1 Department of Thoracic Surgery, Xi’an International Medical Center Hospital, 710000 Xi’an, Shaanxi, China
2 Department of General Medicine, Xi’an International Medical Center Hospital, 710000 Xi’an, Shaanxi, China
3 Department of Nephrology, The Second Affiliated Hospital of Xi’an Jiaotong University, 710006 Xi’an, Shaanxi, China
Abstract
Non-small cell lung cancer (NSCLC) accounts for approximately 80–85% of all lung cancers, and lymph node metastasis is a key determinant of patient prognosis, often leading to poorer clinical outcomes. Although immune checkpoint inhibitors (ICIs) have transformed the therapeutic landscape of advanced NSCLC, their specific efficacy in patients with lymph node metastases remains unclear. This study aimed to evaluate the efficacy and safety of ICI monotherapy versus chemotherapy in previously treated, unresectable NSCLC patients with lymph node metastasis.
A retrospective analysis was performed on patients with unresectable, previously treated NSCLC with lymph node metastasis who received treatment at Xi’an International Medical Center Hospital between April 2019 and October 2022. Following 1:1 propensity score matching (PSM), 112 patients were included in the final analysis, comprising 56 in the ICI group (treated with pembrolizumab) and 56 in the chemotherapy group (treated with docetaxel). The primary endpoint was objective response rate (ORR), while secondary endpoints included disease control rate (DCR), overall survival (OS), and safety.
The ICI group demonstrated a significantly higher ORR compared with the chemotherapy group (50.0% vs. 26.8%, p = 0.012). No significant difference in DCR was observed between the groups (p = 0.057). Kaplan-Meier analysis revealed significantly longer OS in the ICI group (p = 0.009). Multivariate analysis indicated a high risk of death in the chemotherapy group compared to the ICI group (hazard ratio [HR] = 1.796; 95% confidence interval [CI]: 1.112–2.900; p = 0.017). In terms of safety, leukopenia occurred more frequently in the chemotherapy group (p = 0.039), while immune-related adverse events (irAEs), including hypothyroidism (p = 0.027) and rash (p = 0.008), were more common in the ICI group.
In previously treated patients with unresectable NSCLC and lymph node metastasis, real-world evidence from this study suggests that pembrolizumab monotherapy offers superior efficacy and survival benefits compared with docetaxel chemotherapy. Moreover, the two therapies display distinct safety profiles, with ICI therapy associated with fewer severe hematologic toxicities.
Keywords
- immune checkpoint inhibitors
- pembrolizumab
- non-small cell lung cancer
- lymph node metastasis
- survival
Lung cancer remains the leading cause of cancer-related deaths globally, with non-small cell lung cancer (NSCLC) representing the majority of cases, accounting for approximately 80–85% of all lung cancer diagnoses [1, 2]. Prognosis and treatment strategies for NSCLC are largely determined by disease stage at diagnosis, with lymph node metastasis serving as a critical prognostic factor [3]. As the predominant route of NSCLC dissemination, lymphatic spread complicates both diagnosis and therapeutic decision-making, often resulting in poorer clinical outcomes.
The advent of immune checkpoint inhibitors (ICIs), particularly those targeting the Programmed Cell Death Protein 1/Programmed Cell Death Ligand 1 (PD-1/PD-L1) pathway, has fundamentally transformed the therapeutic landscape for advanced NSCLC [4, 5]. Landmark clinical trials have established the superiority of ICI monotherapy over conventional chemotherapy, especially in selected populations such as patients with high PD-L1 expression, where significant survival benefits have been demonstrated [6].
However, the specific efficacy of ICIs in patients with lymph node metastases remains unclear. Although pivotal trials have included individuals with metastatic disease, outcomes stratified by the extent of nodal involvement (N1–N3) have not been consistently reported. This constitutes a significant knowledge gap, as the tumour immune microenvironment within lymph nodes may differ immunologically and potentially influence responsiveness to immunotherapy [7, 8].
Given that NSCLC patients with lymph node metastasis represent a complex and prognostically heterogeneous subgroup, it is clinically relevant to determine whether ICI monotherapy provides sustained benefit in this population. The current lack of targeted evidence has created uncertainty regarding the optimal application of ICI use in these patients.
