1 Department of Epidemiology and Biostatistics, School of Public Health, Anhui Medical University, 230032 Hefei, Anhui, China
2 Inflammation and Immune Mediated Diseases Laboratory of Anhui Province, 230032 Hefei, Anhui, China
3 Preventive Medicine Experimental Teaching Center, School of Public Health, Anhui Medical University, 230032 Hefei, Anhui, China
Abstract
Autoimmune diseases (ADs) impose a significant and growing burden on global health. Emerging studies indicate that dysregulation of the mucosal immunity is not merely a consequence of disease progression but often represents a critical sentinel event that triggers autoimmune responses. This review explores a paradigm shift in the understanding of ADs pathogenesis, moving away from an exclusive emphasis on systemic immune imbalance towards a perspective centered on mucosal immune abnormalities characterized by mucosal barrier dysfunction and microbiota dysbiosis. We synthesize the principal mechanisms through which mucosal immunity contributes to the onset and progression of ADs and critically evaluate emerging therapeutic strategies. These insights offer new opportunities for precision medicine approaches that intervene at the mucosal origins of ADs.
Keywords
- autoimmune diseases
- immune mechanisms
- targeted therapy
- microbiota
- gut-kidney axis
- mucosal immunity
Autoimmune diseases (ADs) are a group of disorders caused by pathological immune responses in which B cells and T cells react against self-antigens, leading to the targeting and destruction of normal tissues. The etiology of these diseases remains unclear. However, their global incidence is increasing, affecting about 4% of the population, especially women, and imposing a complex and chronic burden on healthcare systems globally [1]. Mucosal immunity refers to localized immune responses that occur within mucosal tissues, which includes the gastrointestinal, respiratory, and urogenital tracts, as well as the oral cavity. These tissues constitute the body’s first line of defense against external pathogens [2]. Traditional research on the pathogenesis and treatment strategies for ADs has primarily focused on systemic immune dysregulation and global immune suppression. However, recent studies are driving a paradigm shift in this understanding. An increasing body of evidence suggests that dysfunction of the mucosal immune system, distributed across the gut, respiratory tract, oral cavity, and urinary tract, often serves as a common precursor and driving factor in the occurrence and progression of ADs [3, 4]. Immunoglobulin A nephropathy (IgAN) is a typical representative of the mucosal-systemic axis pathogenic mechanism, and its onset is associated with dysregulation of the mucosal immune system in multiple sites. This realization expands the focus of ADs research from mere systemic immune suppression to a deeper understanding and precise regulation of immune homeostasis at the mucosal level. Accordingly, this review aims to systematically elaborate on the core mechanisms of mucosal immune dysregulation in ADs by analyzing both shared and site-specific pathological pathways in key mucosal sites. Notably, we emphasize emerging therapeutic strategies targeting mucosal immunity, including the restoration of microbial homeostasis, localized immune modulation, and precision-targeted interventions. Together, these approaches provide a new theoretical framework and potential intervention strategies for the precise prevention and treatment of ADs.
Mucosal immunity encompasses the localised immune responses that occur within mucosal tissues, including the respiratory, gastrointestinal, oral, and urogenital tracts [2]. This system depends on the synergistic coordination between innate and adaptive defense mechanisms. The innate immune system is formed by the mucosal epithelial layer together with its associated physical, chemical, and biological barriers. Within this framework, the physical barrier is primarily composed of secretory mucus and epithelial tight junctions. Furthermore, distinct mucosal sites possess specialised clearance mechanisms [5]. For example, ciliated cells in the respiratory tract facilitate the expulsion of mucus and foreign particulates through coordinated ciliary beating, while the intestinal tract utilizes peristalsis and rapid epithelial cell turnover to substantially reduce the risk of pathogen colonization [6, 7]. Adaptive immunity, characterized by antigen specificity and immunological memory, is largely mediated by mucosa-associated lymphoid tissue (MALT), which can be functionally divided into inductive sites and effector sites [8]. Inductive sites are responsible for initiating antigen-specific T cell and B cell responses. Effector sites such as the epithelium and the lamina propria execute the downstream defensive functions [4]. This section illustrates the specific mechanism of mucosal immunity using the intestinal mucosa as an example (Fig. 1). Epithelial cells, antimicrobial peptides, and mucins secreted by goblet cells form a physical and chemical barrier. Once this barrier is breached, dendritic cells (DCs) activate antigen-specific T cells and assist B cells in differentiating into plasma cells. The immunoglobulin A (IgA) secreted by these plasma cells then travels through the epithelium into the intestinal lumen to coat pathogens for immune clearance, which ultimately maintains intestinal homeostasis [9, 10].
