1 Sheffield Teaching Hospitals NHS Foundation Trust, S5 7AU Sheffield, UK
2 Academic Unit of Gastroenterology, Sheffield Teaching Hospitals, S5 7AU Sheffield, UK
Abstract
Sedation plays a crucial role in gastrointestinal endoscopy, balancing patient comfort and procedural safety. With the increasing complexity of gastrointestinal endoscopic procedures, comes the inherent risk of sedation-related complications. This review, based on the 2024 British Society of Gastroenterology (BSG) guidelines, outlines updated strategies to address the increasingly diverse clinical needs. This review article focuses on case studies to highlight key recommendations for different patient groups. Case analyses highlight unique sedation considerations for patients with comorbidities such as obstructive sleep apnoea (OSA), cardiac dysrhythmia, and chronic kidney disease, emphasizing the need for thorough pre-procedural assessment and individualized sedation plans. Special considerations for the choice of sedation in pregnant and breastfeeding patients are explored. In emergency endoscopies, particularly for upper gastrointestinal bleeding, close collaboration with anaesthetics is essential to determine sedation choice, airway management, and the ceiling of care. The review also examines the need for deep sedation in complex or combined procedures such as endoscopic ultrasound (EUS) and endoscopic retrograde cholangiopancreatography (ERCP), advocating for resource investment to support safe and effective sedation practices.
Keywords
- sedation
- gastrointestinal endoscopy
- patient safety
- breastfeeding
- pregnancy
- obstructive sleep apnoea
In the last three decades, significant advancements in endoscopy have expanded its role in therapeutic procedures, particularly in neoplasia resection, bridging the gap with surgical interventions. This has led to increasingly complex and prolonged procedures. Due to these advancements, sedation requirements have increased, with longer and deeper sedation often needed to ensure patient comfort and procedural success [1].
Sedation is commonly used in gastrointestinal endoscopy, but practices vary globally. In the UK, conscious sedation is the standard, primarily using benzodiazepines and opioids. In the USA and Australia, benzodiazepines (mainly midazolam) and opioids are the most common sedatives for moderate sedation [1], but deep sedation is readily available. However, many European countries favour propofol monotherapy or combined with opioids over the use of benzodiazepines [2].
Gastrointestinal endoscopy carries inherent risks, including sedation-related adverse events such as hypotension, hypoxia, and inadvertent oversedation, all of which can lead to significant respiratory compromise. The increasing complexity and duration of endoscopic procedures have resulted in greater sedation use, which raises safety concerns. Fortunately, significant respiratory depression requiring reversal agents remains relatively infrequent. Data from the National Endoscopy Database (NED) indicate that the use of reversal agents for sedated colonoscopy procedures is 0.02% per annum, while for gastroscopy, it is 0.05% per annum [3].
The recently published British Society of Gastroenterology guidelines provide a comprehensive overview of recommendations for delivery of sedation and monitoring and training requirements for staff delivering sedation [4].
This article presents five fictional clinical cases constructed by the authors to reflect realistic clinical scenarios commonly encountered in endoscopy services. These cases are used to illustrate key principles and practical applications of the updated British Society of Gastroenterology (BSG) sedation guidelines. This review explores management strategies and comprehensive guidance on patient management, drug selection and intra-procedural recommendations for the specific patient groups. This review would serve as a useful guide to endoscopy nurses and allied healthcare professionals, junior and senior endoscopists to adopt into their daily clinical practice.
A 40-year-old man is admitted to the hospital to have an ileo-colonoscopy as he
has been having persistent diarrhea. He has a past medical history of anxiety but
otherwise is normally well. His blood results showed a slightly raised C-reactive
protein (CRP) of 16 mg/L [0–5 mg/L] and faecal calprotectin of 300 mg/kg [
He had the colonoscopy under conscious sedation using fentanyl and midazolam. During the procedure, he was uncomfortable and in distress despite being given additional doses of sedation. His observations remained normal throughout. The procedure was completed with constant reassurance. After the procedure, he informed the staff, “I expected to be asleep throughout the procedure.”.
Patient dissatisfaction occurs when there is a mismatch between patient expectations and the real experience, which can cause concern and anxiety during the procedure. This situation can occur when patients are not appropriately informed about the procedure at the time of booking. In addition, the study has shown that a large portion of patients, especially those who have not had endoscopies in the past, tend to believe that they will be asleep during the procedure or that it will be painless [5]. They believe that having “sedation” is equivalent to “being asleep”. Studies found that in many instances, this misconception was not cleared up until the procedure had started [6, 7].
