1 Department of Otolaryngology-Head & Neck Surgery, University Hospitals of Derby and Burton NHS Foundation Trust, DE22 3NE Derby, UK
2 Department of Otolaryngology-Head & Neck Surgery, Nottingham University Hospitals NHS Trust, NG5 1PB Nottingham, UK
Abstract
Head and neck cancer (HNC) is a complex and increasingly prevalent group of malignancies with high mortality and significant treatment-related morbidity. Risk factors include tobacco, alcohol, and human papillomavirus (HPV), with HPV-positive oropharyngeal cancers showing improved prognosis. HPV vaccination is reducing oncogenic strains and future disease burden. Common symptoms include swallowing and speech difficulties, airway obstruction, and neck masses. Multidisciplinary care remains the cornerstone of management, encompassing surgery, radiotherapy, chemotherapy, and emerging immunotherapy. This article aims to provide hospital-based clinicians with a concise overview of current innovations in HNC management. Key developments include transoral robotic surgery, radiogenomics, liquid biopsy, and the integration of artificial intelligence in diagnostics and treatment planning. The review also considers survivorship, caregiver burden, and the growing importance of coordinated community-based care. Emphasising patient-centred, multidisciplinary approaches, these advances aim to improve outcomes and quality of life for individuals affected by HNC across healthcare settings.
Keywords
- head and neck neoplasms
- squamous cell carcinoma of head and neck
- laryngeal neoplasms
- HPV
- liquid biopsy
- tumour biomarkers
- targeted therapy
- radiation therapy
Although head and neck cancer (HNC) encompasses a range of malignancies arising from tissues including thyroid, salivary gland, skin and epithelium of the upper aerodigestive tract, over 90% of HNC cases are squamous cell carcinomas originating from the squamous epithelium lining the oral cavity, pharynx and larynx [1], and these are the cancers discussed herewith. HNC is the seventh most common cancer worldwide, responsible for approximately 500,000 deaths annually [2, 3]. In the UK, HNC ranks as the eighth most common cancer overall and the fifth most common in men [4]. The incidence of HNC has increased by over 30% since the 1990s, largely due to a rise in human papillomavirus (HPV)-associated oropharyngeal cancers [1, 5, 6]. Despite improvements in survival driven by advances in treatment, mortality rates remain high: UK-based data show a 28–67% 5-year survival rate depending on tumour stage and subsite [5]. Early-stage disease (stage I or II) has long-term survival rates of 70–90% [2]; however, advanced disease (stage III/IV) has a 5-year survival rate of well below 50% [2, 3]. Alarmingly, 60% of HNC patients present with advanced disease, a figure that has remained unchanged in the UK for decades—in stark contrast to early detection in more common cancers [5]. As with other malignancies, early detection is the single best prognostic factor.
The purpose of this review is to explore key innovations, including transoral robotic surgery, immunotherapy, biomarker development, and artificial intelligence, as well as the broader context of community follow-up, caregiver burden, and system-wide service delivery. These advances are particularly pertinent in secondary care settings, where prompt diagnosis, individualised treatment planning, and integration with community services can significantly influence outcomes. These developments are particularly relevant for hospital clinicians, offering evidence-based guidance on diagnostics and treatment with direct implications for acute care, multidisciplinary collaboration, and coordinated discharge planning.
Historically, smoking and alcohol were identified as the primary independent risk factors for developing HNC, with their synergistic carcinogenic effects well established. Men are two to four times more likely than women to develop HNC [3, 7], with peak incidence occurring in the seventh decade of life [4]. Tobacco consumption in the UK has decreased by 50% since 1980 across both genders, which correlates with a reduction in laryngeal cancer, the subsite most strongly linked with tobacco use [1, 8]. In contrast, the rise in HPV-associated oropharyngeal squamous cell carcinoma (OPSCC) in recent decades has led to a shift in the typical sociodemographic profile of HNC patients [6, 7]. Although still predominantly affecting males, HPV-associated OPSCC is increasingly diagnosed in a younger population with absence of tobacco or alcohol use [6]. HPV is transmitted through oro-genital contact, and studies suggest that an increased number of oral sex partners significantly raises the risk of developing OPSCC [1, 6]. Although this phenomenon is observed globally, the increase in incidence has been most pronounced in Europe and the USA, with HPV-associated OPSCC now more common than cervical cancer in the UK [1, 6, 8]. Unlike laryngeal cancer, where most are diagnosed at an early stage, oral cavity and OPSCC are most commonly diagnosed at a late stage, which has huge implications for patient outcome [1]. Oral cavity cancers, traditionally associated with alcohol and smoking, have continued to rise, albeit less dramatically than OPSCC. Interestingly, UK studies have found that a higher proportion of patients with oral cavity cancer do not use tobacco or alcohol, and are female, without the same history of oro-genital contact [8, 9]. Additional risk factors include betel quid chewing (particularly prevalent in Asian and Australasian populations) [10], poor oral hygiene, low body mass index and a diet deficient in fresh fruit and vegetables [1]. While the incidence of oral cavity cancer has not surged as dramatically as OPSCC, the continued rise in cases may suggest a distinct patient group for whom HPV does not appear to be the primary driver [9].
Despite these changes, HNC remains disproportionately prevalent in lower socioeconomic groups. Even after adjusting for tobacco and alcohol consumption, household income has been shown to independently affect survival outcomes in HNC patients [11]. Ensuring equitable access to healthcare and support services is crucial in addressing this disparity.