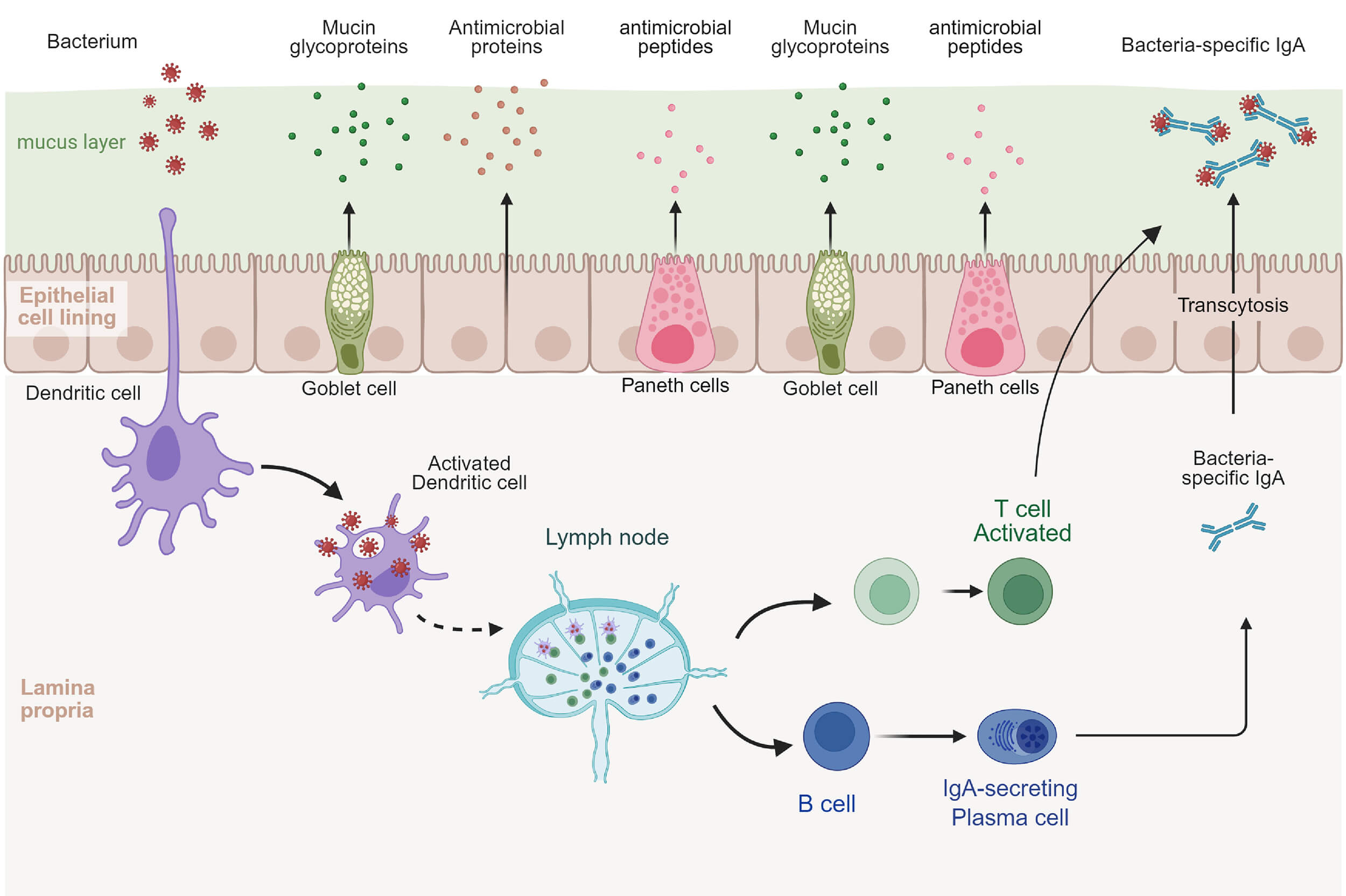 Fig. 1.
Fig. 1.
Immune mechanisms of the intestinal mucosa. Notes: IgA, immunoglobulin A. This figure was created using BioRender.com (https://www.biorender.com/).
The pathophysiology of ADs is related to intestinal mucosal immunity. The underlying mechanisms include dysbiosis of the intestinal microbiota, impaired intestinal barrier function, and abnormal activation of local immune cells in the intestine. In this section, we clarify how intestinal mucosal immune responses contribute to, and regulate the occurrence and progression of ADs.
Systemic lupus erythematosus (SLE) is a chronic autoimmune disease characterized by persistent inflammatory response and immune-mediated tissue damage, which can involve multiple organ systems such as the skin, mucosa, musculoskeletal, blood and kidney [11]. Patients with SLE often exhibit gut microbiota dysbiosis, manifested in the alterations in the Firmicutes-to-Bacteroidetes ratio, increased pro-inflammatory bacteria (i.e., Streptococcus), and reduced anti-inflammatory species belonging to Bifidobacterium [12]. Katsushima et al. [13] found that gut bacteria-derived adenosine triphosphate (ATP)-binding cassette (ABC) transporters can serve as cross-reactive antigens, inducing the production of anti-dsDNA antibodies. This highlights the crucial role of bacterial translocation and molecular mimicry in driving the autoimmune response in SLE. Additionally, increased intestinal permeability, marked by elevated levels of zonulin, has been found to correlate with disease activity in SLE patients [14].
Rheumatoid arthritis (RA) is a chronic immune-mediated inflammatory disease primarily characterized by invasive synovitis. Clinically, it commonly presents as symmetric polyarthritis with joint pain, swelling, and morning stiffness, and can progressively lead to irreversible destruction of articular cartilage and bone [15]. Impairment of the intestinal mucosal barrier is regarded as an early event in RA pathogenesis. A study has shown that abnormalities in intestinal permeability are present before the onset of overt clinical symptoms of arthritis. Consistent with this, animal experiments have demonstrated that mice with collagen-induced arthritis (CIA) exhibit early-stage gut microbiota dysbiosis and intestinal mucosal inflammation [16]. These changes may be associated with aberrant activation of signalling pathways involving intestinal tight junction proteins [17]. In addition, the gut microbiota may play an important role in the initiation and maintenance of systemic inflammation in RA. For instance, Prevotella copri promotes the expression of interleukin-17 (IL-17) and T helper 17 (Th17)-related cytokines, thereby exacerbating disease progression [18]. Conversely, Parabacteroides distasonis has been shown to alleviate arthritis symptoms, potentially through modulation of bile acid metabolism [19].
