1 School of Medical Sciences, College of Medicine and Health, University of Birmingham, B15 2TT Birmingham, UK
2 Department of Radiology, Guy’s and St Thomas’ NHS Foundation Trust, SE1 7EH London, UK
3 Department of Radiology, Royal Glamorgan Hospital, Cwm Taf Morgannwg, CF72 8XR Llantrisant, UK
4 Department of Radiology, University Hospital of Wales, CF14 4XW Cardiff, UK
Abstract
Multiple myeloma is a haematological malignancy of plasma cells, typically characterised by a monoclonal immunoglobulin protein detectable in the blood. Myeloma requires treatment when it affects specific end-organs, including lytic bone disease. The accurate detection of bone disease is therefore crucial in the diagnosis and management of myeloma, and advanced imaging with whole-body magnetic resonance imaging or positron emission tomography is now recommended as the standard of care. For patients with non-secretory myeloma and those with extramedullary disease, imaging is the only way to monitor response to treatment and to detect relapse. Whilst there are challenges in delivering advanced imaging modalities to patients with myeloma, the clinical and financial advantages mean that improving access should be a priority. This review discusses the importance of advanced imaging in the management of myeloma across its disease course, and outlines current barriers and potential future directions in this rapidly evolving field.
Keywords
- multiply myeloma
- whole-body imaging
- diffusion magnetic resonance imaging
- positron emission tomography computed tomography
Multiple myeloma is a highly treatable tumour of plasma cells that originates in the bone marrow (hence the Greek-derived name, myelo- marrow -oma tumour), and is characterised by bone damage, anaemia, renal impairment and infection [1]. Myeloma is almost exclusively seen in adults; the exceptionally rare manifestations in children are not discussed further. The tumour produces a monoclonal immunoglobulin protein (a single serum immunoglobulin and/or a serum free light chain) which acts as a tumour marker in the blood. The non-specific nature of symptoms at presentation means there is often a delayed diagnosis, and diagnosis of myeloma in the context of an emergency hospital admission is associated with poorer overall survival [2, 3, 4]. Although still incurable, myeloma typically has a relapsing remitting course over years, and there has been a significant advance in treatments in the last 25 years with a range of new targeted therapies (immunomodulatory drugs, proteasome inhibitors, stem cell transplant, monoclonal antibodies, bi-specific antibodies and chimeric antigen receptor T cell therapy). Tumour evolution occurs such that the patient eventually becomes resistant to their current treatment, and they then get switched to an alternative available treatment, until ultimately their tumour is refractory to all treatment options [5] (Fig. 1, Ref. [6]). For younger and fitter patients, a median survival of 5–10 years can be expected. However, there is marked heterogeneity in tumour biology, as evidenced by at least 25% of patients living longer than 10 years and 25% of patients living less than 3 years [7]. Despite modern treatment options, the rate of early mortality (within six months of diagnosis) is around 5–10%, commonly related to infection [8, 9]. For the significant proportion of older frail patients, treatments are less well tolerated, dose reductions are frequently required, and patients often receive just 1–3 lines of treatment [10].
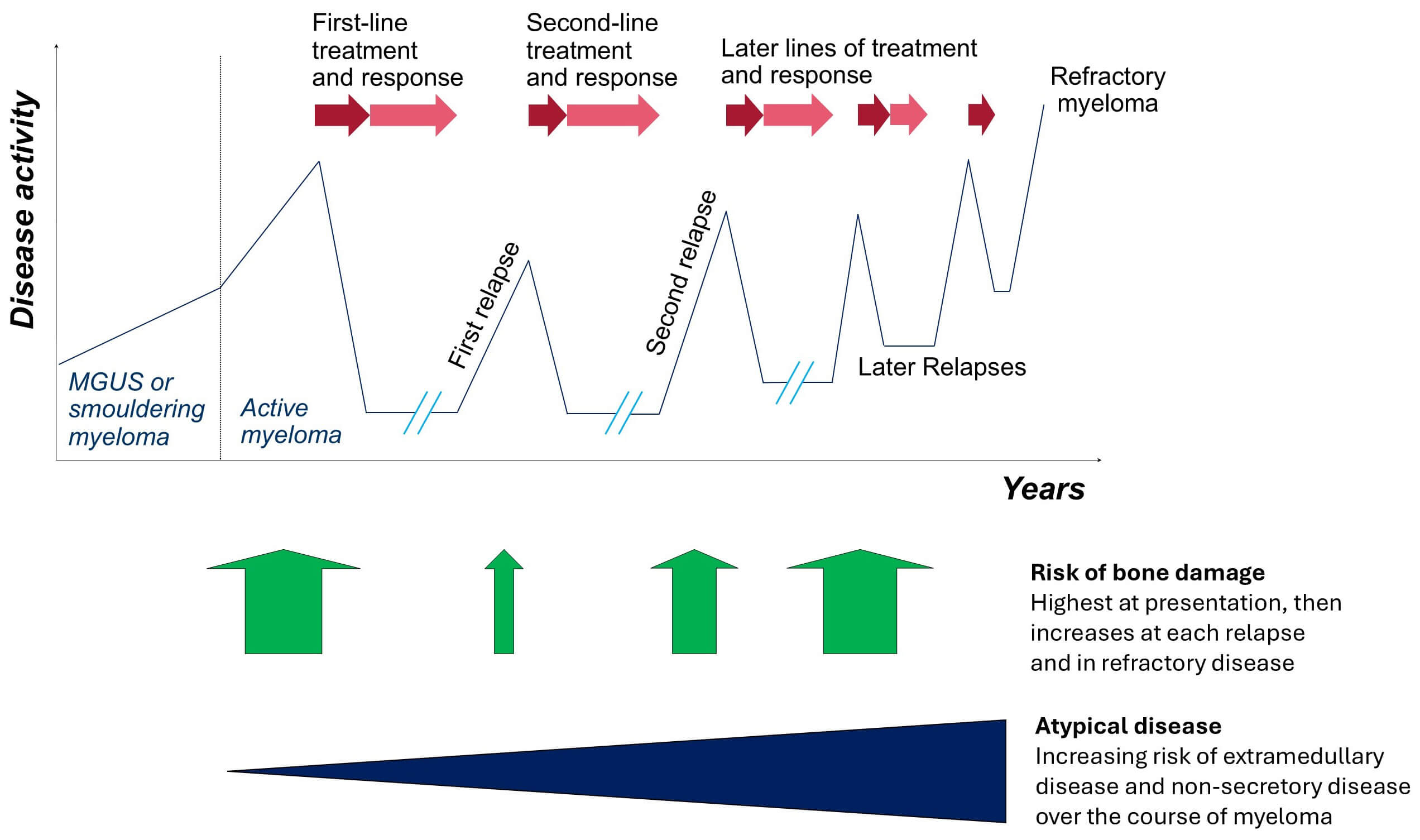 Fig. 1.