Randomised control trials (RCTs) have shown that patient-based education
significantly reduces pre- and post-procedural anxiety (7.09
These studies highlight that pre-procedural education is essential in mitigating
anxiety, aligning patient expectations, and improving overall satisfaction.
Patients who are more satisfied or have better experiences are found to be
adherent to post-procedural care or future follow-up and willingness for further
endoscopies [11]. Decreased satisfaction is also associated
with further symptoms and worsened physical health (59.7 vs. 68.4; p
Patients who receive detailed sensory information about sedation, bowel preparation, and the sensations they may experience during the procedure tend to feel less anxious before, during, and after the procedure. This clear understanding of what to expect greatly alleviates pre- and peri-procedural anxiety [13, 14, 15, 16].
British Society of Gastroenterology (BSG) guidelines recommend assessing patient comfort continuously throughout the procedure.
The Patient-Reported Scale for Tolerability of Endoscopic Procedures (PRO-STEP) is a validated Patient-Reported Experience Measure (PREM) designed to assess patients’ physical tolerability by evaluating their discomfort and awareness during the procedure and for endoscopy-related symptoms post-procedure. Unlike other PREMs, PRO-STEP prioritizes symptom assessment over holistic patient experience [11]. It has been validated in studies, including one with 255 patients, where its results were consistent with other established PREMs. Its simple and practical six-question template makes it easy to record, analyse, and interpret tolerability for various procedures such as gastro-duodenal endoscopies, colonoscopies and endoscopic retrograde cholangiopancreatography (ERCP) [11]. However, PRO-STEP does not measure pre-procedural anxiety, patient emotions or overall patient experience. Therefore, it is best used as a quick, symptom-focused assessment tool alongside other PREMs to provide a comprehensive evaluation of the patient’s experience [11].
The Newcastle ENDOPREM is a more holistic PREM designed to assess patient comfort and overall experience before, during, and after endoscopic procedures which may be useful. It was validated through 799 patient responses across National Health Service (NHS) trusts, followed by rigorous psychometric analysis. As a detailed patient-centred questionnaire, it evaluates the entire patient journey, from appointment scheduling to post-procedural care. This makes it a valuable tool for identifying any areas for improvement throughout the service [17].
By continuously monitoring patient comfort throughout the process, these PREMs are invaluable for ensuring that patient comfort is continuously monitored and improved, leading to better patient outcomes and satisfaction.
Auditory and visual distractions have been found to be effective in alleviating patients’ anxiety and distress during endoscopic procedures.
The use of music can be used to significantly reduce the patient’s reported levels of pain and anxiety, while also enhancing their satisfaction and comfort during the procedure [18].
This patient experienced distress due to unmet expectations, highlighting the importance of pre-procedure education. Inadequate education led to him feeling increased anxiety during and after the procedure. This can negatively affect his experience and recovery. Clear education through booklets, videos, and other multimedia resources can help him understand the procedure and set realistic expectations, thus reducing his fear of the unknown. PREMs can be used to assess his satisfaction and identify areas for improving communication and education. Auditory and visual distractions can be used during the procedure to alleviate his distress.
A 60-year-old man presented with persistent fatigue. His blood results showed
low ferritin of 14
Bidirectional endoscopy was planned to investigate his iron deficiency anaemia (IDA).
In patients with obstructive sleep apnoea, a formal pre-assessment is required.
In patients such as this with complex co-morbidities, a comprehensive pre-assessment is essential to mitigate sedation-related risks. Individuals in higher American Society of Anaesthesiologists (ASA) classes and older age groups are at increased risk for serious adverse events during procedures. A detailed pre-assessment, including a thorough patient history, allows for effective risk stratification and the identification of risk factors and comorbidities that may predispose patients to circulatory or respiratory failure during deeper sedation [19, 20, 21]. This process ensures that sedation plans are tailored to enhance patient safety and that patients are fully informed about the procedure’s purpose, benefits, possible risks, clinical preparations, and sedation options along with their associated risks [22].
In this case, particular attention should be given to the patient’s OSA and cardiac dysrhythmia, as these conditions significantly impact the cardiac and respiratory safety and management of sedation during the procedure.
Patients at high risk of OSA are recommended to be screened for it before their endoscopic procedure.