Inflammatory bowel disease (IBD) is a chronic, recurrent immune-mediated disorder that includes Crohn’s disease (CD) and ulcerative colitis (UC). Its main clinical manifestations are abdominal pain, diarrhea, bloody stools and weight loss [20]. Beneficial commensal bacteria, such as Faecalibacterium prausnitzii and Roseburia species, are essential for maintaining intestinal homeostasis and alleviating inflammatory symptoms. In contrast, the proliferation of pathobionts, including Escherichia coli and Clostridium difficile, is associated with disease exacerbation and more severe clinical manifestations [21]. Notably, the impairment of intestinal barrier integrity represents a critical pathogenic element of IBD. This impairment arises from multiple mechanisms, including epithelial cell death, abnormalities in intercellular tight junctions, dysfunction of the mucus layer, and dysbiosis of the symbiotic microbiota, collectively contributing to sustained intestinal inflammation [22].
Type 1 diabetes mellitus (T1DM) is a T-cell-mediated AD characterized by
progressive destruction of pancreatic
Multiple sclerosis (MS) is a chronic inflammatory disease of the central nervous system characterized by immune-mediated demyelination, neurodegeneration, and progressive functional disability [27]. In recent years, the gut–brain axis has attracted considerable attention as a pivotal pathway linking intestinal health with neurological wellbeing. Studies have shown that MS patients frequently exhibit significant alterations in gut microbiota composition [28]. Notably, Gargano et al. [29] discovered that dysbiosis of the intestinal fungal community triggers the activation of mucosal-associated invariant T (MAIT) cells. These activated cells subsequently cross the blood–brain barrier, infiltrate the central nervous system, and secrete pro-inflammatory cytokines, thereby directly contributing to and exacerbating the neuroinflammatory process in MS. Furthermore, gut dysbiosis influences the production of microbial metabolites, including various secreted molecules, membrane components, neurotransmitters, and hormone-like substances. These signalling molecules can modulate central nervous system functions by regulating immune and inflammatory responses, playing a crucial regulatory role in the pathophysiology of MS [30].
IgAN is the most common type of glomerulonephritis driven by an autoimmune response against galactose-deficient immunoglobulin A1 (Gd-IgA1) [31]. Compared with healthy individuals, patients with IgAN exhibit alterations in both the diversity and composition of the gut microbiota. These changes are correlated with clinical indicators such as estimated glomerular filtration rate (eGFR) and urine albumin-creatinine ratio (UACR), suggesting a link between intestinal dysbiosis and disease severity [32]. Gut dysbiosis may exacerbate the overproduction of pathogenic Gd-IgA1 by activating toll-like receptor 4 (TLR4) signalling pathway and promoting the expression of B-cell-activating factor (BAFF) [33]. Recent research has further revealed that the deficiency of the polypeptide N-acetylgalactosaminyltransferase 14 (GalNAc-T14) leads to aberrant O-glycosylation of intestinal mucins. This disruption interferes with the normal homing of B cells to mucosal sites, ultimately resulting in the large-scale entry of pathogenic Gd-IgA1 into the systemic circulation, where they form immune complexes that deposit in the kidneys [34]. These findings highlight the central role of the gut-kidney axis in the pathogenesis of IgAN.
Autoimmune thyroid disease (AITD) is an organ-specific autoimmune disorder that involves complex interactions among different components of thyroid tissue. Based on clinical outcomes and immune phenotypes, it can be categorized into two main types, Hashimoto’s thyroiditis (HT) and Graves’ disease (GD) [35]. Intestinal mucosal immunity may play a role in the inflammatory induction process of AITD. Specifically, the impairment of intestinal barrier integrity can lead to dysbiosis, resulting in a decrease in beneficial metabolic products such as SCFAs alongside increased exposure to harmful substances. These changes can ultimately promote excessive production of pro-inflammatory cytokines [36]. Furthermore, structural similarities between protein sequences in Lactobacillus and Bifidobacterium and thyroid autoantigens (peroxidase and thyroglobulin), may influence immune responses through molecular mimicry [37].
Gut mucosal immunity plays a dual role as both an amplifier and a regulator in ADs. Local barrier dysfunction and dysbiosis form a common pathogenic foundation, while specific microorganisms, metabolic products, or immune pathways interact with host genetic susceptibility to determine the specific target organs and clinical phenotypes of the autoimmune response. This provides a unified framework for understanding the common pathological basis of ADs and their individualized clinical manifestations. It also suggests that while restoring the integrity of the gut barrier is a universally applicable strategy, personalized management for different ADs still requires precise regulation of specific microbial strains and their metabolic products.
In addition to the gut, the respiratory mucosal immune system serves as a primary interface between the body and the external environment. Beyond serving as a physical barrier against pathogens, it plays a crucial role in the pathogenesis of ADs as discussed in this section.
