1 Department of Cardiology, Tianjin Fourth Central Hospital, The Affiliated Hospital of Tianjin Medical University, 300192 Tianjin, China
2 Research Department, Characteristic Medical Center of PAP, 300162 Tianjin, China
3 Department of Military Patient Management, The 987th Hospital of the Joint Logistics Support Force of PLA, 721000 Baoji, Shaanxi, China
4 Medical Service Department, The 980th Hospital of the Joint Logistics Support Force of PLA, 050082 Shijiazhuang, Hebei, China
Abstract
Pharmacological therapy can enhance the therapeutic efficacy of radiofrequency ablation (RFA) for atrial fibrillation (AF) and reduce the risk of recurrence. Dronedarone and amiodarone are two commonly prescribed adjuvant medications. However, no consensus exists on which agent demonstrates superior efficacy. This study aimed to compare the effects of dronedarone and amiodarone on the prognosis of patients undergoing RFA for AF, thereby providing evidence-based guidance for clinical medication selection.
A retrospective analysis was conducted on AF patients who underwent RFA in Tianjin Fourth Central Hospital between August 2022 and May 2023. Patients were assigned to a dronedarone group (n = 50), and an amiodarone group (n = 59) based on the postoperative medication received. Postoperatively, patients were treated with either dronedarone hydrochloride tablets or amiodarone hydrochloride tablets for 6 months. Long-term electrocardiographic monitoring (7-day duration via 5G network) was performed at 1, 3, 6, and 12 months postoperatively to assess recurrence of atrial arrhythmias. Follow-up assessments at the same intervals included thyroid, liver, and renal function, left atrial diameter (LAD), left ventricular ejection fraction (LVEF), and QT interval.
No statistically significant difference was observed in AF recurrence between the two groups (p > 0.05). The incidence of thyroid dysfunction was significantly lower in the dronedarone group compared with the amiodarone group (p < 0.05), whereas other adverse events showed no significant difference (p > 0.05). In both groups, postoperative LVEF was significantly higher than preoperative values (p < 0.05), and LAD was significantly reduced (p < 0.05). Comparing the two groups, postoperative LAD was significantly smaller in the dronedarone group than in the amiodarone group (p < 0.05), while postoperative LVEF exhibited no statistically significant difference (p > 0.05).
Dronedarone and amiodarone are similarly effective in reducing the AF recurrence after RFA. However, dronedarone is associated with a lower risk of thyroid dysfunction and confers greater improvement in left atrial remodelling.
Keywords
- dronedarone
- amiodarone
- atrial fibrillation
- thyroid function
- radiofrequency ablation
As one of the most common arrhythmias, atrial fibrillation (AF) is characterised by rapid and irregular atrial fibrillatory waves replacing coordinated electrical activity. This results in severe atrial electrical dysfunction and significantly increases the risk of death, stroke, heart failure, and dementia [1]. From 2014 to 2016, the prevalence of AF in the Chinese population aged 45 years and above was 1.8% (1.9% in men and 1.7% in women), and prevalence rises progressively with age [2]. Radiofrequency ablation (RFA) has emerged as a standard therapeutic approach for AF due to its high efficacy and minimally invasive profile [3]. The procedure relies on precise catheter manipulation to ablate arrhythmogenic foci, thereby restoring normal cardiac electrophysiology, alleviating symptoms, and improving quality of life for patients [4]. Despite its efficacy, some patients remain at risk of recurrence even after successful ablation [5]. Pharmacological adjunctive therapy is thus indispensable: preoperative medication stabilises heart rate and reduces procedural risks, while postoperative drugs consolidate therapeutic outcomes and mitigate recurrence.
Among available antiarrhythmic agents, dronedarone and amiodarone are commonly used in AF management and as RFA adjuncts [6]. Dronedarone exhibits superior AF suppression through its unique pharmacological mechanisms [7, 8], whereas amiodarone remains a cornerstone therapy due to its potent antiarrhythmic properties [9]. However, robust evidence directly comparing their efficacy in the RFA setting is still limited.