Therefore, this study aimed to address this clinical question by directly comparing the efficacy and safety of ICI monotherapy with standard chemotherapy in a real-world cohort of previously treated, unresectable NSCLC patients with lymph node metastasis following failure of platinum-based therapy. By evaluating objective response rate (ORR), disease control rate (DCR), and survival outcomes, this study aimed to clarify the therapeutic value of ICIs and support evidence-based treatment decisions for this challenging patient subgroup.
This study retrospectively analysed data from a cohort of patients diagnosed with non-small cell lung cancer (NSCLC) with lymph node metastasis at Xi’an International Medical Center Hospital between April 2019 and October 2022. The study protocol was approved by the Ethics Committee of Xi’an International Medical Center Hospital (Approval No. 2024034) and conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all patients or their legal guardians.
Patient selection followed a structured process. Initially, 155 patients who met the preliminary diagnostic criteria were identified from hospital records, including 70 patients who received ICI and 85 patients treated with chemotherapy. Propensity score matching (PSM) was subsequently performed in a 1:1 ratio, yielding a final analysis cohort of 112 patients, with 56 assigned to each group (ICI and chemotherapy).
Patients were eligible for the study if they met the following criteria:
age
Exclusion criteria included: prior surgical treatment for NSCLC; concurrent malignancies or severe interstitial lung disease; presence of autoimmune diseases or requirement for corticosteroid or immunosuppressive therapy; and any other contraindications to immunotherapy or chemotherapy.
The final matched cohort was divided into two treatment groups. The ICI group (n = 56) received pembrolizumab monotherapy (JS20180031, MSD Ireland, Berlin, Germany) at a fixed dose of 200 mg administered intravenously every 21 days. The chemotherapy group (n = 56) received docetaxel monotherapy (H20020543, Hengrui Co., Ltd., Nanjing, China) at 75 mg/m2, administered every 21 days.
Clinical variables, including age, gender, histological subtype, ECOG performance status, PD-L1 expression level, smoking history, tumour-node-metastasis (TNM) staging (N classification), epidermal growth factor receptor (EGFR) and anaplastic lymphoma kinase (ALK) mutation status, and the number of prior systemic therapy lines, were retrospectively retrieved from the electronic medical records of the hospital.
The primary efficacy endpoint was the objective response rate (ORR). Secondary endpoints included the disease control rate (DCR) and survival outcomes.
Efficacy assessment: tumour response was radiologically evaluated every two treatment cycles (approximately every 6 weeks) according to RECIST version 1.1 [10]. Tumour responses were classified as complete response (CR), partial response (PR), stable disease (SD), or progressive disease (PD). ORR was defined as the proportion of patients achieving CR or PR:
DCR was defined as the proportion of patients achieving CR, PR, or SD:
Survival follow-up: from treatment initiation, patient survival status was evaluated every three months through outpatient visits or telephone interviews. Follow-up continued until death or the last recorded contact.
Safety assessment: adverse events (AEs) were monitored from treatment initiation until 30 days after the final dose. Collected safety data included common AEs (e.g., anaemia, leukopenia, thrombocytopenia, nausea, vomiting, and elevated transaminases) as well as immune-related adverse events (irAEs). All AEs were graded according to the Common Terminology Criteria for Adverse Events (CTCAE) [11].
Statistical analyses were performed using SPSS software (version
26.0, IBM Corp., New York, NY, USA), while graphical presentations were generated
with GraphPad Prism (version 6.0, GraphPad Prism Software Inc., San Diego, CA,
USA). A two-sided p-value
To minimise selection bias and balance baseline characteristics, 1:1
nearest-neighbour propensity score matching (PSM) was applied. Matching variables
included age, gender, histological subtype, ECOG performance status, PD-L1
expression level, smoking history, number of prior systemic therapies, N stage
status, EGFR mutation status, and ALK rearrangement status. The calliper width
was set at 0.05. In addition to p-values, standardized mean differences (SMDs)
were calculated to assess the balance of baseline covariates between groups.
An SMD
Categorical variables are presented as counts (n) and proportions (%).
Group comparisons were selected based on the minimum expected frequency (T) in
contingency tables: the chi-square test was applied if T
A total of 155 patients with unresectable NSCLC and lymph node
metastasis were initially identified, including 85 patients in the chemotherapy
group and 70 in the ICI group. Following 1:1 PSM, a final matched cohort of 112
patients was established, with 56 participants in each treatment group (Fig. 1).
