1 Department of Respiratory Medicine, Kaihua County Traditional Chinese Medicine Hospital, 324300 Quzhou, Zhejiang, China
2 Department of Respiratory Medicine, The Second Affiliated Hospital, Zhejiang University School of Medicine, 310000 Hangzhou, Zhejiang, China
Abstract
Lambert-Eaton myasthenic syndrome (LEMS) is a rare autoimmune disorder of the neuromuscular junction that is frequently associated with small-cell lung cancer (SCLC). It is characterized by proximal muscle weakness, reduced deep tendon reflexes, and autonomic symptoms, all of which can significantly impair quality of life and complicate oncologic management. Early recognition and timely intervention are crucial to improving clinical outcomes.
We report a case of a 76-year-old man with extensive-stage SCLC and chronic renal insufficiency whose LEMS worsened during durvalumab immunotherapy. Despite continued chemotherapy and immunotherapy, his symptoms progressively worsened. Chemotherapy failed to improve his neurological condition, with subsequent recurrent episodes of muscle weakness. Subsequent administration of intravenous immunoglobulin (IVIG) combined with pyridostigmine led to marked neurological improvement and restoration of muscle strength to grade 5, enabling continuation of antitumor therapy. The patient subsequently received three cycles of second-line lurbinectedin chemotherapy, during which inflammatory responses, anemia, and venous thrombosis developed but were effectively managed with supportive treatment.
Following chemotherapy and immunotherapy, the patient’s limb muscle strength recovered to grade 5, followed by recurrent muscle weakness symptoms. Hematologic and biochemical parameters gradually normalized, and inflammatory markers returned to baseline. At discharge, his clinical condition was stable.
This case highlights the importance of early recognition and standardized management of paraneoplastic LEMS in enhancing functional recovery and enabling the safe continuation of antitumor therapy.
Keywords
- small-cell lung cancer
- Lambert-Eaton myasthenic syndrome
- immune checkpoint inhibitors
- paraneoplastic syndrome
- case report
Lambert-Eaton myasthenic syndrome (LEMS) is a rare autoimmune disorder of the neuromuscular junction that primarily targets voltage-gated calcium channels on the presynaptic membrane, resulting in reduced acetylcholine release and impaired neuromuscular transmission [1]. Clinically, it is characterized by proximal muscle weakness, diminished or absent deep tendon reflexes, and autonomic symptoms such as dry mouth, constipation, and erectile dysfunction. These manifestations can markedly impair patients’ quality of life, leading to difficulties in walking, dysphagia, and, in severe cases, respiratory failure due to diaphragmatic involvement, thereby increasing morbidity and mortality [2].
LEMS often occurs as a paraneoplastic neurological syndrome and is strongly associated with malignancy, with approximately 50–60% of cases linked to small-cell lung cancer (SCLC) [3]. SCLC is a highly aggressive neuroendocrine carcinoma. When LEMS coexists with SCLC, it typically reflects a neuroendocrine tumor phenotype in which cross-reactive antibodies—such as anti-P/Q-type voltage-gated calcium channel antibodies—trigger autoimmune attacks. This paraneoplastic association complicates cancer management and may delay tumor diagnosis, as LEMS symptoms can precede tumor detection by several months [4, 5, 6].
Immune checkpoint inhibitors (ICIs) have become a cornerstone of SCLC treatment. By blocking the programmed cell death protein 1/programmed death-ligand 1 (PD-1/PD-L1) pathway, ICIs enhance T-cell-mediated antitumor immunity and significantly improve objective response rates and progression-free survival [7]. For example, durvalumab, a PD-L1 inhibitor, combined with chemotherapy, has demonstrated improved survival outcomes in patients with advanced SCLC [8]. However, while ICIs potentiate antitumor immunity, they may also provoke excessive immune activation, resulting in immune-mediated adverse events (imAEs) that can affect nearly all organ systems [9]. Common imAEs include dermatitis, colitis, hepatitis, pneumonitis, and endocrine disorders such as thyroiditis and hypophysitis, which can be life-threatening in severe cases [10]. Therefore, early recognition, accurate diagnosis, and standardized management of imAEs are critical during immune checkpoint inhibitor (ICI) therapy.
We report the case of a 76-year-old man with extensive-stage SCLC who developed LEMS during durvalumab immunotherapy. He presented with progressive limb weakness and reduced reflexes, and was diagnosed with LEMS based on electrophysiological testing, elevated inflammatory markers, and increased serum pro-gastrin-releasing peptide levels. Following treatment with intravenous immunoglobulin and pyridostigmine, his muscle strength improved to grade 5. He subsequently received three cycles of lurbinectedin chemotherapy, achieving partial remission of inflammation and tumor burden. The distinctive feature of this case is that LEMS partially improved after initial antitumor therapy but continued to relapse after discharge; however, following lurbinectedin treatment, the lung cancer did not recur, suggesting that lurbinectedin demonstrated good tumor control in this case of small-cell lung cancer complicated by LEMS. The case underscores the importance of early detection and appropriate management of LEMS to optimize functional recovery and maintain continuity of oncologic therapy. The report follows the case report (CARE) guidelines to ensure completeness and transparency; the corresponding checklist is provided as Supplementary Material [11].
A 76-year-old male was admitted in February 2025 with a six-month history of generalized limb weakness that had markedly worsened in the preceding week. On admission, he was alert, with fair mental status, stable weight, preserved appetite, normal sleep, and intact bowel and urinary function. He had a 10-year history of hypertension under regular medical control, chronic renal insufficiency with multiple renal cysts and nephrolithiasis, and a known diagnosis of extensive-stage small-cell lung cancer. He had previously received combination chemotherapy with etoposide (Batch No. ABIK4006; Qilu Pharmaceutical Co., Ltd., Jinan, China) and carboplatin (Batch No. 4E029CA4; Qilu Pharmaceutical Co., Ltd., Jinan, China), followed by durvalumab (Imfinzi; Batch No. BBKN; AstraZeneca, Cambridge, UK) maintenance immunotherapy. During treatment, he developed progressive limb weakness. Treatment with intravenous immunoglobulin (IVIG) (Batch No. 202307060; Shandong Taibang Biological Products Co., Ltd., Tai’an, China) and pyridostigmine (Batch No. C1212024223; Shanghai Zhongxi Pharmaceutical Co., Ltd., Shanghai, China) led to significant symptomatic improvement. The patient also had a history of cerebral infarction with residual multiple lacunar lesions on imaging, but no apparent neurological deficits. There was no history of diabetes mellitus or coronary artery disease. He was a long-term resident of Wenzhou and a former manual laborer, with a long-standing history of smoking and occasional alcohol consumption. He had no known drug allergies. Both parents had died from cerebrovascular and cardiovascular diseases, and there was no family history of neuromuscular disorders or malignancy.