During the high-risk period and early stages of RA, abnormal T cell–driven activation of B cells has been detected within the lung mucosa of patients, accompanied by high-frequency of somatic mutations and antibody class transitions. These processes facilitate the generation of citrulline-reactive autoantibodies [38]. This evidence supports the concept that the respiratory mucosa may serve as an important starting site for the autoimmune response of RA. Meanwhile, RA patients frequently exhibit mucosal dysbiosis, with the abundance of specific microbial taxa being correlated with anti-cyclic citrullinated peptide (anti-CCP) antibody levels, as well as various disease markers such as erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) [39]. Furthermore, in both individuals at high risk of RA and patients, neutrophils in the lungs are more prone to undergoing neutrophil extracellular trap formation (NETosis), thereby releasing a substantial array of citrullinated autoantigens [40], further underscoring the pivotal role of the local respiratory immune microenvironment in the pathogenesis of RA.
Respiratory mucosal immune dysregulation may directly contribute to the pathogenesis of IgAN through alterations in microbial structure and the aberrant activation of associated signalling pathways. A study has demonstrated that the respiratory microbiota in IgAN patients undergoes significant shifts, specifically characterised by an increased abundance of genera such as Neisseria and Streptococcus, alongside a reduction in Prevotella. Notably, immunosuppressive therapy has been shown to reduce the levels of Neisseria and Actinobacillus [41]. Kano et al. [42] found that the continuously activated toll-like receptor 9 (TLR9) signalling pathway can up-regulate the expression of key factors of B cells such as BAFF and a proliferation-inducing ligand (APRIL), thereby driving abnormal activation of B cells and the production of pathogenic Gd-IgA1. Similarly, nasal antigens can also activate nasal-associated lymphoid tissue (NALT) through the TLR9 signalling pathway, making it an important source of abnormal glycosylated IgA, ultimately leading to the deposition and damage of renal immune complexes [43].
RA and IgAN share several pathogenic features at the level of the respiratory mucosal immune system, including dysbiosis, breakdown of immune tolerance, and abnormal B cell activation. Both conditions highlight the importance of mucosa-associated lymphoid tissue. However, their core mechanisms differ. RA is primarily driven by the anti-citrullinated protein immune response and NETosis, both of which are initiated within the pulmonary mucosa, while IgAN is characterized by TLR9-driven excessive production of defective IgA. This illustrates the heterogeneity of diseases arising from the same mucosal immune system, influenced by the affected site, the nature of the antigens involved, and the dominant pathogenic pathways.
Similar to the gut and respiratory tract, the oral cavity contains a complex mucosal immune system, the disruption of which is related to the pathogenesis of ADs.
An existing study has found that anti-citrullinated protein antibody (ACPA),
anti-carbamylated protein (anti-CARP), and IgA-type anti-aminoacyl protein
antibodies (AAPA) can be detected in the saliva of RA patients, suggesting that
there may be local secretion of autoantibodies in their oral mucosa [44].
Furthermore, the prevalence of periodontal disease is higher in RA patients
compared to healthy individuals, and effective periodontal treatment can help
improve RA’s disease activity [45]. These observations imply that oral mucosal
immunity may be associated with RA through mechanisms related to periodontal
inflammation. Citrullinated oral bacteria caused by periodontitis repeatedly
enter the bloodstream, not only activating the inflammatory monocyte subsets
related to synovitis, but also continuously stimulating the ACPA immune response,
promoting the spread of autoimmune reactions to endogenous citrullinated
antigenic epitopes, thereby aggravating the progression of RA [46]. Notably,
Moentadj et al. [47] reported that the abundance of
Streptococcus species is elevated in the oral cavity of patients with
RA. Moreover, further research has confirmed that Streptococcus
parasanguinis strains can induce the production of pro-inflammatory cytokines,
such as tumour necrosis factor-
Oral lesions, as common clinical manifestations of SLE, have been included in the diagnostic criteria of the American College of Rheumatology (ACR), among which oral ulcers are one of the important indicators [48]. A meta-analysis revealed that approximately 31% (95% confidence interval [CI]: 28%, 35%) of SLE patients develop oral mucosal lesions. These lesions manifest in various forms, including typical ulcers, oral erythema, candida infection and purpura [49]. Moreover, the degree of oral microbiota dysbiosis in SLE patients correlates with clinical indicators and disease activity, suggesting that the local oral immune microenvironment is involved in the disease process [50].
Sjogren’s syndrome (SS) mainly affects the salivary glands and lacrimal glands. Chronic inflammation of the glands can cause functional disorders, thereby triggering clinical symptoms mainly characterized by dry mouth and dry eyes [51]. Singh et al. [52] reported that levels of Veillonella parvula in both subgingival and supragingival plaque samples from SS patients were elevated compared to healthy controls, suggesting that this bacterium may serve as a potential specific microbial biomarker for the disease. Notably, Alam et al. [53] further proposed that oral dysbiosis might lead to bacterial invasion into salivary gland ductal cells, thereby interfering with salivary gland epithelial function and activating the type I interferon (IFN) signalling pathway. Furthermore, bacterial infection of the salivary glands may persistently exacerbate local inflammation and secretory dysfunction, ultimately driving the pathological progression of SS.
