1 Department of Trauma and Orthopaedics, Guy’s and St Thomas’ NHS Foundation Trust, SE1 9RT London, UK
2 Department of Trauma and Orthopaedics, East Kent Hospitals NHS Trust, CT1 3NG Ashford, UK
3 Department of Trauma and Orthopaedics, Medway NHS Foundation Trust, ME7 5NY Gillingham, UK
Abstract
Back pain is a common presentation in healthcare settings. The most serious cause is cauda equina syndrome (CES), a surgical emergency caused by acute compression of the lumbosacral nerve roots. In the UK, diagnosis requires a thorough clinical examination and emergency magnetic resonance imaging (MRI). Once confirmed on imaging, definitive treatment is urgent surgical decompression usually within 24 hours. Delays in diagnosis or treatment can lead to life-changing consequences including bowel and bladder incontinence, sexual dysfunction, lower limb paralysis and chronic pain. This narrative review provides a 2025 update to the 2023 review published in this journal. It covers the anatomy, epidemiology, aetiology, pathophysiology, classification, clinical presentation, examination, investigations, differentials, treatment, and management of CES. Major updates are based on the new UK 2025 Getting It Right First Time (GIRFT) interactive pathway. Important changes include: (1) the standardisation and timing of red flag symptoms, (2) the use of bladder scans as a rule-in rather than rule-out investigation, (3) aim for 24-hour MRI local diagnostic capabilities with standardized imaging sequences and proximal scanning in absence of cauda equina compression, (4) declassification of CES, (5) emphasis on all patients with established CES to receive urgent decompressive surgery as National Confidential Enquiry into Patient Outcome and Death (NCEPOD) level 1/2. This review uses the authors’ experience of suspected CES pathway improvement to propose a pathway for the management of suspected CES in the emergency department at district general hospitals without 24-hour MRI capability based on the most recent national GIRFT guidelines.
Keywords
- cauda equina syndrome
- spinal cord compression
- intervertebral disc herniation
- magnetic resonance imaging
- decompression
Back pain is a common presentation in primary and secondary healthcare settings. It accounts for 4% of general practitioner (GP) consultations [1] and 2% of emergency department presentations [2]. The aetiology can vary greatly and so it is vital to rule out the most serious causes which occur in 1% of cases [3, 4].
One of those is cauda equina syndrome (CES), a surgical emergency which involves
an acute or rapidly progressive compression of the lumbar and/or sacral nerve
fibres (the cauda equina) within the spinal canal [5]. Patients typically present
with back pain alongside new onset (
A major shift in understanding and management of CES has occurred within the last 5–10 years, with many reviews of CES conducted in the wider literature including within this journal. This paper provides an update to the review by Miller et al. [9] by incorporating the latest UK national guidelines for CES including British Association of Spine Surgeons (BASS) [10] and GIRFT [7]. Since this journal’s most recent review [9] notable changes include the standardisation and timing of red flag symptoms, the use of bladder scans as a rule-in rather than rule-out investigation, recommendation of 24-hour MRI local diagnostic capabilities with standardized imaging sequences, and emphasis on all patients with established CES to receive urgent decompressive surgery within 24 hours. There remains a scarcity of UK specific reviews following recent national guidance, hence this paper’s goal to provide an updated comprehensive overview of CES and provide key updates for its management within hospitals in the UK.
Within the UK, CES is managed inconsistently based on the clinical setting. The overall median time from hospital presentation to surgical decompression ranges from 17 hours to 29 hours. More specifically, the median time to MRI in emergency spinal units is 4 hours, 7 hours if transferred from an outside hospital, and 13 hours if the MRI is performed at a referring hospital [11]. Associated delays in diagnosis and treatment of CES have led to £186,134,049 worth of negligence claims against the National Health Service (NHS) between 2008–2018 [12]. This accounts for 23% of all claims related to spinal surgery [7]. Therefore, it is important for all healthcare providers to be able to recognise and manage CES, complying with these new national guidelines within their local set up.
This review aims to summarise current knowledge of CES in order to help healthcare providers understand, recognise, investigate and manage CES appropriately given their local setting in the context of updated national UK guidelines. This review will also propose a pathway for the management of suspected CES in the emergency department at district general hospitals without 24-hour MRI capability based on the most recent national guidelines [7] and the authors’ experience of suspected CES pathway improvement at local level [13].