In July 2024, the patient presented to the Department of Neurology at the Second Affiliated Hospital, Zhejiang University School of Medicine with limb weakness but did not receive specific treatment at that time. A high-resolution computed tomography (HRCT) scan of the chest without contrast (Fig. 1) revealed a mass lesion in the left upper lobe and patchy consolidation in the right lower lobe. Bronchoscopic biopsy showed atypical naked nuclei in the mediastinal 4R lymph node and the anterior segment of the right upper lobe, consistent with small cell carcinoma. Immunohistochemistry (IHC) demonstrated positivity for pan-cytokeratin (CK-PAN), thyroid transcription factor-1 (TTF-1), synaptophysin, chromogranin A, cluster of differentiation (CD) 56, and insulinoma-associated protein 1 (INSM1), with a Kiel-67 (Ki-67) index of approximately 60%, confirming the diagnosis of SCLC (Fig. 2) [12]. Cytological examination of the 4R lymph node also revealed small cell carcinoma cells. Electromyography performed on 5 August 2024, demonstrated a low-frequency decrement and high-frequency increment across multiple muscles; reduced amplitudes and absent responses in motor and sensory nerves of the right lower limb; and neurogenic changes in several examined muscles (Table 1). The patient received 2 cycles of etoposide plus carboplatin (EC), followed by 4 cycles of EC combined with durvalumab, and subsequently 3 cycles of durvalumab maintenance therapy, after which muscle strength improved.
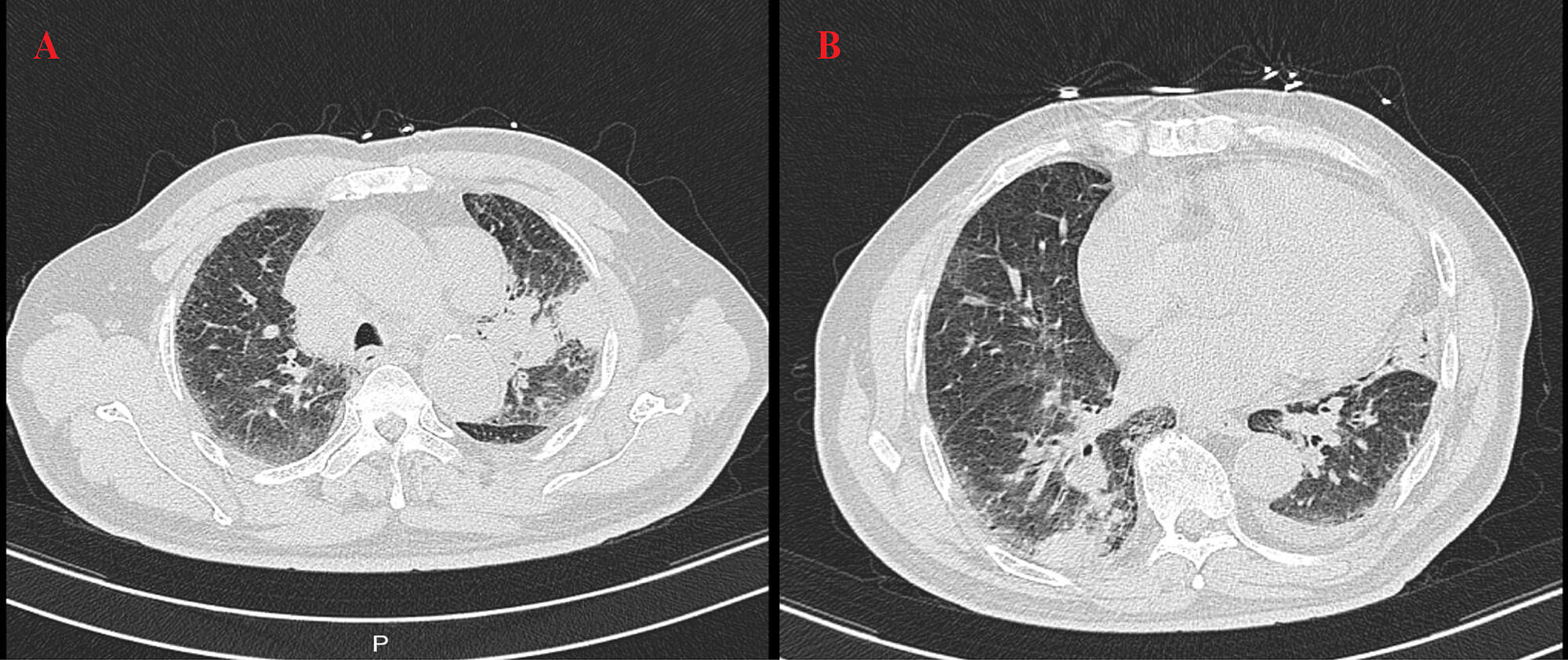 Fig. 1.
Fig. 1.
Baseline chest high-resolution computed tomography (HRCT) findings. HRCT of the chest without contrast performed on 6 July 2024 showed a mass lesion in the left upper lobe (A) and patchy consolidation in the right lower lobe (B).
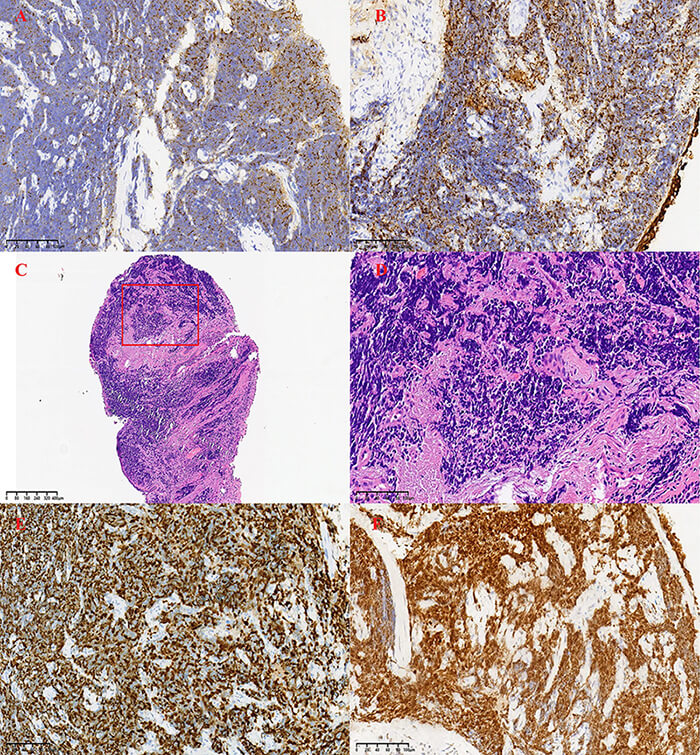 Fig. 2.
