1 Department of Cardiology, Imperial College NHS Foundation Trust, W12 0HS London, UK
2 National Heart and Lung Institute, Imperial College London, SW3 6LY London, UK
Abstract
Infective endocarditis (IE) is a serious and potentially life-threatening condition characterised by inflammation and infection of the endocardium. While cardiac manifestations are well-recognised, extracardiac manifestations of IE, particularly in the gastrointestinal tract, are less commonly reported. We report the first known case of IE presenting with colitis-like symptoms in the absence of identifiable colonic pathology.
A 34-year-old male patient presented to primary care with gastrointestinal symptoms, including fever, weight loss, and bloody diarrhoea. There was an absence of detectable intestinal pathology on colonoscopy, and subsequent findings revealed a link between the gastrointestinal symptoms and IE-related complications. The patient later developed right-sided flank pain, a pansystolic murmur, and splinter haemorrhages in two fingernails. Imaging studies indicated renal infarcts suspicious for septic emboli, and echocardiography revealed severe mitral regurgitation with an anterior leaflet vegetation. Blood cultures were negative, likely due to prior antibiotic administration.
The patient was therefore treated with empirical intravenous antibiotics (amoxicillin, flucloxacillin and gentamicin), resulting in the resolution of gastrointestinal symptoms.
This case highlights the importance of considering IE in patients with unexplained gastrointestinal symptoms and systemic features, even in the absence of visible colonic lesions. Early recognition and appropriate management may improve outcomes.
Keywords
- infective endocarditis
- gastrointestinal bleeding
- cardiology
- microbiology
- case report
Infective endocarditis (IE) is a serious and potentially life-threatening condition characterised by microbial infection and inflammation of the endocardial surface, most commonly involving cardiac valves [1]. The epidemiology of IE has been evolving, with an overall incidence of 1.5–9.6 per 100,000 people, and rising rates among elderly patients, those with prosthetic valves [0.5–3.1% per patient-year after transcatheter aortic valve implantation (TAVI)], cardiac implantable devices (now up to 10% of IE cases), and healthcare-associated settings (34% of cases) [2]. The diagnostic standard is the modified Duke criteria, which integrate major findings from blood cultures and echocardiography with various minor criteria to confirm infective endocarditis. Treatment consists of prolonged courses of intravenous antibiotics and often requires surgical intervention [3]. IE continues to carry substantial morbidity and mortality, even with modern diagnostic and therapeutic advances. While neurological and renal manifestations of IE are well-documented, gastrointestinal involvement is less commonly reported [3].
Gastrointestinal manifestations, while relatively rare, can include splenic infarction, hepatosplenic abscesses, mesenteric ischaemia and gastrointestinal bleeding secondary to embolic or immune-mediated mechanisms [3]. These symptoms may be the presenting complaint of the patient. Without awareness of the association between IE and gastrointestinal manifestations, the diagnosis and treatment of this condition can be delayed, leading to worse outcomes.
We present a case of a 34-year-old male patient who initially presented with unexplained gastrointestinal symptoms—fever, weight loss and change in bowel habit (frequent bloody diarrhoea with tenesmus and mucus). There was an absence of detectable intestinal pathology on colonoscopy and subsequent findings revealed a link between the gastrointestinal symptoms and IE-related complications.
IE presenting as clinical colitis has not previously been described in the literature. Whilst a link between Streptococcus gallolyticus (formerly Strep.bovis) IE and colonic neoplasms have been well-described [4], our case describes a link between IE and gastrointestinal symptoms without macroscopic colonic pathology.
A 34-year-old man presented to his general practitioner with 6 weeks of fever, weight loss and change in bowel habit (frequent bloody diarrhoea with tenesmus and mucus). He had no past medical or surgical history of note, had no risk factors for IE and was taking no regular medications. The patient was referred under a two-week-wait pathway for further outpatient investigation. Colonoscopy showed external haemorrhoids but no macroscopic colonic pathology; computed tomography (CT) of the chest, abdomen and pelvis demonstrated uncomplicated sigmoid diverticulosis but no cause of symptoms.
Approximately two weeks after undergoing outpatient colonoscopy, the patient
represented to the Emergency Department with new-onset right-sided flank pain.
