1 Department of Cardiology, Northumbria Healthcare NHS Foundation Trust, NE23 6NZ Northumberland, UK
Abstract
Chronic heart failure (HF) is a complex clinical syndrome and a major cause of morbidity and mortality, which affects more than 64 million people worldwide. HF is associated with decreased life expectancy and is characterised by increased hospitalisations, which increase in frequency and severity as the disease progresses. Patients with advanced HF experience a poorer health status and quality of life. Advanced HF increases the risk of premature death (80% of patients die within 5 years), as well as increasing symptoms, psychosocial and spiritual burden. The trajectory of the disease is unpredictable and person-specific, influenced by a multitude of factors including age of diagnosis, ethnicity, gender, comorbidity and socioeconomic status. In advanced HF, symptom management, psychosocial support and assistance with advanced care planning are important aspects of delivering holistic care. Dyspnoea, fatigue, pain, depression, anxiety, insomnia and worsening cognitive function are frequently seen in these patients. The symptoms experienced by the patient can be caused by the HF itself, comorbidities and treatment side effects, which all require careful consideration. While many aspects of palliative care are universal, HF patients have unique needs, challenges, and opportunities. Palliative care is much less likely to be offered to patients with HF than to those with other diagnoses. In this article, we explore the rationale for improving patient access and the key opportunities and challenges for delivering high-quality palliative care in this group.
Graphical Abstract
Keywords
- heart failure
- palliative care
- cardiac implantable electronic devices
Chronic heart failure (HF) is a complex clinical syndrome and a major cause of
morbidity and mortality, which affects more than 64 million people worldwide
[1]. Chronic HF is categorised into three main types
based on left ventricular ejection fraction (LVEF): heart failure with reduced
ejection fraction (LVEF
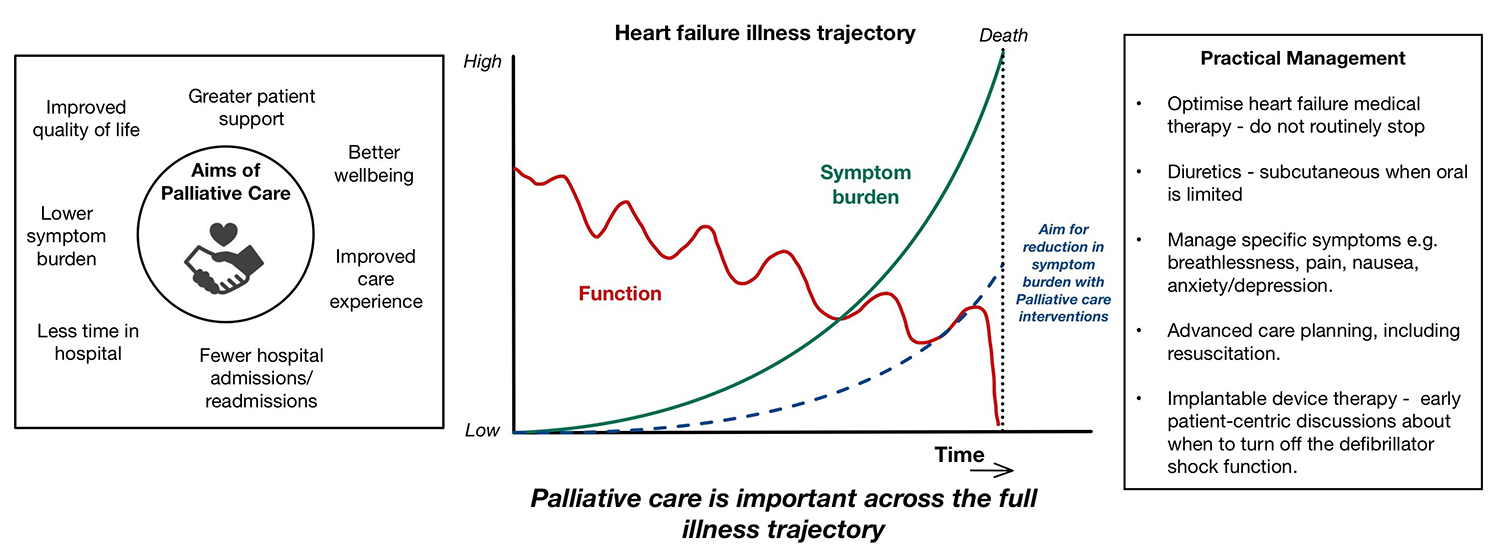
Chronic illnesses such as HF generally follow an erratic, unpredictable trajectory [4]. Patients experience frequent acute exacerbations of their disease, each having the potential to be fatal. Most exacerbations are overcome with appropriate management; however, this typically results in gradual deterioration in health over time.