The spinal cord terminates in the conus medullaris at the vertebral levels L1–L2 [14]. Spinal nerve roots then descend distally from this structure to form the cauda equina between L2–S5 (Fig. 1). The cauda equina is protected by the three meningeal layers, the dura mater, arachnoid mater, and pia mater, filled with cerebrospinal fluid within the lumbar cistern of the spinal canal [14].
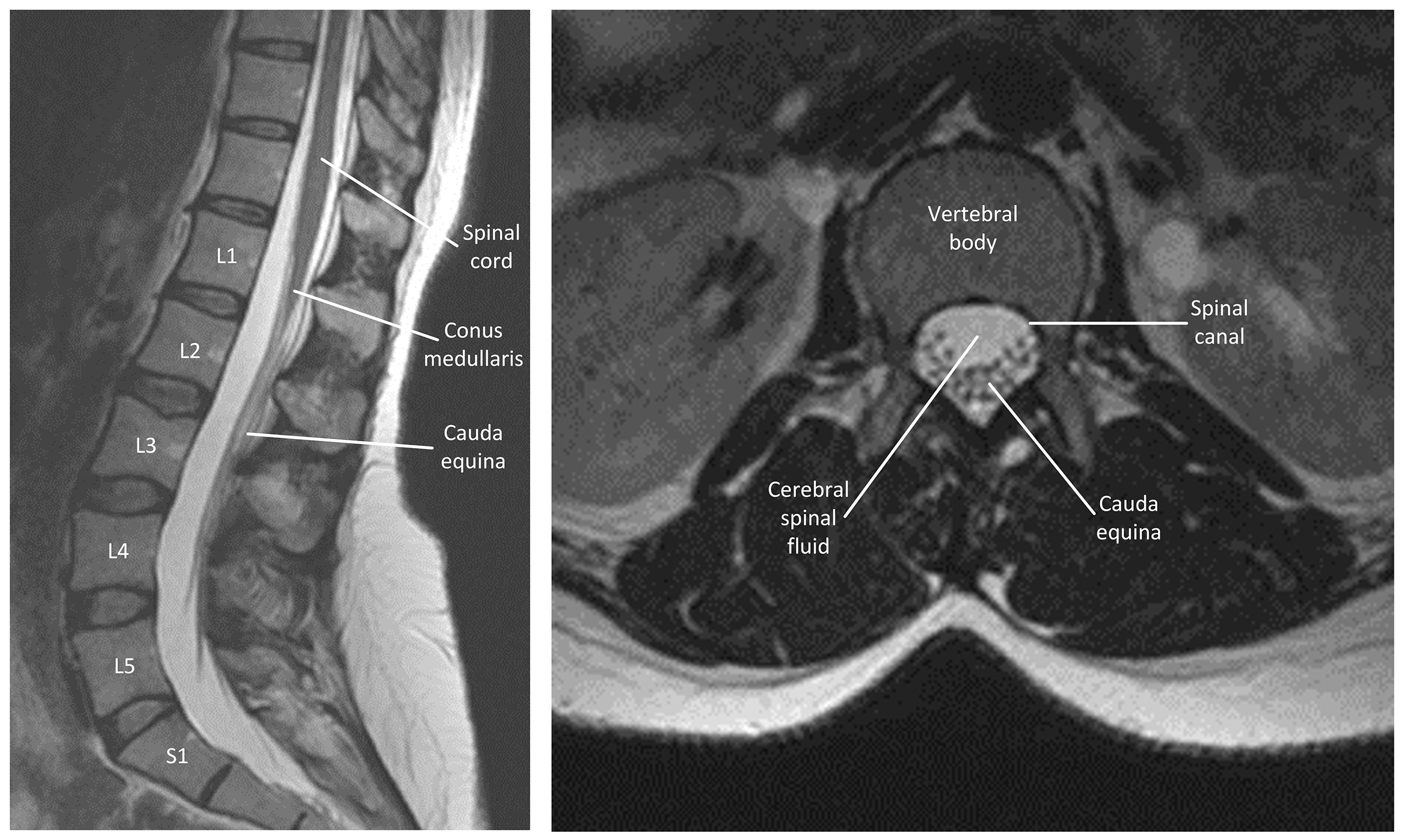 Fig. 1.
Fig. 1.
T2 weighted magnetic resonance images of a normal lumbar spine. The left sagittal image shows the spinal cord, conus medullaris, cauda equina and normal lumbar spine. The right axial image shows the L3 vertebral body and cauda equina in the vertebral canal. The image does not contain any identifiable patient data, therefore informed consent is not required.
