1 Department of Gastroenterology, University Hospital “Tsaritsa Yoanna – ISUL”, Medical University of Sofia, 1527 Sofia, Bulgaria
2 Department of Diagnostic Imaging, University Hospital “Tsaritsa Yoanna – ISUL”, Medical University of Sofia, 1527 Sofia, Bulgaria
Abstract
Gastrointestinal duplication cysts are rare congenital anomalies that are often discovered incidentally. Although asymptomatic cases may not require immediate intervention, optimal management remains controversial, particularly due to concerns regarding malignant potential. This report aims to highlight the diagnostic role of contrast-enhanced endoscopic ultrasound (CE-EUS) and discuss management strategies for asymptomatic gastrointestinal duplication cysts.
A 54-year-old woman was found to have an asymptomatic subepithelial lesion during a routine oesophagogastroduodenoscopy (OGD). Further evaluation with endoscopic ultrasound (EUS) demonstrated a characteristic “gut signature sign”. Subsequent contrast-enhanced EUS revealed enhancement limited to the cyst wall, supporting the diagnosis of a duplication cyst. The patient remained asymptomatic throughout the evaluation period.
Over two years of follow-up, serial EUS examinations demonstrated no significant changes in the size or characteristics of the lesion. No clinical symptoms developed, and no features suggestive of malignant transformation were identified.
This case illustrates the utility of CE-EUS as a safe and effective diagnostic alternative to EUS-guided fine needle biopsy for gastrointestinal duplication cysts. The findings support a conservative management approach with careful EUS surveillance for asymptomatic lesions, reserving surgical intervention for symptomatic cases. A tailored strategy based on clinical presentation and lesion characteristics may optimize patient outcomes.
Keywords
- duplication cysts
- EUS
- CE-EUS
- case report
Duplication cysts are rare congenital anomalies of the gastrointestinal tract formed during embryonic development. They include foregut duplication cysts, small bowel duplication cysts, and large bowel duplication cysts as discussed by Diehl et al. [1]. The foregut stretches from the oral cavity to the proximal duodenum. Duplication cysts are most frequently found in the ileum, esophagus, and colon. They can be located either within the wall of the gastrointestinal tract or outside of it. Gastric duplication cyst account for 4% to 9% of gastrointestinal cysts as reported by Wang et al. [2]. To date, fewer than ten cases of gastric cancer originating from gastric duplication cysts have been documented. Given the potential for cancer to develop, some researchers advocate for surgical intervention in asymptomatic patients. Conversely, other experts support a conservative management approach, citing that instances of malignant transformation in these lesions are largely anecdotal as discussed by Gandhi et al. [3]. Magnetic resonance imaging (MRI) and endoscopic ultrasound (EUS) are commonly utilized for assessing and diagnosing duplication cysts. EUS is the preferred diagnostic method for examining these cysts because it can differentiate between solid and cystic lesions. EUS can also determine the cyst’s location to nearby tissues. Duplication cysts typically appear on EUS as anechoic, homogeneous lesions with smooth edges, originating from the submucosal layer or located outside the gut wall, although they may also present a hypoechoic echo pattern as reported by Liu and Adler [4]. Contrast-enhanced endoscopic ultrasound (CE-EUS) is an advanced imaging technique that combines traditional EUS with the use of contrast agents to enhance the visualization of structures within the gastrointestinal (GI) tract, which is effective in distinguishing submucosal tumors. EUS-guided fine needle aspiration (EUS-FNA) of duplication cysts should be approached with caution, especially if the endosonographer suspects that the lesion is a duplication cyst due to concerns about the potential adverse events following the procedure.
We present a case of a patient with an asymptomatic small duplication cyst of the stomach. This case underscores the utility of CE-EUS as a safer alternative to EUS-guided fine needle biopsy (EUS-FNB) for the diagnosis and follow-up of patients with duplication cysts of the upper GI tract, particularly when the lesion remains asymptomatic and stable, with surgical intervention reserved for symptomatic cases. The findings contribute to the literature by showing that CE-EUS is a perfect non-invasive additional tool for the evaluation of duplication cysts of the GI tract especially when the diagnosis is uncertain and should be preferred to EUS-FNB due to safety reasons.
