95) | ||||
| 49 (18.99) | 209 (81.01) | ||||
| 41 (10.90) | 335 (89.10) | ||||
| 5 (4.63) | 103 (95.37) | ||||
| Disease type (n, %) | Fisher’s Exact Test | 0.299 | |||
| Disseminated IFI | 12 (16.67) | 60 (83.33) | |||
| Pneumonia | 134 (15.51) | 730 (84.49) | |||
| CNS infection | 26 (14.21) | 157 (85.79) | |||
| Bloodstream infection | 8 (7.84) | 94 (92.16) | |||
| Endocarditis | 1 (16.67) | 5 (83.33) | |||
| Nasosinusitis | 0 (0.00) | 17 (100.00) | |||
| Arthritis | 0 (0.00) | 4 (100.00) | |||
| Keratitis | 0 (0.00) | 1 (100.00) | |||
| Peritonitis | 0 (0.00) | 1 (100.00) | |||
| Pathogen (n, %) | Fisher’s Exact Test | 0.301 | |||
| No identified pathogen | 107 (13.99) | 658 (86.01) | |||
| Candida | 19 (15.45) | 104 (84.55) | |||
| Aspergillus | 15 (13.04) | 100 (86.96) | |||
| Cryptococcus | 12 (11.21) | 95 (88.79) | |||
| Pneumocystis | 25 (24.04) | 79 (75.96) | |||
| Mucor | 2 (10.00) | 18 (90.00) | |||
| Histoplasma | 1 (25.00) | 3 (75.00) | |||
| Talaromyces | 0 (0.00) | 6 (100.00) | |||
| Blastomyces | 0 (0.00) | 3 (100.00) | |||
| Sporotrichum | 0 (0.00) | 3 (100.00) | |||
IFI remains a major cause of morbidity and mortality in immunocompromised children. Its global burden is rising, driven by the increased use of immunosuppressive therapies and the presence of more complex underlying conditions [2, 18]. Children represent a considerable proportion of IFI cases, with incidence and associated healthcare costs continuing to escalate worldwide [3]. This large nationwide epidemiological study of paediatric IFI was conducted across 30 tertiary hospitals in seven regions of China.
Between January 2016 and December 2022, 1250 paediatric IFI cases were diagnosed in China, corresponding to a prevalence of 0.14‰ among hospitalised patients, which is lower than global data [19]. To date, no epidemiological studies on paediatric IFI have been reported in China. Internationally, incidence rates in adults are higher: Spain (0.243‰, 2017–2021), the Netherlands (0.186‰ in 2017), France (0.216–0.236‰, 2012–2018), and the United States (approximately 0.272‰, 2006–2015) [20, 21, 22, 23]. The lower incidence in children is largely attributed to reduced exposure to risk factors such as chronic obstructive pulmonary disease, diabetes, and malignant tumours, which are more common in adults. These comorbidities significantly compromise immune function, creating a favourable environment for fungal infections [24]. Despite the overall low prevalence, hospitalisation rates for IFI rose from 0.09‰ in 2016 to 0.24‰ in 2022. This upward trend, particularly pronounced after 2019, suggests shifting epidemiological patterns and highlights global concerns over the growing complexity and vulnerability of hospitalised children [25]. The marked increase after 2019 may be partially linked to the COVID-19 pandemic. During this period, some children likely delayed medical visits for non-COVID-19 conditions, including those predisposing to IFI. Moreover, several studies show that severe COVID-19 increases IFI risk, particularly Aspergillus- and Candida-related infections, due to factors such as dual immune damage from both virus and host, intensive care unit-related complications, widespread immunosuppressive therapy, microbial imbalance, and antibiotic overuse [26]. Additionally, the growing use of advanced diagnostic technologies, including metagenomic sequencing in China, may have further contributed to increased IFI detection rates [27].