Fig. 1.
Typical trajectory of myeloma treatment and disease burden. The disease course of myeloma, preceded by a period of asymptomatic monoclonal gammopathy, follows a remitting and relapsing pattern with sequential lines of treatment. As the disease evolves over time, the effectiveness of each line of treatment reduces, the risk of bone damage increases, and patients are more likely to develop extramedullary or non-secretory myeloma. Adapted with permission from Kaiser et al. [6]. MGUS, monoclonal gammopathy of undetermined significance.
A premalignant condition of monoclonal gammopathy of undetermined significance (MGUS) is common and asymptomatic, characterised by a small monoclonal immunoglobulin only detected incidentally, and is associated with a low risk of progression to myeloma [11, 12]. Smouldering myeloma is also asymptomatic and defined by a larger monoclonal protein or bone marrow plasma cell burden. The risk of progression for smouldering myeloma is higher, but a significant minority never develop myeloma or need treatment [13]. Myeloma is ultimately diagnosed when the underlying malignant plasma cells are responsible for specific end-organ damage, namely anaemia, renal impairment due to cast nephropathy, hypercalcaemia, or lytic bone disease. In some patients with extremely adverse biomarkers, the risk of developing end-organ damage is sufficiently high to warrant a diagnosis and treatment of myeloma [14]. In practice, imaging investigations and a bone marrow biopsy are only performed if a patient is suspected to have myeloma based on the clinical picture of potential myeloma-related organ damage.
With lytic bone disease a core feature of myeloma, the requirement for imaging to visualise the skeleton has been essential for decades. Early case series, including X-ray characterisation, demonstrate a range of skeletal abnormalities, including ‘punched-out’ (lytic) lesions and pathological fracture [15]. Over time, a skeletal survey became the standard way to assess for bone disease, comprising plain films of the skull, chest, spine, pelvis and long bones. Radioisotope bone scanning has never had a role in myeloma, missing over half of lesions in one series [16]. The higher resolution of computed tomography (CT) has long been recognised as more sensitive than plain X-rays, the latter requiring far more bone destruction to be visually detectable [17]. Efforts to reduce the exposure to ionising radiation resulted in whole-body low-dose CT protocols, which maintained diagnostic accuracy and were adopted by the International Myeloma Working Group (IMWG) [18, 19]. As a technique that measures the absorption of X-ray radiation, CT can demonstrate structural abnormalities at a snapshot in time, including bone disease and soft tissue deposits. However, it is limited in its ability to reflect dynamic disease processes, which at the least would require serial imaging.
More recently, advanced imaging techniques have been developed and applied to myeloma, using functional measures of tissue dynamics and metabolism, to better understand active disease processes. Magnetic resonance imaging (MRI) is well-suited for the analysis of soft tissues, including abnormalities in the bone marrow of patients with myeloma [20], with most early applications of the technology focusing on disease of the spine. Now, whole-body magnetic resonance imaging (WB-MRI), including measurement of the movement of water molecules through tissue with diffusion weighted imaging (DWI), allows highly sensitive and specific detection of active myeloma [21]. The limitations with WB-MRI are mostly associated with the long scan times that are unsuitable for some patients, and the time and expertise required to evaluate the vast quantity of data produced by each scan. 18F-fluorodeoxyglucose positron emission tomography (FDG-PET) is a nuclear medicine technique that identifies tissues with high glucose uptake, and was shown to identify areas of active and occult myeloma [22, 23]. Coupling with CT (FDG-PET/CT) allows accurate anatomical localisation of areas of active disease, which can inform the use of localised treatments such as radiotherapy. However, the ionising radiation dose of FDG-PET/CT is not trivial, especially when repeated imaging is required during the disease course.
This review describes the necessity of adequate imaging in myeloma, which in current clinical practice requires advanced cross-sectional and functional techniques. The optimal modalities are discussed in relation to diagnosis, response assessment and ongoing monitoring. The importance of advanced imaging is reflected by inclusion in recent UK national and existing international guidelines. Whilst barriers exist to their implementation, it is crucial for patients that these are overcome, and advances in computing may provide part of the solution.
Imaging is critical for detecting myeloma within bone, marrow and as extramedullary disease at diagnosis, at suspicion of relapse, and increasingly for assessment of response. Importantly, the burden of disease on imaging does not always correlate well with changes in the monoclonal protein in individual patients, especially as disease biology changes over time. The use of imaging and the techniques available have changed significantly in the last few decades, yet access to modern imaging remains a significant challenge. The majority of patients presenting with myeloma have widespread bone disease at diagnosis, and for a significant proportion, this leads to symptomatic bone disease causing major morbidity with pain, fractures, spinal cord compression, functional impairment and reduced quality of life [6]. Early diagnosis greatly reduces the risk of severe organ impairment, allows effective treatment to be delivered more easily and is associated with better outcomes. When diagnosed late, which is common in myeloma [2, 24], bone pain is often severe enough to require regular opioid pain relief (Non-Steroidal Anti-Inflammatory Drugs are avoided due to renal toxicity), and catastrophic fractures and cord compression are frequently seen.