HNC disproportionately affects older adults, many of whom present with comorbidities and reduced physiological reserves. Chronological age alone is a poor predictor of treatment tolerance; thus, comprehensive geriatric assessments (CGAs) are increasingly used to evaluate frailty, cognition, nutrition, and social support. These tools guide personalised care, helping to balance treatment benefit with potential harm. Frailty is associated with increased toxicity and poorer outcomes, highlighting the need for tailored treatment strategies, including possible de-intensification. Incorporating geriatric principles into HNC management improves decision-making, care planning, and patient quality of life [12].
Symptoms of HNC can be quite varied and are dependent on the affected subsite, but typically include dysfunction or disruption to breathing, voice, and swallow. Neck lumps, oral lesions and persistent sore throats are additional common presenting features [13]. The UK National Health Service (NHS) ‘2 week wait’ system was introduced at the turn of the century and remains the mainstay for primary care referrals for HNC services [14]. Referral criteria generally focus on persistent symptoms such as hoarseness, mouth ulcers, and non-resolving lumps, but conversion rates to diagnosis of malignancy remain less than 3%, highlighting the non-specific nature of symptoms or lack of general practitioner confidence in excluding cancer [15]. The planned evolution of a patient-reported symptom-based risk stratification system to redesign the suspected head and neck cancer referral pathway (EVEREST-HN) trial aims to address these challenges by using a patient-reported symptom-based risk stratification system to improve the suspected HNC referral pathway [16]. The NHS Long Term Plan aims for 75% of all cancers to be diagnosed at an early stage (I or II) by 2028, a target that poses a significant challenge for HNC, given the lack of reliable screening tools and the typically delayed nature of its clinical symptoms [17]. Despite improvements in treatment outcomes, early detection rates for HNC have not significantly improved over the past decade, with 60% of cases still diagnosed at an advanced stage [5].
Current screening strategies for head and neck cancer remain limited, particularly for high-risk groups such as young, non-smoking individuals with HPV-associated disease. This population often presents at an advanced stage due to the absence of traditional risk factors. There is a growing need to develop and validate risk-based screening algorithms tailored to emerging epidemiological patterns. Early detection tools such as circulating tumour DNA (ctDNA) and exosomes show promise as minimally invasive biomarkers, but further research is needed to assess their utility in routine surveillance and pre-symptomatic screening [18].
In an acute setting, patients may present with absolute dysphagia or with airway obstruction. The overlap in risk factors between HNC, cardiovascular, and pulmonary diseases can complicate diagnosis. Good clinical examination with emphasis on the progressive nature of symptoms, as well as eliciting additional locoregional symptoms and red flags, is essential [19].
The burden of HNC on the emergency department, both for patients presenting with a first diagnosis and advanced pathology as well as those suffering with the sequelae of disease or treatment, is significant [20]. Educating healthcare professionals about the relevant symptoms and risk factors is key to ensuring early identification, assessment, and treatment, which are crucial for optimal patient outcomes.
Decisions surrounding assessment and treatment are guided by the UK Head and Neck Cancer National Multidisciplinary Guidelines and the American Joint Committee on Cancer (AJCC) Staging Manual [21, 22]. Tumour staging is dependent on histological assessment obtained from biopsy, extension into surrounding structures as well as nodal disease or distant metastatic spread. The specific staging and classification of disease can vary by HNC subsite [21]. Due to the complexity and range of multimodal options available, treatment should ideally take place in a tertiary centre, following multidisciplinary team (MDT) discussions that incorporate individual patient factors, such as performance status, comorbidities, and treatment preferences [22]. UK guidance is for weekly MDT meetings that must be attended by ‘core’ members defined as ear, nose and throat (ENT) and oral and maxillofacial surgeons including those trained in reconstruction, oncologists, pathologists, head and neck radiologists and other allied healthcare professionals; dieticians, speech and language therapists, clinical nurse specialists, specialist restorative dentists [21, 22].
Patients with HNC have complex care needs that extend across hospital and community settings, placing significant demands on healthcare providers, families, and carers. These include specialised airway and nutritional support, tracheostomy or laryngectomy care, pain management, and psychological support.
Family members and carers play a crucial role in managing dietary modifications, enteral feeding via gastrostomy tubes, monitoring hydration and nutritional intake, and assisting with tracheostomy or laryngectomy care, which requires regular suctioning, stoma cleaning, and humidification. The emotional and psychological burden on carers is substantial, as they must adapt to complex caregiving tasks while supporting the patient’s social and emotional well-being.
Care coordination between hospital specialists, general practitioners, and allied health professionals, providing holistic and multidisciplinary approaches, is essential to addressing the physical, emotional, and practical challenges faced by both patients and their carers [23].
The Union for International Cancer Control (UICC)/American Joint Committee (AJC) 8th Edition tumour, lymph node, metastasis (TNM) Classification of Malignant Tumours [24] explains the system for describing the anatomical extent of disease (tumour) is based on the assessment of three components: T—the extent of the primary tumour, N—the absence or presence and extent of regional lymph node metastasis, and M—the absence or presence of distant metastasis. The addition of numbers to these three components indicates the extent of the malignant disease. All cases should be confirmed histo-cyto-pathologically. Table 1 demonstrates the overall staging based on tumour and node stage. Any evidence of metastasis (M1) will produce a staging of IV.
| N0 | N1 | N2 | N3 | |
| Tis | 0 | |||
| T1 | I | III | IVA | IVB |
| T2 | II | III | IVA | IVB |
| T3 | III | III | IVA | IVB |
| T4a | IVA | IVA | IVA | IVB |
| T4b | IVB | IVB | IVB | IVB |
HNC, head and neck cancer; Tis, Carcinoma in situ; TNM, tumour, lymph node, metastasis.