Overall, these findings suggest that although dysbiosis and barrier dysfunction in the oral cavity are common features of RA, SLE, and SS, the downstream pathogenic pathways differ significantly. RA is primarily driven by oral pathogen-induced molecular mimicry and the systemic dissemination of autoantibodies. SLE is characterised by a correlation between typical mucosal ulcerations and disease activity, whereas SS involves direct bacterial invasion into ductal cells and the subsequent activation of the IFN signalling pathway, ultimately leading to glandular dysfunction. This difference highlights that while restoring oral microbial homeostasis is a universal therapeutic goal, it is essential to address the unique mucosal immunopathology of each disease.
Beyond the gut, respiratory tract, and oral mucosa, urogenital tract stands as another critical immune interface. Dysbiosis in its microbiota and associated local immune dysregulation have also been demonstrated to correlate with certain ADs. In RA patients, the urinary microbiome exhibits characteristic alterations, marked by a significant increase in the relative abundance of Proteus, Enterococcus, and Bacteroides. Notably, the enrichment of these genera correlates positively with the disease activity score in 28 joints-erythrocyte sedimentation rate (DAS28-ESR) and peripheral plasma B-cell levels [54]. Similarly, in the bladder microbiota of patients with SLE, comparable associations between microbial composition and immune markers have been observed. Specifically, the abundance of Megasphaera and Phocaeicola correlates negatively with serum complement component 3 (C3) levels, whereas Streptococcus shows a positive correlation with serum immunoglobulin G (IgG) levels [55]. These findings suggest that the urogenital mucosa may modulate local and systemic immune responses through its specific microbial composition, thereby influencing the pathological progression of ADs.
The above findings have indicated that mucosal immune disorders are involved in the occurrence and development of ADs. Although the mucosal sites and specific immune mechanisms involved in different diseases have their own focuses, there are also important common pathways and interrelationships among them. The key mechanisms of mucosal immunity in ADs are shown in Table 1 (Ref. [12, 13, 14, 16, 17, 18, 19, 21, 22, 24, 25, 26, 28, 29, 30, 32, 33, 34, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 49, 50, 52, 53, 54, 55]). Furthermore, we have provided an integrative diagram of cross-mucosal mechanisms in ADs (Fig. 2).
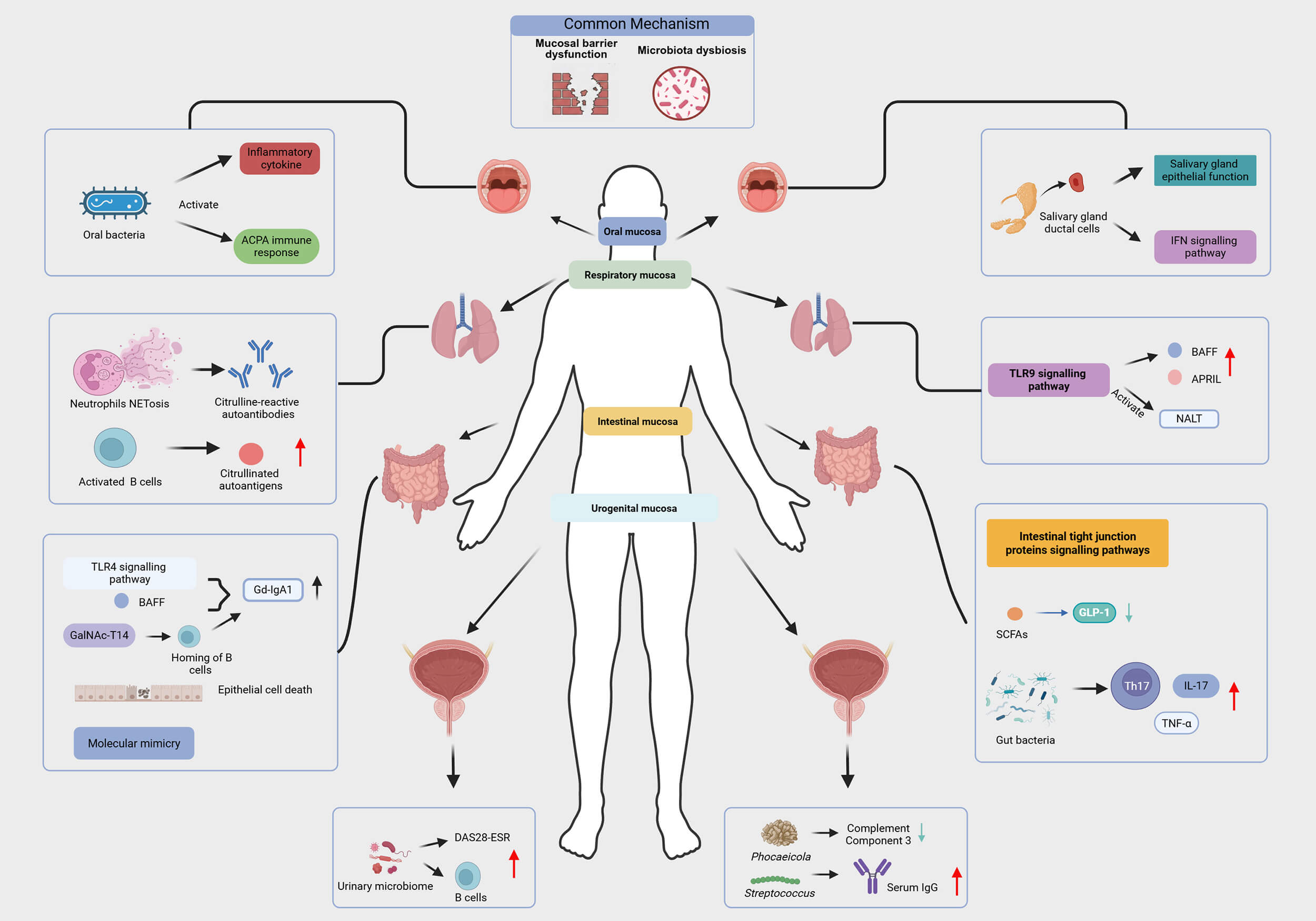 Fig. 2.