Patients known to be diagnosed with OSA, such as the one above, or those with a body mass index (BMI) greater than 35 kg/m2, have a significantly greater risk of hypoxia and respiratory failure when undergoing sedation. For patients who have not been diagnosed with OSA but are of high risk, the Berlin and Snoring, Tiredness, Observed apneas, Pressure (high blood pressure), BMI, Age, Neck circumference, and Gender (STOP-BANG) questionnaires can be utilised as quick and easy tools to screen for OSA so that peri-procedural preparations can be made to ensure the patient’s safety [21, 23]. Details of the STOP-BANG questionnaire are provided in Table 1 (Ref. [23]).
| Parameter | Yes | No | |
| Do you snore loudly? | 1 | 0 | |
| Do you feel tired, fatigued or sleepy during daytime? | 1 | 0 | |
| Has anyone observed you stopped breathing or gasping for air during sleep? | 1 | 0 | |
| Do you have high blood pressure? | 1 | 0 | |
| BMI |
1 | 0 | |
| Age |
1 | 0 | |
| Neck circumference | 1 | 0 | |
| Male | |||
| Female | |||
| Are you a male? | 1 | 0 | |
OSA low risk: Total score 0–2; OSA intermediate risk: Total score 3–4; OSA high risk: Total score 5–8.
BMI, body mass index; OSA, obstructive sleep apnoea; STOP-BANG, Snoring, Tiredness, Observed apneas, Pressure (high blood pressure), BMI, Age, Neck circumference, and Gender.
Meta-analyses have shown that the STOP-BANG has a higher sensitivity than the Berlin Questionnaire (BQ) for detecting OSA. Determining OSA severity is also crucial, as moderate to severe cases pose greater risks for peri- and post-procedural complications. Studies have found that the STOP-BANG questionnaire has a higher sensitivity than BQ for identifying moderate and severe OSA, with sensitivities of 93% vs. 78% and 100% vs. 87%, respectively. However, the BQ has greater specificity than STOP-BANG for moderate and severe OSA, with 50% vs. 43% and 46% vs. 37%, respectively [24, 25].
OSA can lead to frequent hypercapnia due to apnoea. In healthy patients, hypercapnia triggers ventilation, but repeated episodes can cause carbon dioxide retention and type 2 respiratory failure. Sedatives for endoscopy can worsen this by causing respiratory depression. Therefore, minimal hypnotics and careful opiate titration are advised in this patient group [20, 22, 26, 27].
The use of capnography is recommended for high risk groups and those with OSA.
Capnography is a non-invasive method to measure end-tidal carbon dioxide and has a higher sensitivity to detect adverse respiratory events compared to standard monitoring with pulse oximetry alone. Meta-analysis from dental studies has shown benefits in procedural sedation and reducing complications [28].
Patients with OSA require extended post-procedural monitoring for at least 90 minutes from the end of the procedure.
Patients with obstructive sleep apnoea (OSA) are at increased risk of postoperative complications such as ventilatory and airway compromise, which can lead to hypoxia and hypercapnia. The likelihood of these adverse events is heightened in prolonged procedures. Due to the increased risk of hypoxaemia and hypercapnia in patients with OSA undergoing endoscopic procedures, especially oral route endoscopy, this patient should have prolonged monitoring post-procedure for at least 90 minutes after procedure completion. Monitoring of oxygen saturation, blood pressure, and heart rate is essential, with pulse oximetry as a minimum requirement. If desaturation occurs, there should be a low threshold for escalation, including the initiation of non-invasive ventilation (NIV) if necessary [26, 29, 30].
Patients with cardiac dysrhythmia require continuous electrocardiogram monitoring throughout the procedure.
The patient’s cardiac rhythm should be closely monitored during the procedure. Cardiac arrhythmias can be exacerbated by factors such as airway manipulation, electrolyte imbalances, and the effects of sedatives, all of which are commonly encountered during endoscopic procedures. In this specific patient who was being assessed for a cardiac defibrillator, continuous electrocardiogram (ECG) monitoring is essential interprocedurally allowing for prompt intervention if necessary [31].
Dose reduction and individual titration are required in patients with chronic kidney disease.
