1 Department of Respiratory and Critical Care Medicine, The First Affiliated Hospital of Anhui Medical University, Anhui Province Public Health Clinical Center, 230012 Hefei, Anhui, China
2 Department of Epidemiology and Biostatistics, School of Public Health, Anhui Medical University, 230032 Hefei, Anhui, China
Abstract
This study aimed to evaluate the predictive value of the hemoglobin, albumin, lymphocyte, and platelet (HALP) score for pulmonary infections in patients with non-small cell lung cancer (NSCLC) undergoing chemotherapy.
A total of 180 NSCLC patients admitted to Anhui Public Health Clinical Center between January 2021 and December 2023 were enrolled. Patients were divided into an infection group (n = 65) and a non-infection group (n = 115) based on the occurrence of pulmonary infection during chemotherapy. Univariate and multivariate binary logistic regression analyses were performed to identify factors associated with pulmonary infections in NSCLC patients. Pearson correlation analysis was used to examine relationships between variables, and the predictive value of the HALP score was assessed using receiver operating characteristic (ROC) curve analysis.
There were no statistically significant differences between the two groups in age, gender, body mass index (BMI), hypertension, NSCLC subtype, serum potassium and calcium levels, pathological stage, total protein, or white blood cell count (WBC) (p > 0.05). However, significant differences were observed for the presence of diabetes, hemoglobin (Hb), albumin (ALB), platelet count (PLT), lymphocyte count (LYM), and HALP score (p < 0.05). Multivariate logistic regression analysis identified diabetes and HALP as independent predictors of pulmonary infections in NSCLC patients during chemotherapy (p < 0.05). ROC analysis showed that the area under the curve (AUC) for HALP was 0.812 (standard error = 0.031; 95% confidence interval (CI): 0.752–0.873; p < 0.05), with a Youden index of 0.55. At the optimal cutoff value, the sensitivity and specificity were 89.23% and 66.09%, respectively. Patients with HALP scores <17.89 had a significantly higher incidence of pulmonary infection compared to those with HALP scores ≥17.89 (p < 0.05).
The HALP score is a valuable predictor of pulmonary infections in NSCLC patients undergoing chemotherapy and may aid in early risk stratification.
Keywords
- hemoglobin
- albumin
- lymphocyte
- platelet
- chemotherapy
Non-small cell lung cancer (NSCLC), one of the malignant tumors with high incidence and mortality rates globally, has seen diversified treatment modalities that bring more hope for patient survival [1, 2]. Chemotherapy, as a crucial approach in the management of NSCLC, significantly prolongs patient survival and improves their quality of life by inhibiting the proliferation and spread of tumor cells [3, 4]. However, while chemotherapy drugs eradicate tumor cells, they also damage normal tissue cells, leading to compromised immune function and an increased risk of various complications, with pulmonary infections being among the most common and severe. Pulmonary infections not only interrupt the smooth progression of chemotherapy but also pose a serious threat to patient survival. Therefore, effectively predicting and preventing pulmonary infections in NSCLC patients during chemotherapy has become a current focus and challenge in clinical research [5].
The hemoglobin, albumin, lymphocyte, and platelet (HALP) score was selected to predict pulmonary infection during chemotherapy in patients with non-small cell lung cancer. Because it integrates four indicators, hemoglobin, albumin, lymphocytes, and platelets, it can comprehensively reflect the nutritional and immune status of the patient, which is closely related to infection risk. The study methodology may involve collecting blood data from patients before chemotherapy, calculating HALP scores, and determining their association with pulmonary infection through statistical analysis. An advantage of the HALP score is that it is simple, cost-effective, and can be derived from routine blood tests. Additionally, it demonstrates high predictive accuracy, allowing for early identification of high-risk patients and implementation of targeted preventive strategies. Previously, the HALP score has been used to evaluate the prognosis of various tumors, including surgical outcomes in NSCLC and the efficacy of first-line therapy in small cell lung cancer, both showing promising clinical value [6, 7]. Its major research significance lies in providing a new evaluation tool for infection prevention and control during chemotherapy in NSCLC patients, potentially reducing infection rates and improving patient prognosis.