The objective of any treatment strategy for HNC is to achieve the highest possible cure rate with the lowest risk of morbidity. Treatment proposals should integrate both objective tumour parameters (e.g., tumour location, tumour histology, T stage, N stage) and patient parameters (e.g., age, performance status, comorbidities, previous history of cancer, occupation, expected functional outcome, personal preference).
Each patient with a new primary, or a recurrence/metastatic squamous cell carcinoma of the head and neck should be presented to a Head and Neck Cancer MDT where a treatment plan is agreed [25]. Broadly patients following TNM staging can be categorised into 3 groups. Early-Stage Disease (Stage I and Stage II, oral cavity, larynx, hypopharyngeal and p16-negative oropharyngeal cancer or T1N0/T2N0 p16-positive oropharyngeal cancer according to the UICC TNM 8th edition). Locally Advanced Disease (Stage III or IV oral cavity, larynx, hypopharynx and p16-negative oropharyngeal cancer, or T3/T4 with a nodal status between 0–3, and T1–4 p16-positive oropharyngeal cancers with nodal staging between N1–3 according to the UICC 8th edition). Or finally, Recurrent/Metastatic Disease (all patients with M1 disease) [26]. Treatment strategies for each group are presented in Figs. 1,2,3 respectively.
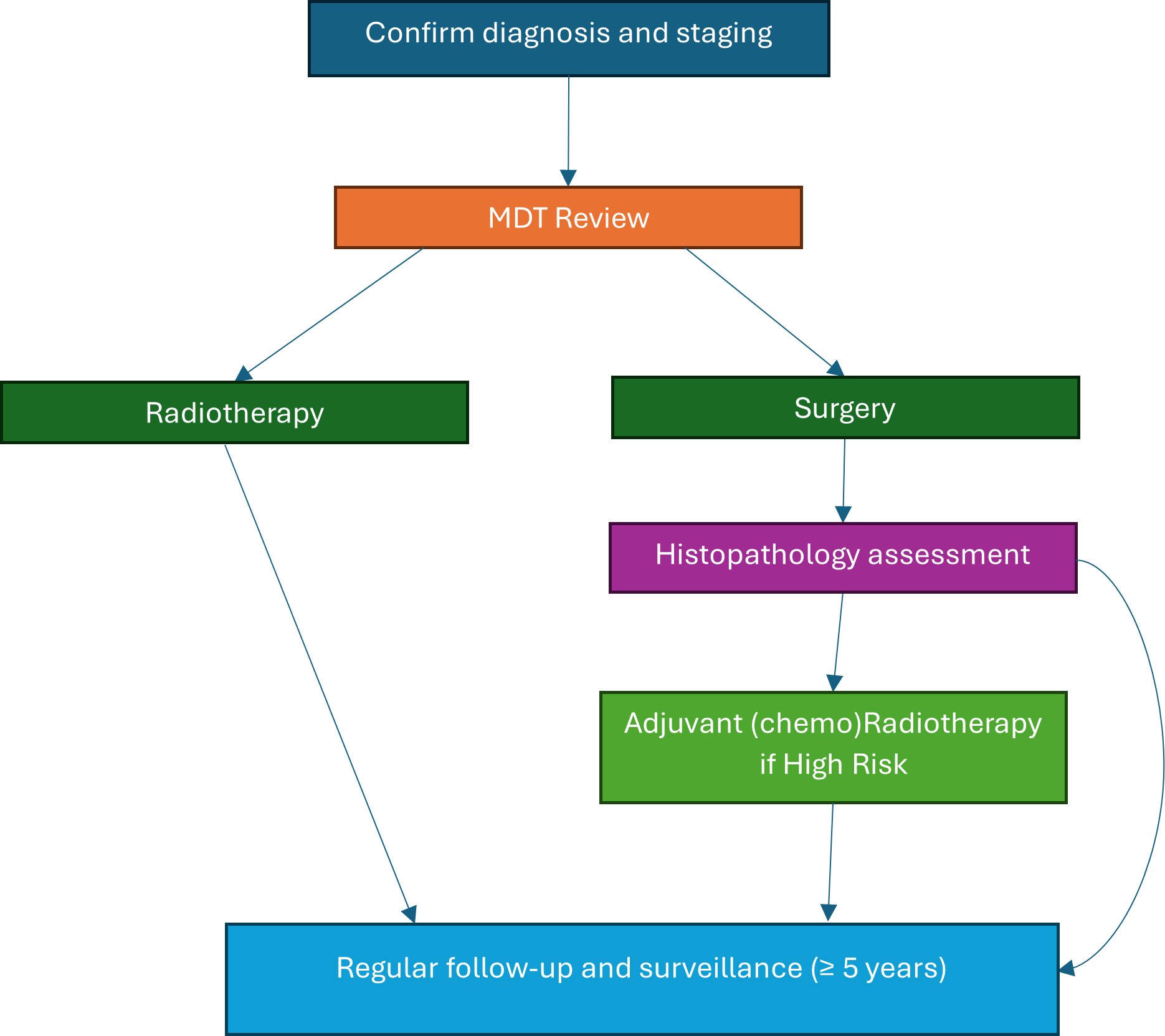 Fig. 1.
Fig. 1.
Treatment algorithm for early-stage HNC. The figure was created using Microsoft Word (version 2504, manufactured by Microsoft Corporation). MDT, multidisciplinary team.