Further investigation into the differential effects of dronedarone and amiodarone as adjunctive treatments may improve ablation success and optimise long-term patient outcomes. This study aimed to provide a clinical reference by systematically comparing the efficacy of these treatments in AF patients undergoing RFA.
A retrospective analysis was conducted on AF patients who underwent RFA in the department of cardiology of Tianjin Fourth Central Hospital between August 2022 and May 2023. The study was conducted following the principles outlined in the Declaration of Helsinki and approved by the Ethics Committee of Tianjin Fourth Central Hospital (Approval number: SZXLL-2023-K019). Informed consent was obtained from all patients or their legal representatives.
Inclusion criteria: (1) Diagnosis of AF consistent with the 2020 European Society of Cardiology (ESC) Guidelines for the Diagnosis and Management of Atrial Fibrillation and successful restoration of sinus rhythm following RFA; (2) Normal thyroid function.
Exclusion criteria: (1) Severe cardiac, hepatic, or renal dysfunction; (2) Pulmonary fibrosis; (3) Malignant tumours; (4) Pregnancy or lactation; (5) Contraindications or allergic reactions from the study medications.
Patients were assigned to groups based on postoperative medication: the dronedarone (Sanofi Winthrop Industrie, HJ20210084, Ambarès-et-Montcare, France; 400 mg twice daily, n = 50) or the amiodarone (Sanofi Winthrop Industrie, HJ20181050, Ambarès-et-Montcare, France; 200 mg once daily, n = 59). All patients discontinued the assigned drug after completing 6 months of treatment.
Long-term (7-day) electrocardiographic monitoring (CarePatch IMED-T103, Hangzhou Proton Technology Co., Ltd., Hangzhou, China) via a 5G network was performed at 1, 3, 6, and 12 months postoperatively to assess recurrence of atrial arrhythmias. The single-lead module, weighing only 16 grams, continuously records bioelectric signals with a 500 Hz sampling rate, enabling detection of subtle heart rate changes. Using intelligent health analysis algorithms, the electrocardiographic waveform can be analysed within 30 seconds and uploaded to a mobile application through the built-in 5G network, facilitating patient-clinician management of health data.
Recurrence was defined as the detection of tachyarrhythmia (atrial flutter, AF,
or atrial tachycardia) lasting
At 1, 3, 6, and 12 months postoperatively, patients in the dronedarone and amiodarone groups were followed to monitor left atrial diameter (LAD) and left ventricular ejection fraction (LVEF). At 12 months, the incidence of adverse reactions was recorded, including sinus bradycardia, QT interval prolongation, thyroid dysfunction, atrioventricular block, hepatic dysfunction, and renal dysfunction. The effects of the two drugs on these parameters were evaluated to determine their safety profiles.
Statistical analyses were performed using the Scientific Platform Serving for
Statistics Professionals (SPSSPRO) (https://www.spsspro.com/). SPSSPRO is an
online data analysis platform that differs from the traditional client-based
software, such as Statistical Product and Service Solutions (SPSS) and
Statistical Analysis System (SAS). It supports all commonly used clinical
statistical analyses and provides a clearer, user-friendly interface. Continuous
variables were expressed as mean
No statistically significant differences were observed between the two groups in clinical characteristics, including gender, age, body mass index (BMI), duration of AF, AF type, presence of other arrhythmias, structural heart disease, history of peripheral vascular disease, transient ischemic attack (TIA), smoking, hypertension, alcohol consumption, low-density lipoprotein cholesterol (LDL-C), total cholesterol, and N-terminal pro-brain natriuretic peptide (NT-proBNP). Detailed results are presented in Table 1.