Prognostication is not an exact science and is a challenge for any disease. The trajectory of HF is unpredictable and person-specific, influenced by a multitude of factors including age of diagnosis, ethnicity, gender, comorbidity and socioeconomic status. Prognosis in HF is holistic and multifactorial, taking into account clinical assessment; biomarkers, e.g., N-terminal pro B-type natriuretic peptide (NT-proBNP); renal function; heart rate; and pulmonary function tests [5, 6]. Significant interventions such as cardiac device implantation, mechanical assist devices and cardiac transplantation, if felt to be appropriate, may also dramatically influence prognosis in an advanced HF patient. Clinical risk scores may be used to predict patient outcomes during acute hospitalisations; however, they remain unvalidated in large-scale clinical trials to predict longer-term outcomes. Advances in technology, artificial intelligence and machine learning will most likely help shape the future of prognostication, which may provide a more personalised approach to management [7, 8].
Palliative care is an approach that focuses on providing relief from the symptoms and stress of a serious illness, aiming to improve the quality of life for both the patient and their family [9]. In contrast, end-of-life care has typically referred to the specific care provided during the final phase of a patient’s life, often involving hospice services that prioritise comfort and quality of life over curative treatments. Palliative care can be integrated at any stage of illness, including but not restricted to the end of life. For cancer patients, early palliative care consultations lead to better outcomes, such as reduced aggressive treatments and improved survival rates, which prompts consideration of whether this could be extrapolated to those with a HF diagnosis.
Diagnosing patients who are approaching the final stages of their illness is a complex process that requires careful consideration of clinical signs, patient history, and communication with both the patient and their family. This is not a simple process, requiring significant experience and empathy. Many clinicians often struggle to identify frail older patients as candidates for end-of-life diagnoses, which can delay the initiation of palliative care [10]. Physician-centric barriers in recognising when a patient is nearing the end of life, including timely and adequate decision making and communication deficits, can hinder timely discussions about palliative care options, which may directly impact patient end-of-life care [11]. This highlights the need for improved training and support for healthcare providers to enhance their confidence in discussing end-of-life issues and palliative care options with patients and families [12].
The General Medical Council [13] defines end-of-life care as:
‘people are approaching the end of life when they are likely to die within the
next 12 months’. This includes people whose death is imminent (expected within a
few hours or days) and those with advanced, progressive, incurable conditions;
general frailty and co-existing conditions that mean they are expected to die
within 12 months; existing conditions if they are at risk of dying from a sudden
acute deterioration in their condition; life threatening acute conditions caused
by sudden catastrophic events. NICE guidelines describe general indicators of
decline and increasing needs, which include repeated unplanned hospitalisations,
significant multi-morbidity, declining functional status, e.g., Rockwood scale,
poor response to treatment, progressive weight loss, reduced serum albumin (
Palliative care interventions in general are recognised for their significant impact on patient and caregiver outcomes, particularly in improving quality of life, reducing symptom burden, fewer hospital admissions, patient satisfaction, well-being and enhancing overall care experiences for patients with serious illnesses [14, 15]. Engaging HF patients with palliative care programs toward the end of life may also increase their chance of dying at home, rather than in an institution [16]. Furthermore, palliative care helps facilitate smoother transitions from hospital to home care, as it helps ensure that patients receive appropriate support and resources, thereby reducing the likelihood of readmissions [17]. This is particularly crucial for older adults and those with complex health needs.