Our findings showed that children aged
The pathogen detection rate in this study was low (38.8%), consistent with the findings of Puerta-Alcalde et al. [30], highlighting persistent challenges in diagnosing paediatric IFI. Contributing factors include the limited sensitivity of available diagnostic tests, difficulty in obtaining adequate paediatric samples, especially from sterile body sites, and prior antifungal therapy masking pathogen detection. Reliance on medical records for pathogen identification presents limitations, as complex clinical scenarios or insufficient emphasis on microbiological confirmation may result in underdiagnosis. The high proportion of missing pathogen data raises concerns about selection and information bias, as confirmed cases may disproportionately represent patients with severe disease or obvious clinical features. Thus, caution is warranted when interpreting pathogen frequency or age-related distribution, since these patterns may not accurately reflect the true epidemiology.
However, our findings align with previous reports [18] identifying Candida, Aspergillus, Cryptococcus, Pneumocystis, and Mucor as the predominant paediatric IFI pathogens. Candida species are most common, especially in neonates and infants [25]. Aspergillus is the leading invasive mold among children aged 1–3 years and in immunocompromised populations, while non-Aspergillus molds such as Mucorales are increasingly reported in paediatric cancer and diabetes. Rising rates of Cryptococcus and Pneumocystis among older children may reflect age-related changes in immunity, environmental exposure, and comorbidities. Given the shifting pathogen spectrum, the emergence of antifungal resistance, especially among Candida and Aspergillus, represents a growing clinical concern. Recent surveillance studies highlight increasing azole resistance in Aspergillus fumigatus and the global spread of multidrug-resistant Candida strains, including C. auris, which complicate management and may worsen outcomes in paediatric IFI [31, 32]. These trends underscore the importance of ongoing antifungal susceptibility monitoring and integration of resistance data into therapeutic decision-making.
In our cohort, 94.24% of paediatric IFI were non-disseminated, with invasive fungal pneumonia being the most common presentation, confirming the respiratory tract as a principal entry point for fungal pathogens in immunocompromised children [33]. CNS and bloodstream infections were the next most frequent, consistent with previous reports identifying these sites as highly vulnerable in high-risk populations. Pathogen distribution correlated with disease syndromes: Candida and Cryptococcus predominated in disseminated IFI and CNS infections, consistent with multicentre studies highlighting their systemic invasiveness and the need to consider them in children presenting with neurological symptoms [34, 35]. In invasive fungal pneumonia, Pneumocystis was the predominant pathogen, followed by Aspergillus, reflecting recent data on immunocompromised paediatric patients [36, 37]. Bloodstream infections were primarily caused by Candida species, the leading fungal bloodstream pathogen in hospitalised children, especially in neonates and infants [38]. These findings highlight the critical need for targeted diagnostic algorithms and empiric antifungal regimens tailored to address the most likely pathogens across distinct clinical presentations.
This study reports a high prevalence of IFI risk factors, particularly haematological malignancies (36.80%) and myelosuppression (18.08%), consistent with previous studies and clinical guidelines that identify severe neutropenia and haematological malignancies as key risk factors [2, 39, 40]. Both conditions impair host immunity, increasing susceptibility to opportunistic fungi such as Candida and Aspergillus. The study also confirms established epidemiological trends: Pneumocystis is common in patients with immunodeficiencies or organ transplant recipients. Candida remains a leading pathogen in preterm infants and surgical patients [41], while Mucor is closely associated with diabetes, reflecting global evidence of its rising incidence among children with hyperglycaemia and ketoacidosis [42].
Cost analysis indicates that non-neonatal paediatric IC in the United States
incurs an average incremental hospital charge of approximately
In our study, the mortality rate during hospitalisation was 14.48%, falling within the lower range of reported values (10–70%) [2]. This comparatively lower rate may partly reflect the lack of post-discharge follow-up, as only in-hospital outcomes were captured in our analysis. Disease type strongly influences prognosis, with previous studies reporting higher mortality in subgroups such as patients with disseminated invasive fungal disease or CNS involvement [2]. Our findings are consistent with these reports, likely due to diagnostic delays, multi-organ dysfunction, and inadequate drug penetration, particularly in CNS infections. In our cohort, pathogen-specific mortality for pneumocystis pneumonia was 24.04%, exceeding the 11.7% reported in a national study from the United States [45], underscoring the urgent need for effective prevention and early intervention in immunodeficient children in China.