Therefore, this study aims to explore the predictive value of the HALP score for pulmonary infections in NSCLC patients undergoing chemotherapy, with the goal of providing a scientific basis for early identification of high-risk individuals and developing personalized prevention and treatment strategies. By investigating the relationship between the HALP scores and pulmonary infections, a deeper understanding of the pathogenesis of these infections during chemotherapy may be achieved, thereby guiding clinical practice more effectively and improving both therapeutic outcomes and the quality of life of NSCLC patients.
A total of 180 patients with non-small cell lung cancer (NSCLC) admitted to the Anhui Public Health Clinical Center between January 2021 and December 2023 were selected for this study. Patients were categorized into an infection group (n = 65) and a non-infection group (n = 115) based on the occurrence of pulmonary infections during chemotherapy (Fig. 1). The inclusion criteria were as follows: (1) all patients met the clinical diagnostic criteria for non-small cell lung cancer [8]; (2) patients had no pre-existing lung complications caused by other diseases. The exclusion criteria were as follows: (1) patients with concomitant lung diseases; (2) patients with psychiatric disorders; (3) patients on long-term anticoagulant therapy; (4) patients experiencing severe adverse reactions during chemotherapy or unable to undergo chemotherapy due to other reasons; (5) patients with concurrent other infectious diseases.
 Fig. 1.
Fig. 1.
Flowchart of patient selection and grouping.
Patients exhibited symptoms such as coughing and sputum production, with or without fever, during the first four cycles of chemotherapy. Additionally, imaging findings on lung computed tomography (CT) scans showed infection-related changes, including pulmonary infiltrates, consolidation, or cavitation. Bacterial cultures of respiratory secretions were performed, and pathogenic organisms were successfully cultured. The identification and culture of respiratory secretions in this study were conducted using the fully automated bacterial identification system available in the Anhui Public Health Clinical Center’s laboratory (VITEK 2 COMPACT (bioMérieux, Marcy-l’Étoile, France); BD M-50 (Becton, Dickinson and Company, Franklin Lakes, NJ, USA). Further testing was performed using antigen detection methods. Based on this combined information, the presence of a pulmonary infection in each patient was determined.
All patients underwent fasting and early-morning blood sampling before chemotherapy. Pre-chemotherapy screening included the following parameters: blood potassium, blood calcium, hemoglobin (Hb), albumin (ALB), lymphocyte count (LYM), platelet count (PLT), total protein (TP), and white blood cell count (WBC). Two to three milliliters of fasting venous blood were collected from each patient.
Complete Blood Count (CBC) test (including WBC, Hb, LYM, and PLT): Blood samples were collected using disposable vacuum blood collection tubes (Jiangsu Rongye Technology Co., Ltd., Suzhou, China; Suzhou Medical Equipment Permit No. 20152220233, 2 mL) under negative pressure for analysis.
Biochemical tests (including albumin, total protein, blood potassium, and blood calcium): Blood was collected using disposable vacuum blood collection tubes (Suzhou Medical Equipment Permit No. 20152220233, 5 mL) under negative pressure for biochemical analysis.
HALP scores were calculated using the following formula:
HALP [9] = (Hemoglobin × Albumin × Lymphocyte Count) / Platelet Count.