Detailed demographic and clinical characteristics of patients before and after
PSM are presented in Table 1. Before matching, ECOG performance status differed
significantly between the two groups (p = 0.017). However, other
baseline variables, including age, gender, histological subtype, PD-L1
expression, smoking history, number of prior therapies, TNM stage, and driver
gene status (EGFR/ALK), were comparable between groups (all p
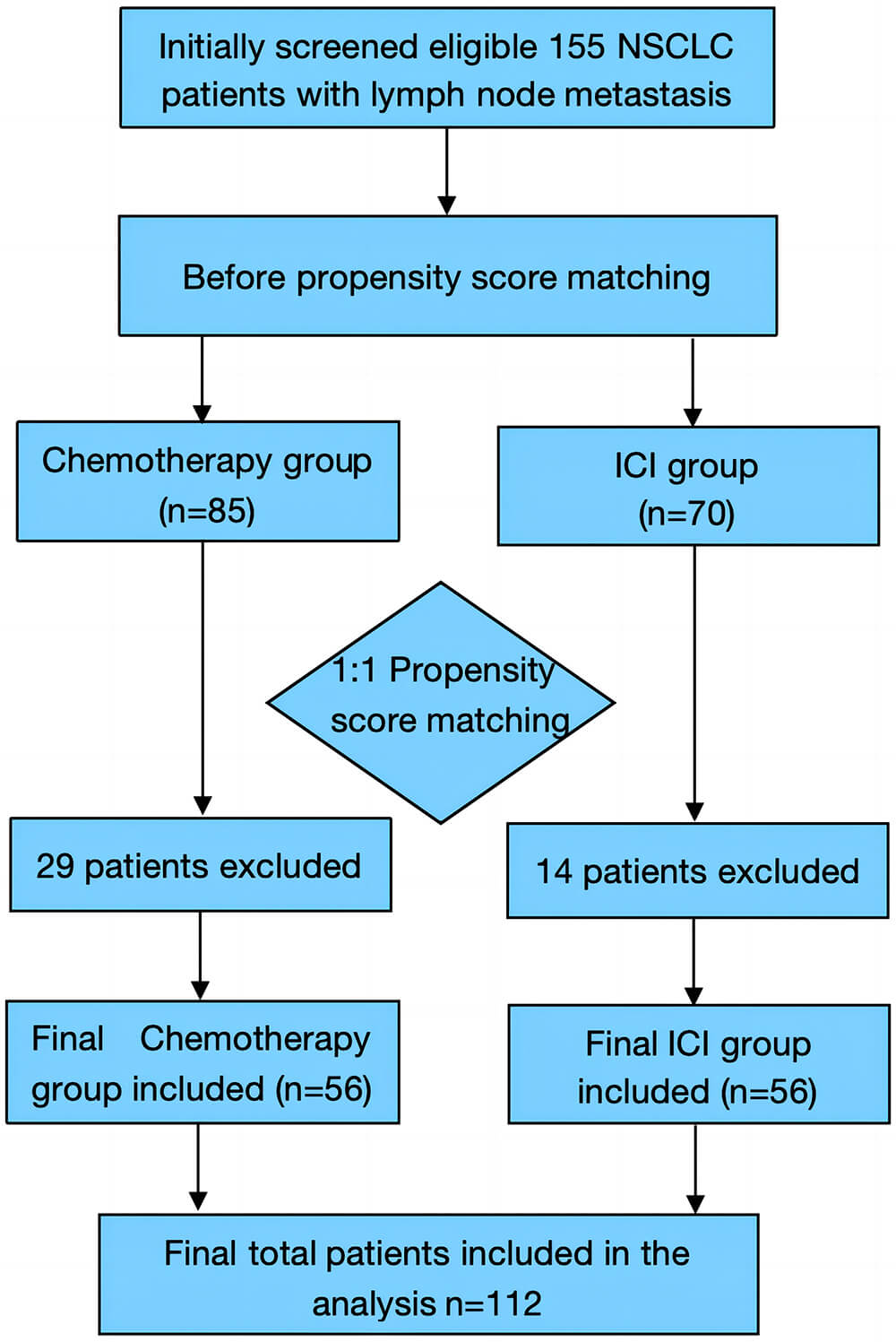 Fig. 1.
Fig. 1.