Fig. 2.
Mechanisms of cross-mucosal immunity in autoimmune diseases.
Notes: ACPA, anti-citrullinated protein antibody; IFN, interferon; TLR, toll-like
receptor; GLP-1, glucagon-like peptide-1; Th17, T helper 17; IL-17,
interleukin-17; DAS28-ESR, disease activity score in 28 joints-erythrocyte
sedimentation rate; IgG, immunoglobulin G; BAFF, B-cell-activating factor; NALT,
nasal-associated lymphoid tissue; APRIL, a proliferation-inducing ligand;
Gd-IgA1, galactose-deficient immunoglobulin A1; NETosis, neutrophil extracellular
trap formation; GalNAc-T14, polypeptide N-acetylgalactosaminyltransferase 14;
TNF-
| Mucosal site | ADs | Mechanisms | References |
| Intestinal mucosa | SLE | Gut microbiota dysbiosis; Bacterial translocation and molecular mimicry; The elevation of zonulin levels. | [12, 13, 14] |
| Intestinal mucosa | RA | Abnormal activation of intestinal tight junction protein-related signaling pathways; Gut microbiota promotes the expression of IL-17 and Th17-related cytokines and modulate bile acid metabolism. | [16, 17, 18, 19] |
| Intestinal mucosa | IBD | Dysbiosis of the intestinal commensal microbiota; Impairment of intestinal barrier integrity. | [21, 22] |
| Intestinal mucosa | T1DM | Dysbiosis of the gut microbiota impairs SCFA production and subsequently diminishes GLP-1 secretion and drives increased inflammation. | [24, 25, 26] |
| Intestinal mucosa | MS | Microbiota dysbiosis activates MAIT cells, which then enter the central nervous system and produce pro-inflammatory cytokines. | [28, 29, 30] |
| Intestinal mucosa | IgAN | Gut microbiota dysbiosis activates the TLR4 signalling pathway and promotes the expression of BAFF; Abnormal glycosylation of intestinal mucins interferes with the proper homing of B cells to mucosal sites. | [32, 33, 34] |
| Intestinal mucosa | AITD | Microbial dysbiosis drives the secretion of pro-inflammatory factors; Molecular mimicry. | [36, 37] |
| Respiratory mucosa | RA | Abnormal B-cell activation in lung mucosa leading to anti-citrullinated antibodies; Neutrophils in the lungs undergo NETosis, thereby releasing large amounts of citrullinated autoantigens. | [38, 39, 40] |
| Respiratory mucosa | IgAN | Abnormal activation of the TLR9 signaling pathway. | [41, 42, 43] |
| Oral mucosa | RA | Oral bacteria trigger inflammatory cytokine production and drive ACPA immune responses. | [44, 45, 46, 47] |
| Oral mucosa | SLE | Oral mucosal lesions; Oral microbiota dysbiosis correlates with disease indicators. | [49, 50] |
| Oral mucosa | SS | Bacteria invade salivary gland ductal cells, thereby interfering with salivary epithelial function and activating the IFN signalling pathway. | [52, 53] |
| Urogenital mucosa | RA | Urinary microbiota is positively correlated with the DAS28-ESR and peripheral plasma B-cell levels. | [54] |
| Urogenital mucosa | SLE | Bladder microbiota is associated with C3 and IgG levels. | [55] |
Notes: ADs, autoimmune diseases; SLE, systemic lupus erythematosus; RA, rheumatoid arthritis; IBD, inflammatory bowel disease; T1DM, type 1 diabetes mellitus; SS, sjogren’s syndrome; MS, multiple sclerosis; IgAN, immunoglobulin A nephropathy; AITD, autoimmune thyroid disease; ACPA, anti-citrullinated protein antibody; IFN, interferon; TLR, toll-like receptor; GLP-1, glucagon-like peptide-1; Th17, T helper 17; IL-17, interleukin-17; DAS28-ESR, disease activity score in 28 joints-erythrocyte sedimentation rate; IgG, immunoglobulin G; BAFF, B-cell-activating factor; MAIT, mucosal-associated invariant T; C3, component 3; SCFA, short-chain fatty acid.
In the clinical management of ADs, systemic immunosuppressive therapy remains the basic approach. However, more targeted mucosal intervention strategies are increasingly attracting attention and becoming important supplementary means and cutting-edge directions. These therapies aim to restore immune tolerance, repair mucosal barriers or precisely regulate specific immune pathways.
Probiotics, an adjunct therapy, have garnered increasing attention for their
potential to promote health. They have also demonstrated promising applications
in the treatment of ADs, exerting positive effects by modulating intestinal
immune balance and systemic inflammatory responses [54, 55, 56, 57, 58, 59, 60, 61] (Table 2, Ref.