Typically, bone disease is patchy in its anatomical distribution within the marrow of the axial skeleton and proximal long bones; involvement of the hands and feet is rare. Bone marrow disease includes both focal areas and a diffuse infiltrative pattern. The lytic bone lesions in myeloma are due to the local effect of the tumour, leading to dysregulation of normal bone turnover, with a massive increase in osteoclast bone resorption and a corresponding inhibition of new bone formation by osteoblasts [25]. This lack of new bone formation is characteristic of a lytic lesion and distinguishes it from most forms of bony metastases, where a sclerotic rim is seen due to an osteoblast reaction. Some bone healing does occur with treatment response [26], but structural damage is permanent, and there is always a risk of vertebral collapse or fracture even when the disease has responded to treatment.
The principal strategy for treating myeloma bone disease is starting the definitive management of the underlying myeloma, using systemic therapy, which is highly effective in newly diagnosed patients and the majority of patients at relapse. Radiotherapy is useful for a bone lesion that remains painful despite starting myeloma treatment. Regular bisphosphonates inhibit osteoclast activity, and given prophylactically, reduce bone-related events (pain and fractures); zoledronate is recommended [27, 28]. Spinal disease specifically is outside the scope of this review, but it is an area of high unmet need yet completely lacking in quality evidence, with surgery required for unstable disease and radiotherapy, bracing and vertebroplasty/kyphoplasty as possible options. Lesions in long bones may be at risk of fracture and require an orthopaedic assessment and prophylactic fixation where indicated.
Myeloma deposits can grow as soft tissue extensions from marrow or arise as a tumour outside of the marrow. Such extramedullary disease is uncommon at diagnosis but becomes increasingly common as the disease relapses and is only detectable by cross-sectional imaging. As with all cancers, myeloma plasma cells are genetically unstable and subclonal evolution leads eventually to a tumour that is more independent of the bone marrow niche, more resistant to treatment and often more aggressive [29].
Non-secretory disease is rare at diagnosis, affecting around 1% of patients [30, 31]. A further 10% of tumours produce only low levels of monoclonal protein, which is challenging to monitor because concentrations are below the lower limit of quantification of routinely available assays [32, 33]. However, over time, tumours frequently evolve to produce less monoclonal protein (oligo-secretory) or none at all (non-secretory), as there is no competitive advantage to the malignant plasma cell clone in secreting the immunoglobulin protein [34].
Both extramedullary disease and tumours producing low levels of immunoglobulins are often seen in patients who have had multiple lines of therapy. Extramedullary disease is missed when biopsy is only taken from bone marrow, and quantification of serum monoclonal protein becomes unreliable, where tumours secrete less immunoglobulin. This highlights how critical modern imaging has become for monitoring and managing patients in the current era.
Rarely, malignant plasma cells can be found in substantial numbers circulating in the blood. Plasma cell leukaemia can come late in the myeloma disease course, and is associated with poor prognosis [35]. The diagnosis of plasma cell leukaemia is made through immunophenotyping of peripheral blood. However, appropriate imaging is still required for a complete assessment of disease burden in such patients.
WB-MRI with diffusion weighting and FDG-PET/CT are the two advanced imaging techniques that have been recommended in numerous guidelines over the last decade, with the exception of the 2019 IMWG imaging guidelines [36] that recommended whole-body CT as first-line (as a triaging tool, determining who should receive WB-MRI). The recent UK British Society for Haematology (BSH) guidelines [6] did not recommend any other imaging technique apart from WB-MRI and FDG-PET/CT, and advocated for the ambition to implement access to the most sensitive and appropriate imaging for all patients. One reason that WB-CT retains prominence in the IMWG guidelines is its relative availability internationally, acknowledging that WB-MRI and FDG-PET/CT are simply not accessible in many institutions worldwide. In contrast, the UK guideline sets out the optimal approach within this healthcare setting, also informed by independent economic modelling relevant to the UK [28]. The advantages and disadvantages of the various imaging techniques are shown in Table 1 [6]. It is clear that WB-MRI is the optimal technique, but decisions will also depend in part on certain disease characteristics, patient wishes, and availability of imaging. Key considerations are described below, and summarised in Table 2.
| Modality | Quality | Radiation and contrast | Scan time and time in hospital | Recommended by BSH | Contraindications and cautions | Current issues |
| WB-MRI | The most sensitive imaging technique. Able to visualise bone marrow infiltration. Can be used for monitoring. | No radiation and no contrast | 45–60 minutes, but likely to reduce with better technology | Yes strongly | Claustrophobia, inability to lie flat. Most implantable cardiac devices and pacemakers. Some types of metalwork, including aneurysm clips. | Access and capacity. Time to report is significant and educational need for radiologists. |
| FDG-PET/CT | Less sensitive than WB-MRI, particularly for diffuse disease detection. | High radiation and intravenous tracer | 4–5 hours in hospital, with scan time of 10 minutes | Yes, as an alternative to WB-MRI | Excess radiation to be considered. More expensive than an MRI. | Access and capacity. Most expensive test. |
| Can be used for monitoring and is recommended for assessment of minimal residual disease status. | ||||||
| Low-dose WB-CT | Recommended by NICE where WB-MRI is unsuitable. | Moderate radiation and no contrast | 10 minutes scan time | No | Excess radiation to be considered. | Low-dose WB-CT is readily available but will miss disease and is hard to interpret in follow-up. |
| Lesions are only visible where 50% cortical bone destruction, i.e., an insensitive test. | Potential role in assessment of vertebral fracture stability and spinal surgery planning. | |||||
| Lesions persist following treatment and therefore not suitable for response assessment or in the relapse setting. | ||||||
| X-ray skeletal survey | Poor sensitivity and poor for follow-up imaging. | Moderate radiation and no contrast | 20 minutes scan time | Not recommended | Excess radiation to be considered. Not easy to perform. |
Limited role in assessing fracture risk in long bones. |
The imaging modalities available in myeloma are shown in decreasing order of preference; only WB-MRI and FDG-PET/CT are recommended. Abbreviations: WB, whole-body; MRI, magnetic resonance imaging; FDG-PET/CT, 18F-fluorodeoxyglucose positron emission tomography/computed tomography; CT, computed tomography; NICE, National Institute for Health and Care Excellence; BSH, British Society for Haematology.