HF is a progressive and increasingly debilitating condition; thus, offering a strategy to improve quality of life in these specific patient groups would be appealing. However, there is limited data in this specific group. Palliative care, by its very definition, is a multidisciplinary intervention which centres around the patient’s symptoms, psychosocial and spiritual requirements. This, in turn, makes defining a true ‘intervention’ one of the largest challenges to overcome when organising randomised clinical trials to formally test its efficacy. Gelfman et al. [18] recently highlighted several key systematic reviews and meta-analyses of the efficacy of palliative care for HF patients, showing that, despite relatively low inclusion numbers, palliative care in HF does positively impact patient quality of life. This data aligns with others, who show palliative care interventions across a range of diagnoses improve symptom burden, comfort, patient satisfaction, and reduce requirements for aggressive therapy as the patient approaches end of life [16, 19, 20].
Frequent hospitalisation is a hallmark of disease progression for HF patients. This is not only impactful on patient quality of life and morbidity trajectories, but has a significant impact on healthcare services and the cost of patient management. HF patients who uptake palliative care interventions, which provide home care support, had a 42% reduction in hospitalisations (odds ratio 0.58, 95% confidence interval [CI] 0.44–0.77) [21]. Furthermore, 30-day re-admission rates fell by 54% (odds ratio 0.46, 95% CI 0.41–0.52) for HF patients who engaged with the palliative care team, and for those who were discharged to care facilities with palliative care input, the 30-day readmission rates were reduced by 73% [22].
The implementation of palliative care early in the disease trajectory of HF may be beneficial and is associated with better management of symptoms such as pain and breathlessness [23, 24]. A proactive approach allows for ongoing assessments and adjustments in care and fosters a collaborative model that includes healthcare professionals from various disciplines [25]. This, in turn, enhances patient satisfaction and improves overall care experiences [26]. The holistic approach of palliative care, which includes addressing spiritual and psychosocial needs, further contributes to enhanced HF patient outcomes [27].
Barriers to the availability and utilisation of palliative care in the UK are multifaceted, and palliative care services are also not uniformly distributed, leading to significant inequities. The National Confidential Enquiry into Patient Outcome and Death (NCEPOD) [28] showed that only 12.5% of people diagnosed with HF were known to specialist palliative care services before death. The reviewers considered that 30% of final admissions could have been avoided if patients had received palliative care. Patients with HF are more likely to die in hospital than those diagnosed with cancer [29, 30]. This is influenced by a number of factors, including greater comorbidity burden, number of hospitalisations, age and socioeconomic status [30]. Furthermore, HF patients are less likely to receive hospice services and may not have a full understanding of their disease prognosis [29, 30].
There is a common misconception that palliative care is only relevant in the last few weeks or days of life. This misperception in itself acts as a significant barrier to access to palliative care for patients with HF and could potentially hinder its integration into standard treatment protocols. Educational initiatives aimed at healthcare professionals are essential to dispel these myths and promote a better understanding of the role of palliative care in chronic illness management [31].
Patients with greater social deprivation are more reliant on hospital-based care at the end of life and may lack access to community-based palliative care options [32]. Black and ethnic minority communities may further be impacted by different cultural preferences regarding end-of-life care [33]. Women are subject to gender inequality and inequity when it comes to their cardiovascular health [34]. Women are known to be underrepresented in most clinical trials for cardiovascular medicine. However, women generally live longer and utilise healthcare services more than men, which may be important when considering palliative care requirements. Studies focusing on gender disparities in end-of-life and palliative care are few and are limited by sample sizes; however, trends suggest that there remain significant gender biases in the uptake of such services by women [35].