Patient selection flowchart. NSCLC, non-small cell lung cancer; ICI, immune checkpoint inhibitor.
| Variable | Before PSM | After PSM | |||||||||
| Chemotherapy (n = 85) | ICI group (n = 70) | SMD | Chi-square value | p-value | Chemotherapy (n = 56) | ICI group (n = 56) | SMD | Chi-square value | p-value | ||
| Age, years, n (%) | 0.144 | 1.673 | 0.196 | 0.152 | 1.328 | 0.249 | |||||
| 56 (65.9%) | 39 (55.7%) | 36 (64.3%) | 30 (53.6%) | ||||||||
| 29 (34.1%) | 31 (44.3%) | 20 (35.7%) | 26 (46.4%) | ||||||||
| Gender, n (%) | 0.207 | 3.314 | 0.069 | 0.050 | 0.146 | 0.703 | |||||
| Female | 44 (51.8%) | 26 (37.1%) | 23 (41.1%) | 25 (44.6%) | |||||||
| Male | 41 (48.2%) | 44 (62.9%) | 33 (58.9%) | 31 (55.4%) | |||||||
| Histological type, n (%) | 0.197 | 3.168 | 0.075 | 0.025 | 0.036 | 0.850 | |||||
| Non-squamous carcinoma | 58 (68.2%) | 38 (54.3%) | 31 (55.4%) | 30 (53.6%) | |||||||
| Squamous carcinoma | 27 (31.8%) | 32 (45.7%) | 25 (44.6%) | 26 (46.4%) | |||||||
| ECOG score, n (%) | 0.253 | 5.689 | 0.017 | 0.000 | 0.000 | 1.000 | |||||
| 0 | 20 (23.5%) | 29 (41.4%) | 16 (28.6%) | 16 (28.6%) | |||||||
| 1 | 65 (76.5%) | 41 (58.6%) | 40 (71.4%) | 40 (71.4%) | |||||||
| PD-L1 expression, %, n (%) | 0.073 | 0.405 | 0.525 | 0.000 | 0.000 | 1.000 | |||||
| 1–49% | 42 (49.4%) | 31 (44.3%) | 27 (48.2%) | 27 (48.2%) | |||||||
| 43 (50.6%) | 39 (55.7%) | 29 (51.8%) | 29 (51.8%) | ||||||||
| Smoking history, n (%) | 0.116 | 1.611 | 0.447 | 0.110 | 0.995 | 0.608 | |||||
| Never smoked | 29 (34.1%) | 18 (25.7%) | 19 (33.9%) | 17 (30.4%) | |||||||
| Former smoker | 26 (30.6%) | 27 (38.6%) | 17 (30.4%) | 22 (39.3%) | |||||||
| Current smoker | 30 (35.3%) | 25 (35.7%) | 20 (35.7%) | 17 (30.4%) | |||||||
| Previous systemic treatments, n (%) | 0.063 | 0.309 | 0.578 | 0.051 | 0.145 | 0.704 | |||||
| 1 | 46 (54.1%) | 41 (58.6%) | 30 (53.6%) | 32 (57.1%) | |||||||
| 39 (45.9%) | 29 (41.4%) | 26 (46.4%) | 24 (42.9%) | ||||||||
| N stage, n (%) | 0.012 | 0.033 | 0.984 | 0.152 | 3.021 | 0.221 | |||||
| N1 | 13 (15.3%) | 11 (15.7%) | 9 (16.1%) | 9 (16.1%) | |||||||
| N2 | 59 (69.4%) | 49 (70.0%) | 37 (66.1%) | 43 (76.8%) | |||||||
| N3 | 13 (15.3%) | 10 (14.3%) | 10 (17.9%) | 4 (7.1%) | |||||||
| EGFR mutation, n (%) | 0.067 | 0.591 | 0.442 | 0.126 | 1.465 | 0.226 | |||||
| No | 72 (84.7%) | 56 (80.0%) | 48 (85.7%) | 43 (76.8%) | |||||||
| Yes | 13 (15.3%) | 14 (20.0%) | 8 (14.3%) | 13 (23.2%) | |||||||
| ALK rearrangement, n (%) | 0.031 | 0.425 | 0.792* | 0.126 | - | 0.495** | |||||
| No | 82 (96.5%) | 66 (94.3%) | 56 (100.0%) | 54 (96.4%) | |||||||
| Yes | 3 (3.5%) | 4 (5.7%) | 0 (0.0%) | 2 (3.6%) | |||||||
Notes: PSM, propensity score matching; ECOG, Eastern Cooperative Oncology Group; ICI, immune checkpoint inhibitor; EGFR, ̵epidermal growth factor receptor; SMD, standardised mean difference; ALK, anaplastic lymphoma kinase; PD-L1, Programmed Cell Death Ligand 1. *p-values were calculated using chi-square corrected test; **p-values were calculated using Fisher’s exact test.