Given the abdominal pain and elevated inflammatory markers [white cell count
(WCC) 13
A CT abdomen with contrast (Fig. 1) again demonstrated uncomplicated sigmoid diverticulosis but did not identify a gastrointestinal cause of bleeding. It also showed multiple right renal infarcts suspicious for septic emboli. On subsequent assessment, the patient had a pansystolic murmur loudest at the apex and splinter haemorrhages present in two fingernails.
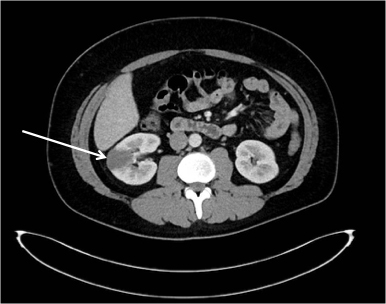 Fig. 1.
Fig. 1.
CT abdomen with contrast. A CT abdomen with contrast was performed which demonstrated a right renal infarct (white arrow) and uncomplicated sigmoid diverticulosis. No cause for gastrointestinal bleeding was identified. CT, computed tomography.
Echocardiography showed severe mitral regurgitation with an anterior leaflet vegetation (Fig. 2). Initial blood cultures were negative for growth, which was likely due to earlier antibiotic administration.
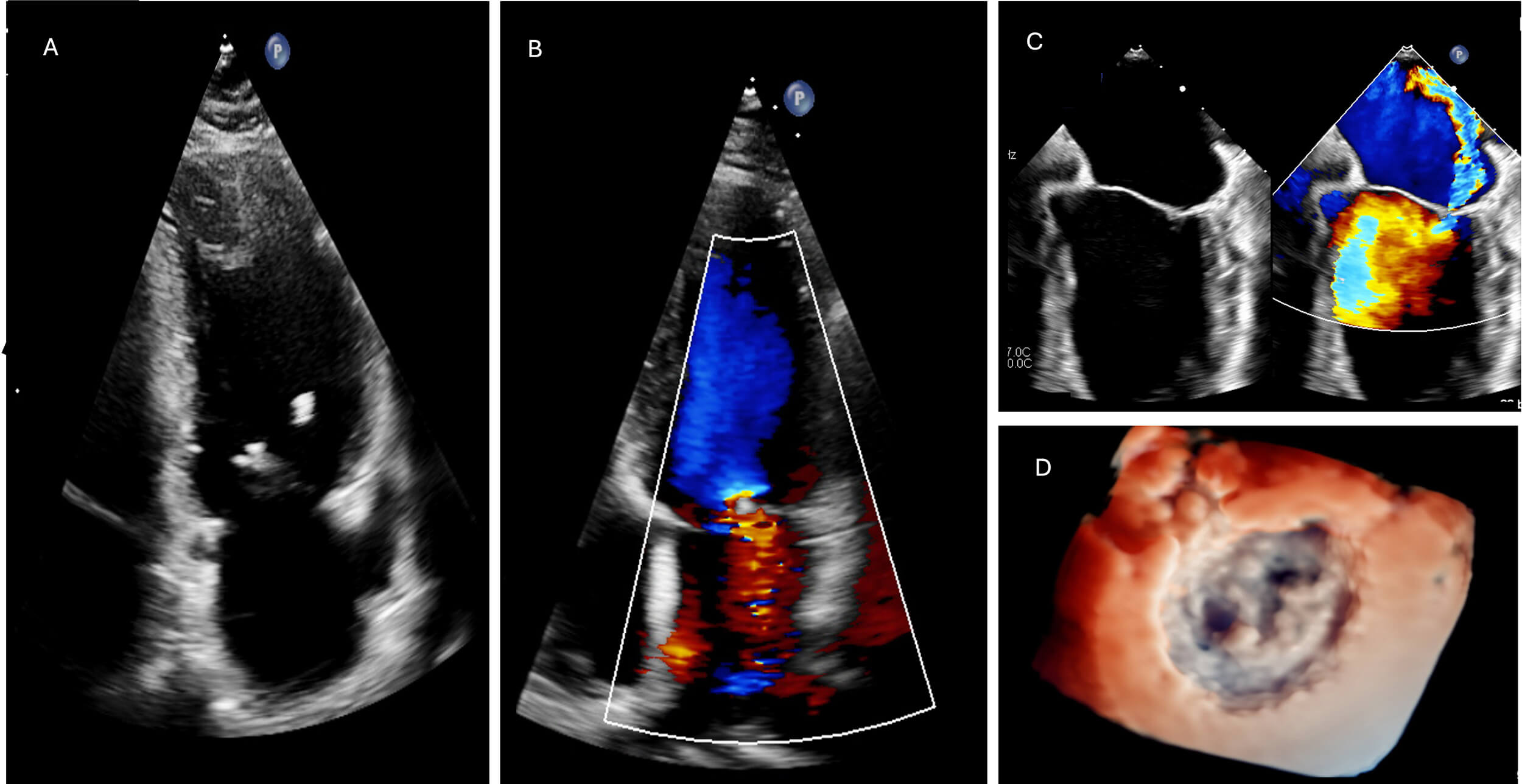 Fig. 2.