This study has several limitations. First, because the data were derived from discharge face sheets, information on the causative pathogen was incomplete in some cases, limiting a full characterisation of the pathogen spectrum. Second, long-term follow-up data for hospitalised patients were unavailable, leading to missing information on severe or fatal cases and affecting evaluations of LOS and associated costs. Third, FSMRs contained only basic patient details, and the absence of clinical information and laboratory test results constrained further analyses. To gain deeper insights into this disease, a prospective multicentre cohort study that integrates comprehensive clinical and laboratory data, pathogen distribution, antifungal resistance dynamics, and structured long-term follow-up is warranted.
In this 7-year nationwide retrospective study of 1250 hospitalised children with IFI across China, hospitalisation rates rose sharply after 2019, with haematological malignancies and bone marrow suppression identified as the primary predisposing factors. Candida, Aspergillus, and Cryptococcus are the most prevalent pathogens, predominantly causing pneumonia, CNS infections, and bloodstream infections. Despite advances in antifungal therapy, IFI-related mortality remains high, particularly in disseminated and CNS infections, imposing significant socioeconomic and healthcare burdens. These findings highlight the urgent need for strengthened epidemiological surveillance and targeted interventions to improve paediatric IFI outcomes in China.
• The FRCPD established the FUTURE database, collecting FSMRs from 30 tertiary children’s hospitals in China to investigate IFI in children between 2016 and 2022.
• A total of 1250 IFI cases were identified among 9,008,615 hospital admissions, with a prevalence of 0.14‰, lower than rates reported in Spain, the Netherlands, France, and the United States.
• IFI hospitalisation rates rose markedly from 0.09‰ in 2016 to 0.24‰ in 2022, with a notable increase post-2019, potentially associated with the COVID-19 pandemic and improvements in diagnostic technology.
• The most common pathogens were Candida, Aspergillus, and Cryptococcus, with Candida predominating in neonates and bloodstream infections, Aspergillus in pneumonia, and Cryptococcus in CNS infections.
• Haematological malignancies (36.80%) and myelosuppression (18.08%) were the leading risk factors for IFI, with Pneumocystis the primary pathogen in immunocompromised patients and Candida in premature infants.
• The median hospitalisation cost reached
Due to privacy restrictions, the raw data cannot be shared publicly; however, aggregated data are available from the corresponding author upon reasonable request.
WYF, XYW and GL conceived and designed the study. WYF and XYW collected the data and designed the analysis. GL, WYF, XYW and GSF interpreted the data. WYF wrote the first draft of the paper. All authors contributed to the important editorial changes in the manuscript. All authors have read and agreed to the published version of the manuscript. All authors had access to the full dataset and agreed to take responsibility for the integrity and accuracy of the research.
This study was conducted following the ethical principles of the Declaration of Helsinki and received approval from the Ethics Committee of Beijing Children’s Hospital, Capital Medical University (Approval Number: [2025]-E-028-R). Owing to the retrospective design of the study, involving only secondary analysis of aggregated and de-identified medical record data, the requirement for informed consent was waived by the ethics committee following national regulations (Measures for the Ethical Review of Biomedical Research Involving Humans in China, 2023 edition).
We are grateful to investigators from members of the Futang Research Center of Pediatric Development (FRCPD).
This work was supported by the Capital’s Funds for Health Improvement and Research (2024–1-2092); 2022 Beijing Major Epidemic Prevention and Control Specially Construction Project (2-1-2-6-15); Beijing Municipal Administration of Hospitals Incubating Program (PX2024042); and Training Plan for High level Public Health Technical Talents Construction Project (Discipline Leader-02-02).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/BJHM53104.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