The cauda equina contains dorsal root afferent fibres to provide sensory transmission for touch, proprioception, temperature, vibration and temperature, and ventral root efferent fibres for motor innervation to sphincters, perineum and legs as well as preganglionic parasympathetic fibres S2–S4 that innervate the bladder and sexual organs [15]. Each nerve root exists distally through the neural foramen directly below the same numbered pedicle.
As nerve roots have a motor and sensory component, damage to these structures will result in a flaccid paralysis of the muscles of the lower limbs dependent on the level of the lesion [16]. Damage will also lead to an areflexic bladder and bowel, reduced or absent ankle jerk reflexes and altered sensation in the perineal region.
Between L2–L4, the cauda equina receives its arterial supply from each corresponding lumbar and radicular artery [17]. Caudal to L5, the arterial supply is derived from a complex anastomosis between the middle sacral, iliolumbar, and lateral sacral arteries, and varies greatly between individuals [17]. As there are no lymphatic capillaries within the cauda equina, cerebrospinal fluid removes waste and carries immune cells to the peripheral lymph nodes.
A Scottish population-based study identified 149 CES cases in one year from a
population of 5.4 million, corresponding to a crude annual incidence of 2.7 per
100,000 [18]. An English epidemiological study of 256 participants with MRI
proven CES or red flag symptoms for CES showed that the median age of incidence
was 58
CES is rare in an asymptomatic population with an incidence of
CES is caused by compression of the nerve roots within the spinal canal (Fig. 2). It occurs in 3% of disc herniations [21], which accounts for 45% of CES cases [22]. This compression arises from a disc herniation at the L4/L5 or L5/S1 level [23]. Those with lumbar stenosis are predisposed to CES upon disk herniation owing to a narrower spinal canal [24]. The most common causes of CES are listed in Table 1 (Ref. [21]) below.
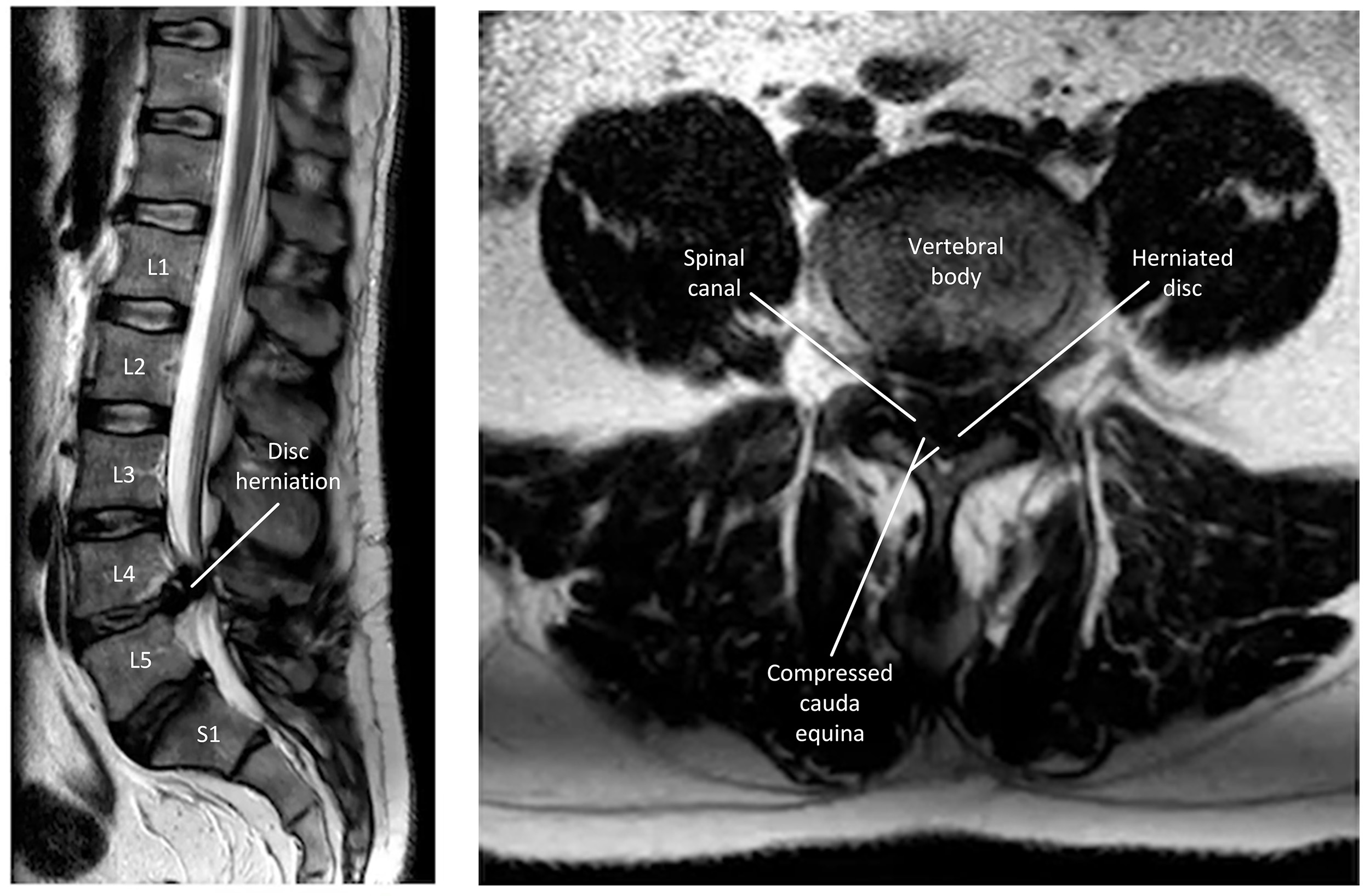 Fig. 2.