A 54-year-old woman was referred to our clinic to evaluate a suspected subepithelial lesion at the oesophagogastric junction, identified during a routine oesophagogastroduodenoscopy (OGD) performed due to reflux symptoms. The examination indicated Grade B gastroesophageal reflux disease (GORD), and the patient was initiated on proton pump inhibitor (PPI) therapy, to which she responded well. Histological examination revealed that she was negative for Helicobacter pylori.
Throughout her hospital stay in our clinic, the patient exhibited normal laboratory results and no symptoms, without any other concomitant diseases or significant family history. From the physical examination (PE) the patient was slightly overweight with a body mass index (BMI)—29 without other pathological findings. The patient was scheduled for an EUS examination, which is the preferred modality for evaluating subepithelial lesions of the upper GI tract. During the EUS examination, an anechoic subepithelial lesion of the stomach was noted just below the oesophagogastric junction, corroborating the findings from the OGD. The lesion was 13.5 mm in size and displayed a typical “gut signature sign”, which is highly suggestive of a duplication cyst (Fig. 1). Given that other subepithelial lesions can appear intensely hypoechoic as lymphoma, leiomyomas, gastrointestinal stromal tumors (GIST), etc. and can mimic anechoic lesions, a CE-EUS was performed revealing enhancement confined to the wall of the lesion (Fig. 2) and confirming the diagnosis of a duplication cyst.
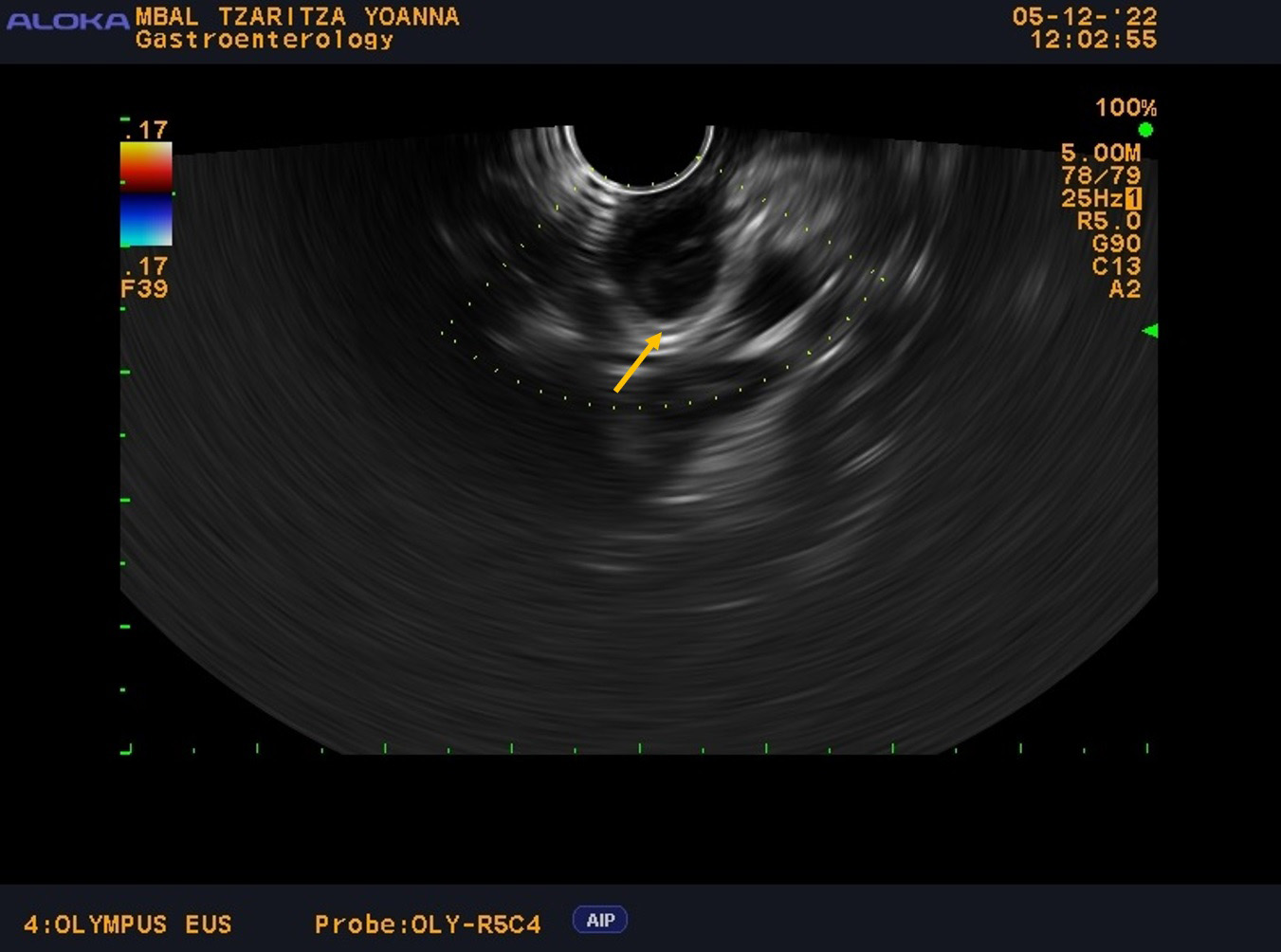 Fig. 1.
Fig. 1.
EUS examination with anechoic subepithelial lesion 13.5 mm in size at OGD and typical “gut signature sign”. Endoscopic ultrasound with the linear probe with anechoic subepithelial lesion 13.5 mm in size originating from the submucosal layer of the wall