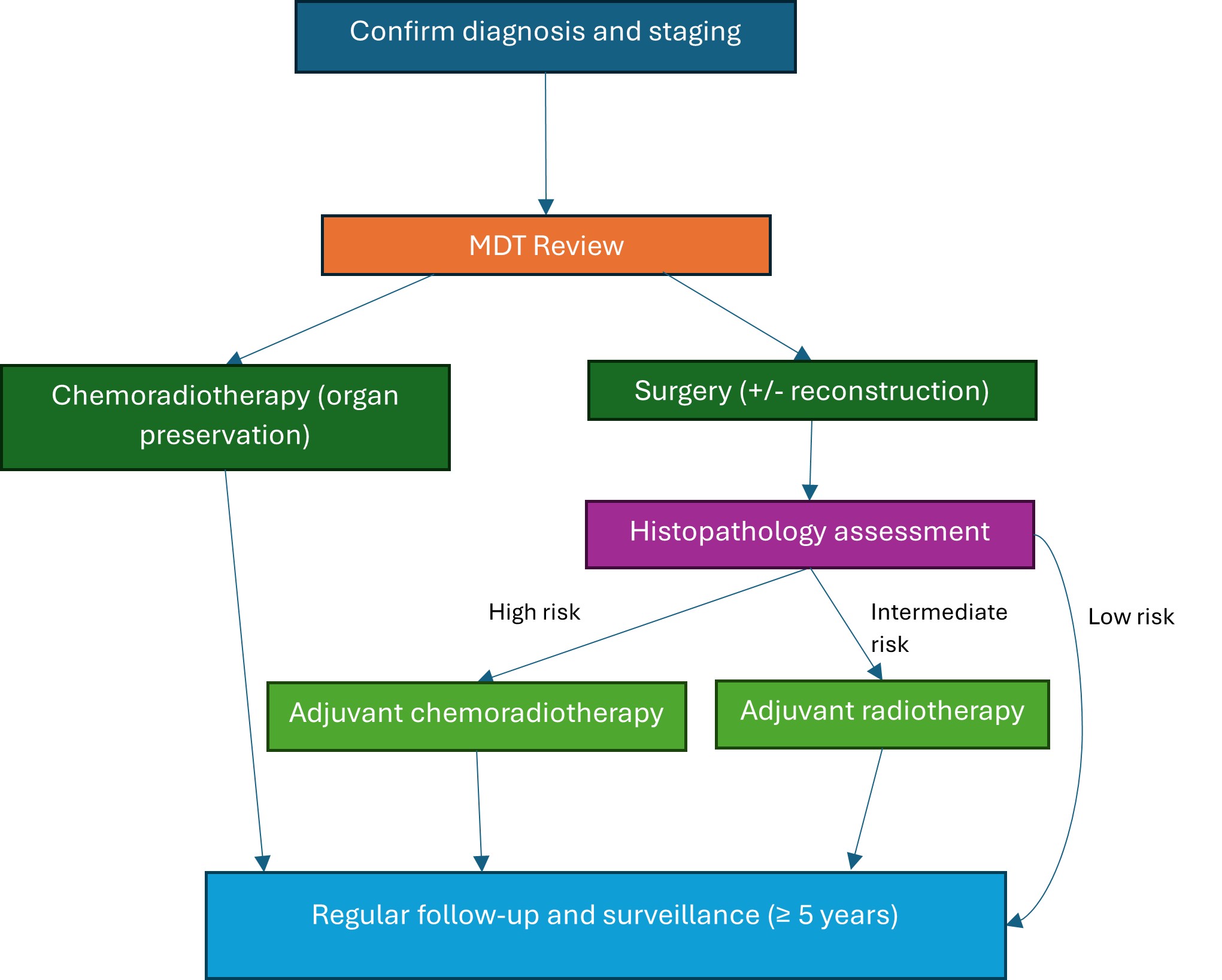 Fig. 2.
Fig. 2.
Treatment algorithm for locally advanced HNC. The figure was created using Microsoft Word (version 2504, manufactured by Microsoft Corporation). MDT, multidisciplinary team.
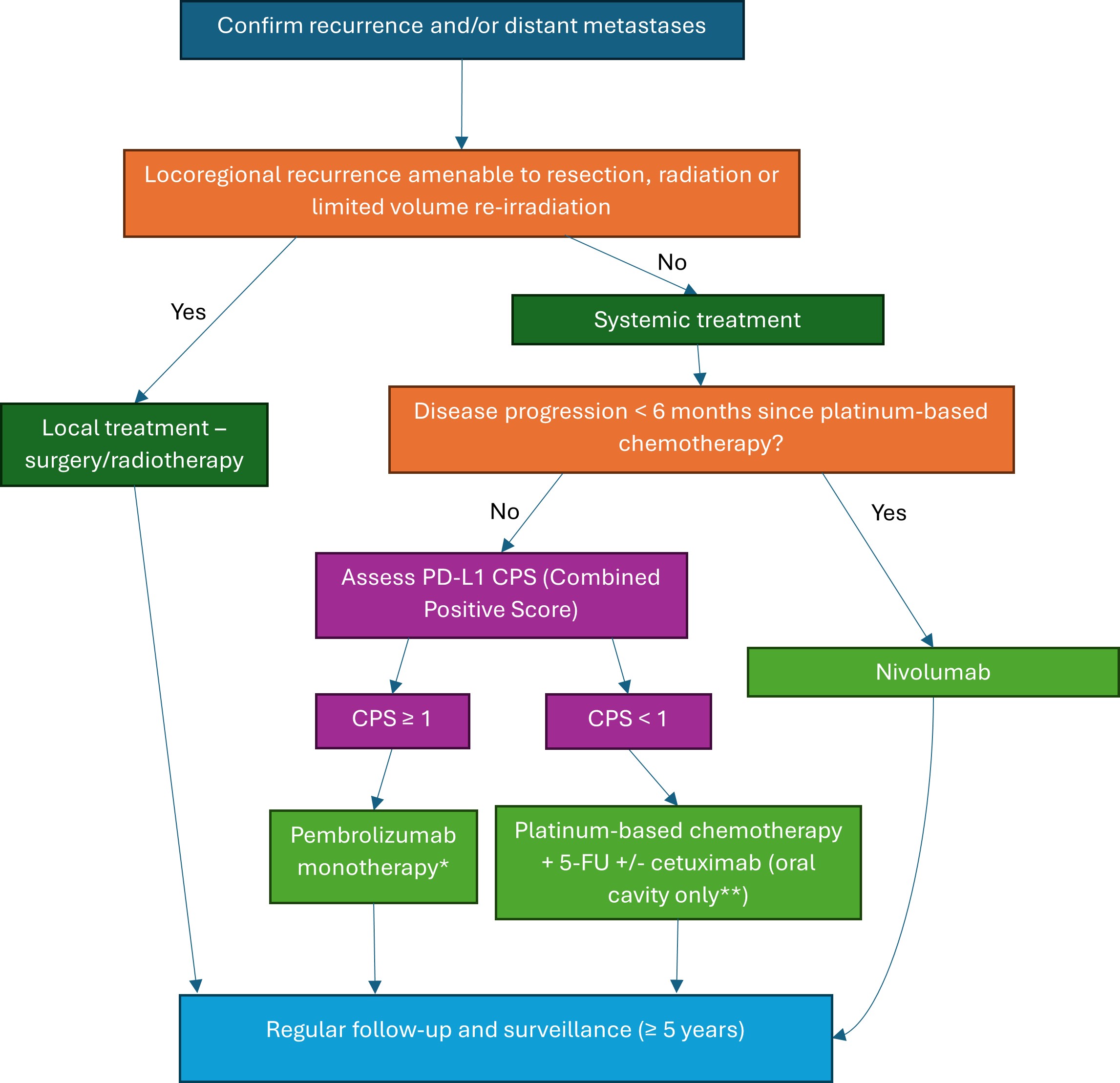 Fig. 3.
Fig. 3.
Treatment algorithm for recurrent and metastatic HNC. The figure was created using Microsoft Word (version 2504, manufactured by Microsoft Corporation). *Pembrolizumab is only approved by the National Institute for Clinical Excellence (NICE) as monotherapy, **Cetuximab is only approved by NICE in unresectable/metastatic oral cavity squamous cell carcinoma. PD-L1, programmed death-ligand 1; CPS, combined positive score; 5-FU, 5-fluoro-uracil.
The first instance of MDT work in HNC can be credited to trials in the 1970s to assess effects when combining surgery with radiotherapy [25]. As radiotherapy techniques and chemotherapeutic agents have advanced, non-surgical treatments have increasingly contributed to the management of HNC and ‘organ preservation’ [27]. Radiotherapy can be used as either singular primary treatment in early-stage disease or, more commonly, in combination with chemotherapy. Seminal clinical trials have demonstrated that concomitant platinum-based chemotherapy and radiotherapy yield better outcomes in locally advanced disease, decreasing 5-year mortality rates by 6.5% vs. radiotherapy alone [2, 22, 27, 28]. OPSCC, in particular, responds well to this multimodal treatment approach, which has become the standard of care for this subsite, as well as for nasopharyngeal cancers and tumours that are not amenable to surgery. Radiotherapy or chemoradiotherapy also has a role in adjuvant treatment post-operatively in high-risk and advanced disease to limit the rate of recurrence [27] and ideally should be started within 6 weeks of surgery [22].
However, the limitations and potential treatment toxicity must be carefully considered, as these can significantly impact a patient’s quality of life and their ability to complete treatment, particularly after major surgery. Notably, the benefit of adding chemotherapy diminishes with age and has no survival benefit over the age of 70 [28]. Both early and late effects of radiotherapy can be debilitating. Common risks include lymphoedema, osteoradionecrosis, skin ulceration and fibrosis, but additionally in HNC include significant dysphagia, xerostomia and hypothyroidism [22]. As many as 20–30% of patients undergoing non-surgical treatment for HNC will require an ‘unplanned admission’ due to complications, most of