Fig. 2.
Immunohistochemical and histological staining results of small-cell lung carcinoma. (A) Chromogranin A showing positive staining (×400). (B) Pan-cytokeratin positive staining (×400). (C) Hematoxylin and Eosin (HE) staining, low magnification (×40). (D) HE staining, high magnification (×400), showing atypical naked nuclei. (E) Kiel-67 (Ki-67) antigen staining with a proliferation index of approximately 60% (×400). (F) Thyroid transcription factor-1 (TTF-1) positive staining (×400). Scale bars = 100 µm in (A,B,D–F) and 400 µm in (C).
| Test category | Abnormal sites | Summary of abnormal findings |
| Repetitive nerve stimulation (RNS) | Bilateral nasalis muscles | Marked decrement at low frequency (~20%+) |
| Bilateral orbicularis oculi muscles | Decrement at low frequency (~10–20%) | |
| Right deltoid | Low-frequency decrement (~15%) | |
| Right trapezius | Low-frequency decrement (~14%) | |
| Right abductor digiti minimi | High-frequency increment (~100%) | |
| Right abductor pollicis brevis | High-frequency increment (70–84%) | |
| Motor nerve conduction | Right peroneal/tibial nerves | Reduced amplitudes in multiple segments |
| Sensory nerve conduction | Right sural nerve | No response elicited |
| Right superficial peroneal and plantar nerves | Reduced amplitudes | |
| Electromyography (EMG) | Right deltoid, first dorsal interosseous, vastus medialis, tibialis anterior | Neurogenic changes |
In February 2025, upon admission, the patient experienced a marked worsening of limb weakness and developed immune-related diabetes. Following treatment with intravenous immunoglobulin combined with pyridostigmine, neurological function improved significantly, and muscle strength returned to normal. Laboratory tests indicated a pronounced systemic inflammatory response, with elevated white blood cell count and C-reactive protein (CRP), anemia, and renal dysfunction. Contrast-enhanced brain magnetic resonance imaging (MRI) in March revealed a metastatic lesion in the left cerebellum, for which whole-brain radiotherapy was performed. In April, further testing revealed markedly elevated CRP, interleukin-6 (IL-6), and pro-gastrin-releasing peptide (ProGRP) levels, suggesting active inflammation and tumor progression. Brain magnetic resonance imaging (MRI) identified a left cerebellar metastatic lesion, which regressed after whole-brain radiotherapy.
In May 2025, the patient developed nausea, vomiting, and dysphagia. Multidisciplinary consultation suggested leptomeningeal metastasis. Cerebrospinal fluid (CSF) analysis revealed an opening pressure of 140 mmH2O and a closing pressure of 110 mmH2O, with a CSF-to-serum albumin ratio of 1:3.2. Paraneoplastic antibody testing was negative, supporting disease progression of SCLC [13].
From June 2025 onward, the patient received lurbinectedin chemotherapy. Inflammatory markers initially increased but subsequently declined. By July, CRP levels had decreased to 46.3 mg/L and normalized by September. Hemoglobin levels rose to approximately 110 g/L, anemia improved, and renal and electrolyte function stabilized. Overall, these findings indicated resolution of systemic inflammation, reduction in tumor burden, and significant clinical improvement.
At the time of discharge in late September 2025 (Table 2), CRP had further
decreased to 5 mg/L (reference range
| Parameter | IVIG pulse therapy for LEMS (February 2025) | Tumor progression and complication management (April 2025) | 1st Lurbinectedin (12 June) | 2nd Lurbinectedin (22 July) | 3rd Lurbinectedin (3 September) | Discharge (September 2025) | Reference range |
| WBC ( |
17.8 | 12.6 | 17.0 | 11.4 | 3.2 | 5.0 | 3.5–9.5 |
| Neutrophil ratio (%) | 81–92 | 88.3 | 92.7 | 78.6 | 93.5 | 65 | 40–75 |
| Lymphocytes ( |
0.22–0.78 | 0.35 | 0.15 | 0.70 | 0.69 | 1.5 | 1.1–3.2 |
| Hemoglobin (g/L) | 114 | 102 | 88 | 84 | 100 | 110 | 130–175 |
| Albumin (g/L) | 26–34 | 30.1 | 31.5 | 30.8 | 30.5 | 35 | 40–55 |
| A/G ratio | 0.59–1.08 | 0.80 | 0.92 | 1.05 | 0.65 | 1.10 | 1.2–2.4 |
| Sodium (mmol/L) | 126.9–134.5 | 131.2 | 133.8 | 133.9 | 128.0 | 136.0 | 137–147 |
| Calcium (mmol/L) | 2.00–2.10 | 2.05 | 2.02 | 2.04 | 2.03 | 2.10 | 2.1–2.6 |
| Magnesium (mmol/L) | 0.57–0.73 | 0.65 | 0.68 | — | — | 0.75 | 0.75–1.02 |
| Urea (mmol/L) | 8.52 | 9.8 | 17.7 | 12.2 | 9.35 | 8.0 | 3.10–8.00 |
| Creatinine (µmol/L) | 123 | 120 | 119.8 | 122.3 | 101.9 | 90 | 57–111 |
| CEA (ng/mL) | 5.8 | 4.0 | 6.3 | 14.2 | 8.1 | 6.0 | |
| CA19-9 (U/mL) | 65.8 | — | — | 67.8 | — | 50 | |
| CYFRA 21-1 (ng/mL) | — | 6.9 | — | 17.2 | 8.9 | 6.0 | |
| ProGRP (pg/mL) | 69.77 | 283.59 | 354.05 | 339.42 | 342.95 | — | |
| CRP (mg/L) | 63.5 | 58.2 | 74.8 | 46.3 | 9.8 | 5 | |
| IL-6 (pg/mL) | — | 465 | — | 1235 | — | ||
| PCT (ng/mL) | 0.64 | 0.52 | 0.68 | — | 0.15 | Normal | |
| NSE (ng/mL) | — | 10.9 | — | 14.6 | 16.6 | — | |
| SCC antigen (ng/mL) | — | 2.4 | — | 2.3 | 1.6 | — |
WBC, white blood cell; A/G, Albumin/Globulin; CA 19-9, carbohydrate antigen 19-9; IVIG, intravenous immunoglobulin; LEMS, Lambert-Eaton myasthenic syndrome; CEA, carcinoembryonic antigen; CYFRA 21-1, cytokeratin 19 fragment antigen 21-1; ProGRP, pro-gastrin-releasing peptide; CRP, C-reactive protein; IL-6, interleukin-6; PCT, procalcitonin; NSE, Neuron-Specific Enolase; SCC, squamous cell carcinoma.