Fig. 2.
Initial transthoracic echocardiography (TTE) with mitral valve regurgitation and echogenic mitral valve lesion. (A) TTE apical 4 chamber view of the left ventricle (LV) revealing an echogenic mobile structure connected to the mitral valve, indicative of vegetation. (B) TTE apical 4 chamber colour doppler demonstrating associated mitral regurgitation (MR), further highlighting the valvular pathology. (C) Transoesophageal echocardiography (TOE) image of a zoomed 4-chamber view, providing a detailed visualization of a flail mitral valve with associated mitral regurgitation, emphasizing the severity of valvular dysfunction. (D) TOE 3D reconstruction displaying a comprehensive view of the vegetation, clearly visualizing its attachment to the ventricular side of the mitral valve.
After consultation with the hospital microbiology team and evaluation at the specialised endocarditis multidisciplinary team (MDT) meeting, the patient was started on intravenous antibiotics, completing a six-week course of amoxicillin, flucloxacillin and gentamicin for subacute bacterial endocarditis with septic emboli.
The patient’s clinical condition, including notably gastrointestinal symptoms, improved and resolved with intravenous antibiotics. The gastrointestinal symptoms were presumed to result from endocarditis-related microinfarcts or vasculitic changes, although this was a clinical diagnosis in the absence of histological confirmation. Biopsy was not pursued given the patient’s clinical improvement with antibiotic therapy. This case demonstrates that endocarditis does not need to be associated with a colonoscopically identifiable lesion to cause lower gastrointestinal symptoms.
Following the course of antibiotics, the patient’s symptoms considerably improved. He was discharged to the endocarditis clinic for close monitoring. From a cardiac and infective perspective, he was asymptomatic and inflammatory markers remained low (Fig. 3)—because of this, further microbiological testing was not attempted. The patients index left ventricular ejection fraction at presentation was 60%, there was no decrease on subsequent examinations. Echocardiography six months after presentation (Fig. 4), revealed persisting severe mitral regurgitation with an eccentric jet (regurgitant volume 66 mL), which persisted at a 20-month follow-up (Fig. 5). He continues to be closely monitored and will be evaluated as an outpatient for possible mitral valve repair/replacement.
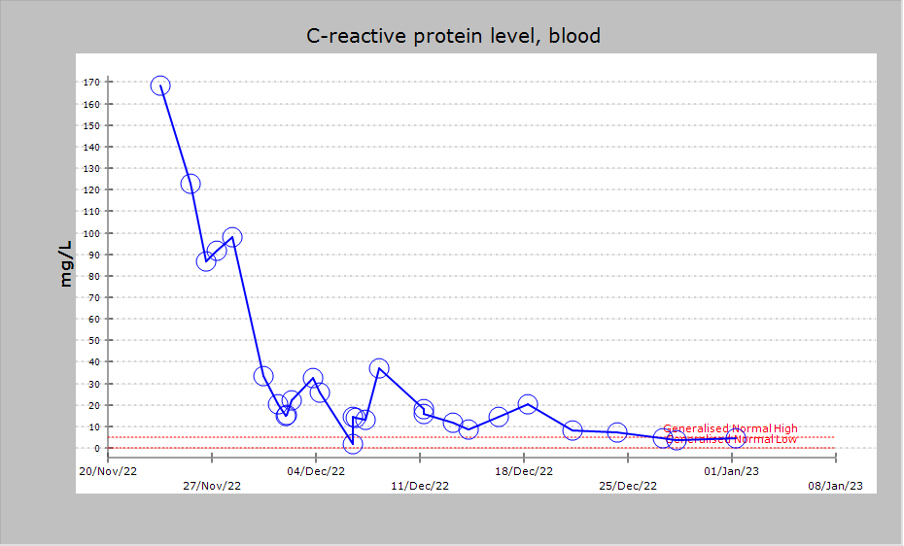 Fig. 3.