with “gut signature sign” highly suspicious for duplication cyst (the arrow). EUS, endoscopic ultrasound; OGD, oesophagogastroduodenoscopy.
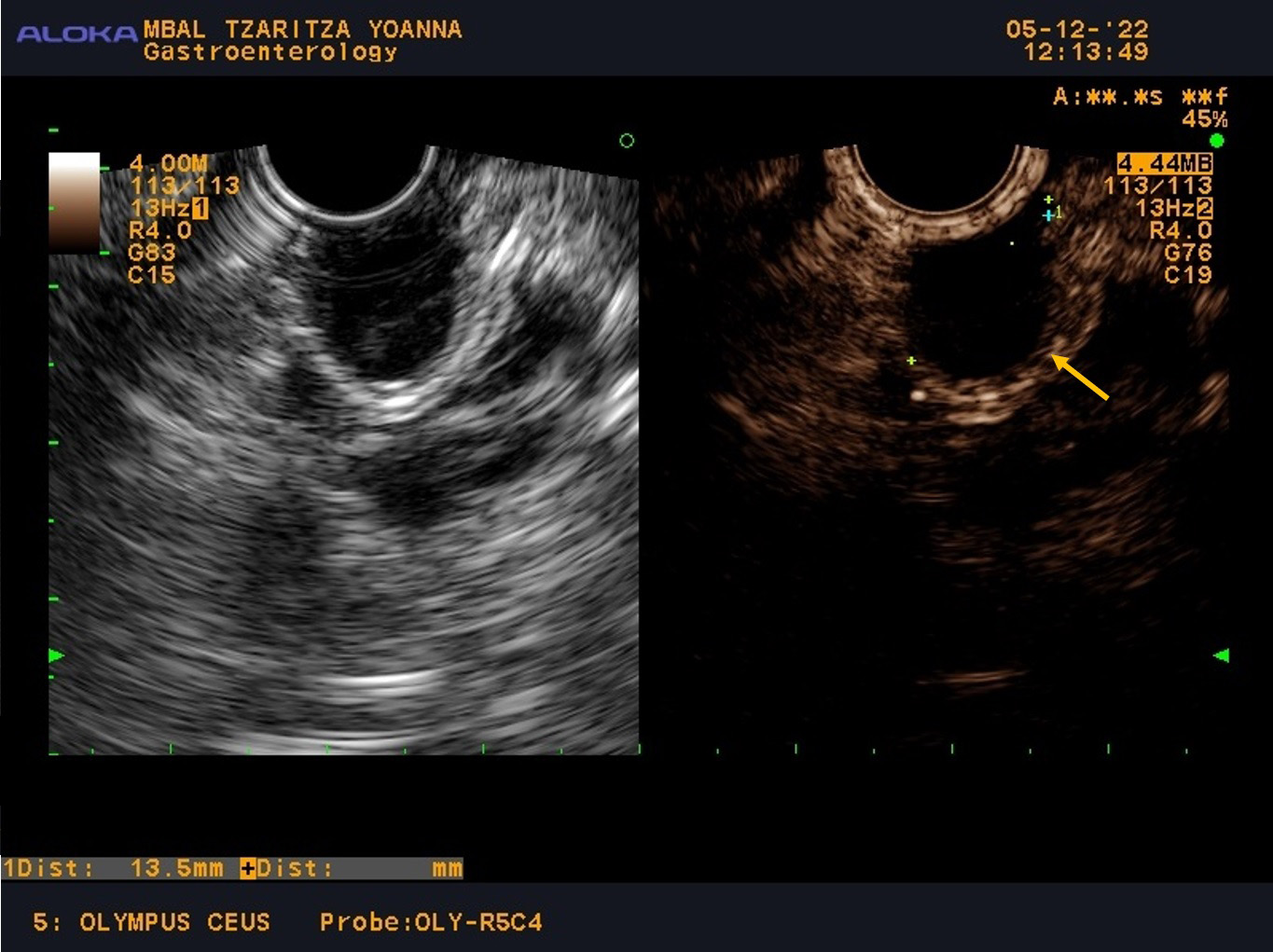 Fig. 2.
Fig. 2.
CE-EUS with contrast-enhancing only of the wall of the cyst and size 13.5 mm. CE-EUS was performed with 4.8 mL SonoVue—contrast-enhancing only of the wall of the cyst (the arrow) and size of the lesion—13.5 mm. CE-EUS, contrast-enhanced endoscopic ultrasound.
Due to the small size of the cyst and the absence of symptoms, the patient was scheduled for follow-up with EUS examinations every six months during the first year, with the interval subsequently extended to one year. Over the two years of follow-up (Fig. 3), there was no change in size (the cyst remains 13.5 mm) or any concerning features of the cyst, and the patient is slated for her next EUS examination after one year.
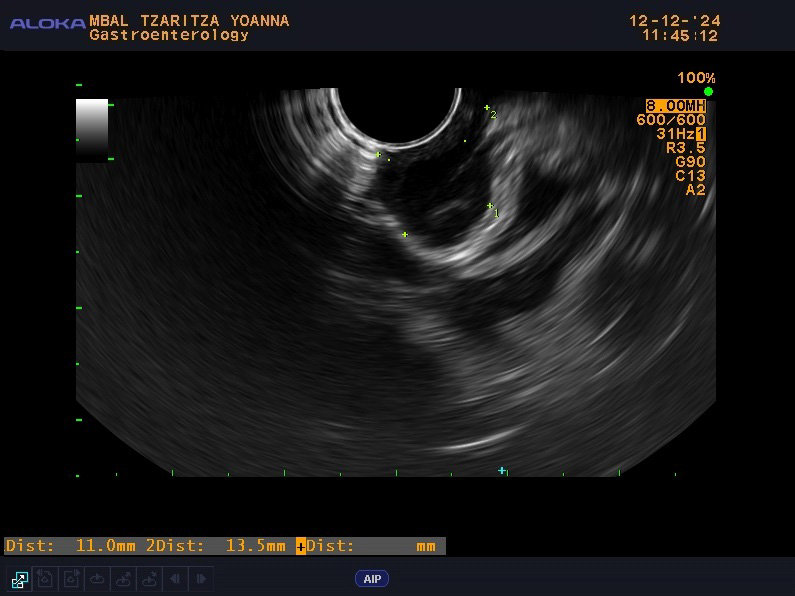 Fig. 3.