To better illustrate the clinical course and treatment response, serial laboratory parameters were continuously monitored throughout hospitalization. As shown in Table 2, inflammatory markers and tumor markers exhibited dynamic fluctuations corresponding to disease progression and treatment phases, while hemoglobin, albumin, and renal function indices gradually improved following immunotherapy and lurbinectedin chemotherapy.
High-resolution computed tomography (HRCT) (Fig. 3) revealed patchy opacities in the left upper and right lower lobes, accompanied by multiple enlarged hilar and mediastinal lymph nodes (Fig. 4), interstitial inflammation in both lungs (Fig. 5), and a small pleural effusion. These findings were consistent with residual tumor and local progression following treatment for extensive-stage SCLC, demonstrating characteristic imaging features of SCLC.
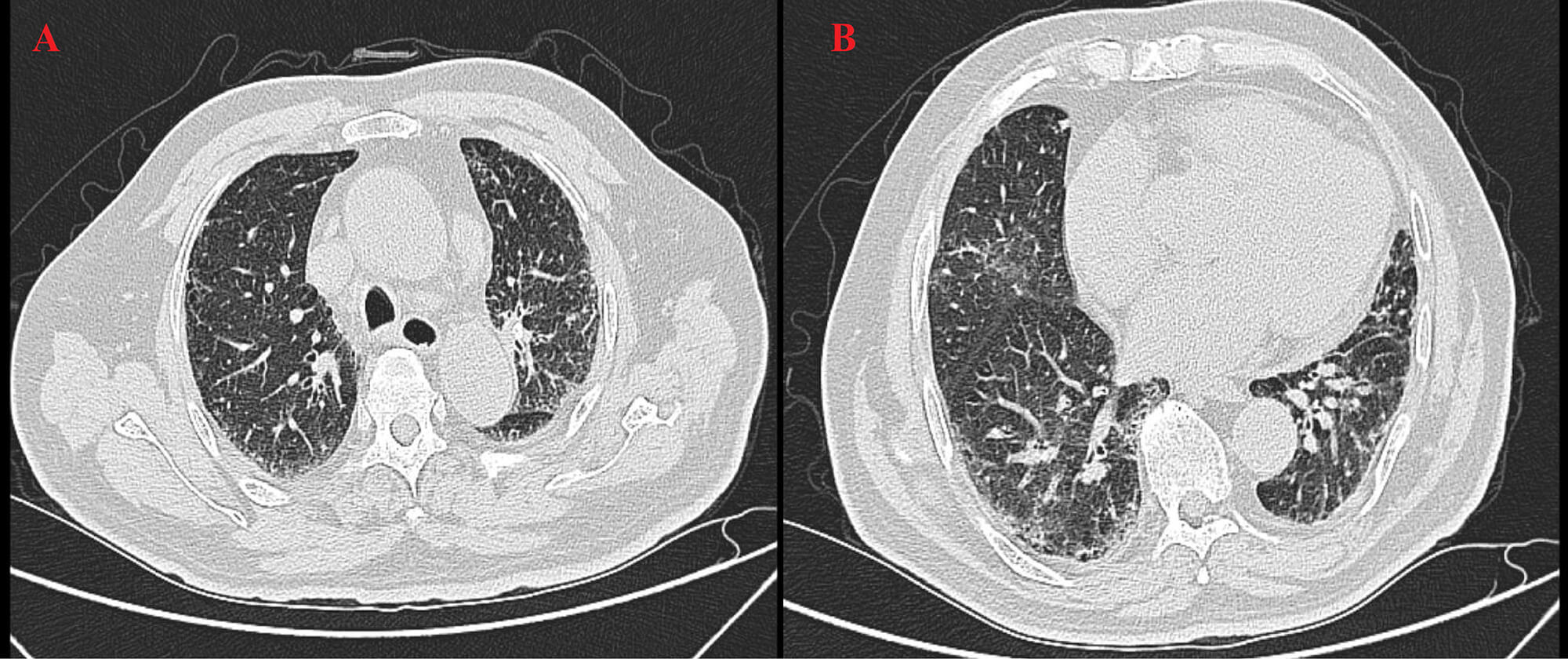 Fig. 3.
Fig. 3.
HRCT on 23 April 2025. This chest scan showed patchy consolidation in the left upper lobe (A) and right lower lobe (B), with mediastinal lymphadenopathy.