The experimental data collected were analyzed using SPSS version 27.0 (IBM
Corporation, Armonk, NY, USA). The Shapiro-Wilk test was applied to test for
normality of data. For metric data that followed a normal distribution, results
were expressed as mean
Comparisons of age, gender, body mass index (BMI), hypertension, NSCLC subtype,
blood potassium, blood calcium, pathological stage, total protein, and WBC showed
no statistically significant differences between the infection and non-infection
groups (p
| Variable | Infection group (n = 65) | Non-infection group (n = 115) | t/ |
p-value | |
| Age (years) | 58.26 |
56.98 |
1.119 | 0.265 | |
| Gender | 0.033 | 0.856 | |||
| Male | 33 | 60 | |||
| Female | 32 | 55 | |||
| BMI (kg/m2) | 21.12 |
21.08 |
0.145 | 0.885 | |
| Hypertension | 0.073 | 0.787 | |||
| Yes | 10 | 16 | |||
| No | 55 | 99 | |||
| Diabetes | 3.966 | 0.046 | |||
| Yes | 21 | 22 | |||
| No | 44 | 93 | |||
| NSCLC type | 4.091 | 0.252 | |||
| Adenocarcinoma | 41 | 55 | |||
| Squamous cell carcinoma | 16 | 43 | |||
| Large cell carcinoma | 5 | 10 | |||
| Other | 3 | 7 | |||
| Potassium (mmol/L) | 4.26 |
4.22 |
1.677 | 0.095 | |
| Calcium (mmol/L) | 2.24 |
2.25 |
0.449 | 0.654 | |
| Pathological stage | 1.791 | 0.181 | |||
| Ⅰ–Ⅱ | 30 | 65 | |||
| Ⅲ–Ⅳ | 35 | 50 | |||
| Hb (g/L) | 124.66 |
142.66 |
18.848 | ||
| ALB (g/L) | 31.14 |
35.39 |
9.619 | ||
| LYM ( |
0.65 |
0.72 |
3.348 | 0.001 | |
| PLT ( |
174.17 |
185.08 |
8.646 | ||
| HALP score | 14.43 |
19.69 |
8.103 | ||
| Total protein (g/dL) | 5.50 |
5.48 |
0.443 | 0.659 | |
| WBC ( |
7.51 |
7.37 |
0.387 | 0.699 | |
Note: NSCLC, non-small cell lung cancer; BMI, body mass index; Hb, hemoglobin; ALB, albumin; LYM, lymphocyte count; PLT, platelet count; WBC, white blood cell count; HALP, hemoglobin, albumin, lymphocyte, and platelet.
Collinearity tests were performed on the significant factors identified in the
univariate analysis, resulting in the exclusion of Hb, ALB, LYM, and PLT due to
multicollinearity. HALP and diabetes were used as independent variables, with
values assigned as follows: actual HALP values and diabetes coded as yes = 1, no
= 0. The dependent variable was pulmonary infection during chemotherapy in NSCLC
patients (infection = 1, non-infection = 0). The results of the binary logistic
regression analysis indicated that HALP and diabetes were significant influencing
factors for pulmonary infections in NSCLC patients undergoing chemotherapy
(p
| Factor | SE | Wald | p-value | Exp ( |
95% CI | Collinearity | |||
| Lower limit | Upper limit | Tolerance | VIF | ||||||
| HALP | –0.313 | 0.050 | 38.631 | 0.731 | 0.663 | 0.807 | 0.996 | 1.004 | |
| Diabetes | 1.136 | 0.456 | 6.207 | 0.013 | 3.114 | 1.274 | 7.609 | 0.969 | 1.032 |
| Constant | 4.425 | 0.815 | 29.487 | 83.494 | - | - | - | - | |
Note: HALP was included in the multivariate model as a composite index to account for collinearity among its components. CI, confidence interval; SE, standard error; VIF, variance inflation factor.
ROC analysis results indicated that the area under the curve (AUC) for HALP was
0.812, with a standard error of 0.031 (95% confidence interval (CI):
0.752–0.873, p
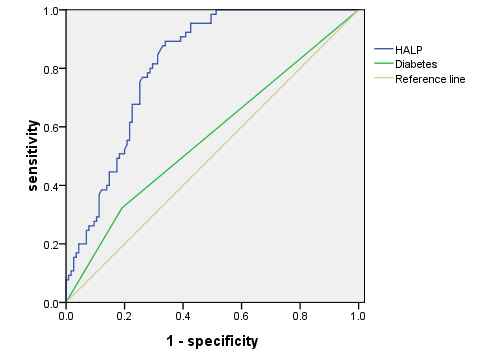 Fig. 2.
Fig. 2.