Fig. 2.
T2 weighted magnetic resonance images of cauda equina syndrome (CES). The left sagittal image shows L4 disc herniation leading to compression of the cauda equina. The right axial image shows the L4 disc occupying the space within the vertebral canal, compressing the cauda equina. The image does not contain any identifiable patient data, therefore informed consent is not required.
| Mechanism | Period prevalence per 100,000 |
| (1) Disc herniation | 5.2 |
| (2) Spinal fracture | 4.8 |
| (3) Other aetiologies | 1.3 |
| (4) Spinal tumours | 0.8 |
| (5) Iatrogenic lesions | 0.7 |
The table was extracted from Podnar [21].
Within the spinal canal, the cauda equina accounts for 44% of the available space within the dural sac [22]. A reduction of available space causes nerve root crowding, with progression leading to nerve compression. At the nerve compression stage, small changes to the pressure gradient in the neural compartment compromises blood supply within the microvasculature [25]. This leads to intraneural ischaemia and oedema, followed by demyelination and axonal Wallerian degeneration. Following the nerve injury, the chance of nerve regeneration is limited [26].
CES has had several classifications which aimed to guide timing of intervention and prognosis, however, in the most recent GIRFT guidance, classification has not been referred to. In line with the latest evidence, the authors agree that classification is not helpful for the assessment and management of emergency presentations [27, 28].
The original CES classification has had several adaptations to reflect the importance of timing of surgery [29]. Early CES (CESE) and incomplete CES (CESI) were prioritised for surgery as it is thought that they have the best chance of reversing neurological damage [30]. Comparatively, the need for immediate surgical decompression in retention CES (CESR) and complete CES (CESC) was felt to be less owing to permanent and likely irreversible nerve damage. Subsequent subclassifications were intended to inform how urgently patients should receive surgical decompression and are listed in Table 2 (Ref. [31]) below .
| Name | Definition |
| Suspected CES | Normal bladder, bowel, genital, perineal control and sensation |
| Bilateral sciatica or motor/sensory loss in the legs or magnetic resonance imaging confirmed disc herniation | |
| Early CES (CESE) | Normal bladder, bowel, genital control |
| Sensory loss in perineum or change in urinary frequency | |
| Incomplete CES (CESI) | Altered bladder function and sensation with maintained bladder control |
| With or without perineal sensory changes, or sexual, or bowel sensory, or functional changes | |
| Retention CES (CESR) | Painless urinary retention and overflow incontinence |
| With or without perineal sensory changes, or sexual, or bowel sensory, or functional changes | |
| Complete CES (CESC) | Painless urinary incontinence, complete loss of perianal sensation, anal tone, and anal squeeze |
The table was modified from Lavy et al. [31].
However, poor interobserver reliability and unpredictable speed of progression between different stages of CES [28, 32, 33] has led to calls for subclassifications to be abandoned for clinical decision making [27]. The GIRFT interactive pathway no longer refers to CES categories [6]. Instead, all patients with red flag symptoms for CES require an “emergency MRI as soon as possible, and certainly within 4 hours of request”, and all patients with MRI confirmed CES require surgery as urgently as any National Confidential Enquiry into Patient Outcome and Death (NCEPOD) 1/2 emergency—time sensitive and life-changing, but not life-threatening.