| Indicator | Dronedarone group (n = 50) | Amiodarone group (n = 59) | p-value | ||
| Age (years) | 53.26 |
54.56 |
–0.918 | 0.361 | |
| Gender, n (%) | 0.127 | 0.721 | |||
| Male | 33 (66.00) | 37 (62.71) | |||
| Female | 17 (34.00) | 22 (37.29) | |||
| BMI (kg/m2) | 24.60 |
23.45 |
1.250 | 0.214 | |
| Duration of atrial fibrillation (years) | 5.74 |
5.85 |
0.233 | 0.816 | |
| LDL-C (mmol/L) | 3.18 |
3.26 |
–0.585 | 0.560 | |
| Total cholesterol (mmol/L) | 4.98 |
4.79 |
0.821 | 0.413 | |
| NT-proBNP (pg/mL) | 238.45 |
246.43 |
–0.890 | 0.376 | |
| Atrial fibrillation type, n (%) | 0.550 | 0.908 | |||
| Paroxysmal | 23 (46.00) | 28 (47.46) | |||
| Persistent | 13 (26.00) | 12 (20.34) | |||
| Long–standing persistent | 8 (16.00) | 11 (18.64) | |||
| Permanent | 6 (12.00) | 8 (13.56) | |||
| Other arrhythmias, n (%) | 5 (10.00) | 14 (23.73) | 3.544 | 0.060 | |
| Structural heart disease, n (%) | 18 (36.00) | 18 (30.51) | 0.369 | 0.544 | |
| Peripheral vascular disease history, n (%) | 4 (8.00) | 9 (15.25) | 1.356 | 0.244 | |
| TIA history, n (%) | 8 (16.00) | 11 (18.64) | 0.131 | 0.717 | |
| Smoking history, n (%) | 21 (42.00) | 24 (40.68) | 0.020 | 0.889 | |
| Hypertension history, n (%) | 34 (68.00) | 38 (64.41) | 0.156 | 0.693 | |
| Alcohol consumption history, n (%) | 27 (54.00) | 33 (55.93) | 0.041 | 0.840 | |
| LAD (mm) | 47.26 |
47.48 |
–0.307 | 0.759 | |
| LVEF (%) | 46.02 |
45.76 |
0.171 | 0.865 | |
Note: BMI, body mass index; LDL-C, low-density lipoprotein cholesterol; NT-proBNP, N-terminal pro-brain natriuretic peptide; TIA, transient ischemic attack; LAD, left atrial diameter; LVEF, left ventricular ejection fraction.