[56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75]). Nevertheless, several clinical trials indicate that probiotics may be
ineffective or even lead to adverse effects in certain conditions. For instance,
a randomized controlled trial revealed that Lactobacillus rhamnosus GG and Bifidobacterium lactis Bb12 strains did not
significantly maintain residual pancreatic
| Representative method | Primary effect | ADs | References |
| Probiotics | Increase levels of IL-2, FOXP3, IL-10, and TGF- |
SLE | [56] |
| Probiotics | Suppresses serum inflammatory cytokines in rats with CIA; Reduce joint swelling and cartilage tissue damage in rats with AIA. | RA | [57, 58] |
| Probiotic Capsules | Improve fasting and postprandial blood glucose and lipid levels. | T1DM | [59] |
| Probiotics | Improve hemoglobin, hematocrit, CRP, IgA, and IL-10 levels in UC patients. | UC | [60] |
| Probiotics | Increase BDNF levels in MS patients and reduce IL-6 level. | MS | [61] |
| Probiotics | Reduce IgA complex deposition and mesangial proliferation. | IgAN | [62] |
| FMT | Correct abnormal hypomethylation; Reduce SLEDAI-2K scores and the level of serum anti-dsDNA antibody. | SLE | [63] |
| FMT | Lower the DAS28 score. | RA | [64] |
| FMT | Control blood sugar levels and improve insulin resistance. | T1DM | [65] |
| FMT | Restore the patient’s gut microbiota to alleviate the disease. | IBD | [66] |
| FMT | Alleviate neurological symptoms in MS patients; Enhance neural function, reduce IL-17 levels, and restore intestinal homeostasis. | MS | [67, 68] |
| FMT | Reduce urinary protein levels in patients. | IgAN | [69] |
| FMT | Elevate levels of free thyroxine and free triiodothyronine. | AITD | [70] |
| Mediterranean diet | Modulate the composition of the gut microbiota and maintain the integrity of the gut barrier. | T1DM | [71] |
| Diet rich in pectin and inulin | Modulate the immune-inflammatory response. | RA | [72] |
| High-fiber diet | Rebalance the dysfunction of the gut-immune system-vascular wall axis. | SLE | [73] |
| Insoluble dietary fibers combined with polyphenols | Increase beneficial bacteria counts and reduce harmful bacteria abundance to improve disease prognosis. | UC | [74] |
| Gluten-free diet | Reduce the deposition of immune complexes. | IgAN | [75] |
Notes: ADs, autoimmune diseases; FOXP3, forkhead box protein 3; TGF-
Fecal microbiota transplantation (FMT) aims to restore gut microbiota balance and improve clinical outcomes by transferring the fecal microbiota from healthy donors to recipients [79]. This therapy, through the pivotal process of resetting the gut microbiome, has shown potential to alleviate symptoms across various ADs. Existing evidence spans from animal studies to clinical observations, supporting its value as an emerging therapeutic strategy, with specific positive effects (Table 2). Nonetheless, in practical applications, FMT still faces numerous unresolved issues and risks. Common adverse reactions include gastrointestinal symptoms such as bloating, diarrhea, and constipation [80]. The associated risks are particularly significant for immunocompromised individuals, as FMT may lead to severe adverse events like bacteremia, sepsis, and even multiple organ failure [81]. Notably, the effectiveness of FMT in ADs is not always positive. For instance, in a RA mouse model, FMT did not demonstrate a significant improvement in arthritis symptoms [82]. Additionally, germ-free mice that received fecal microbiota from donors with RA or IBD exhibited pathological changes such as cartilage alterations, claw deformities, elevated inflammatory mediators, and T lymphocyte activation, alongside behavioral abnormalities, occult bleeding, and gut dysfunction [83]. These findings suggest that the therapeutic outcome of FMT is not guaranteed and can be paradoxically detrimental, depending critically on the disease-specific composition and functional state of the donor microbiota.
Dietary patterns play a complex and crucial role in the management of ADs. In
T1DM, the Mediterranean diet may help delay disease progression and reduce the
risk of cardiovascular complications by modulating gut microbiota composition,
preserving intestinal barrier integrity, and thereby regulating host immune
responses [71]. Similarly, a high-fiber diet rich in pectin and inulin can
alleviate arthritis symptoms in CIA mice by modulating the gut microbiota and
regulating the abundance of butyrate [72]. This suggests that a high-fiber diet
may have the potential to modulate the immune-inflammatory response in RA. In the
case of SLE, dietary fiber interventions that adjust the composition of the gut
microbiota can rebalance the dysfunction of the gut-immune system-vascular wall
axis, thereby preventing the onset of hypertension [73]. Insoluble dietary fibers
combined with polyphenols improve outcomes in dextran sulfate sodium
(DSS)-induced UC in mice by increasing beneficial bacteria and reducing harmful
bacteria abundance [74]. However, not all high-fiber diets are universally
beneficial. Unfermented
Mucosal administration through nasal, oral, sublingual and rectal routes are non-invasive systemic approach to drug delivery. It offers several advantages, including ease of self-administration, rapid onset of action, and reduced first-pass metabolism, thereby facilitating prompt symptom control [86]. Additionally, this approach may induce more potent immune responses than traditional administration routes. By activating the shared mucosal immune system, this strategy can simultaneously trigger both effective systemic and localized mucosal immune responses, an outcome that is difficult to achieve with systemic delivery methods such as intravenous injection. Effective mucosal immune induction can also prevent pathogens from invading the body via mucosal pathways [87]. This strategy has demonstrated potential in both clinical trials and animal studies of ADs. In the experimental autoimmune encephalomyelitis (EAE) mice models, nasal administration enables the drug to pass the blood-brain barrier through the naso-brain pathway, allowing direct delivery to the central nervous system, thereby enhancing the therapeutic effect [88]. Clinically, targeted-release formulations like budesonide (Nefecon) are used to treat IgAN. These formulations are designed for precise delivery to the Peyer’s patches in the ileum, thereby inhibiting the local immune response while minimizing systemic adverse effects [89]. Notably, for the treatment of UC, the chitosan-modified microemulsion encapsulated nobiletin (NOB-CS-ME) can penetrate deep into the intestinal mucus layer, overcoming the limitations imposed by the intestinal mucus barrier. This approach enhances targeted delivery to the colonic epithelium, promoting the reconstruction of the intestinal barrier function and balancing M1/M2 polarization of macrophages, while inhibiting local inflammatory cascades [90].