Shortcomings of CES subclassifications are: (1) it does not change the management according to the GIRFT guidelines, (2) the emphasis for CES management for healthcare providers is recognition of warning signs and diagnosis, not classification, and (3) some classifications may lead to a reduced sense of urgency. The positive aspect of the classification is that there is potential for prognostic benefits and consenting for surgery given that CESR has a reduced likelihood to recover functionality compared to CESE or CESI. Considering these arguments, the authors urge healthcare providers not to focus on classifications when diagnosing CES, but rather focus on identifying the red flag symptoms, and if present, refer for an emergency MRI. If CES is then confirmed, the healthcare provider should try to facilitate surgery as soon as possible.
Patients may present with back pain in primary or secondary clinical settings. Therefore, it is crucial that all healthcare providers know how to take a detailed history and conduct a thorough clinical examination to identify red flag features of CES.
A patient presenting with any of the red flag symptoms of CES must be referred to the nearest facility with MRI provision [7]. In practice, this means that patients who present in GP or community are sent immediately to the nearest emergency department.
Red flag symptoms are new onset (
• Difficulty initiating micturition or impaired sensation of urinary flow.
• Altered perianal, perineal, genital sensation (S2–S5 dermatome).
• Severe or progressive neurological deficit of both legs.
• Loss of sensation of rectal fullness.
• Sexual dysfunction.
The latest national guidelines highlight the warning signs for CES [7]. These
include sudden onset bilateral leg pain (sciatica), or unilateral leg pain that
has become bilateral. If these patients do not have any of the other red flag
symptoms for CES, they require an urgent (
Patients who are found not to have CES must be safety-netted so that they know when to seek help if their condition deteriorates. The national guidelines advocate for the distribution of a leaflet produced by the Musculoskeletal Association of Chartered Physiotherapists [7, 34]. This leaflet is available online in 35 languages and also includes a video. The leaflet highlights CES symptoms for patients as:
Pain radiating down one or both legs and/or severe lower back pain (any combination, seek help immediately):
• loss of feeling pins and needles between your inner thighs or genitals.
• numbness in or around your back passage or buttocks.
• altered feeling when using toilet paper to wipe yourself.
• increasing difficulty when you try to urinate.
• increasing difficulty when you try to stop or control your flow of urine.
• loss of sensation when you pass urine.
• leaking urine or recent need to use pads.
• not knowing when your bladder is either full or empty.
• inability to stop a bowel movement or leaking.
• loss of sensation when you pass a bowel motion.
• change in ability to achieve an erection or ejaculate.
• loss of sensation in genitals during sexual intercourse.
Not covered by the GIRFT guidelines are symptoms of “grumbling” CES, the gradual onset of CES symptoms in older adults with spinal stenosis [35]. Symptoms of saddle paraesthesia, urinary or bowel incontinence may fluctuate with time given the change in spinal position and may not always be acute in nature [33]. Clinicians must be aware of this presentation and ensure any new or changed symptoms are appropriately investigated through either an emergency or urgent elective pathway dependent on the acuity of symptoms in line with GIRFT CES guidance.
CES is a clinical diagnosis based on patient symptoms and signs supported by MRI findings [7]. Positive examination findings only have a 30% positive predictive value of diagnosing CES [36]. This has led to disagreement in the literature as to which tests to include when examining a patient with suspected CES—most notably whether to include anal tone [37].
The GIRFT pathway red flags suggest a subjective report of altered perianal, perineal, genital sensation is sufficient to warrant an emergency MRI [6]. However, given that CES presents as a varied combination of signs and symptoms [27], the authors advocate for a comprehensive neurological examination to include perianal sensation, anal tone, anal squeeze, and if the patient is catheterised, a catheter tug test for bladder sensation or the bulbocavernosus reflex [36, 38]. A patient that produces a single positive test suggestive of CES should receive an emergency MRI [27]. A combination of negative examination findings have provided a negative predictive value of 96–100% in a small case series of ten patients [36].