Compared with the amiodarone group, the dronedarone group showed no significant difference in either early or late postoperative recurrence. Detailed results are presented in Table 2.
| Group | No recurrence | Early recurrence | Late recurrence |
| Dronedarone group (n = 50) | 41 (82.0%) | 6 (12.0%) | 3 (6.0%) |
| Amiodarone group (n = 59) | 46 (78.0%) | 9 (15.2%) | 4 (6.8%) |
| 0.289 | |||
| p-value | 0.865 | ||
During follow-up, no statistically significant differences were observed between
the two groups in the incidence of sinus bradycardia, QT interval prolongation,
atrioventricular block, hepatic dysfunction, or renal dysfunction. However,
thyroid dysfunction occurred significantly more frequently in the amiodarone
group than in the dronedarone group (p
| Group | Sinus bradycardia | QT interval prolongation | Thyroid dysfunction | Atrioventricular block | Liver dysfunction | Renal dysfunction | Total adverse reactions |
| Dronedarone group (n = 50) | 2 (4.0%) | 1 (2.0%) | 2 (4.0%) | 4 (8.0%) | 2 (4.0%) | 3 (6.0%) | 14 (28.0%) |
| Amiodarone group (n = 59) | 3 (5.1%) | 1 (1.7%) | 11 (18.6%) | 4 (6.8%) | 1 (1.7%) | 5 (8.4%) | 25 (42.4%) |
| 0 | 0 | 4.219 | 0 | 0.021 | 0.016 | 1.848 | |
| p-value | 1.000 | 1.000 | 0.040* | 1.000 | 0.884 | 0.900 | 0.174 |
Note: *p
At baseline, there were no statistically significant differences in LAD or LVEF
between the dronedarone and amiodarone groups (all p
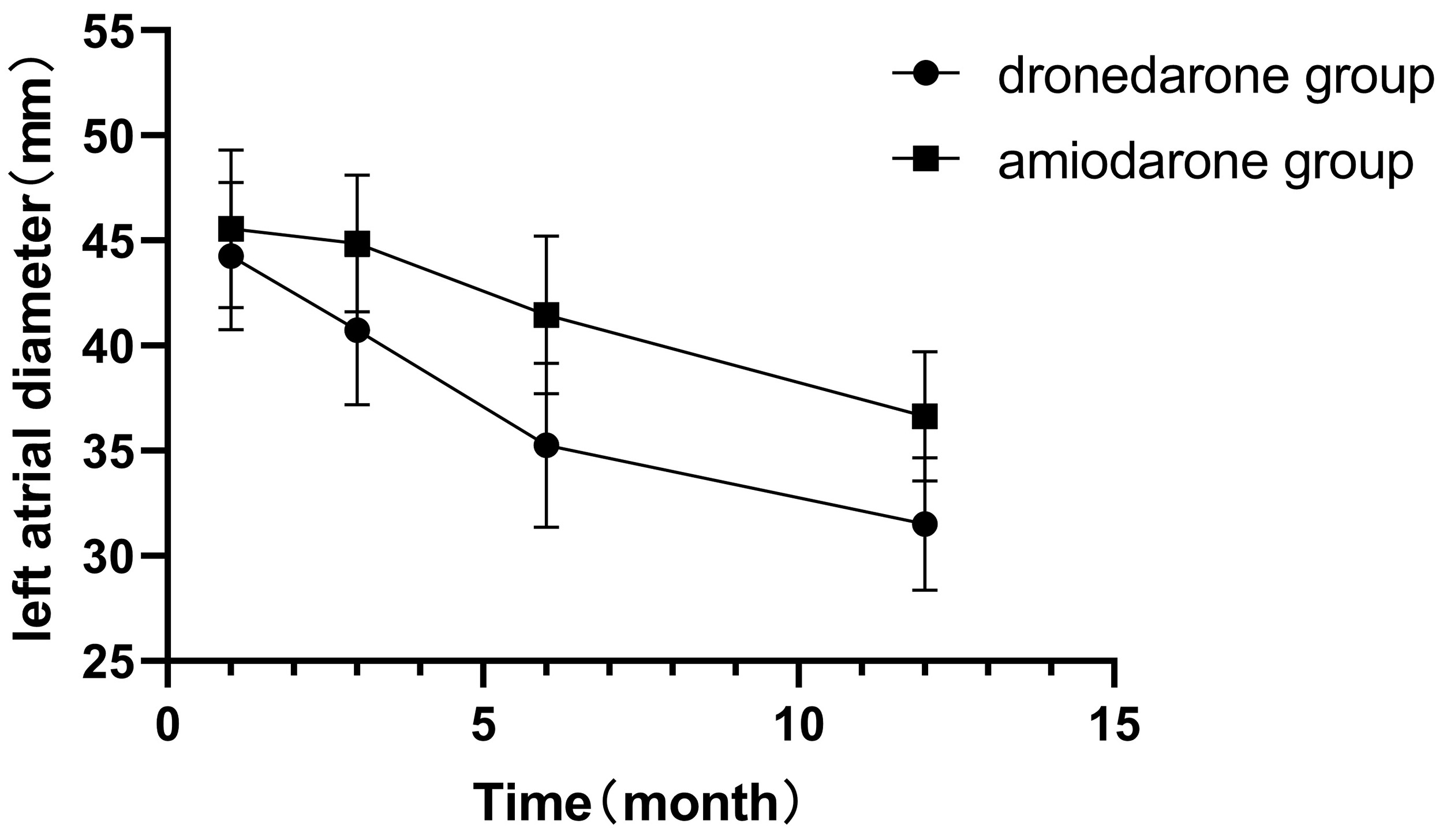 Fig. 1.
Fig. 1.
Changes in left atrial diameter (LAD) before and after treatment.