The number of individuals in Europe affected by HF is 10 million and rising [36]; therefore, any system changes for delivering palliative care interventions need to be both effective and scalable. Clear national and international guidance has been developed to provide the best palliative care input for those patients with worsening HF. The European Society of Cardiology guidelines for palliative care emphasise the integration of palliative care into the management of patients with HF, particularly as the disease progresses to advanced stages. These guidelines advocate for a multidisciplinary approach, recognising the complex needs of patients and the importance of symptom management, shared decision-making, and caregiver support throughout the course of the illness [2, 26]. National Institute for Health and Care Excellence (NICE) guidance similarly suggests utilisation of the multi-disciplinary team including community nurses, HF specialist nurses, a palliative care team, occupational therapists, physiotherapists, dietetics, out-of-hours services, and hospice care to help coordinate an appropriate care plan [3]. A regularly reviewed advanced care plan is important to address medical, psychosocial and spiritual care needs, which may be tailored to the patient’s specific preferences. The National Health Service (NHS) England have recently published a document outlining a revised framework for integrated care systems to address palliative care needs in HF populations [37]. A responsive and integrated service with a needs-based approach for all patients should be the goal. Such a guidance needs to be taken together with appropriate education and training in palliative care for all healthcare professionals involved in HF patient care in order to maximise the benefit to our patients [26].
HF therapies remain a crucial component of symptom management and improvements in quality of life in all patients with HF. This remains important in advanced HF and at a time when palliation rather than life prolongation may have become the main goal [2]. Therapies should not be routinely stopped when patients enter a phase of their disease where palliative care is being considered, or their prognosis is felt to be limited, as they still have potential to reduce symptoms and improve quality of life. This is perhaps at odds with the approach to rationalisation of medications, which is common in managing palliation in other diseases and frailty.
Diuretics help maintain euvolaemia in all types of HF, facilitating better control of breathlessness and oedema, thus should not be withdrawn in the absence of a clear indication [2]. If oral intake reduces or the patient is felt to be fluid-depleted for an alternative reason, then a dose reduction or interruption may be considered. In addition, the use of parenteral diuretics may be considered if oral therapy is not possible or effective. There is some evidence supporting the use of subcutaneous furosemide (unlicensed) as an effective alternative to intravenous therapies. It has been shown to improve symptoms and is associated with reductions in oedema. This can be effectively delivered in the community using a 24-hour continuous subcutaneous infusion using techniques and equipment familiar to palliative care and community teams [38].
In the setting of HFrEF, renin angiotensin aldosterone inhibition (RAAS-i) drugs, which include angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, angiotensin-neprilysin inhibitors (e.g., sacubitril/valsartan), and mineralocorticoid receptor antagonists, may all be important for the reduction of symptoms and hospitalisations [39]. These drugs may exacerbate hypotension, and changes in renal function may be attributed to them. There is no specific value for blood pressure which necessitates dose reduction, and decisions should be weighed against both the presence and quality of life impact of any hypotension. All decisions regarding dose reductions or drug cessation need to be based on an individualised risk-benefit analysis. Other drugs that reduce blood pressure with no HF benefits should be discontinued ahead of RAAS-i. Similarly, deterioration in renal function is common as HF progresses. There is no absolute value for discontinuation of these medications, particularly if the patient has no uraemic symptoms, and a rise in creatinine in isolation should not prompt cessation. HF medications (RAAS-i and sodium glucose transporter-2 [SGLT-2]) are all shown to slow renal decline in the setting of chronic kidney disease over the longer term, which further cautions against ceasing these medications in response to changes in creatinine. Beta blockers have also been shown to reduce mortality and HF hospitalisations in HFrEF and may improve quality of life [39, 40]. These medications may need to be reduced in the setting of symptomatic hypotension, but in the setting of atrial arrhythmias or angina, the consequences of this and the use of alternate medications (such as digoxin or blood pressure neutral anti-anginals) should be considered. Current evidence also supports the use of SGLT-2 inhibitors [39], which reduce mortality and HF hospitalisations alongside improving quality of life in both HFpEF and HFrEF [41]. These drugs should be continued where possible. Any therapies causing significant side effects and those with indications that are no longer relevant, such as statins for long-term prevention, may be considered for withdrawal [42].