which are related to nausea and vomiting (25%) or reduced intake and dehydration (30%). As expected, the majority of this cohort (58%) are those undergoing combination chemoradiotherapy rather than single modality treatment [29]. Modern techniques like intensity-modulated radiotherapy (IMRT) minimise damage to surrounding healthy tissues, reducing side effects and improving quality of life [30].
Resistance to standard chemotherapy and radiotherapy remains a major challenge in head and neck cancer. Key molecular alterations have been implicated in treatment failure. Tumor protein p53 (TP53) mutations, frequently found in HPV-negative HNC, are associated with poor response to radiotherapy and increased tumour aggressiveness. Epidermal growth factor receptor (EGFR) amplification, although a common target in HNC, has shown variable treatment response, and overexpression often correlates with resistance to both cisplatin and radiation. Similarly, Rat sarcoma (RAS) mutations—particularly in harvey rat sarcoma viral oncogene homolog (HRAS)—are linked to reduced sensitivity to conventional therapies and are under investigation as targets for novel inhibitors. Understanding these molecular drivers allows for better risk stratification and may inform the use of combination therapies or biomarker-guided de-escalation in selected patients [31].
Surgical approaches to HNC are determined by tumour size, the accessibility of the affected subsite, and the need to preserve function. In addition to resecting the primary tumour, ipsilateral neck dissection is routinely performed, even in clinically node-negative necks, to address the high risk of occult metastases. This approach has been shown to improve both overall survival and disease-free survival [2, 3, 22]. With the rise of use of chemoradiotherapy for advanced disease with superior organ function preservation, the role of surgery is now primarily limited to single-modality treatment of small primary tumours [2]. The exception to this is offering total laryngectomy for select advanced stage primary laryngeal or hypopharyngeal carcinomas or in the use of ‘salvage’ surgeries with disease recurrence or failure to respond post-chemoradiotherapy [3]. Treatment for oral cavity cancers, however, remains primarily surgical due to its inherent easy access, which allows for preservation of function and the avoidance of scars. Additionally, non-surgical alternatives have a significant risk of toxic damage to the nearby bony tissue of the maxilla and mandible, including osteoradionecrosis, infection and fractures [21, 22].