| Timepoint | Recommended imaging modality | Comments |
| Baseline assessment | WB-MRI | First-line option. |
| FDG-PET/CT | Alternative to WB-MRI. | |
| WB-CT | Where WB-MRI and FDG-PET/CT are unavailable or contraindicated. | |
| Response assessment (e.g., day +100 post-autologous stem cell transplantation) | FDG-PET/CT | Evidence from clinical trials shows MRD-negativity by PET complements marrow assessments. |
| WB-MRI | ||
| Can also assess disease response. | ||
| Using the same modality as pre-treatment can help decision-making at relapse. | ||
| Relapse | WB-MRI or FDG-PET/CT | As at baseline. |
| No role for WB-CT, as historic (non-active) lesions are likely to be detected. | ||
| Extramedullary or non-secretory disease | WB-MRI or FDG-PET/CT | Regular advanced imaging is required to monitor response and detect relapse. Modality and scanning interval should be patient-specific, according to disease risk and radiation exposure. |
| Assessment of fracture risk and planning surgery | CT | Option for spinal fractures. |
| Plain film X-ray | Option for long bone fractures. |
Imaging is recommended across a range of disease timepoints in myeloma. Abbreviations: WB, whole-body; MRI, magnetic resonance imaging; FDG-PET/CT, 18F-fluorodeoxyglucose positron emission tomography/computed tomography; CT, computed tomography; MRD, measurable residual disease.
The criteria for suspecting myeloma are not defined, but include patients with
any monoclonal immunoglobulin protein and symptoms or signs concerning for
myeloma end-organ damage. In asymptomatic patients, there is a need to define a
monoclonal protein threshold above which further investigation is needed;
however, this is challenging as myeloma can occur at any level of paraprotein
(Immunoglobulin G
The two advanced imaging methods for patients with suspected myeloma, WB-MRI or
FDG-PET/CT, are able to detect myeloma-associated changes both inside and outside
the bone marrow, and importantly prior to the occurrence of irreversible,
structural damage. Lytic bone disease requires at least 50% cortical bone loss
to be detectable on X-ray, at which point the risk of pathological fracture is
high and may not be completely reversible despite treatment for myeloma. In
addition to lytic bone disease, focal lesions of the bone or marrow detected by
MRI are independently associated with disease progression [37]. Such is the strength of this association, the presence of more than 1
focal lesion
Other staging investigations, such as serum protein biomarkers and genetic testing, are important complementary tests that help define prognosis. However, staging investigations based on blood tests are unable to indicate if a patient has bone disease or extramedullary disease, and can misrepresent the overall tumour burden.
The wrong imaging techniques are often requested for patients with suspected myeloma, which can result in patients being investigated with multiple modalities (including many using ionising radiation), and the introduction of unnecessary delays in diagnosis and treatment. Selected imaging of single anatomical areas is wasteful, as repeat imaging is still required to cover the rest of the skeleton. Plain imaging is vastly inferior at detecting bone disease and not recommended [38, 39], except in assessing the fracture risk of a long bone. Whole-body CT scan is also insufficiently sensitive and only provides anatomical information [40], as opposed to functional imaging techniques WB-MRI and FDG-PET/CT that also highlight tumour activity.
Of the two advanced techniques, WB-MRI is superior to FDG-PET/CT at detecting
myeloma bone disease, and there is a higher false negative rate with FDG-PET/CT
[41]. In around 10% of patients, a false negative FDG-PET
is due to a deficiency in the enzyme hexokinase in the tumour, which leads to a
lack of glycolytic activity [42]. Superior sensitivity of
WB-MRI over FDG-PET/CT was confirmed in head-to-head clinical trial comparisons
[43, 44, 45].