The lower limb neurological examination should include tone, power, reflexes and sensation [39]. Tone and power must be tested across myotomes L2–S1, sensory innervation across dermatomes L1–S5, and reflex testing to include L3–L4 and S1–S2 for the ankle jerk reflex [39]. Table 3 summarises the testing areas across dermatomes, myotomes and reflexes.
| Dermatome | Sensory testing |
| L1 | inguinal region and proximal medial thigh |
| L2 | anterior and lateral aspect thigh |
| L3 | medial femoral condyle |
| L4 | medial malleolus |
| L5 | dorsum of the foot at the third metatarsophalangeal joint |
| S1 | lateral aspect calcaneus |
| S2 | popliteal fossa |
| S3 | horizontal gluteal crease |
| S4 | perianal area |
| S5 | anal verge/anal skin |
| Myotomes | Motor testing |
| L2 | hip flexion |
| L3 | knee extension |
| L4 | ankle dorsiflexion |
| L5 | hallux extension |
| S1 | ankle plantar flexion |
| Reflexes | |
| L3–L4 | knee jerk |
| S1–S2 | ankle jerk |
| S2–S4 | bulbocavernosus reflex |
In practice, only perianal sensation and anal tone are commonly tested, however, a recent large study of 1005 patients found that only 35% of MRI positive CES had reduced anal tone and 51.5% of MRI positive CES had abnormal perianal sensation [37]. Examinations with a higher positive predictive value of up to 100% in a small case series of ten patients include the anal squeeze test and bulbocavernosus reflex [36, 40].
The following examinations are conducted in the left lateral position to visualise the anus. Perianal sensation is tested by sweeping a gloved finger from the outside towards the sphincter on both the left and right buttocks [41]. A folded paper clip or a blunt red fill needle can be used to check pinprick sensation. In CES, the patient may have reduced or totally impaired sensation. Anal tone is tested by inserting a gloved finger into the anus and assessing the compliance of the anal musculature to palpation as normal, abnormal or absent. Voluntary anal contraction is tested by asking the patient to squeeze the inserted finger. Absent anal tone or anal squeeze is suggestive of CES. The bulbocavernosus reflex is tested by squeezing the glans penis, clitoris or tugging the catheter whilst observing for anal contraction. This examination is controversial and often not accepted in non-catheterised patients [42]. In CES the sacral nerve roots are compressed, resulting in a lack of anal contraction.
Babinski reflexes and clonus are commonly tested with spinal examinations—though CES cannot include these upper motor neurone signs anatomically, the presence of upgoing Babinski or clonus in the context of CES may prompt the provider to expand their examination or imaging consideration to include the whole spine. Rare causes such as conus medullaris syndrome (compression at L1) or traumatic spinal cord injury (where multi-level injury is not uncommon) may present with upper motor neurone signs in the lower limb alongside urinary symptoms. The authors feel it is pragmatic to consider including these additional examinations within routine CES examination.
National guidelines include a post-void bladder scan as a necessary
investigation for suspected CES [6]. A post-void residual volume
However, a bladder scan must not be used in isolation in deciding whether a
patient receives an MRI scan [7]. This follows a study by Woodfield et al. [32]
who found that 60% of patients who underwent emergency decompressive surgery for
CES had post-void residual volume
In practice, if the patient is unable to void then they should receive a bladder
scan followed by catheterisation and catheter tug if the bladder volume is
When taken in combination, negative signs across neurological examination,
perianal sensation, anal tone, anal squeeze, bulbocavernosus reflex, post void
residual bladder volume
MRI is the gold standard investigation to rule out CES [7]. The request for MRI does not need to be discussed with the on-call spinal services as this may delay receiving an MRI, but should include a senior decision maker which GIRFT identifies as the level of Specialty Trainee 4 (ST4) or above [7].
The patient should be kept nil by mouth in case surgery is required and the patient should be prioritised over all other elective or routine cases. The on-call surgical teams can review out-of-hours MRI scans if a radiologist report is unavailable. If there is an absolute contraindication to MRI, such as non-compatible implanted metalwork or pacemakers, then a computed tomography (CT) scan can be used.
Despite national guidelines setting June 2024 as the deadline for local 24-hour MRI provision most hospitals in the UK still do not have this capability [7]. Instead, all hospitals should be part of one of 14 Spinal Services Clinical Networks [45]. Part of this network includes the blue light transfer of patients with suspected CES between hospitals in order to facilitate an emergency out-of-hours MRI capability.
The MRI protocol should be based on a sagittal T2 weighted sequence as defined by GIRFT [7] and designed in collaboration with the Royal College of Radiologists. If cauda equina compression is identified by the radiographer, then additional images of an axial T2 and sagittal T1 weighted image should be acquired. If no cauda equina compression is identified, then a single T2 sagittal sequence including the cervical and thoracic spine is needed, as more proximal pathology could be identified. Surgical teams can interpret the MRI report out of hours to save time waiting for a radiologist report. A report should be made available within 1 hour of scan completion.