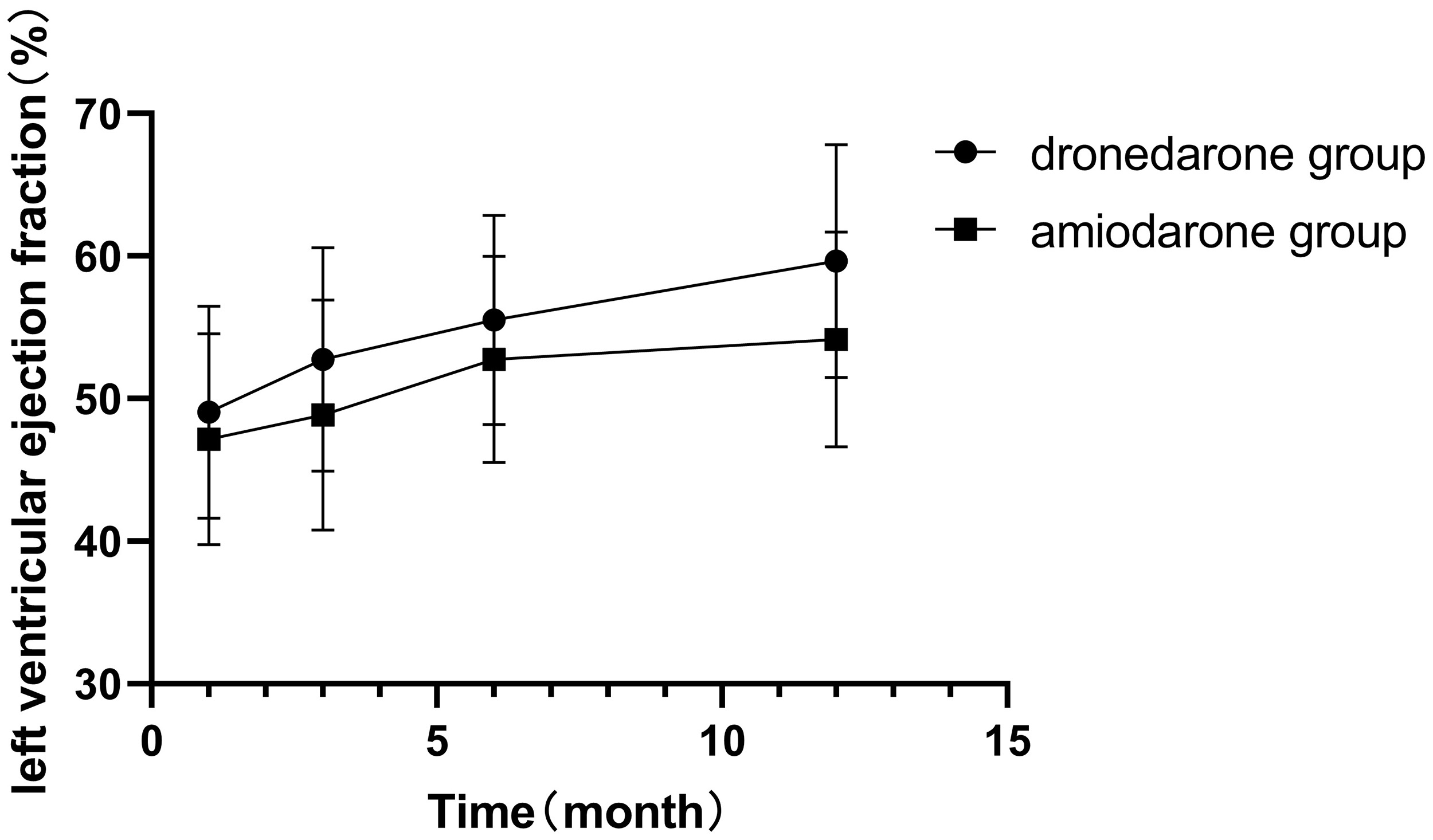 Fig. 2.
Fig. 2.