WB-MRI is also superior to FDG-PET-CT at showing diffuse infiltrative disease,
which correlates with the degree of plasma cell infiltration. A diffuse
infiltration pattern without focal disease is more likely to be associated with
high-risk genetics [46]. The presence of multiple large
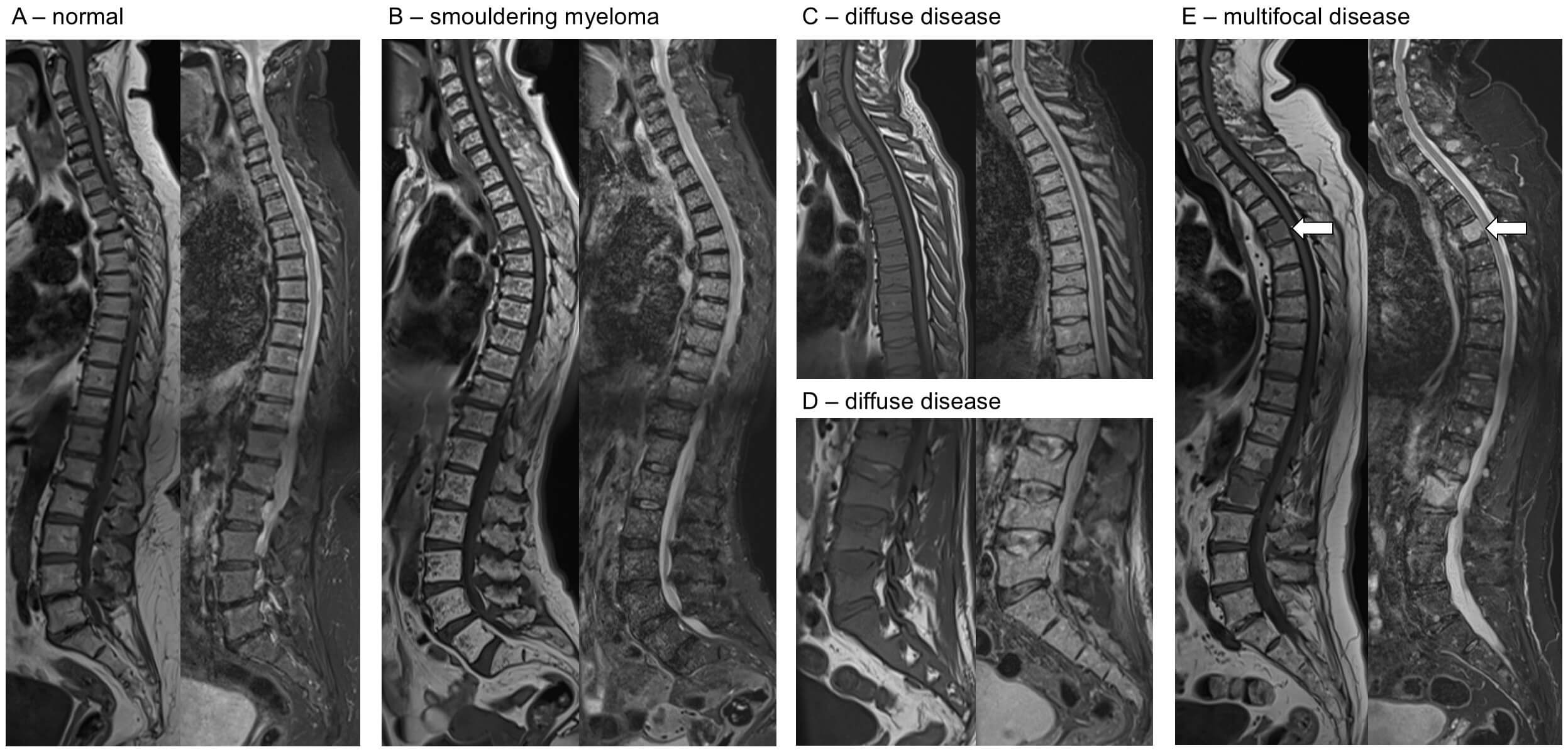 Fig. 2.
Fig. 2.
Example MRI images across the spectrum of disease. For each pair of images, T1 sequences are on the left, short tau inversion recovery (STIR) on the right. (A) Normal T1 sequences are fat sensitive, and bone marrow is bright. STIR sequences are fluid-sensitive fat fat-saturated. (B) Smouldering myeloma, with innumerable small foci seen on T1 (low signal, pepper) and STIR (high signal, salt) throughout the spine, but not reaching the diagnostic threshold for myeloma. (C,D) Diffuse disease is shown by reduced marrow signal on T1 and increased signal on STIR (C, thoracic; D, lumbar). (E) Multifocal disease identified by hypointense areas on T1 with corresponding increase in signal in STIR sequences (e.g., as identified by the arrows).
Whilst WB-MRI is the preferred imaging modality, some patients are unable to tolerate the relatively long scan times, for example, due to claustrophobia or pain, the latter an important consideration in patients with active myeloma.
Solitary plasmacytoma is an isolated tumour of plasma cells which can be either in association with bone marrow, or entirely outside of the bone marrow (extramedullary), but by definition, with no evidence of myeloma elsewhere. Extramedullary plasmacytomas can occur anywhere, but most commonly in head and neck (Fig. 3). Both solitary bone and extramedullary plasmacytomas are treated with radiotherapy or surgical resection, however most patients will later develop myeloma, with the highest risk amongst those with bone plasmacytomas [48, 49] (Fig. 4). Plasmacytomas may or may not be associated with a monoclonal immunoglobulin protein and imaging is always key for detection of relapse.
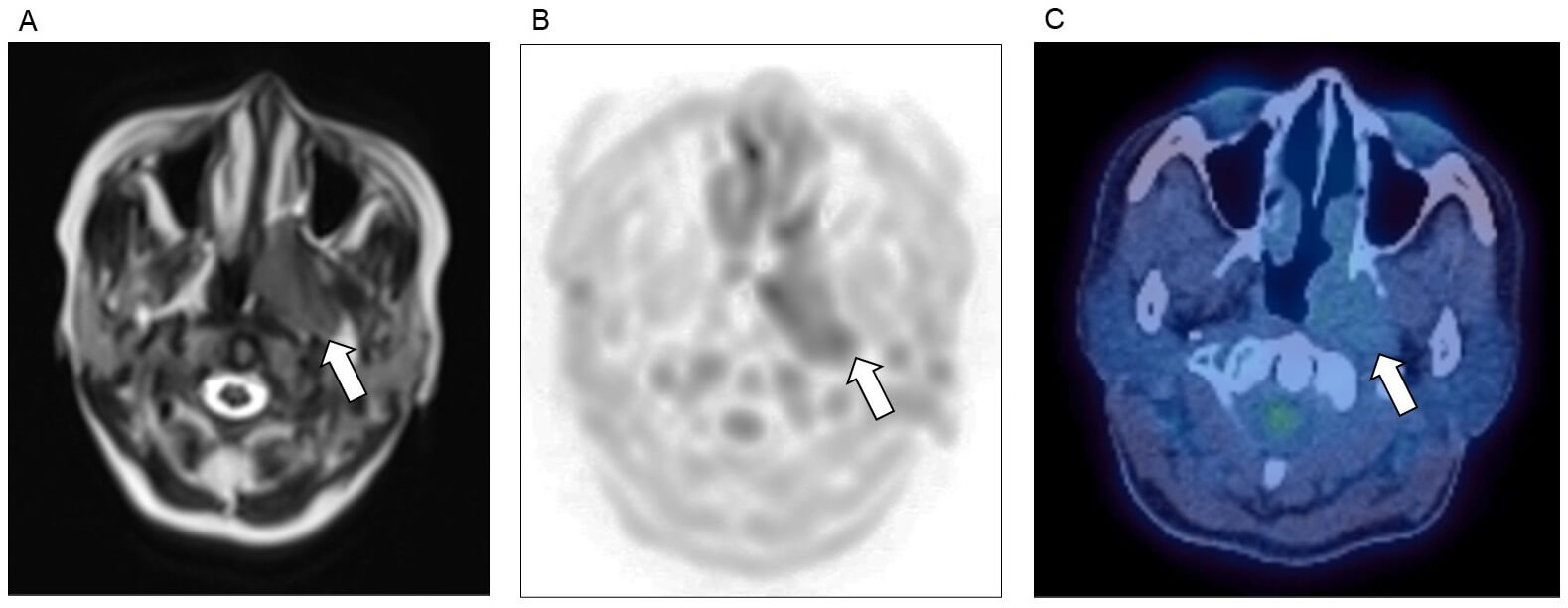 Fig. 3.