There are a limited number of non-compressive differentials which may mimic symptoms of CES. They are listed in Table 4 below.
| Category | Condition | Distinguishing features | UMN/LMN signs | Timing of symptoms |
| Combination | Cauda equina syndrome | Back or bilateral leg pain and one of Bilateral neurological leg dysfunction, bowel/bladder incontinence, sexual dysfunction, altered perineal/genital sensation | LMN | Acute |
| Lumbosacral plexopathy | Asymmetric LMN signs, pelvic tumour, radiation history | LMN | Variable | |
| Conus medullaris syndrome | Saddle anaesthesia, mixed signs, earlier bladder involvement | Mixed UMN & LMN | Acute or subacute | |
| Autoimmune | Guillain-barré syndrome | Ascending symmetrical weakness, areflexia, recent viral illness | LMN | Subacute (days–weeks) |
| Multiple Sclerosis | Upper limb, visual, or facial involvement, relapsing course | UMN | Variable | |
| Congenital | Spinal arteriovenous malformation | Sudden or progressive myelopathy, may mimic CES | UMN or LMN | Variable |
| Infective | Human immunodeficiency viruses-associated myelopathy | Slowly progressive, spasticity, ataxia | UMN | Chronic |
| Vascular | Spinal cord infarct | Sudden paraplegia, may follow hypotension or embolism | UMN | Acute |
| Inflammatory | Transverse myelitis | Sensory/motor/autonomic dysfunction, often post-viral | UMN | Subacute (days–weeks) |
| Psychiatric | Functional neurological disorder | Non-dermatomal sensory loss, inconsistent exam | None/Variable | Sudden or fluctuating |
UMN, upper motor neurone; LMN, lower motor neurone.
Definitive management is emergency surgical decompression. This intervention is time critical as the symptoms of CES develop in a progressive fashion, and patients will more likely retain, rather than regain function at time of decompressive surgery [46]. Those patients who receive definitive surgery faster tend to have better outcomes [32].
National guidelines advocated for decompression within 24 hours of MRI [7]. This is a common theme in the literature and stems from a large metanalysis [47]. Since that publication, there continues to be agreement that the earlier the intervention, the better [10, 48, 49].
Whilst there is an acknowledgement that patients with painless urinary retention and overflow incontinence or symptoms of complete CES are less likely to benefit from an emergency intervention [30] and likelier to suffer a poorer prognosis overall, around 70% of these patients will still benefit from decompression [7]. Hence GIRFT recommends all patients with CES to still undergo emergency decompressive surgery within 24 hours.
The authors have experience of implementing the latest GIRFT [7] national guidelines in the setting of a district general hospital in the UK. This was as part of a quality improvement project to reduce the time from emergency department presentation to MRI scan. Owing to the lack of 24-hour-MRI availability in the local setting, two pathways were designed for use in-hours and out-of-hours by the doctors in the emergency department. If an out-of-hours MRI is needed, the patient is sent to the neurological centre with 24-hour MRI provision by blue light transfer. Such interhospital transfers are associated with significant financial costs. These pathways are shared in Fig. 3 below, and we recommend other district general hospitals implement pathways adapted to their local context based on the GIRFT national pathway to help doctors efficiently manage patients with suspected CES [13].