Introduced in the 2000s, transoral robotic surgery (TORS) has revolutionised the surgical management of oropharyngeal, supraglottic, and hypopharyngeal tumours [27]. This minimally invasive technique offers superior access and tumour visualisation, allowing precise tumour excision while avoiding the risks and morbidity of traditional open surgery and chemoradiotherapy. This translates to faster recovery, shorter hospital stays, fewer tracheostomies, and improved functional outcomes, preserving speech and swallowing.
Early-stage OPSCC outcomes following TORS are comparable to those achieved with non-surgical treatments [6]. TORS versus other surgical approaches is superior in terms of survival outcomes, improving 5-year survival rates in early-stage OPSCC from 80% to 84% [32]. Although some patients may then go on to require adjuvant treatment, there is growing research into more targeted and reduced doses as a result of primary resection [6]. TORS may also facilitate organ-preserving surgery in cases where curative or functional laryngectomy would otherwise be required [33]. Induction chemotherapy followed by TORS resection is a novel treatment paradigm being explored for locally advanced tumours with promising preliminary results [34].
In early-stage laryngeal cancers, trans-oral laser/robotic surgery or primary radiotherapy is frequently curative. Radiotherapy also plays a crucial role in postoperative settings to eliminate residual microscopic disease and improve survival [35, 36].
Where loco-regional recurrence occurs, curative options may exist, including ‘salvage’ or revision surgery in those who have received primary non-surgical or surgical treatment, respectively, or radical (chemo)radiotherapy where this has not been used already. Otherwise, treatment options are limited and often yield poor outcomes, especially when neither salvage surgery nor radiotherapy is viable. In these cases, the basis for treatment has been chemotherapy and the monoclonal antibody Cetuximab [2, 22]. The 5-year survival for these patients is approximately 10–50% [1, 37]. Immunotherapy has emerged in the last 10 years as an alternative to chemotherapy, demonstrating improved survival and quality of life [37, 38, 39]. The two drugs currently approved by the National Institute for Clinical Excellence (NICE), pembrolizumab and nivolumab, are both immune checkpoint inhibitors that act on programmed cell death protein 1 (PD-1). PD-1 is an immune checkpoint responsible for dampening the body’s immune response and preventing apoptosis. These antigens can also be expressed on cancer cells, and this overexpression prevents the body’s immune response from killing cancer cells. By blocking the action of PD-1, immunotherapy can restore the T-cell-mediated anti-tumour response for destroying cancer cells [37, 38]. In recurrent cases post-chemotherapy, nivolumab improved two-year survival from 6% to 17% [38]. Pembrolizumab is licensed in the UK for use in untreated metastatic or recurrent disease, with a high tumour expression of PD-1, as a monotherapy with a better response rate (14.6% vs. 10%) and survival (8.4 months vs. 6.9 months) than chemotherapy [38]. Predictive biomarkers such as programmed death-ligand 1 (PD-L1) expression and tumour mutational burden (TMB) are becoming critical in immunotherapy decision-making. The combined positive score (CPS) is used to assess eligibility for anti-PD-1 therapy in HNC patients—this represents the percentage of tumour cells on a histopathological slide which demonstrate PD-L1 positivity [38, 40].
Fig. 3 demonstrates an algorithm for management of recurrent or metastatic cases.
Further studies to look at alternative antigens and identification of tumour biomarkers to target are ongoing. Despite promising results, response rates to immunotherapy remain relatively low even among patients with high PD-1 expression, suggesting that the immunological drivers of HNC are not fully understood. Although there is emerging strong evidence for the use of immunotherapy alongside standard treatments in both primary and adjuvant settings, this is currently not available in the UK due to the prohibitive costs [39].
HPV is a group of over 200 viruses spread through sexual contact [6, 41]. Infection is common, with an estimated 8 in 10 people affected in their lifetime. While most infections are benign, high-risk strains, particularly HPV-16, contribute to cancer by altering cellular regulation [6, 41]. Low-risk strains cause warts, whereas high-risk strains contain E6 and E7 oncogenes, which suppress tumour suppressor genes TP53 and retinoblastoma protein (Rb), leading to uncontrolled DNA replication and genetic instability [6, 41].
Oncogenic HPV strains prefer mucosal epithelium, explaining their link to ano-genital and oropharyngeal cancers. The structural features of these tissues allow viral entry and persistence, evading immune detection. For instance, HPV-positive tonsil tumours originate in the tonsillar crypts, unlike HPV-negative tumours, which arise from surface epithelium [42]. HPV-16 is the primary oncogenic virus in head and neck cancer (HNC), responsible for around 90% of HPV-positive oropharyngeal squamous cell carcinoma (OPSCC) [41, 42].
In the UK, 52% of OPSCC cases are now HPV-positive [42], a distinct subgroup with different pathophysiology and improved survival rates. Reflecting this, recent AJCC staging separates HPV-positive and negative OPSCC, allowing for downstaging and better prognostic accuracy [43, 44]. HPV-positive disease responds better to treatment [6], with five-year survival rates of 73.9% in non-smokers compared to 33.9% in HPV-negative cases. Among smokers, survival remains higher in HPV-positive cases at 55.2% vs. 22.6% [43]. Given that HPV-positive tumours mainly affect younger patients, reducing treatment toxicity is key, with trials exploring lower-dose chemoradiotherapy showing promising results [3, 42].