Changes in left ventricular ejection fraction (LVEF) before and after treatment.
| Characteristics | Dronedarone group (mm) | Amiodarone group (mm) | Time | Group | Time | |||
| F-value | p-value | F-value | p-value | F-value | p-value | |||
| 0 months | 47.26 |
47.48 |
205.037 | 50.249 | 9.778 | |||
| 1 month | 44.26 |
45.56 |
||||||
| 3 months | 40.74 |
44.85 |
||||||
| 6 months | 35.26 |
41.46 |
||||||
| 12 months | 31.50 |
36.64 |
||||||
| Characteristics | Dronedarone group | Amiodarone group | Time | Group | Time | |||
| F-value | p-value | F-value | p-value | F-value | p-value | |||
| 0 months | 46.02 |
45.76 |
27.399 | 2.246 | 0.135 | 1.122 | 0.340 | |
| 1 month | 49.06 |
47.14 |
||||||
| 3 months | 52.76 |
48.85 |
||||||
| 6 months | 55.50 |
52.76 |
||||||
| 12 months | 59.66 |
54.15 |
||||||
Atrial fibrillation (AF), one of the most common arrhythmias, has made RFA a major therapeutic approach [3]. Adjunctive pharmacotherapy plays an essential role in improving surgical success rates. Dronedarone, an antiarrhythmic drug, exerts its therapeutic effects by reducing the excitability of cardiac cells, and its efficacy in AF treatment has been confirmed in multiple studies [10, 11, 12]. Amiodarone, with its long half-life and potent antiarrhythmic properties, also holds a significant position in AF management [13]. However, further research is warranted to validate and compare the therapeutic outcomes of these two drugs. Accordingly, we conducted a comparative analysis of dronedarone versus amiodarone following RFA for AF.
Our results demonstrated no statistically significant difference in recurrence rates between the two groups, suggesting comparable therapeutic efficacy. Extended postoperative surveillance is critical for cardiac monitoring and requires effective and real-time long-term assessment of the cardiac electrical activity of the patients. Conventional ECG and 24-hour Holter monitoring remain the most widely used clinical methods for arrhythmia detection. However, their short recording duration limits their potential to capture infrequent arrhythmias, and hospital visits reduce convenience. Portable remote ECG monitoring devices are increasingly accepted and applied in AF screening and follow-up. A stable and high-speed network is fundamental. By leveraging the low-latency advantage of 5G technology, our research team enabled real-time acquisition and transmission of the physiological data of the patients to the servers, allowing prompt predictive analysis and therapeutic intervention by the backend system [14].
The safety profile of the administered drugs represents a critical aspect of this study. Adverse reactions to the two medications during radiofrequency ablation for AF were systematically monitored and recorded, followed by analysis of their types and incidence rates. This approach enabled a comprehensive assessment of drug safety and provided robust evidence to support clinical decision-making. The results demonstrated that the incidence of thyroid dysfunction was significantly lower in the dronedarone group than in the amiodarone group. Abnormal thyroid function was defined by deviations in thyroid-stimulating hormone, free triiodothyronine, and free thyroxine levels. No statistically significant differences were observed in other adverse reactions or in the overall adverse event rates. Given the complex mechanism of action for amiodarone, which involves multiple ion channels and receptors, its therapeutic efficacy may demonstrate reduced stability and reliability [15]. In contrast, dronedarone lacks iodine components, thereby avoiding iodine-related extracardiac adverse effects associated with amiodarone [16, 17, 18]. This observation aligns with previous studies reporting no significant differences in AF recurrence rates or rehospitalisation for re-ablation between amiodarone and dronedarone. Nevertheless, the dronedarone group exhibited a higher incidence of adverse drug events, primarily gastrointestinal reactions and bradycardia [11]. Conflicting findings have also been reported: one study found that among patients hospitalised with incident AF, those discharged on dronedarone had an increased risk of cardiovascular-related rehospitalisation or death compared with patients prescribed amiodarone [11].