In this patient with chronic kidney disease (CKD) and an eGFR of 28 mL/min/1.73m2, impaired
renal function must be considered when selecting sedatives and opioids, as it
affects drug clearance. Midazolam, a lipophilic benzodiazepine, is primarily
metabolised in the liver. However, end-stage renal failure (eGFR
When selecting opioids, it is essential to differentiate between hydrophilic and lipophilic agents. Hydrophilic opioids such as morphine and codeine are renally excreted. Administering hydrophilic drugs such as this can result in drug accumulation, prolonged sedation, and increased risk of respiratory depression in CKD patients. In contrast, lipophilic opioids such as fentanyl and buprenorphine are primarily metabolised by the liver, making them safer choices in renal impairment [33].
However, even lipophilic opioids undergo some renal excretion. Therefore, in advanced CKD, it is recommended to start at 50% of the usual dose and titrate carefully to minimize toxicity [32, 33].
Due to this patient’s extensive medical history, particularly OSA, an anaesthetic pre-assessment is required to assess their severity using the STOP-BANG questionnaire. This is crucial as OSA increases the risk of hypercapnia. Continuous oxygen saturation monitoring and ideally capnography should be used during and post-procedure for this patient. He should also have continuous ECG monitoring due to his dysrhythmia. Given his impaired renal function (an eGFR of 28 mL/min/1.73m2), lipophilic opioids such as fentanyl are preferred, while midazolam can be administered at a standard dose for sedation.
A 50-year-old man presented to the emergency department with haematemesis, confusion, distended abdomen and new onset of jaundice. He has a past medical history of alcoholic liver cirrhosis, gastric ulcer, and depression. On assessment, his blood pressure is 92/63 mmHg and his heart rate is 114 beats per minute (bpm). He was noted to be encephalopathic. His blood results showed:
Haemoglobin (Hb): 58 g/L [131–166 g/L]
Platelets: 98
White cell count: 6.7
Creatinine: 200
Urea: 16 mmol/L [2.5–7.8 mmol/L]
Prothrombin time: 16 seconds [9.8–11.6 seconds]
He was resuscitated and a restrictive transfusion policy was advocated.
Whilst in the department, he had further haematemesis and remained tachycardic, hence an emergency endoscopy was planned.
Discussion with Anaesthetics is required on the choice of sedation and management of airway.
Assessment of Hepatic encephalopathy needs to be done prior to endoscopy and sedation.
In upper gastrointestinal bleeding (UGIB), there is a high risk of aspirating gastric contents, especially in patients with hepatic encephalopathy (HE). While fasting minimizes aspiration risk in elective procedures, urgent endoscopy in HE patients presents airway management challenges [34].
The West Haven grading system should be used to assess HE severity. Patients with grade 3 or 4 HE require prophylactic intubation to protect the airway. Sedation should be minimized to prevent further neurological depression [34]. Benzodiazepines should be used with caution as they are lipophilic and metabolised by the liver. If the liver function is impaired, it can result in impaired benzodiazepine elimination resulting in abnormal accumulation and prolonged sedation [35]. Patients with HE and ASA grade 4 or higher should also be discussed with the anaesthetic team to determine the safest airway strategy. Early anaesthetic involvement ensures proper sedation and airway management, including consideration for intubation if necessary [34, 36].
The ceiling of care needs to be documented prior to emergency endoscopy.
Emergency endoscopy poses significant risks, including hemodynamic instability, aspiration, and the potential need for intensive resuscitation. Establishing a ceiling of care before the procedure ensures that interventions align with the patient’s best interests and desires. Discussing escalation levels with the patient in advance respects their autonomy and allows them to participate in decision-making, clarifying their preferences.
A standardized locally modified template should be provided to document the patient’s preferences regarding resuscitation, intubation, and post-endoscopy intensive care unit (ICU) transfer. The template will also include the clinical decision on escalation and ceiling of care that is appropriate for the patient. The documented escalation plan will aid the clinical team in appropriate interventions should the patient deteriorate [37, 38, 39, 40].
As this patient is encephalopathic, an immediate discussion with the anaesthetic team is required to determine his ceiling of care and appropriate interventions. If full escalation is warranted, airway management and prophylactic intubation should be considered. Additionally, due to his liver impairment, his sedation plan must be carefully reviewed, as benzodiazepine metabolism may be impaired, making it a less suitable option.
A 42-year-old female presented with a two-week history of persistent diarrhoea with blood per rectum and cramping left iliac fossa pain but with an absence of urinary symptoms. She has a past medical history of ulcerative colitis (UC) on mesalazine, migraine, and hypertension. She had a baby 12 weeks ago, whom she is breastfeeding. She is fatigued but is otherwise systemically well with no pyrexia. Her blood results showed:
Haemoglobin (Hb): 118 g/L [131–166 g/L]
Platelets: 220
White cell count: 6.3
Creatinine: 98
Urea: 7.8 mmol/L [2.5–7.8 mmol/L]
C-reactive protein (CRP): 12 mg/L [0–5 mg/L]
Stool cultures were negative for any growth.