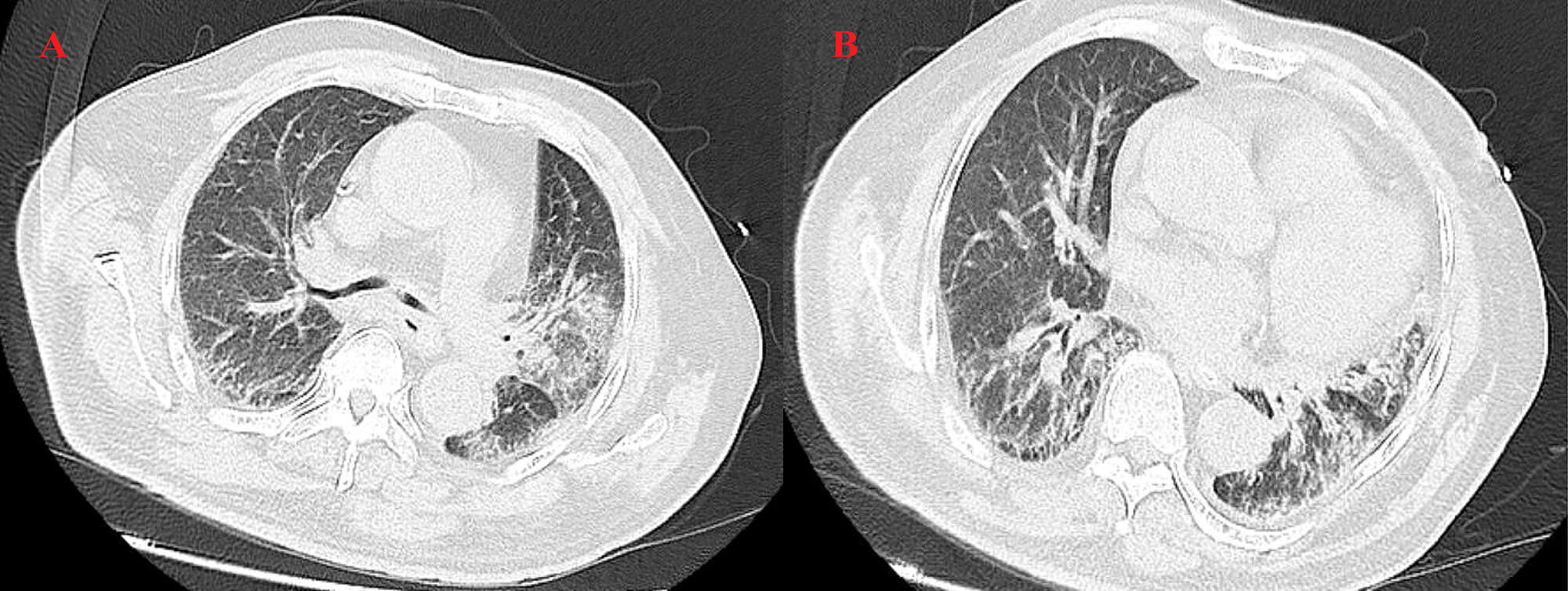 Fig. 4.
Fig. 4.
HRCT on 6 May 2025. (A) The left upper lobe shows a patchy high-density shadow with interstitial changes, indicating post-treatment inflammation and tumor activity. (B) The right lower lobe shows consolidation with diffuse interstitial thickening and mediastinal lymphadenopathy, suggesting inflammatory progression.
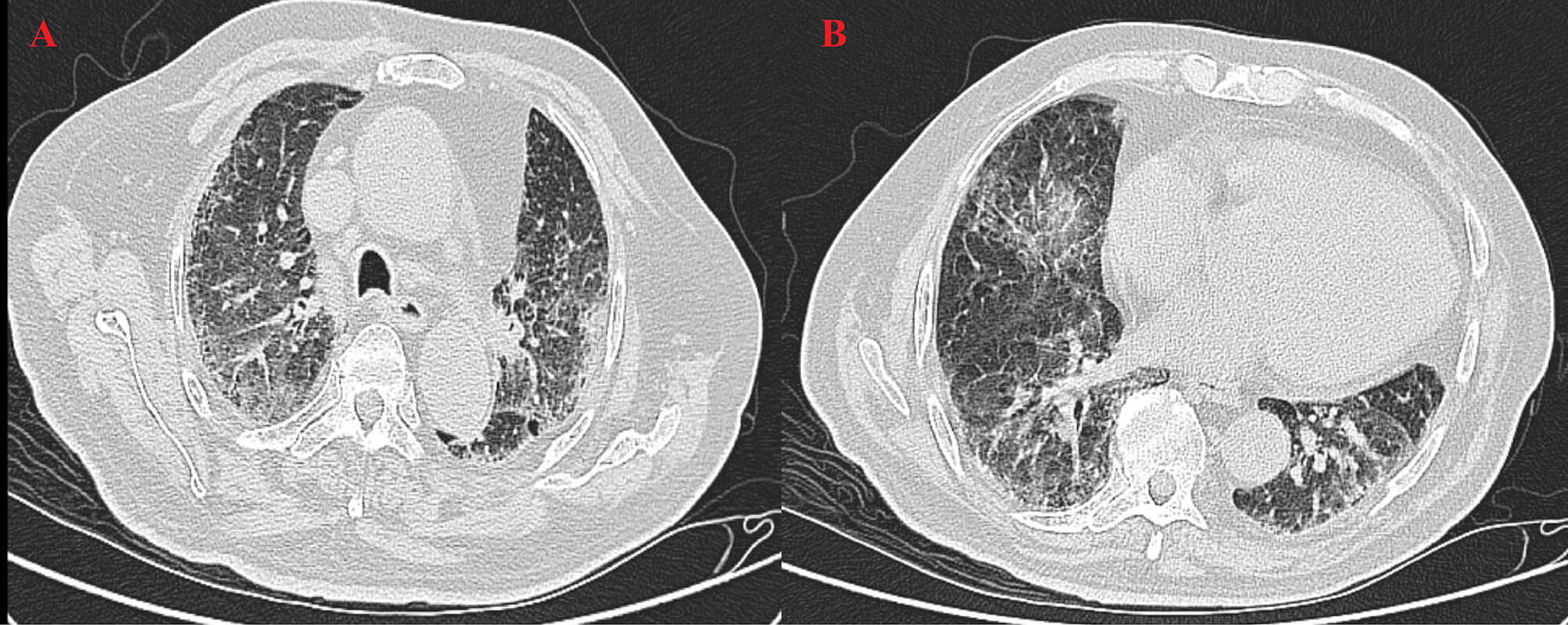 Fig. 5.
Fig. 5.
HRCT on 22 August 2025. (A) The left upper lobe shows fibrotic changes and partial absorption, indicating post-treatment improvement. (B) The right lower lobe shows persistent high-density areas and mild pleural effusion, indicating residual inflammation and need for follow-up.
A follow-up brain MRI in March 2025 showed a metastatic lesion in the left cerebellar hemisphere, for which whole-brain radiotherapy was subsequently administered. Contrast-enhanced brain MRI (Fig. 6) later demonstrated gradual resolution of the left cerebellar metastasis (no visible lesion was detected in August). Multiple lacunar foci were observed in the basal ganglia, corpus callosum, brainstem, and left thalamus, along with white matter rarefaction, confirming the efficacy of whole-brain radiotherapy and excluding alternative neurological causes.