Fig. 3.
Plasmacytoma affecting the head and neck. Nasopharyngeal plasmacytoma (arrows), as detected on (A) MRI T2 sequence, (B) MRI diffusion weighted imaging (DWI), and (C) FDG-PET/CT imaging.
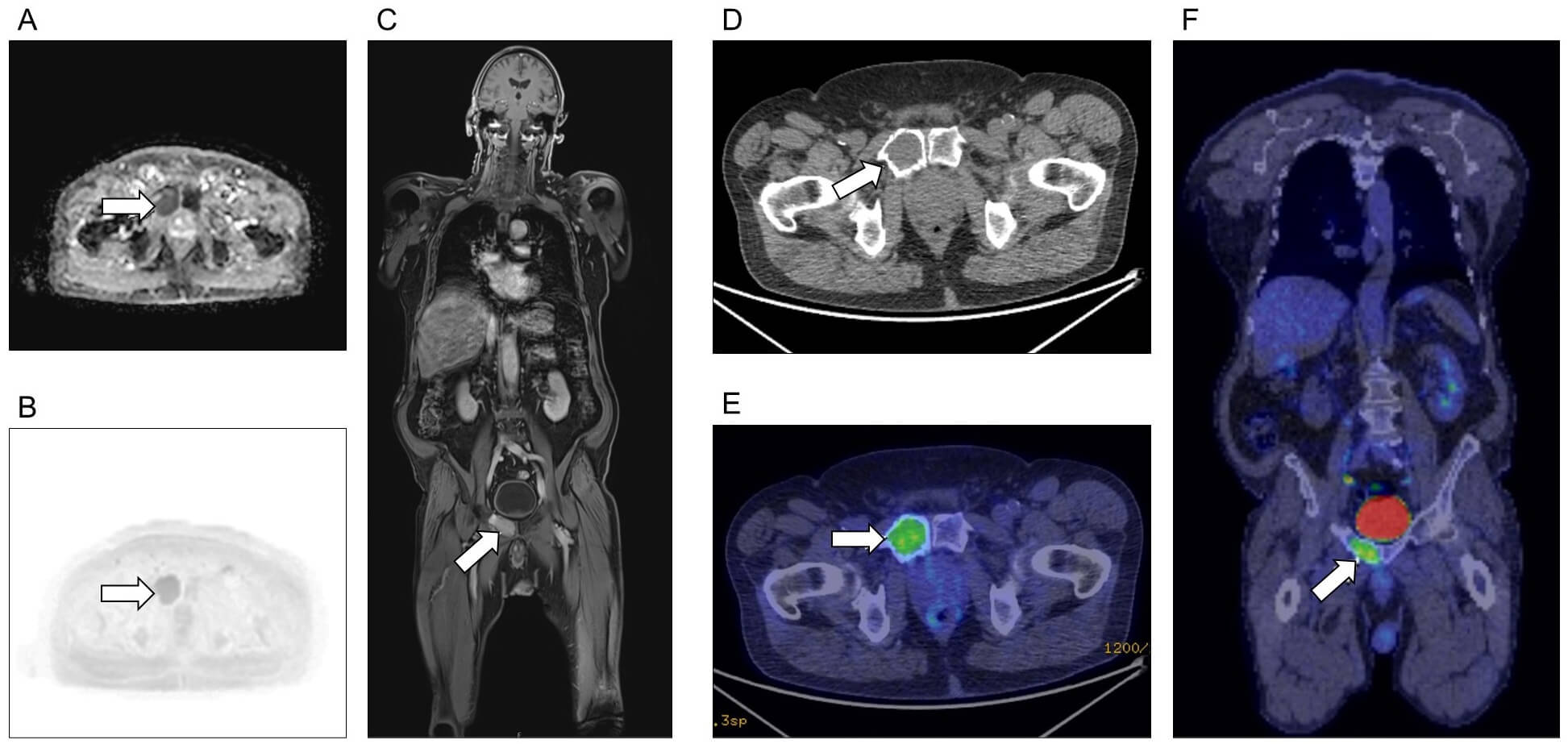 Fig. 4.
Fig. 4.
Plasmacytoma affecting bone. Solitary plasmacytoma (arrows) of the right pubic bone, as seen on (A–C) MRI and (D–F) FGD-PET/CT. (A) Apparent diffusion coefficient axial image. (B) DWI axial image. (C) T1 post-contrast coronal image. (D) CT only axial image. (E) Fused CT and FDG-PET axial image. (F) Fused coronal image. Imaging of both modalities identified no additional sites of disease.