Fig. 3.
Trend in C-reactive protein. Trend in C-reactive protein during inpatient admission and treatment with intravenous antibiotics. This diagram was generated by the programme cerner.
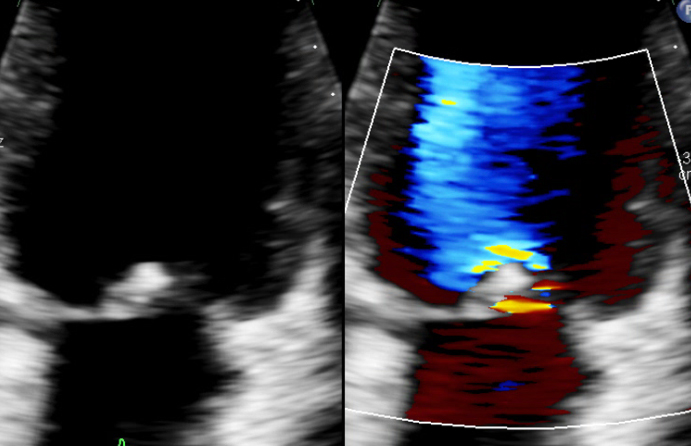 Fig. 4.
Fig. 4.
Transthoracic echocardiography (TTE) 6 months from index admission. TTE with residual mitral valve regurgitation and echogenic structure connected to the mitral valve.
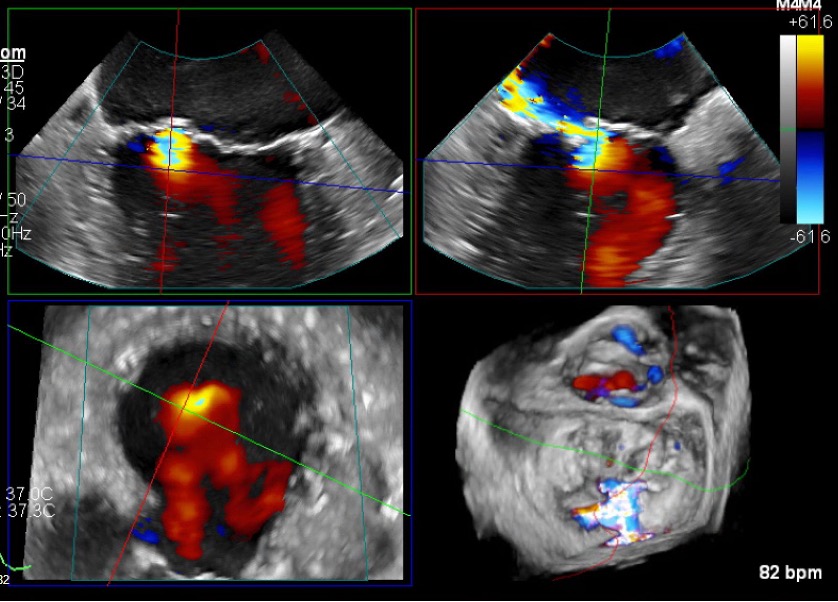 Fig. 5.
Fig. 5.
Transoesophageal echocardiography (TOE) 20 months from index admission. Top left: 4-chamber view demonstrates persistent mitral regurgitation. Top right: alternative 2D plane with colour doppler confirming a turbulent regurgitant jet directed into the left atrium. Bottom left: cross-sectional (short-axis) view with colour doppler showing the regurgitant jet spreading within the atrium. Bottom right: TOE 3D reconstruction displaying persistent mitral regurgitation.