Her findings indicate a likely flare-up of her ulcerative colitis (UC) and she was treated with steroids. She required a flexible sigmoidoscopy to further assess her disease severity and extent prior to considering biological therapy. However, as she is currently breastfeeding, it is important to determine what types of laxatives and sedation are safe for patients with breastfeeding.
Adequate hydration is essential during breastfeeding, as breast milk is over 80% water. This patient, who is both breastfeeding and experiencing fluid loss from diarrhoea due to her flare-up, faces an increased risk of dehydration. Additionally, laxatives and phosphate enemas used for flexible sigmoidoscopy can further contribute to fluid loss. It is therefore crucial to ensure that breastfeeding women like this patient are well-hydrated before and throughout the procedure to reduce the risk of dehydration and acute kidney injury. For a flexible sigmoidoscopy in this setting, a phosphate enema would suffice instead of full purgatives [41].
Endoscopic Interventions should only be done for pregnant and breastfeeding patients if there is a clear indication and the benefits outweigh the risks.
Pregnancy increases the risk of aspiration especially in the further terms as the size of the foetus grows. Additionally, certain sedation drugs can be harmful to the foetus and the mother and must be used with caution. As aforementioned, dehydration is also a huge risk, especially in breastfeeding patients. Due to these risks, endoscopic procedures should be carefully considered with benefits and risks weighed with discussion with the patient [42, 43, 44].
In patients who are pregnant, single doses of midazolam are safe, but diazepam and other benzodiazepines should be avoided.
Opioids, propofol, nitrous oxide and local anaesthetic throat spray are safe to use in pregnancy. However, benzodiazepines can pass through the placenta and affect the foetus. Diazepam is contraindicated due to the risk of teratogenicity. Thus, if benzodiazepines are required for sedation, then a single dose of midazolam is recommended. It also has a shorter half-life, resulting in decreased systemic bioavailability [45, 46].
In patients who are breastfeeding, midazolam, opioids, fentanyl and propofol are safe, but diazepam should only be used as a one-off dose if needed.
Although this patient is breastfeeding, most standard sedatives and anaesthetics used in endoscopy are safe for her. Nitrous oxide, being an inhaled agent, remains minimally systemic, is rapidly metabolised, and is primarily cleared through exhalation. As a result, negligible amounts are transferred to breast milk. However, its use is debated due to environmental concerns [47].
Propofol and midazolam appear in small amounts in breast milk and so are deemed safe. Only about 0.025% of propofol is transferred to breast milk, regardless of whether it is used for induction or maintenance of anaesthesia. Midazolam has a short half-life of 3 hours and a milk-to-plasma concentration ratio of 0.15, with values below 1 considered safe for lactating women. Therefore, both propofol and midazolam allow mothers to breastfeed as soon as they are alert. Diazepam, however, has a much longer half-life of 43 hours and a milk-to-plasma concentration ratio of 2.7; therefore, it should be used cautiously, ideally limited to a single dose if necessary. After receiving these sedatives or anaesthetics, there is no need for the patient to pump and discard breast milk before feeding [47]. This patient’s fluid status must be closely monitored as she has fluid loss from both breastfeeding and diarrhoea. If signs of dehydration or acute kidney injury develop, intravenous (IV) fluids should be administered. For her flexible sigmoidoscopy, midazolam or propofol can be safely used for sedation.
A 60-year-old man presented to emergency department (ED) with severe right-sided upper abdominal pain and new onset of jaundice. On further questioning, he reports his urine has been darker than usual with associated pale stools. His pain is worse after eating and he thinks he has potentially lost some weight over the last few months. He has a past medical history of depression, alcohol excess, and oesophagitis. He has a history of being an intravenous drug user (IVDU), having last injected 6 months ago. He is haemodynamically stable with no pyrexia.