Breathlessness is a common symptom in advanced HF. The mainstay of treatment should be optimal medical therapy as outlined above. However, if these medications are ineffective or limited by adverse effects, additional therapies may be helpful. Low-dose opiates have been shown to have some benefit in reducing dyspnoea in HF, although not all studies have been positive [43, 44]. It is suggested that low-dose morphine (10 mg) should be used for this indication with weekly increases in doses as required [45]. In patients with renal dysfunction, oxycodone should be considered as an alternative to minimise adverse effects. A Cochrane review of benzodiazepine use in non-cancer palliative care found they may help relieve breathlessness but should be second-line agents [46]. Evidence suggests that supplemental oxygen should be reserved for patients who have breathlessness in the presence of demonstrable hypoxia [47]. Cardiac rehabilitation and exercise training may improve exercise capacity and reduce breathlessness, and are often underused in this patient group [48]. Simple measures such as breathing exercises, use of a fan and anxiety management may also improve subjective sensations of breathlessness [24].
Pain is perhaps a surprisingly common symptom in advanced HF, with a prevalence increasing with the New York Heart Association classification and rates of 89% reported in class IV HF [49]. Research has shown that medical comorbidities are associated with pain in advanced HF, including degenerative joint disease, chronic back pain and depression, suggesting this is a multifactorial problem [23, 50]. However, rates of opiate use remain much lower in advanced HF compared with cancer (22% vs. 50%) [51]. The reasons for this are not well understood. Pain should be specifically assessed in this patient group, and in the absence of any specific clinical evidence, the general principles of pain management should be applied.
Nausea, anorexia and weight loss are common in advanced HF, with almost half of patients reporting nausea during their last 6 months of life [52]. Causes include gut and hepatic congestion, renal dysfunction and medications. If symptoms persist despite HF management, in the absence of specific guidance in this patient group, the general principles of antiemetic use should be applied.
Depression is very common in advanced HF patients, with approximately 20% of patients meeting the criteria for a major depressive episode and greater numbers reporting depressive symptoms [53]. There is limited data to guide therapy in this patient group. The Sertraline Against Depression and Heart Disease in Chronic Heart Failure (SADHART-CHF) trial found no difference in depression or cardiovascular endpoints with sertraline treatment for 12 weeks versus placebo [54]. The MOOD-HF trial comparing escitalopram with placebo was also negative [55]. Cognitive behavioural therapy has been evaluated in a randomised trial, which showed benefits in the Beck depression inventory score [56]. Despite the unclear efficacy, it is common for patients to receive treatment for depression in a similar way to the general population. Patients with HF also experience significant physical, psychological and spiritual distress [57]. Approaches to the spiritual issues surrounding patients’ and caregivers’ losses and sources of grief should be similar to those in other advanced illnesses.
Implantable cardiac defibrillators (ICDs) are used in patients who are felt to be at high risk of sudden cardiac death from an arrhythmic cause. An ICD can deliver either rapid burst pacing or a shock with the aim of terminating a ventricular arrhythmia. Most ICDs also function as a pacemaker to either prevent slow heart rates or as part of cardiac resynchronisation therapy (CRT, aka biventricular pacing). CRT devices synchronise contraction of the left and right ventricles and thereby reduce symptoms in some people with HF. A CRT device may function solely as a pacemaker (CRT-P) or also function as an ICD; these are referred to as cardiac resynchronization therapy with defibrillator (CRT-D) devices. The pacemaker and ICD functions of each device are programmable independently of each other.
A patient with an ICD may clinically deteriorate either with worsening cardiac failure, another non-cardiac condition, or general frailty and develop a limited prognosis. An ICD does not improve symptoms and can create an additional burden on patients related to therapies and procedures. At this stage, the ability of the device to increase overall survival becomes more limited and the risk-benefit of an active device shifts [58]. Therefore, to ensure that the person receives high-quality end-of-life care, they should have the opportunity to consider and discuss the option to deactivate the shock function of their ICD. If the ICD shock therapy is not deactivated, there is an increased risk that, as a person reaches the last days of life, the ICD may deliver multiple, painful shocks, which are distressing [59, 60, 61, 62]. There is also a risk that the device may delay the person’s natural death, which the person would not have chosen if they had been given the opportunity to discuss discontinuation. Whilst anti-tachycardia pacing may be used to treat some episodes of ventricular arrhythmia without major distress, it is not possible to programme this therapy alone without the addition of some shock treatments, which could result in distressing therapy.