ROC curve of HALP score for predicting pulmonary infections.
| Variable | AUC | Standard error | 95% CI | Youden index | Sensitivity | Specificity | p-value | Optimal cutoff value |
| HALP | 0.812 | 0.031 | 0.752–0.873 | 0.55 | 89.23 | 66.09 | 17.89 | |
| Diabetes | 0.566 | 0.045 | 0.477–0.655 | 0.13 | 32.31 | 80.87 | 0.142 | - |
Note: AUC, area under the curve; ROC, receiver operating characteristic; CI, confidence interval.
The infection rate among patients with HALP
| Group | HALP |
HALP |
p-value | |
| Infection rate (%) | 44.33 | 26.51 | 6.159 | 0.013 |
| Number of infections | 43 | 22 | ||
| Number without infection | 54 | 61 |
This study aimed to investigate the predictive value of HALP score for pulmonary infections during chemotherapy in non-small cell lung cancer (NSCLC) patients. By conducting univariate analysis, binary logistic regression analysis, and receiver operating characteristic (ROC) curve analysis on clinical data of 180 NSCLC patients, several significant conclusions were obtained.
In this study, the HALP score and its components, Hb, ALB, and PLT, were found to play significant roles in the occurrence of pulmonary infections during chemotherapy in NSCLC patients. Notably, patients in the infection group had significantly lower levels of Hb, ALB, and PLT compared to those in the non-infection group, and the HALP score was also significantly lower in the infection group. These findings suggest that NSCLC patients undergoing chemotherapy with lower levels of Hb, ALB, and PLT may face a higher risk of developing pulmonary infections. Hemoglobin, a key protein responsible for oxygen transport in red blood cells, is critical for maintaining tissue oxygenation [10]. A decrease in its levels may lead to tissue hypoxia, impairing the normal function of immune cells, and thereby increasing the risk of infection [11]. Previous research by Ding et al. [12] also identified albumin as a factor influencing postoperative pulmonary infections in NSCLC patients, which is consistent with the present study and provides further support for this association. ALB, the most abundant plasma protein, serves multiple physiological functions, including maintaining plasma colloid osmotic pressure and transporting nutrients and metabolic waste [13, 14]. A decline in ALB levels may indicate malnutrition or compromised immune function, which can also elevate the risk of infection [15]. PLT, as a vital blood component, participates in hemostasis and plays a significant role in inflammatory and immune responses. A decrease in PLT count during chemotherapy may weaken the defense capacity against infections. Previous research by Unal et al. [16] showed that PLT could serve as a prognostic indicator during chemotherapy and may be independently associated with chemotherapy outcomes. These observations further support the relevance and consistency of the findings of this study.
Previous studies have focused on individual laboratory indicators as standalone factors. However, considering the aforementioned influencing factors, it can be observed that these variables are closely associated with the HALP score. The HALP score, as an important indicator reflecting the overall nutritional status and immune condition of patients, may reflect a decline in immune function when decreased, making patients more susceptible to pulmonary infections [17, 18]. This study further examined the relationship between various factors and the HALP score and analyzed its value in predicting pulmonary infections. Through ROC curve analysis, the predictive value of the HALP score for pulmonary infections during chemotherapy in NSCLC patients was further evaluated. The results indicated that the area under the curve (AUC) for HALP was 0.812, with a standard error of 0.031 (95% CI: 0.752–0.873), and a Youden index of 0.55. At this threshold, the sensitivity was 89.23%, and the specificity was 66.09%. These findings suggest that the HALP score has relatively high sensitivity in predicting pulmonary infections during chemotherapy in NSCLC patients, while its specificity is comparatively lower. Sensitivity reflects the potential of the model to correctly identify infected patients, while specificity reflects its ability to correctly identify non-infected patients. The high sensitivity of the HALP score in this study indicates a strong ability to identify patients at risk of developing pulmonary infections during chemotherapy. However, the relatively lower specificity suggests certain limitations in distinguishing non-infected individuals, likely due to the complexity of HALP as a composite index integrating multiple interrelated components. Future studies may focus on optimizing the composition and calculation of the HALP score to improve its specificity and overall predictive performance.