International guidelines recommend WB-MRI to assess patients with newly diagnosed bone plasmacytoma, and FDG-PET/CT for extramedullary plasmacytoma [36], although there are data showing WB-MRI has higher sensitivity also for extramedullary disease [50].
Response to treatment is typically measured by changes in the monoclonal
immunoglobulin concentration detected in the blood, and the level of bone marrow
infiltration by malignant plasma cells down to the current level of measurable
residual disease (MRD) which with modern techniques (flow cytometry or next
generation sequencing) is 1
When assessing response, both WB-MRI and FDG-PET/CT, as functional techniques, provide additional independent prognostic information [52, 53], but are rarely performed due to cost and access. For example, early change in lesional fat, detected by MRI, is predictive of a favourable clinical response to treatment [54, 55]. The functional information provided by WB-MRI, including apparent diffusion coefficient (ADC) and relative fat fraction, can also be used to identify patients with inadequate response to treatment [56], a group for whom improved and intensified treatment algorithms are urgently needed. Plain imaging and CT scans are much less sensitive and typically cannot distinguish new activity from old changes.
Compared with WB-MRI, the earlier access and longer experience with FDG-PET/CT have meant this modality has been incorporated into guidelines for assessing response [57]. In this setting, as at diagnosis, WB-MRI has a higher sensitivity of residual disease [58]. However, this is balanced against lower specificity, potentially increasing the number of non-viable residual lesions (false positives) being interpreted as evidence of sub-optimal treatment response. Consequently, post-treatment FDG-PET/CT has been shown to be a better predictor of outcomes than WB-MRI [59]. Given the lower cost and lack of radiation, there is a strong argument for WB-MRI in assessing response and monitoring, particularly in patients whose baseline imaging was by that modality. But despite the inclusion of imaging as part of IMWG response criteria, this has not translated into imaging being regularly incorporated into clinical trials, and far less into routine clinical practice.
There is now a major focus in clinical trials for assessment of treatment response to be based on bone marrow MRD, using that result as a trial endpoint (as a surrogate of survival), and subsequently using MRD results to escalate or de-escalate treatment [60, 61]. This move reflects the unprecedented depth and duration of responses that come with current first-line treatments and continuous maintenance therapy. However, given the patchy nature of myeloma, it is not infrequent to have active disease away from the bone marrow biopsy site, and these sites can only be detected by imaging. The importance of imaging is that it gives additional independent information in predicting the outcome. This will become clinically relevant if MRD becomes incorporated into routine clinical decision-making. Bone marrow MRD methods can have limitations around sensitivity, specificity and lack of reproducibility; therefore, combination with imaging assessments may allow for more confidence in clinical decision-making. Given the greater experience with FDG-PET/CT, this modality has been recommended to complement bone marrow MRD assessment in patients achieving complete response to treatment [62].
During monitoring, imaging is generally reserved for when there is a suspicion of relapse, most commonly because of a rising monoclonal protein or occasionally due to new pain symptoms. Generally, routine imaging in a well patient with stable monoclonal protein is not recommended. However, for an increasing number of patients, imaging may be the only way of monitoring disease, for example, with oligo- or non-secretory myeloma and extramedullary disease. In the recent BSH guidelines [6] the recommendation is as follows: “For patients with oligo- or non-secretory myeloma, and some types of extramedullary disease, regular advanced whole-body imaging should generally be accessible on a 3–4 monthly basis, as in solid cancer monitoring; if deemed clinically appropriate, clinical teams may opt for larger scanning intervals at an individual patient level. Cumulative radiation exposure should be considered regarding the choice of imaging modality”. It is increasingly common to see patients developing poorly-secretory or extramedullary tumours after treatment, emphasising the importance of imaging as a method of disease monitoring in such patients (Fig. 1). As at suspected diagnosis, failure to perform imaging in a timely way during monitoring can lead to catastrophic fractures or development of significant bone pain.
In 2016, the UK National Institute for Health and Care Excellence (NICE) published guidelines for myeloma, which included the first cost-effectiveness analysis for diagnostic imaging [28]. The guidance recommends advanced imaging with WB-MRI or FDG-PET/CT for all patients with suspected myeloma. Their cost-effectiveness analysis showed that it is more cost-effective to do WB-MRI as a single test as first line, rather than sequentially perform low-sensitivity tests that yield equivocal results (Fig. 5, Ref. [6, 28]). National, European and international guidelines on imaging for patients with suspected multiple myeloma have all strongly advocated the need for whole-body cross-sectional imaging [6, 14, 63]. As discussed, the IMWG also incorporated imaging into international response criteria [57]. Despite these guidelines, three UK surveys have shown widespread practice outwith recommendations, with patients frequently referred for multiple and inappropriate imaging tests [64, 65, 66].