Beyond the cancer diagnosis itself, HPV-related disease adds complexity due to concerns around transmission. Clinicians must provide sensitive information, reassuring patients that lifestyle changes are unnecessary with long-term partners and that the risk of cancer transformation remains low. A holistic approach helps reduce stigma and ensures continued engagement with healthcare services [45].
With no validated screening for OPSCC, long-term strategies focus on HPV vaccination. Introduced nationally in 2008 for girls aged 12–13 and expanded to boys in 2019 [6], it has significantly reduced oncogenic HPV strains [46]. Post-vaccination, HPV-16 and 18 rates have dropped from 8.2–16.4% to 0.6–1%. However, HPV-positive OPSCC cases will likely continue rising for decades until the vaccine’s full impact is seen. Vaccine hesitancy post-coronavirus disease 2019 (COVID-19) has led to declining uptake: in 2022/23, only 71.3% of girls received their first dose, down from 88% in 2017/18, with similar declines in boys [47, 48]. A new single-dose regimen and awareness efforts, particularly around the higher OPSCC rates in men, aim to boost future uptake. However, vaccine hesitancy remains a significant public health challenge, limiting uptake and potentially undermining long-term efforts to reduce the incidence of HPV-positive OPSCC, particularly in populations with low health literacy or limited access to preventive care [49].
Approximately 25% of all HNC patients experience recurrence, predominantly within two years post-treatment [22]. Follow-up is therefore an essential component of HNC oncology; UK-based guidelines state a patient should be reviewed every 2 months for the first 2 years post-treatment and to be seen regularly in the first 5 years [22, 50]. The benefit of detecting recurrence early is significant; however, there is no strong evidence to suggest that routine, regular follow-up impacts outcome. Despite close surveillance, the majority of recurrence is still advanced stage disease, limiting the role for salvage surgery or non-surgical options in treatment [50]. Interestingly, a higher percentage of HNC recurrences are detected when patients present with symptoms or request expedited appointments [22, 50], prompting the idea to explore patient-initiated follow-up (PIFU) [51]. Although there is understandable hesitancy from patients, there is ongoing research into this becoming the new mode of follow-up, particularly in combination with interval imaging. Given that most recurrences are detected symptomatically, patient-initiated follow-up (PIFU), possibly combined with interval imaging like positive emission tomography (PET) scans, may represent a future approach in HNC monitoring [52].
Ongoing tobacco and alcohol consumption post-treatment continues to be a major risk factor for recurrence. As part of follow-up, patients should have good access to support for alcohol and tobacco cessation services. 1–4 years after stopping smoking reduces the risks of HNC to a similar level to people who had quit over 20 years ago. Comparatively, the benefits of stopping alcohol may take more than 20 years to emerge [9].
Functional outcomes are an important measure in assessing treatment of HNC, particularly in younger patients. Six functional domains (swallow, speech, breathing, pain, mood, neck/shoulder mobility) were assessed one year post-treatment in a large cohort study in Austria [53]. More than 40% of patients experienced significant functional deficit in at least one area post-treatment, and almost 10% had a complete loss of function in one domain. Worse functional outcomes are linked to advanced disease, recurrent disease and poor patient baseline health. Oral intake is the most commonly affected function in HNC treatment; approximately 1 in 5 patients are limited to a soft/liquid diet or require feeding tubes [53].
As treatment outcomes vary widely depending on the stage of disease at presentation, a critical component of care for many HNC patients is palliative management. Survival rates are widely variable, reaching as low as 20% in hypopharyngeal cancers [54]. With a median age of diagnosis around 65 years, the HNC patient population includes many older adults, and this demographic is expected to grow over time [55]. Given the complex symptoms and heightened care needs, early integration of palliative care in HNC treatment plans can substantially enhance quality of life [56]. Therefore, clinicians should be considering from as early as diagnosis about the role for end-of-life care and the particular challenges raised by HNC that increase care needs compared with other terminal cancers.
Beyond common symptoms such as pain, anxiety, and nausea, HNC patients face risks of acute airway obstruction and haemorrhage, which are critical to discuss with patients and their families [22]. Management of this will be guided by individual preference and can range from anticipatory medication to surgical tracheostomy. Advanced discussion and planning are essential as rapid deterioration can easily occur in HNC [55]. Even in the terminal phase, patients with HNC may present to the emergency department due to the acuity of their symptoms [20] and are more likely to have complex, specialist care requirements due to the common need for tracheostomy care and feeding tubes [55]. Consequently, patients are more likely to die in a hospice than at home, in contrast to other cancers [55]. Ensuring a ‘good death’—one with comfort, minimal symptom burden and dignity—requires coordinated care across acute and community settings, which should be central to HNC palliative management [54].
Advances in tumour genetics, treatment accessibility, and technology offer new opportunities to improve HNC outcomes [57]. Molecular and protein pathway analysis has identified key biomarkers and targeted therapies.
‘Liquid biopsy’ involves analysing tumour-related material in blood, saliva, or urine [58, 59]. Circulating tumour DNA (ctDNA) is gaining recognition as a biomarker for diagnosis, prognosis, and treatment monitoring, with high specificity (96%) and sensitivity (86%) in distinguishing HNC from disease-free cohorts [59]. ctDNA levels correlate with tumour burden, making it a promising tool for assessing treatment response and recurrence [18, 60, 61]. Beyond ctDNA, exosomes, microRNAs, and circulating tumour cells provide additional insights into tumour evolution [18], particularly relevant in the era of immunotherapy, where rapid and accurate assessment of tumour evolution is crucial [62].