Extra-cardiac symptoms are common in IE, and this is one of the reasons diagnosis is often delayed. In this case, the patient initially presented with gastrointestinal symptoms, including fever, weight loss and bloody diarrhoea. These symptoms prompted colonoscopic evaluation to rule out colitis.
The pathophysiology of gastrointestinal involvement in IE is multifactorial. Embolic phenomena, immune complex deposition and vasculitic processes can all contribute to the development of gastrointestinal symptoms [5, 6, 7]. In this case, the presence of renal infarcts and splinter haemorrhages helped confirm the diagnosis of IE with systemic involvement.
While the association between Streptococcus gallolyticus (formerly Strep.bovis) endocarditis and colonic neoplasia is well established [4], such cases typically involve identifiable colonic lesions serving as a portal of entry. In contrast, our patient presented with gastrointestinal symptoms without macroscopic colonic pathology, and blood cultures were negative. This suggests an alternative mechanism, potentially related to microinfarction or vasculitic phenomena secondary to embolic complications of IE.
The prompt initiation of appropriate antibiotic therapy was crucial in managing this patient. Intravenous amoxicillin, flucloxacillin and gentamicin were administered. The resolution of both cardiac and gastrointestinal symptoms with antibiotics underscored the link of IE with the gastroenterological symptoms, which may have been caused by colonic microinfarcts or vasculitis [8].
In this case, a causative bacteria was never identified. This is due to the fact that the patient had already received intravenous antibiotics empirically to treat presumed intraabdominal sepsis. Three doses of intravenous co-amoxiclav were given prior to any blood cultures being taken. A broad differential should be considered in the absence of a causative organism. HACEK organisms—Haemophilus, Aggregatibacter, Cardiobacterium, Eikenella, and Kingella species—are well-known causes of endocarditis and may require prolonged culture incubation. In this case, the patient responded promptly to antibacterial therapy and further evaluation was not deemed to be of benefit.
The major strength of this case is the multidisciplinary team approach and long-term follow-up, which permitted an accurate correlation between systemic manifestations and cardiac disease. However, a significant limitation is that gastrointestinal involvement was not histologically confirmed by biopsy because of the patient’s clinical improvement with antibiotics.
This case underscores the need for increased awareness among healthcare providers regarding the diverse clinical presentations of IE. Clinicians should consider IE in patients presenting with unexplained gastrointestinal symptoms, particularly in the absence of detectable colonic lesions, ensuring a comprehensive evaluation is performed including a thorough cardiac examination. Recognising IE’s extracardiac manifestations, such as gastrointestinal involvement, can facilitate early diagnosis, appropriate treatment, and a multidisciplinary approach involving specialities such cardiology, infective diseases and gastroenterology. This can help prevent complications, such as embolic events, and improve patient outcomes.
To ensure comprehensive reporting, this case report was structured according to the case report (CARE) statement guidelines available on the CARE website. The CARE checklist has been provided as Supplementary Material.
• Infective endocarditis can cause a range of extra-cardiac manifestations, including abdominal pathology such as splenic infarct, hepatosplenic abscess, mesenteric ischaemia and gastrointestinal bleeding.
• Perform a thorough cardiac examination and consider infective endocarditis as a potential underlying cause in patients with unexplained gastrointestinal symptoms.
• If considering infective endocarditis, prompt collection of blood cultures prior to antibiotic administration can aid diagnosis and treatment.
The datasets generated or analysed during this case report are available from the corresponding author on reasonable request.
RA drafted the manuscript, performed a literature review, and collected clinical data. MZ contributed to echocardiographic analysis, and clinical interpretation. NK contributed to clinical interpretation. DK contributed to echocardiographic analysis, and clinical interpretation. DL contributed to the conception and design of the case report and supervised the project. All authors contributed to revising the manuscript critically for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Written informed consent has been acquired from the patient for publication of this case and any accompanying images.
The authors sincerely thank the patient for their cooperation and consent to share their clinical case.
This research received no external funding
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/BJHM52972.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