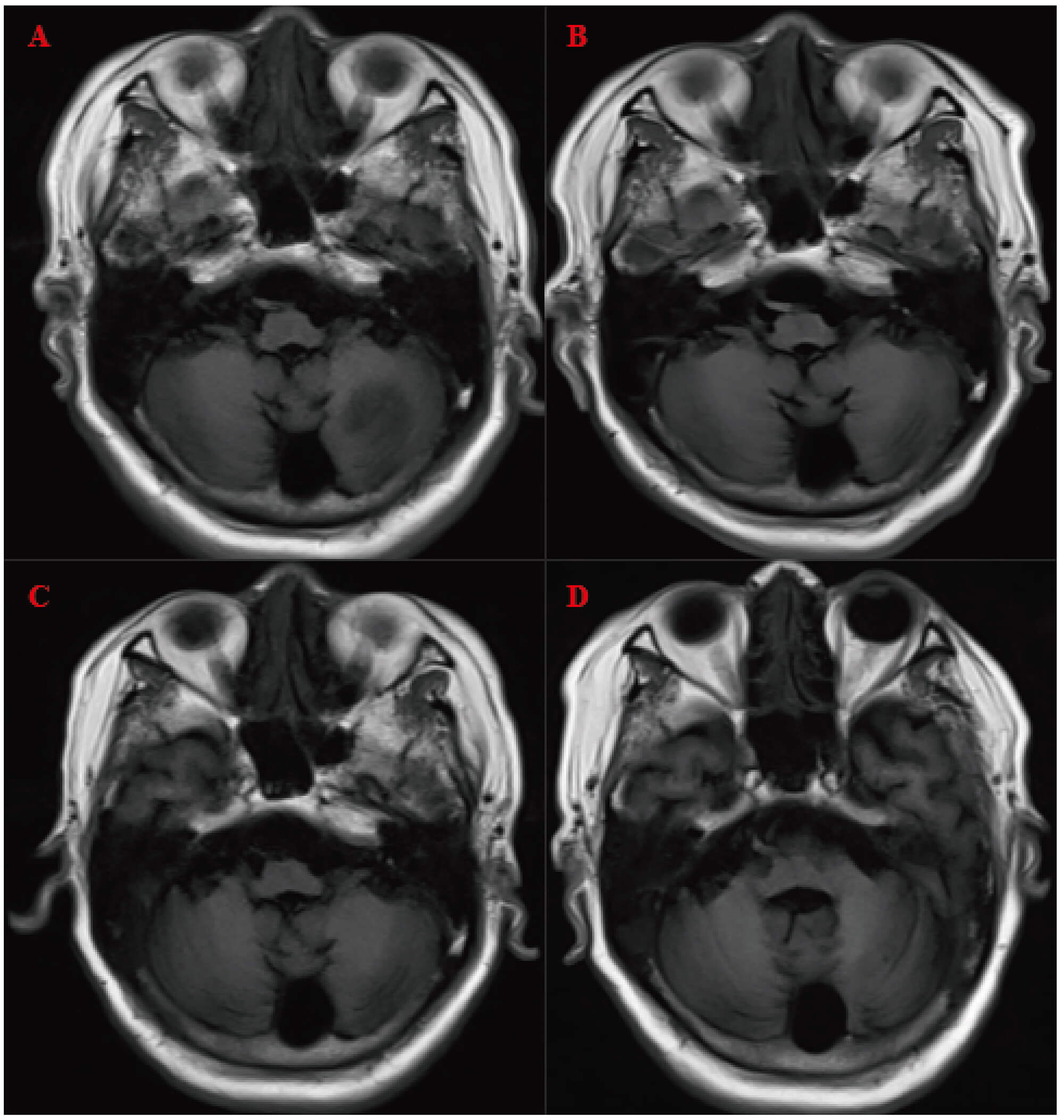 Fig. 6.
Fig. 6.
Contrast-enhanced brain magnetic resonance imaging (MRI). (A–D) were obtained on 18 March, 25 April, 12 May, and 23 August 2025, respectively, demonstrating gradual absorption of the left cerebellar metastatic lesion following whole-brain radiotherapy.
Electromyography (EMG) and evoked potential studies performed on 5 and 6 February 2025, revealed that repetitive nerve stimulation (RNS) produced an amplitude decrement (–11.9% to –13.4%) in the right trapezius at low frequencies (3 Hz, 5 Hz), and a marked amplitude increment (+178% to +350%) in the right abductor digiti minimi and abductor pollicis brevis at high frequencies (20 Hz, 50 Hz). These findings are characteristic of Lambert-Eaton myasthenic syndrome (LEMS), indicating low-frequency decrement and high-frequency facilitation. Motor nerve conduction studies demonstrated decreased amplitudes in the bilateral common peroneal nerves (right, 1.8 mV; left, 0.9 mV), suggesting impairment of the neuromuscular junction. Sensory conduction studies showed mildly reduced amplitudes (6.0–10.3 µV) in the right sural and superficial peroneal nerves with normal latencies, indicating mild sensory nerve involvement.
Tumor marker assays consistently showed elevated levels of pro-gastrin-releasing peptide (ProGRP, 283.59–354.05 pg/mL), carcinoembryonic antigen (CEA, 5.8–14.2 ng/mL), and cytokeratin 19 fragment antigen 21-1 (CYFRA 21-1, 6.9–17.2 ng/mL), consistent with ongoing tumor activity. Following treatment with IVIG (0.4 g/kg/day for 5 days) and pyridostigmine, the patient’s limb muscle strength improved to grade 5, with significant symptom relief. Although the serum anti-P/Q-type voltage-gated calcium channel (VGCC) antibody was negative, the combination of characteristic clinical and electrophysiological findings supported the diagnosis of LEMS.
In February 2025, after follow-up electromyography (EMG) showed worsening of LEMS, IVIG therapy was initiated immediately. The patient received IVIG (0.4 g/kg/day for 5 days) and oral pyridostigmine. After treatment, the patient’s limb muscle strength improved to grade 5, and LEMS-related symptoms marked resolution. In May 2025, a second course of IVIG was administered; however, the therapeutic response was limited, with no significant further improvement in neurological symptoms. Beginning in June 2025, the patient was transitioned to second-line lurbinectedin chemotherapy. During the early treatment phase, mild hematologic abnormalities and inflammatory reactions were observed; these were effectively managed with supportive therapy, enabling continued chemotherapy without interruption. By July, inflammatory markers had declined; hemoglobin levels began to rise in August; and by September, leukocyte counts and CRP levels had normalized. Overall, the patient achieved sustained clinical stability following immunomodulatory and chemotherapy interventions, with significant improvement in both inflammatory status and tumor burden.