The analysis further demonstrated that the LVEF was significantly higher postoperatively than preoperatively in both groups, while the LAD was significantly reduced postoperatively. Over time, following RFA, the LAD index was markedly smaller in the dronedarone group than in the amiodarone group. Patients receiving dronedarone showed a significant decrease in LAD after three months of treatment, which was lower than in the amiodarone group. Even after discontinuation at six months, LAD remained lower in the dronedarone group at the 12-month follow-up after ablation. Conversely, postoperative LVEF was comparable between the two groups, with no statistically significant difference. These findings suggest that both drugs effectively improved myocardial function after radiofrequency ablation, with dronedarone demonstrating comparatively higher efficacy. In patients with AF, ineffective atrial contractions reduce atrial stroke volume, increase intra-atrial pressure, and impose mechanical stretch on cardiac cells, ultimately driving atrial structural remodelling and LAD enlargement, which further promotes AF progression. Surgical elimination of AF, followed by pharmacological prevention of AF recurrence, reduces intra-atrial pressure and mechanical stress on cardiac cells, thereby facilitating atrial reverse remodelling [19, 20].
This study provides innovative insights into post-ablation pharmacotherapy for AF. Beyond contributing to the theoretical framework for post-ablation medication strategies, this comparative study establishes a practical basis for individualised treatment planning. Dronedarone demonstrates superior safety by significantly reducing the risk of thyroid dysfunction while maintaining therapeutic efficacy comparable to amiodarone. Further elucidation of its mechanisms may broaden its clinical utility, benefiting an increasing number of patients.
Nevertheless, several limitations should be acknowledged. First, the relatively small sample size may reduce statistical power, particularly in subgroup analyses where clinically meaningful differences might remain undetected; this warrants cautious interpretation of the results. Second, the inclusion of obese participants introduces a potential confounding factor, as obesity alters drug distribution volume and hepatic metabolism, thereby affecting antiarrhythmic drug concentrations. However, the absence of systematic pharmacokinetic data limits the ability to comprehensively adjust for this confounding effect. Third, as a retrospective study, medication adherence was primarily assessed through outpatient records rather than standardised monitoring methods such as therapeutic drug monitoring or electronic pillboxes, which may have introduced bias in evaluating treatment efficacy. In addition, the retrospective design inherently carries the risk of selection bias. Fourth, due to pharmacokinetic differences between dronedarone and amiodarone, arrhythmia recurrence during the 6–12-month period may have been influenced by treatment discontinuation. Although no significant difference in recurrence rates was observed between the two groups, this finding should still be interpreted with caution. Finally, although single-lead wearable ECG monitoring devices provide portability and high patient compliance, their accuracy in identifying complex arrhythmia waveforms is lower than that of standard multi-lead Holter monitoring. These limitations underscore the need for future prospective studies with larger sample sizes that incorporate obesity-related pharmacokinetic parameters and objective adherence measures to further validate our findings.
This study demonstrates that dronedarone represents a safer alternative to amiodarone for post-RFA management of AF, with comparable antiarrhythmic efficacy and fewer thyroid-related complications. Both agents improve cardiac function. However, dronedarone exhibits superior impact on atrial remodelling, which underscores its potential for long-term structural benefits. These findings support the integration of dronedarone into personalised therapeutic strategies, particularly for patients at risk of thyroid dysfunction. Future investigations involving larger cohorts and extended follow-up are warranted to validate these observations. Clinically, prioritising dronedarone may optimise patient safety without compromising therapeutic effectiveness, thereby advancing post-ablation care in AF patients.
• Dronedarone and amiodarone exhibited no significant difference in preventing AF recurrence post-radiofrequency ablation (RFA), with comparable early and late recurrence rates.
• Dronedarone was associated with a significantly lower risk of thyroid dysfunction compared to amiodarone, while the incidence of other adverse events was comparable between groups.
• Both agents enhanced LVEF and reduced LAD; however, dronedarone achieved superior long-term LAD reduction, suggesting greater efficacy in promoting atrial reverse remodelling.
The data in the current study are available from the corresponding authors on reasonable request.
YS participated in the research design, writing of the paper, performance of the research, data collection and analysis. XS, HL and XG participated in data analysis and interpretation. TZ and GZ participated in the research design, data analysis and paper review. All authors reviewed and provided feedback on the manuscript. All authors contributed to the important editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of the Tianjin Fourth Central Hospital (Approval number: SZXLL-2023-K019). Informed consent was obtained from all patients or their legal representatives.
Not applicable.
The study was supported by Tianjin Municipal Health Commission Science and Technology Project (TJWJ2022MS022).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