‘Breath biopsy’ is another emerging diagnostic tool. Meta-analysis of volatile biomarkers has shown specificity of 85% and sensitivity of 83%, representing a promising avenue for non-invasive surveillance [63]. Current imaging techniques, such as positron emission tomography widely used, have limitations in post-treatment assessment, with positive predictive values as low as 30% in detecting recurrence in OPSCC patients [58]. This underscores the need for improved, non-invasive monitoring methods. Targeted therapy for HNC has focused on inhibition of key signalling pathways, such as EGFR, phosphoinositide 3-kinase/protein kinase B (PI3K/AKT), and Janus kinases/signal transducer and activator of transcription proteins (JAK/STAT) [64]. The challenge remains in overcoming resistance mechanisms and tumour heterogeneity—a particular challenge for HNC. However, this heterogeneity also presents alternative immunotherapy targets, such as chimeric antigen receptor (CAR) T cell therapy, which is well established in haematological malignancies, and dysregulated genes like RAS that promote tumour progression and immune avoidance [65, 66]. Identification of cancer stem cells and mesenchymal stromal cells in HNC and their respective pathways has begun to reveal early cellular targets for treatment [67, 68]. Immunotherapy continues to evolve, particularly in recurrent and metastatic HNC, where immune checkpoint inhibitors such as PD-1 and programmed death-ligand 1 (PD-L1) inhibitors have demonstrated improved survival outcomes [69]. Nanoparticle-based drug delivery systems (nanotechnology) improve targeted therapy efficacy while minimising systemic toxicity [64]. Tumour microenvironment modulation is being explored to alter immune-suppressive cells and hypoxia pathways [69]. Radiomics and radio-genomics are advancing personalised radiotherapy by linking imaging data with genomic information, allowing for tailored treatment approaches [62]. Radiomics also allows non-invasive assessment of response to neo-adjuvant chemoimmunotherapy using magnetic resonance imaging (MRI), helping to determine personalised treatment strategies [70].
Therapeutic cancer vaccines, including dendritic cell and neoantigen vaccines, are being investigated to enhance immune system activation against HNC [69]. Additionally, proton and heavy ion therapy offer highly targeted radiation, reducing damage to surrounding tissues, particularly for tumours near critical structures [64].
Combination therapies are perhaps demonstrating the most promise. Chemoimmunotherapy has been demonstrated to be effective in the metastatic and recurrent setting [71], as well as the neoadjuvant locally advanced HNC setting [72], postulated to occur due to depletion of immunosuppressive cells and cell death permitting antigen release and increased tumour ‘visibility’ to the immune system. and combinations of agents targeting specific mutations along with resistance mechanisms or immune checkpoints. Immunotherapy in combination with targeted therapies such as the EGFR inhibitor Cetuximab or tumour microenvironment modulators is being investigated, with the rationale that they can promote immune destruction of malignant cells by increasing antigen presentation, inhibiting antagonistic signalling pathways, enhancing T-cell infiltration, and promoting a productive tumour microenvironment. Furthermore, these combinations can be tailored to the patient’s specific tumour biology, epigenetics, and biomarkers, providing individualised treatment [73].
The COVID-19 pandemic accelerated risk-stratification models to prioritise HNC evaluations. A validated model combining epigenetic and clinical risk factors helps identify high-risk patients [74], while online triage questionnaires based on red-flag symptoms have shown predictive rates of 77% [75].
The biggest area of potential growth remains artificial intelligence (AI) in HNC [76]. AI-driven self-learning algorithms have been integrated into clinical practice, aiding in the diagnosis and treatment of HNC through radiological imaging analysis and histological tissue assessment. AI can be trained with positive and negative cases to develop pattern recognition or use unsupervised learning to identify previously unrecognised links. Image recognition combined with patient data has improved diagnostic accuracy, such as distinguishing leukoplakia (white patches) from oral squamous cell carcinoma, with accuracy improving from 93% to 95% [76]. AI is also facilitating rapid data interpretation through ‘literature mining’, helping identify biomarkers and therapeutic targets, as well as generating survival prediction models. Ultimately, AI holds the potential to personalise treatment plans by incorporating tumour genomic factors, patient characteristics, and individual genetics to optimise treatment strategies [76].
As research in HNC continues to expand, integrating biomarker-driven diagnostics, targeted therapies, immunotherapy, and AI-driven analytics will likely shape the future of personalised medicine in this field.
HNCs are a diverse group of malignancies requiring multidisciplinary management due to their complex treatment and care needs. It is expected that recent advances and innovations will improve outcomes, while supportive care remains essential in addressing functional, nutritional, and psychosocial challenges.
• Head and neck cancers have a rising incidence rate with poor clinical outcomes, representing an area for further research and development in treatment.
• Recognition of the changing patient demographic to younger patients who do not engage in well-recognised risk factors such as tobacco and alcohol consumption is essential in light of the rise of HPV-associated disease.
• Advanced head and neck tumours are typically managed with chemoradiotherapy, with surgery primarily being limited to smaller, resectable tumours.
• Immunotherapy is an emerging treatment in cases of disease recurrence and has the potential to expand as knowledge of tumour biology and mechanisms improves.
• An array of technological advancements, such as artificial intelligence, liquid biopsy and targeted cell therapy, provides promise in improving care and outcomes for patients with head and neck cancers.
Not applicable.
PB, AI, and AC were responsible for study design and aims. AI, AC and PB performed literature review. AI performed initial draft. AC produced figures. All authors contributed to revising the manuscript critically for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
The authors would like to express gratitude to their loved ones for their unrelenting support, permitting us to undertake work such as this in our personal time.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