Fig. 3.
Control EUS examination during follow up.
The CARE checklist has been provided as Supplementary Material.
Duplication cysts are rare congenital anomalies of the gastrointestinal tract. They are cystic structures lined by gastrointestinal epithelium and have their own muscular walls. These cysts are often located along the gastrointestinal tract, commonly found in the ileum, colon, and esophagus. They can vary in size and may be connected to the bowel or exist as isolated entities. On the other hand, a gastrointestinal cyst is a broader term that refers to any cystic lesion found within the gastrointestinal tract. These cysts can arise from various causes, including congenital anomalies, infections, or neoplastic processes. Gastrointestinal cysts can be classified into several types, including enteric, mesenteric, ovarian, and neoplastic cysts.
Gastric duplication cysts are typically solitary and account for approximately 4% to 9% of all gastrointestinal cysts, as noted by Wang et al. [2]. They are formed during embryonic development and the stomach duplication cysts originate from the foregut. While some patients with stomach duplication cysts may remain asymptomatic, others can experience symptoms such as vomiting, weight loss, epigastric pain, and outlet obstruction as reported by Napolitano et al. [5]. According to Wildi et al. [6], EUS is one of the most effective imaging modalities for assessing subepithelial lesions in the upper GI tract. EUS is a minimally invasive procedure that uses high-frequency sound waves to create images of the digestive tract and surrounding tissues. CE-EUS is an advanced imaging technique that combines traditional EUS with contrast agents for better visualization. In CE-EUS, microbubble contrast agents (in Europe SonoVue—contrast agent that contains tiny bubbles filled with a gas called sulphur hexafluoride, and due to the high mechanical index an entire 4.8 mL vial is required for one procedure) are injected intravenously. These agents enhance the echogenicity of blood vessels and tissues. CE-EUS improves the delineation of lesions, blood flow, and vascular structures. It helps in real-time to differentiate between benign and malignant lesions based on their vascular characteristics. CE-EUS is less invasive compared to other imaging modalities like computed tomography (CT) or MRI, which often require contrast agents that can have more side effects. One of the main limitations of CE-EUS is that it requires specialized equipment and training and although rare, there is a risk of allergic reactions to the contrast agents.
A hallmark EUS finding for the diagnosis of duplication cyst is the presence of the so-called “gut signature sign”, which reflects a double layer of smooth muscle in the wall, as described by Gandhi et al. [3]. The inner layer consists of hyperechoic mucosa, while the outer layer comprises hypoechoic muscle. The bi-layered cystic appearance strongly indicates intestinal duplication as reported by Strouse [7]. However, inflammation can obscure these layers, reducing the clarity of the diagnosis. While the gut signature sign can be a helpful indicator a limitation should be noted that not all gastrointestinal duplication cysts will present with this sign and it should always be interpreted in the context of a comprehensive clinical evaluation. Establishing a definitive diagnosis for a patient with subepithelial lesions can be challenging even during an EUS examination as reported by Varanese et al. [8]. In the case of a duplication cyst, the internal characteristics of the cyst-containing fluid can vary significantly based on factors such as the extent of hemorrhage, protein content, infection, and other fluid components, which can complicate the diagnostic process as noted by Adachi et al. [9]. EUS-FNB is not routinely employed in such cases due to the associated risks of complications and infection. A case in which a patient experienced an infection after undergoing EUS-FNA on an esophageal hyperechoic duplication cyst, which subsequently resulted in the acute development of a pseudoaneurysm of the thoracic aorta is reported by Cevasco et al. [10]. Similarly, an instance of an infected hypoechoic esophageal duplication cyst following EUS-FNA performed on a paraesophageal mass raised suspicions of persistent non-Hodgkin’s lymphoma, highlighting how esophageal duplication cysts can mimic serious conditions like malignant adenopathy as reported by Trojan et al. [11]. While some authors recommend the use of antibiotics to mitigate the risk of infection, a patient who developed an infected esophageal duplication cyst after EUS-FNA despite receiving prophylactic antibiotics before and after the procedure was reported by Diehl et al. [1]. This patient ultimately required a thoracotomy for cyst resection. Overall, EUS-guided FNA of duplication cysts should be approached with caution, especially if the endosonographer suspects that the lesion is a duplication cyst. In certain situations, EUS-FNA may be necessary to characterize the lesion, but if a duplication cyst is identified, appropriate antibiotic prophylaxis should be implemented.