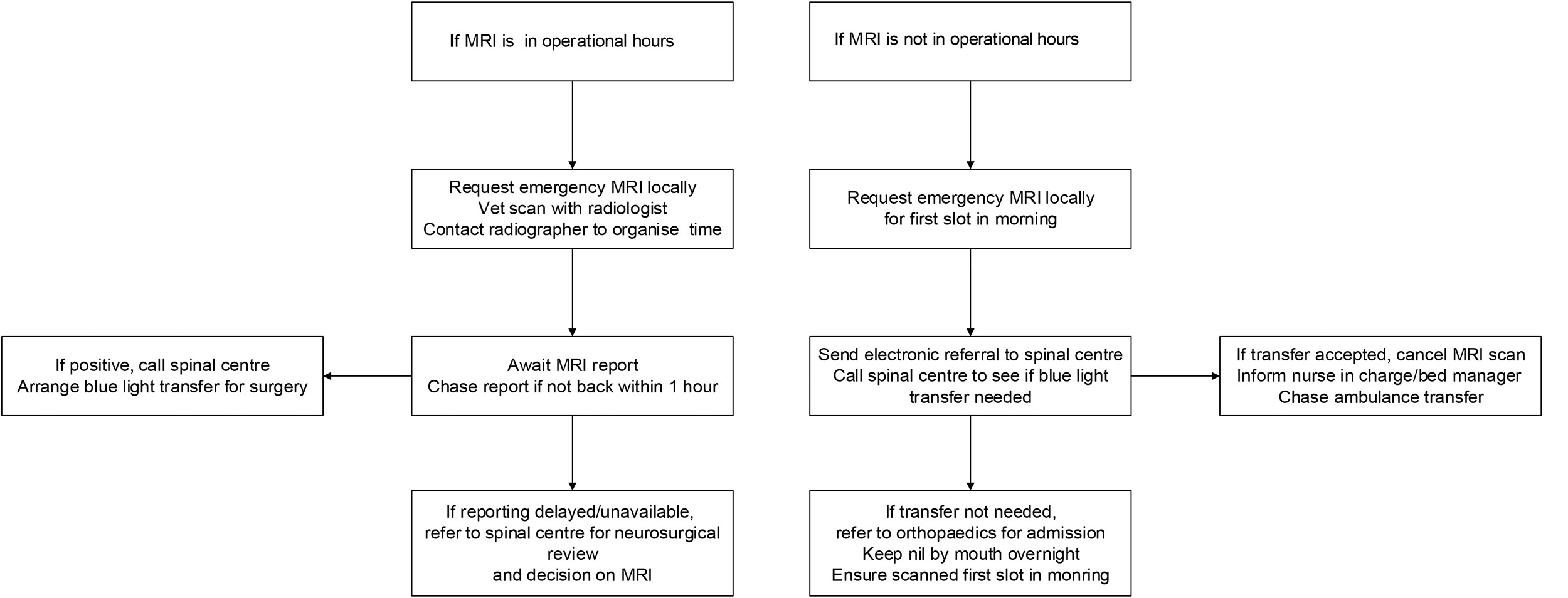 Fig. 3.
Fig. 3.
Suggested emergency department pathway to request a magnetic resonance image for suspected CES in-hours and out-of-hours at a hospital without 24-hour magnetic resonance imaging (MRI) provision.
Whilst in the future triage and diagnosis of CES will likely be supported by artificial intelligence [50, 51], it will remain imperative for healthcare providers to recognise the symptoms of CES, understand the need for timely diagnosis, and facilitate urgent surgical management.
Overall, the GIRFT pathway [6] is a useful tool for standardising and improving the care of patients with suspected CES. However, the authors note two shortcomings and areas for future improvement.
The first is that patients with CES often present in considerable pain [31]. Unfortunately, timely administration of analgesia is not mentioned in the interactive pathway nor the online PDF [6, 7]. Healthcare providers need to be aware of this and make every effort to administer prompt analgesia.
Secondly, GIRFT guidance excludes single leg radiculopathy without other red
flag features as an indication for emergency MRI. Though GIRFT guidance
acknowledges unilateral sciatica progressing to bilateral as a warning sign
warranting urgent musculoskeletal referral (
This review highlights important updates in the understanding and management of CES, particularly the implication of the recent UK national guidance from GIRFT. Recent literature places greater emphasis on early recognition of red flag symptoms, a thorough neurological examination, rapid access to MRI and urgent surgical decompression for all cases, with less emphasis on historical classification systems and bladder scans. Despite clear national recommendations, implementation remains challenging in hospitals without 24-hour MRI provision. Locally adapted clinical pathways and improved coordination within spinal service networks may help streamline investigation, referral and treatment. Continued efforts to improve clinician awareness, optimise diagnostic pathways and evaluate the real-world impact of GIRFT recommendations will be essential to ensure timely management and improve outcomes for patients with suspected CES.
• This review covers cauda equina syndrome (CES), a rare but important spinal condition that has life-changing consequences for back pain sufferers if not promptly diagnosed and treated.
• Identification of red flag symptoms for this condition is key, with targeted neurological assessment and emergent diagnostic MRI scanning for patients with suspected CES.
• New national guidance (GIRFT) provides the framework for local pathways for triage, referrals, diagnosis and surgical management.
• An example emergency department CES pathway is shown for hospitals without 24/7 access to MRI scanning facilities.
All the data of this study are included in the article.
Conceptualisation, AC, RJ, AL, MM; methodology, AC, RJ, AL, MM; software, AC, RJ, AL, MM; validation, AC, RJ, AL, MM; resources, AC, RJ, AL, MM; writing—original draft, AC, RJ, AL, MM; visualisation, AC, RJ, AL, MM; supervision, AL, MM; project administration, AC, AL. All authors contributed to revising the manuscript critically for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was conducted in accordance with the Declaration of Helsinki. The images do not contain any identifiable patient information, so there is no need to obtain the patient’s informed consent.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