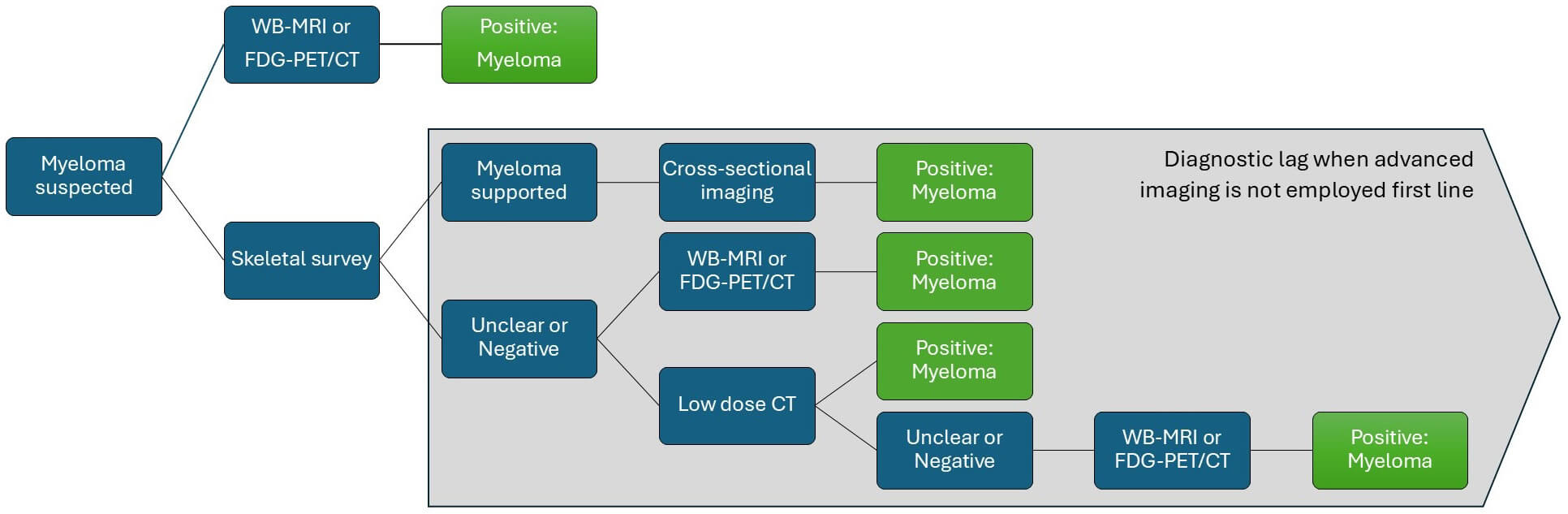 Fig. 5.
Fig. 5.
Decision tree of the imaging modalities required to diagnose myeloma. NICE [28] health economic modelling showed performing multiple imaging techniques to confirm myeloma was cost-ineffective and led to a delay in diagnosis. This increases delay, unnecessary testing, radiation exposure and cost. Adapted with permission from Kaiser et al. [6].
All guidelines strongly recommend against using skeletal surveys, but regrettably, all three UK surveys have shown that plain X-rays are still being used for suspected myeloma, and many patients undergo multiple imaging tests (requiring multiple hospital visits) before eventually reaching their delayed diagnosis of myeloma. There are major issues with scanner availability, reporting time, financial limitations, dedicated radiologists and radiographers, and nuclear medicine technician training. The superiority and benefit of advanced imaging for both patient care and efficient use of NHS resources have been demonstrated, but repeated recommendations on imaging modalities of choice in national/NICE/international guidelines have resulted in little measurable change in practice overall in the UK. Given the barriers to routine adoption of imaging at baseline, it is not surprising that repeat imaging after treatment and during follow-up is seldom completed outside of dedicated clinical trials, despite IMWG recommendations.
The demand for WB-MRI and FDG-PET/CT will increase as the incidence of myeloma is increasing, patients with myeloma are living longer, and imaging is being used more for monitoring and assessment of response. Although the total number of scans is relatively small, the burden on radiology is significant. Imaging departments are experiencing tremendous service and financial pressures, and are inadequately resourced, with demand outstripping both acquisition and reporting capacity. There is a need for more rapid scanner protocols to reduce scanner time, and more training and time allocated to reporting of these complex scans—a degree of inter-rater variability is a recognised limitation of WB-MRI [67]. Given the need for standardised acquisition, interpretation and reporting of WB-MRI in myeloma, there has been work to develop a Myeloma Response Assessment and Diagnosis System (MY-RADS) [68, 69]. It is possible to successfully deliver a multicentre WB-MRI study using the MY-RADS protocol, from sites with a wide range of hardware and prior WB-MRI experience. The challenge is to expand such approaches and access to WB-MRI and FDG-PET/CT across the UK.
With the advent of artificial intelligence (AI), there is increasing interest in exploring how advanced computing can be applied to a range of imaging techniques to improve the accuracy and speed of reporting. Given the volume of data generated by each WB-MRI or FDG-PET/CT scan, there is a need to incorporate machine-assisted learning approaches to myeloma imaging, and efforts in this area are underway [70]. Examples of early application of AI include the automated identification of bone disease, thereby supporting a diagnosis of myeloma [71], and to MRD evaluation following treatment [72]. Research groups have also used machine learning techniques to improve prognostication of patients with newly diagnosed myeloma [73], including with correlation to high-risk genetic profiles [74]. As imaging techniques become more advanced and adopted into routine clinical practice, their interpretation is likely to require support from AI tools [75]. The application of AI towards overcoming the existing barriers to routine use of advanced imaging is a research priority; examples include the UK-based WISER-M study (ISRCTN84990088).
Imaging is essential during the diagnostic work-up of myeloma, and has an increasingly important role in the assessment of treatment response and monitoring. For patients with non-secretory or extramedullary disease, imaging is essential throughout the disease course. Advanced imaging is universally recommended, and the advantages of WB-MRI have made this the preferred modality within UK guidelines. The implementation of WB-MRI represents a substantial challenge, but one worth pursuing, as it brings significant benefits to the patient of an earlier diagnosis and avoidance of unnecessary ionising radiation, and overall cost savings through performing the right test first time.
All data included in this study are available from the corresponding author upon reasonable request.
GM, OW and GP designed and performed the literature review. GM, OW and GP drafted the manuscript. All authors contributed to critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Anonymised images are provided by the authors. In accordance with local policies, specific consent for use is not required. NHS Digital policy states that fully anonymised data are not confidential patient information.
GM, NIHR Academic Clinical Lecturer, is funded by the NIHR. The views expressed in this publication are those of the author(s) and not necessarily those of the NIHR, NHS or the UK Department of Health and Social Care. With thanks to the Guy’s and St. Thomas’ NHS Foundation Trust myeloma imaging and research group.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