On the other hand, CE-EUS allowed for further assessment of subepithelial lesions. The contrast agent is safe for use in patients with renal impairment and has not been associated with serious side effects. CE-EUS in the GI tract has been successful in differentiating submucosal tumors, GIST, neuroendocrine tumors (NET), leiomyomas, etc. In this case, CE-EUS showed no blood flow within the lesion, and no abnormal nodules were seen in the lumen, appearing as an anechoic area. Thus, a diagnosis of a benign esophageal duplication cyst was made. One limitation of CE-EUS is the lack of studies in the literature that evaluate true positive and false positive cases. Most available articles consist of clinical case reports or series that primarily assess the benefits of this method in the diagnostic process. CE-EUS was utilized in a clinical trial and proved effective for both the diagnosis and differentiation of esophageal duplication cysts as reported by Adachi et al. [9]. The authors commented that future studies should explore the utility of CE-EUS across various clinical scenarios to further assess its effectiveness due to a range of imaging characteristics of the duplication cyst.
The debate persists regarding management strategies for patients with duplication cysts of the stomach, especially concerning potential malignant transformation. Surgical resection is often the preferred method for symptomatic patients to alleviate symptoms. The approach for asymptomatic patients is more contentious; while some experts recommend resection due to the potential for malignant transformation, others support a strategy of observation as discussed by Adachi et al. [9]. Since there have been reports of stable duplication cysts under EUS surveillance, this may serve as a viable outpatient follow-up method, with surgical resection considered if symptoms arise. Ultimately, the debate over surgical versus nonsurgical management of asymptomatic duplication cysts is likely to persist until we gain greater insight into the time course and risk factors related to malignant degeneration. In the presented case the cyst remained stable over two years of follow-up with no significant changes or symptoms and a surgical treatment was not needed. As a disadvantage, it can be noted that there is currently no clear algorithm in the literature for the follow-up interval of such patients which can lead to economic disadvantage and additional stress for the patients.
This clinical case contributes to the literature by highlighting the utility of CE-EUS as a safer alternative to EUS-FNB for the diagnosis and the role of EUS for follow-up of patients with duplication cysts of the upper GI tract, particularly when the lesion remains asymptomatic and stable.
In summary, this case report illustrates the effective use of CE-EUS as a diagnostic tool for asymptomatic gastrointestinal duplication cysts. The stable nature of the cyst over a two-year follow-up period reinforces the notion that careful surveillance may be a viable management strategy for asymptomatic cases. This report emphasizes the need for a tailored approach in the management of duplication cysts, balancing the risks of surgical intervention against the potential for malignant transformation. Further research is warranted to enhance our understanding of the long-term outcomes associated with different management strategies for these rare congenital anomalies.
All data included in this study are available from the corresponding author upon reasonable request.
SC and GJ made substantial contributions to the conception and design of the article. TA, PG and BG made substantial contributions to the acquisition of data. TA wrote the first draft. PG and BG made the critical review before the publication of the article. All authors contributed to revising the manuscript critically for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
EUS was performed at the University Hospital “Tsaritsa Yoanna – ISUL”, a tertiary center for performing EUS, CE-EUS, and EUS-FNB, approved by the institution’s ethics committee (approval number: 11/19.04.2021) and conformed to the code of ethics stated in the Declaration of Helsinki. The patient provided written informed consent.
Not applicable.
This research is supported by the Bulgarian Ministry of Education and Science under the National Program “Young Scientist and Postdoctoral Students - 2”.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/BJHM50725.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