The potential for future ICD shock therapy deactivation should be discussed early, including before the device is implanted, as part of a shared decision-making process [62]. All members of the multidisciplinary team should be encouraged to identify patients with an ICD, monitor for signs of clinical deterioration or worsening prognosis, and flag the potential need for device deactivation discussions. Examples of flags that may prompt discussion include frequent therapy delivery, repeated hospitalisation for HF, refractory symptoms, deteriorating quality of life, consideration of a do-not-attempt-resuscitation (DNAR) order or if the patient develops a new condition with a poor prognosis.
Clinicians should be aware of where to obtain and how to use a specific cardiac device magnet to temporarily deactivate ICD shock therapy in an emergency. It is important to note that some manufacturers’ ICDs will reactivate after 8 hours if the magnet is not removed and reapplied periodically. Definitive device deactivation by an appropriately trained individual (this requires a face-to-face interaction) is preferable, and teams should know how to access this locally. Early identification of patients for whom ICD deactivation is appropriate and clear lines of communication between specialities are crucial to facilitate this. The British Heart Rhythm Society [59] has published a practical guide for discontinuation of ICD therapies, which contains useful resources for clinical teams.
Pacemaker and CRT functions typically aim to improve quality of life and reduce symptoms. For this reason, it is unusual to decide to deactivate these functions. Some patients are dependent on their pacing function, and if pacing were deactivated, then this would likely shorten their lives or potentially lead to death immediately. However, if a patient with capacity understands these potential consequences and does wish to deactivate these functions, as with other invasive procedures, withdrawal of this life-sustaining therapy may be appropriate.
Once it has been determined that a patient who is deteriorating despite standard HF medication is not a candidate for cardiac transplantation or mechanical circulatory support, it is important to discuss their likely transition to end-of-life care. It is important to understand the patient’s wishes to ensure that medical decision-making aligns with the patient’s values. A patient with HF is likely to deteriorate over time, but the trajectory can be unpredictable. It is therefore crucial to revisit these decisions frequently to ensure that the care is patient-centred and appropriate at each stage. Evaluation of what is most important to the patient, e.g., prolongation of life, avoidance of admission to hospital, minimising tablets, will help clinicians to work with the patient to provide the most appropriate care.
In advanced HF discussions regarding the appropriateness of cardiopulmonary resuscitation (CPR) should take place early and revisited as the disease progresses. Cardiac arrest may occur in all stages of HF and is associated with poor outcomes. Even when a patient suffers a witnessed, in-hospital arrest, the chance of survival to discharge after CPR is very low. Moderate-to-severe HF is one of the strongest independent predictors of in-hospital mortality after CPR [63]. In addition, consequent neurological disability due to anoxic damage alongside other functional organ impairments may limit quality of life in survivors. Patients (and their advocates, if appropriate) should be involved in discussions about resuscitation as the disease progresses to ensure that their priorities regarding length and quality of life are respected.
The recently passed vote in the UK parliament in favour of proposals to legalise assisted dying in England, if implemented, may have significant effects on the end-of-life care of HF patients. Difficulties in estimating prognosis in HF may prove challenging for patients who may wish to go down this path.
Advanced HF is associated with high rates of morbidity and mortality, and patients have significant palliative care needs. More studies are needed to evaluate the efficacy of specific interventions in this population and refine the tools available to support our patients. Despite clear national and international guidance that patients with worsening HF should receive palliative care input, there remains significant variation in access to palliative care services for advanced HF patients. The number of HF patients worldwide is increasing; thus, any system changes for delivering palliative care interventions need to be both effective and scalable. Palliative care should be seen to complement cardiac care, and not as an alternative to it. A responsive and integrated service with a needs-based approach for all patients should be the goal. Until this exists, every healthcare professional can still work to identify and address the unmet need in the advanced HF patients we care for. This review highlights the steps clinicians may take to optimise management of patients with advanced HF as part of best supportive care for our patients in order to deliver holistic palliative care in this patient population.
All the data of this study are included in this article.
ML and HT conceived the idea and undertook final revisions. ML and HT wrote the first draft. ML undertook multiple revisions. Both authors contributed to the important editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