His blood results showed:
Haemoglobin (Hb): 142 g/L [131–166 g/L]
White cell count: 8.0
Bilirubin: 76
Alanine transferase (ALT): 38 IU/L [0–33 IU/L]
Aspartate transaminase (AST): 42 IU/L [30–32 IU/L]
Alkaline phosphatase (ALP): 428 IU/L [30–130 IU/L]
Gamma-glutamyl transferase (GGT): 124 IU/L [0–40 IU/L]
Features of cholestatic jaundice are found in the above blood test and the patient’s presenting features. A magnetic resonance cholangiopancreatography (MRCP) was arranged which showed a dilated common bile duct (CBD) containing a stone and a possible mass at the head of the pancreas. The hepato-pancreato-biliary (HPB) multidisciplinary team recommended a combined endoscopic ultrasound (EUS) and endoscopic retrograde cholangiopancreatography (ERCP) to investigate the pancreatic mass and remove the obstructing CBD stone.
In the majority of routine ERCP and EUS, moderate sedation with midazolam and fentanyl is adequate; however, in complex HPB or combined procedures, deep sedation is recommended.
The decision to use standard or deep sedation for this patient is significantly influenced by the procedure’s length and complexity. As procedure duration increases, so do the risks of complications and reduced patient tolerance. Deep sedation helps maintain patient immobility, allowing endoscopists to undertake therapeutic intervention as needed. This stability is especially crucial in longer, technically demanding procedures to decrease the risk of complications. For complex procedures like ERCP and EUS, deep sedation should be carefully considered, factoring in the patient’s comorbidities and previous tolerance. However, for more intricate upper and lower gastrointestinal (GI) procedures, such as neoplasia resections (e.g., endoscopic submucosal dissection) or prolonged procedures like device-assisted enteroscopy, deep sedation is generally recommended to enhance safety and procedural success [3, 36, 48].
To induce deep sedation, an initial low-dose bolus of propofol is administered, with incremental adjustments and titration to maintain the sedation required. The dose required is based on the weight, age, comorbidities and the proposed duration of procedure. In the UK, propofol and deep sedation can only be administered by anaesthetists [4].
The main risks of deep sedation are airway compromise, respiratory depression, body temperature drop, and haemodynamic instability. In order to mitigate the risk of these events, it is imperative that the patients have continuous pulse oximetry, ECG, automated blood pressure measurements, core body temperature monitoring and capnography. High-flow oxygen should be available in case the patient becomes hypoxic [4, 49, 50]. This patient’s history as a former intravenous drug user (IVDU) may make gaining intravenous access challenging. Chronic IV drug use can lead to scarring, thrombosis, and vein collapse, complicating IV access. However, intravenous access is necessary for administering sedatives, such as propofol, during the endoscopic procedure. To ensure reliable access, alternative strategies like ultrasound-guided IV cannulation, non-IV sedation routes, or assistance from anaesthesiology may be considered to promote safe and effective sedation management [51, 52].
Ex-IVDU patients, especially those with a history of heroin or other opioids, often develop tolerance to sedatives and analgesics due to prolonged opioid exposure. This desensitization can make standard sedation doses ineffective, often requiring higher or additional doses [52].
For these patients, combining sedatives with analgesics can enhance sedation effectiveness. However, this approach significantly increases the risk of respiratory depression and drug interactions. Therefore, individualized sedation and analgesia plans are essential, with careful assessment and vigilant monitoring to ensure safe and effective sedation is provided while preventing distress or inadequate analgesia [51, 52, 53].
As this patient requires a combined EUS and ERCP, deep sedation with propofol is necessary. To mitigate the risks of respiratory depression and hemodynamic instability, he requires continuous monitoring with pulse oximetry, ECG, automated blood pressure measurements, core temperature monitoring, and capnography. Given his history of IV drug use, which may have led to tolerance to sedatives and opioids, he will need an individually tailored sedation and analgesia plan to ensure adequate but safe sedation.
Fig. 1 depicts the patient journey incorporating key points from the above 5 cases.