Following combined IVIG with pyridostigmine therapy, the patient’s limb muscle strength improved significantly and remained at grade 5, but a recurrence occurred subsequently. The subsequent three cycles of lurbinectedin chemotherapy were completed without major complications. Although the patient developed inflammatory reactions, anemia, and lower-extremity venous thrombosis during treatment, all adverse events were effectively controlled through timely intervention. Hematologic and biochemical parameters gradually improved, inflammatory markers normalized, and renal function showed partial recovery. At discharge, the patient’s clinical condition was stable, with notable improvement in physical performance and quality of life compared with admission. In summary, this case demonstrated favorable short-term outcomes under chemotherapy combined with immunotherapy. However, long-term prognosis and the risk of LEMS recurrence necessitate ongoing monitoring and evaluation. The key clinical events and treatment timeline are presented in Fig. 7.
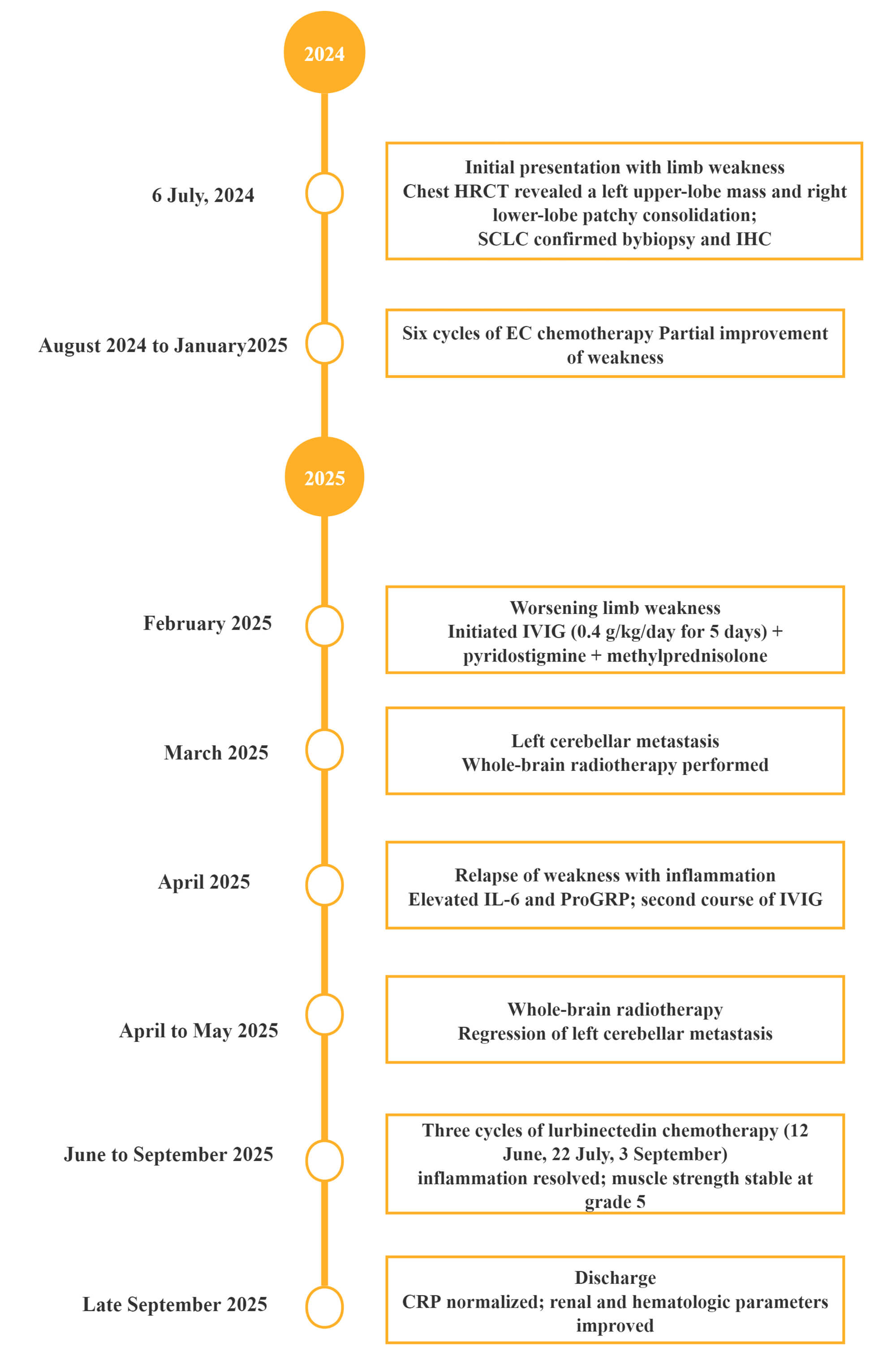 Fig. 7.
Fig. 7.
Timeline of key clinical events and treatments. CT, computed tomography; IHC, immunohistochemistry; EC, etoposide plus carboplatin; IVIG, intravenous immunoglobulin.
LEMS is a rare autoimmune disorder of the neuromuscular junction, characterized by autoantibodies targeting presynaptic P/Q-type VGCCs, resulting in reduced acetylcholine release and impaired neuromuscular transmission [14]. Clinically, patients typically present with proximal lower-limb weakness, diminished or absent deep tendon reflexes, and autonomic symptoms such as dry mouth, constipation, and erectile dysfunction. Respiratory muscle involvement may lead to life-threatening respiratory failure, representing one of the most severe complications [15, 16]. Several case reports have documented the coexistence of LEMS and SCLC, underscoring their clinical complexity and prognostic implications. According to a European multicenter study, approximately half of patients with LEMS are ultimately diagnosed with SCLC, with older age, male sex, and smoking being common risk factors [4]. In some patients, neurological symptoms may appear several months before the tumor is detected, serving as a potential early clinical indicator of malignancy [17]. In addition to this classic paraneoplastic association, recent evidence has identified immune checkpoint inhibitors (ICIs) as potential triggers or amplifiers of LEMS. The current case contributes to this emerging evidence by describing LEMS onset during durvalumab maintenance therapy [18].
One report described a 74-year-old man with extensive-stage SCLC who developed muscle weakness and fatigue after 13 cycles of atezolizumab maintenance therapy; diagnostic workup confirmed ICI-induced LEMS, highlighting neuroimmunologic adverse events related to ICI therapy [19]. Similarly, another case described a 70-year-old man with extensive-stage SCLC who developed LEMS after five cycles of atezolizumab maintenance therapy; cessation of the drug and administration of corticosteroid pulse therapy plus IVIG resulted in marked neurological improvement [20].