Additionally, this study identified that diabetes is another factor influencing pulmonary infections during chemotherapy in NSCLC patients. The proportion of patients with diabetes was significantly higher in the infection group than in the non-infection group, suggesting that diabetes may increase the risk of pulmonary infections during chemotherapy in NSCLC patients. As a metabolic disorder, diabetes is often associated with impaired immune function, hyperglycemia, and other pathophysiological changes that can elevate infection risk [19]. Further analysis of diabetes as a predictive factor for pulmonary infections revealed an AUC of 0.566 with a standard error of 0.045 (95% CI: 0.477–0.655), and a Youden index of 0.13. At this threshold, the sensitivity was 32.31%, and the specificity was 80.87%. Compared to HALP score, diabetes showed lower predictive value and lower sensitivity, further highlighting the advantages of the HALP score in predicting pulmonary infections during chemotherapy in NSCLC patients.
The high sensitivity of the HALP score indicates that patients with actual pulmonary infections can be more accurately identified, reducing missed diagnoses. However, this may be accompanied by an increased number of false positive results. Low specificity suggests that some patients who are unlikely to develop infections may be misclassified as high risk, potentially leading to unnecessary interventions. This trade-off must be interpreted in the context of clinical practice. If the consequences of infection are severe, higher sensitivity may be prioritized to ensure early detection. Conversely, if medical resources are limited or interventions carry significant side effects, lower specificity could impose an added burden. Therefore, the advantages and limitations should be balanced, and additional indicators may be incorporated to optimize the overall evaluation strategy where appropriate.
The results of this study provide new insights and approaches for predicting pulmonary infections during chemotherapy in NSCLC patients. The HALP score, as a simple and feasible predictive tool, can be widely implemented in clinical practice. By regularly monitoring the levels of Hb, ALB, LYM, and PLT in NSCLC patients and calculating the HALP score, clinicians can identify high-risk patients early. This enables the implementation of targeted preventive measures such as enhancing nutritional support, improving immune function, and judicious use of antibiotics to reduce the incidence of pulmonary infections, thereby improving treatment outcomes and quality of life for patients.
Although this research has achieved certain results, there are still some shortcomings. Firstly, no prior power analysis was conducted, and the single-center sample size from our hospital was limited, possibly leading to selection bias and affecting result universality. Secondly, specific chemotherapy details like drug types and dosages weren’t considered, and other potential confounding factors weren’t fully accounted for in variable analysis. Moreover, it was a retrospective analysis with possible information bias. Additionally, exploring the combined application of HALP score with other indicators, its biological mechanisms, and differentiating among infection pathogens, as well as conducting long-term follow-up, could enhance predictive accuracy and comprehensively assess its long-term impact on patients.
In conclusion, this study provides new insights and methods for predicting pulmonary infections during chemotherapy in NSCLC patients by evaluating the predictive value of the HALP score. As a simple and practical predictive tool, the HALP score demonstrates significant potential for identifying high-risk patients and guiding early intervention strategies, making it worthy of broader application and promotion in clinical practice. Future research may further optimize the composition and calculation method of the HALP score to enhance its predictive value. Additionally, integrating HALP with other biomarkers or clinical indicators could provide stronger support for the prevention and management of pulmonary infections in NSCLC patients undergoing chemotherapy.
The data used to support the findings of this study are available from the corresponding author upon request.
FMP and XHC designed the research study. XHC, MW and YH performed the research. MW and MZL analyzed the data. XHC wrote the initial draft. All authors contributed to revising the manuscript critically for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study is in accordance with the Declaration of Helsinki. The study was approved by the Medical Ethics Committee of Anhui Public Health Clinical Center (PJ-YX2024-070). The principle of informed consent was followed throughout the experiment, and information about the study was provided to patients or their families, and then consent was obtained.
Not applicable.
This work was supported by the Anhui Public Health Clinical Center (the North District of the First Affiliated Hospital of Anhui Medical University) Funded Project (Grant No. 2023YKJ05).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