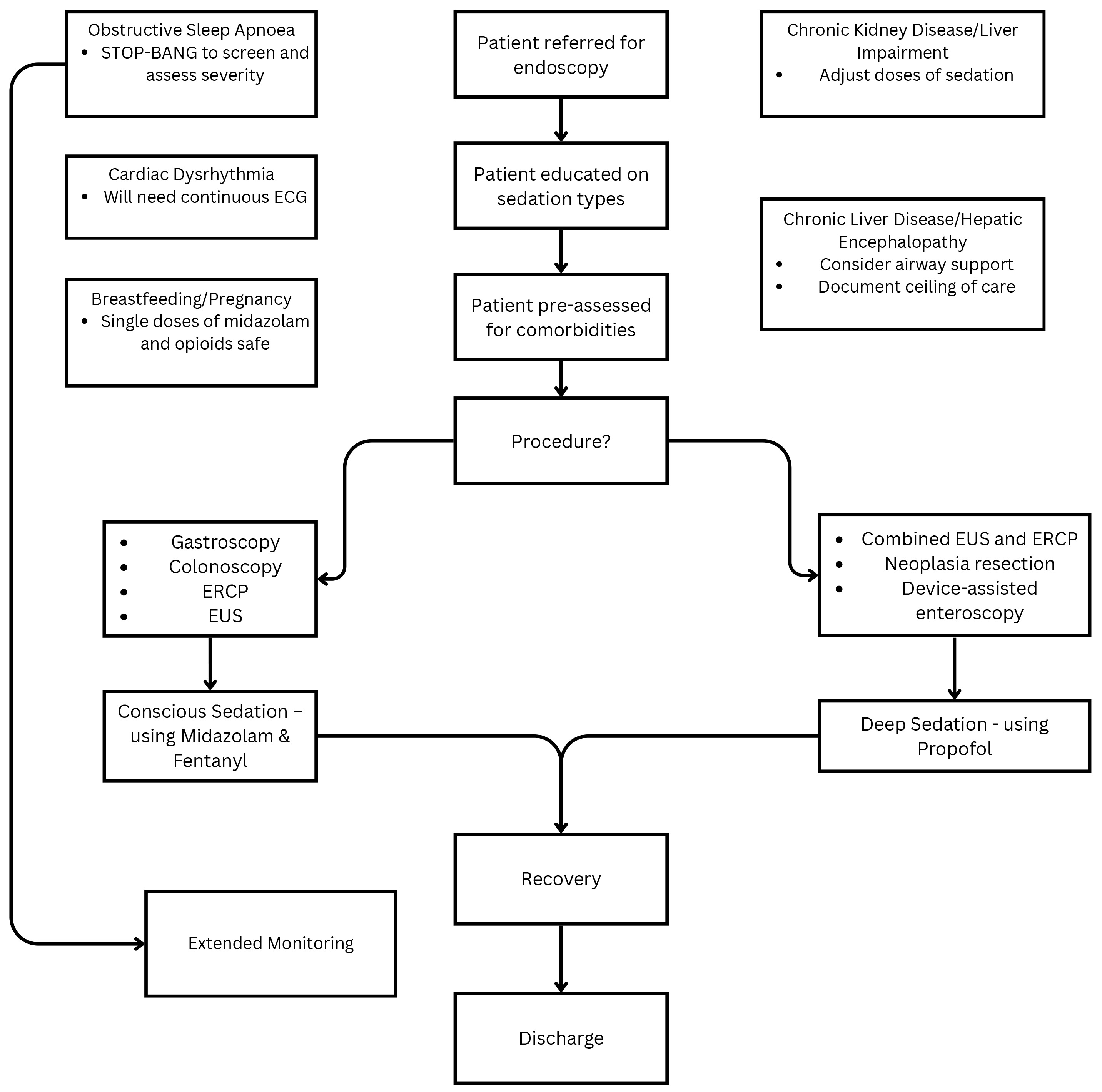 Fig. 1.
Fig. 1.
Patient journey when undergoing endoscopy. The figure was created using the Canva software (version 3.201.2; Canva Pty Ltd., Sydney, NSW, Australia). ECG, electrocardiogram; EUS, endoscopic ultrasound; ERCP, endoscopic retrograde cholangiopancreatography.
Patient expectations have evolved as healthcare has become more patient-centred, emphasizing both clinical safety and patient experience [54]. To enhance patient comfort and satisfaction, endoscopy services must reduce the gap between expectations and actual delivery of care. Clear communication about procedures, sedation, and outcomes helps align patient expectations with reality, improving both experience and clinical outcomes [6, 14]. Monitoring patient experience and comfort needs to be improved using validated tools and feedback incorporated into the service to improve the patient journey.
Pre-assessment plays a crucial role in balancing procedural goals with patient safety. In the UK, pre-assessment varies widely, from telephone-based to face-to-face appointments. In most units, formalised services analogous to surgical pre-assessment are lacking due to funding and the lack of infrastructure and manpower. With rising obesity rates, conditions such as obstructive sleep apnoea (OSA) are becoming more prevalent, increasing the risk of obesity hypoventilation syndrome, airway obstruction, and impaired respiratory function [23, 55]. Due to these factors, early identification and risk stratification are necessary for safe sedation [51, 53, 55].