The advent of ICIs has revolutionized the treatment of SCLC but has also introduced new challenges. Beyond atezolizumab, other PD-L1 inhibitors may elicit similar immune-mediated complications. For example, a 62-year-old woman with extensive-stage SCLC and pre-existing paraneoplastic LEMS received durvalumab with chemotherapy followed by maintenance therapy, which did not exacerbate LEMS; antibody titers decreased substantially, but brain metastases later developed, underscoring the need for vigilant monitoring [21]. Another report described a 73-year-old woman who had achieved long-term remission of LEMS after limited-stage SCLC therapy 22 years earlier; upon later diagnosis of non-small-cell lung cancer (NSCLC) treated with pembrolizumab plus chemotherapy, she experienced recurrent muscle weakness and ptosis, with elevated anti-VGCC antibody titers confirming LEMS relapse—providing key evidence that ICIs can reactivate prior LEMS [22].
In our case, the patient developed LEMS during durvalumab maintenance therapy, suggesting that ICIs may act as either precipitating or aggravating factors. Durvalumab was promptly discontinued following LEMS confirmation. Subsequent treatment with IVIG and pyridostigmine led to rapid neurological improvement, and subsequent chemotherapy with lurbinectedin plus supportive care achieved both symptomatic relief and partial tumor response. These findings are consistent with previous reports, demonstrating that individualized sequential therapy can effectively balance tumor control and neuromuscular recovery in complex clinical settings.
Diagnostic confirmation relies on the integration of clinical manifestations, electrophysiological findings, and serologic testing. Repetitive nerve stimulation (RNS) typically demonstrates low-frequency decremental and high-frequency incremental responses that are characteristic of LEMS, while the detection of anti-VGCC antibodies provides valuable supportive evidence [23]. Early recognition and timely initiation of treatment are essential for optimizing patient outcomes.
LEMS management generally involves two main strategies: (1) symptomatic treatment to enhance neuromuscular transmission and (2) antitumor therapy to control the underlying malignancy [24]. Among symptomatic agents, 3,4-diaminopyridine remains the first-line therapy, as it prolongs presynaptic action potentials and promotes acetylcholine release, thereby improving muscle strength [25]. Pyridostigmine, an acetylcholinesterase inhibitor, may be used as an adjunct but often provides limited efficacy [15]. IVIG and corticosteroids play pivotal roles in immunomodulation by reducing autoantibody levels and suppressing immune responses [26, 27].
Lurbinectedin has been supported by studies for the treatment of relapsed small-cell lung cancer. A phase II clinical trial showed that in patients with disease progression after platinum-based chemotherapy, the objective response rate reached 35.2%, with a median progression-free survival of 3.5 months [28]. Another study targeting relapsed patients also suggested that the drug can provide stable disease control, consistent with the observed tumor stabilization and reduction in inflammatory markers in this case [29].
The fluctuating muscle weakness observed in this case may have been driven by multiple factors. The initial IVIG treatment administered in February 2025 produced marked improvement, whereas the second course in May yielded only limited benefit, suggesting potential IVIG tolerance or resistance. Furthermore, the patient’s fluctuating muscle strength and concomitant diabetes during durvalumab therapy raise the possibility that checkpoint inhibition disrupted immune homeostasis, exacerbating LEMS symptoms [30]. Although causality cannot be definitively established, these observations suggest that variations in IVIG responsiveness, corticosteroid effects, and ICI-induced immune remodeling likely acted synergistically, underscoring the multifactorial nature of neuromuscular instability in patients treated with ICIs.
This case presents several notable strengths. Early recognition and electrophysiological confirmation of LEMS during ICI therapy prevented misdiagnosis and treatment delays. The combined use of IVIG and pyridostigmine facilitated rapid neurological recovery and enabled continuation of antitumor therapy. Additionally, proactive monitoring and management of complications—such as inflammation, anemia, and venous thrombosis—illustrate the importance of multidisciplinary care. However, the study is limited by a relatively short follow-up period and the inability to assess long-term prognosis or recurrence risk. As a single-case observation, the findings cannot be generalized but provide valuable clinical insight, warranting further investigation.
This case underscores the importance of early detection and standardized management of paraneoplastic LEMS in patients with SCLC. Prompt initiation of immunomodulatory therapy not only improves functional outcomes but also allows uninterrupted oncologic treatment. While ICIs have significantly improved survival in SCLC, their immune-activating mechanisms may also induce or exacerbate neuroimmunologic adverse events. Therefore, vigilance for neuromuscular symptoms during immunotherapy is essential, as such manifestations should not be attributed solely to tumor progression or chemotherapy toxicity. A multidisciplinary approach integrating oncology and neurology expertise is crucial for optimizing patient outcomes. As ICI use continues to expand, the establishment of comprehensive monitoring and management systems for neuroimmune toxicity is urgently needed. Continued accumulation of clinical evidence will help refine therapeutic strategies and improve long-term prognosis.
This case reports worsening of LEMS during durvalumab maintenance immunotherapy. Following discontinuation of the drug, treatment with IVIG combined with pyridostigmine was administered, and immune checkpoint inhibitors were not reintroduced, resulting in rapid neurological recovery. Subsequently, lurbinectedin was used as second-line chemotherapy to further improve the patient’s condition. During immunotherapy, monitoring for neurological complications and early recognition and management are of critical importance.
• Even in patients with a prior diagnosis of small-cell lung cancer, LEMS may worsen during treatment with immune checkpoint inhibitors such as durvalumab.
• The close temporal association between durvalumab maintenance and the onset of LEMS, along with improvement after drug withdrawal, possibly supports an ICI-related mechanism.
• This case adds to the limited literature describing immune-related neuroimmunologic complications in small-cell lung cancer, highlighting the need for increased clinical awareness and early diagnostic evaluation.
• Early recognition and treatment with intravenous immunoglobulin and corticosteroids can achieve rapid neurological recovery and allow continuation of antitumor therapy, laying the foundation for second-line lurbinectedin chemotherapy.
• Multidisciplinary collaboration between oncologists and neurologists is essential for optimizing outcomes and ensuring the safe administration of immunotherapy in patients at risk for neuroimmune adverse events.
The data and materials in the current study are available from the corresponding author on reasonable request.
YYF, YY, and LRD contributed to the study design. YY conducted the literature search. YYF and LRD acquired the data. YYF and YY wrote the article. LRD performed data analysis. All authors contributed to revising the manuscript critically for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was approved by the Ethics Committee of the Second Affiliated Hospital, Zhejiang University School of Medicine (Approval No. 2025-1574). The study was conducted in accordance with the principles of the Declaration of Helsinki. Written informed consent for participation and publication of clinical details and accompanying images was obtained from the patient.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/BJHM53025.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.