Mucosal vaccines are immunological agents delivered through mucosal surfaces
such as the respiratory and gastrointestinal tracts, while nanoparticle vaccines
utilize advanced delivery systems based on nano-sized carriers to transport
antigens or immunomodulatory molecules. These nanoparticle vaccines demonstrate
enhanced targeting efficiency for the mucosal immune system and hold promise for
overcoming the limitations of traditional mucosal vaccines [91]. Some studies
indicate that nanoparticle vaccines exhibit unique advantages in the treatment of
ADs. By targeting antigen-presenting cells in mucosal-associated lymphoid
tissues, they can induce antigen-specific immune tolerance rather than
activation. Specifically, poly (lactide-co-glycolide) (PLG) nanoparticles loaded
with protein lipid peptide delivered to the lungs can effectively intercept
autoreactive T cells migrating to the central nervous system, alleviating
symptoms in animal models of MS [92]. Similarly, oral administration of targeting
nanoparticles loaded with heat shock protein 65-6
With the continuous emergence and clinical application of various novel
monoclonal antibodies, the arsenal of precision targeted therapies for IBD
continues to expand. As first-line treatments for IBD, these agents effectively
alleviate intestinal inflammation and promote mucosal healing [94]. In the
treatment of MS, interferon-
Small molecular drugs are expanding the boundaries of ADs treatment through multiple mechanisms. In the field of MS, the dimethyl fumarate (DMF) not only directly modulates immune cell function but also reshapes gut microbiota by reducing the abundance of Lachnospiraceae and Veillonellaceae, while regulating vitamin A metabolic pathways, thereby forming an intestinal-brain axis immune regulation network [96]. In the treatment of CD, the novel drug Upadacitinib has shown potential in promoting intestinal mucus healing and achieving endoscopic remission [97]. These advancements highlight the promising application of small molecule drugs in the treatment of ADs by synergistically regulating the immune system and gut microenvironment, while directly promoting mucosal repair.
This review summarizes that the dysregulation of mucosal immunity, characterized by barrier dysfunction and dysbiosis plays a crucial role in the pathogenesis of ADs. Therapeutic strategies targeting mucosal immunity are therefore expanding the current treatment landscape for ADs. Nonetheless, significant challenges remain, mainly due to the inter-individual heterogeneity in mucosal immune responses and insufficient understanding of mucosal immunopathological mechanisms in ADs. Future research should prioritise several key directions. First, integrating multi-omics technologies to reveal the underlying mechanisms linking mucosal immunity and ADs. Secondly, developing personalized intervention strategies based on individual microbiomes, such as utilizing next-generation probiotics or metabolic products to target specific immune-metabolic pathways. Finally, constructing novel mucosal vaccine carriers and delivery systems to achieve precise localization and controlled release of antigens or drugs at mucosal sites. These advances have the potential to drive a paradigm shift in ADs treatment, moving it from traditional systemic immunosuppression to the precise modulation of mucosal immunity. Such a shift represents a key step towards disease modification and lays an important foundation for establishing long-term and stable disease management pathways.
• Mucosal immune dysregulation is an early and specific driving factor of autoimmune diseases, potentially occurring before the clinical manifestations of the disease appear.
• Although mucosal barrier impairment and microbial imbalance form a common pathological basis, different mucosal regions still shape disease-specific immune pathways, with disease phenotypes determined by region-specific and pathway-specific mechanisms.
• Emerging strategies targeting mucosal immunity through targeted mucosal delivery, nano-vaccines, microbiome interventions, and precision biologicals enable a shift from broad immunosuppression to precise, disease-modifying therapies.
Not applicable.
HFW, HAM, SST, and HFP made substantial contributions to the conception and design of the review. HFW and HAM jointly conceived the study and drafted the initial manuscript. All authors made substantial contributions to conception. All authors contributed to revising the manuscript critically for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This study was funded by grants from the National Natural Science Foundation of China (82504495, 82273710).
Hai-Feng Pan is serving as one of the Editorial Board Members of this journal. We declare that Hai-Feng Pan had no involvement in the review of this article and has no access to information regarding its review. The authors declare no conflict of interest. Full responsibility for the editorial process for this article was delegated to Arianna Di Stadio.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