Although large meta-analyses have not demonstrated increased risk of cardiovascular events with mild or moderate sedation, endoscopists need to be aware of the inherent risk of apnoea. In this group, the guidelines recommend a formal pre-assessment, careful titration of opiates and minimal hypnotics. Peri-procedural monitoring should be enhanced when sedatives are used, and this should be extended into the recovery phase [56, 57]. The evidence for capnography in endoscopy is limited. However, studies have found capnography to detect episodes of hypoxia faster than standard pulse oximetry [28, 58]. Patients should also be advised to bring their CPAP machine if they usually utilise one, should this be required post-procedure.
Endoscopists are scoping an aging population with increasing co-morbidities. While patients with stable atrial fibrillation (AF) do not need ECG monitoring, cardiac monitoring is recommended for patients with active dysrhythmia. This includes patients with recent episodes of arrhythmia or under investigation with cardiology. This approach has resource implications, as these patients need to be scheduled on lists with clinicians skilled in ECG interpretation. However, this strategy allows for safer procedures in this cohort of patients [31].
Upper gastrointestinal bleeding is a common medical emergency. In the context of a patient with chronic liver disease, bleeding from oesophageal or gastric varices with a large amount of gastric content of blood needs to be factored into the decision on the type of sedation for the emergency endoscopy. The guidelines also recommend that a clear escalation or ceiling of care is documented to avoid confusion on the most suitable location for the patient post-procedure [4]. Assessment for hepatic encephalopathy is mandated prior to the use of sedatives and patients with grade 3 or 4 encephalopathy should have their airway protected with formal intubation due to the risk of aspiration [59].
For complex and prolonged endoscopic procedures like combined EUS and ERCP, endoscopic submucosal dissection, and device-assisted enteroscopy, deep sedation is preferred to allow clinicians to focus fully on the procedure [48]. However, many units face challenges in providing general anaesthesia or propofol lists due to financial constraints, necessitating a thorough business case to justify the costs. Key expenses include additional staff, such as an anaesthetist or operating department practitioner, and specialized equipment like infusion pumps, anaesthesia monitoring machines, high-flow oxygen masks and capnography. Additional medications, including propofol and glycopyrrolate, are also required. Post-procedure, patients need close monitoring by resuscitation-trained recovery nurses with oxygen saturation, blood pressure, and ECG monitoring [4, 49, 50]. These requirements entail inherent costs, making a cost-benefit analysis essential to determine the most effective sedation approach [3].
Sedation training in the UK varies, with some elements covered in Joint Advisory Group on GI Endoscopy (JAG) courses and local training provided by certain endoscopy units. A national safe sedation course is also available. The new guidelines recommend that at least one staff member in every endoscopy room be trained in Intermediate Life Support (ILS) [4]. While Basic Life Support (BLS) covers cardiopulmonary resuscitation (CPR) basics—chest compressions, rescue breaths, and automated external defibrillator use—ILS expands on this, including the use of resuscitation drugs, airway management, and arrhythmia recognition and management [60]. To meet these training requirements, local endoscopy units will likely need to deliver additional ILS training for their staff.
The implementations of the recommendations from the new BSG sedation guidelines should be audited locally and nationally to further improve the delivery of sedation to patients.
Sedation plays a crucial role in gastrointestinal endoscopy. It is vital to balance patient comfort, procedural success, and safety. The increasing complexity of endoscopic procedures, particularly for high-risk patients with OSA, cardiac dysrhythmia, CKD, and liver failure, emphasizes the importance of pre-procedural assessment and individualized sedation plans. The BSG guidelines highlight the need for structured risk stratification, optimized monitoring, and appropriate sedation selection to minimize complications.
While deep sedation enhances procedural conditions for complex endoscopies, its implementation is resource-intensive, requiring additional staffing, equipment, and financial investment. Furthermore, the variability in sedation training across UK endoscopy units demonstrates the necessity of standardized training programs, including ILS certification for staff. Continuous audit and quality improvement initiatives will be essential in the future to refine sedation practices, enhance patient outcomes, and ensure safe, effective delivery of care in endoscopy.
Not applicable.
DGNN and RS were both actively involved in shaping the concept and structure of the article. Both authors contributed to writing and revising the manuscript critically for important intellectual content. Both authors gave final approval of the version to be published. Both authors have agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