Treatment outcomes, including ORR and DCR, are summarised in Table 2. The ORR was significantly higher in the ICI group compared with the chemotherapy group (p = 0.012). Conversely, the DCR did not differ significantly between the two groups (p = 0.057).
| Variable | ICI group (n = 56) | Chemotherapy group (n = 56) | Chi-square value | p-value |
| Complete response | 14 | 8 | - | - |
| Partial response | 14 | 7 | - | - |
| Stable disease | 17 | 21 | - | - |
| Progressive disease | 11 | 20 | - | - |
| Objective response rate, n (%) | 28 (50.0%) | 15 (26.8%) | 6.38 | 0.012 |
| Disease control rate, n (%) | 45 (80.4%) | 36 (64.3%) | 3.61 | 0.057 |
Notes: ICI, immune checkpoint inhibitor.
As shown in Fig. 2, subgroup analysis demonstrated that ICI therapy was
associated with higher ORR compared with chemotherapy across several patient
subgroups. Notably, patients with PD-L1 expression
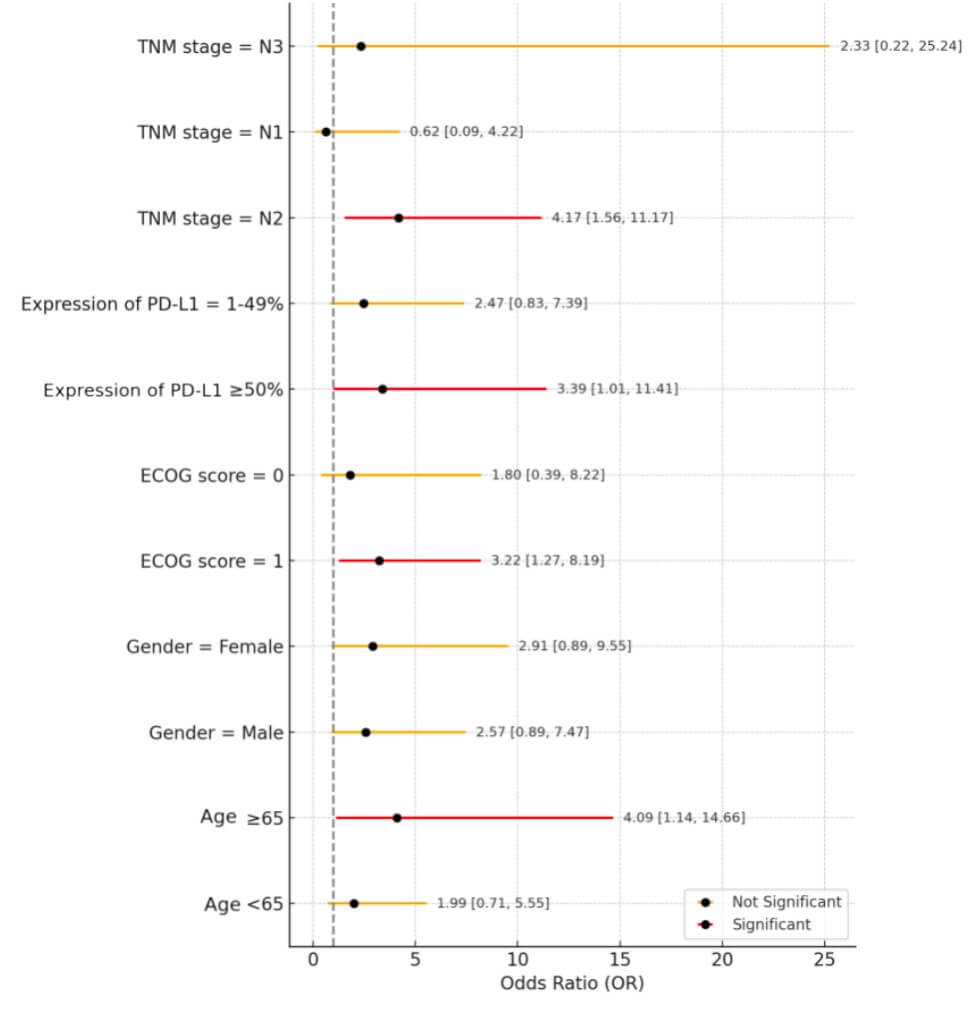 Fig. 2.
Fig. 2.
Forest plot of objective response rate (ORR) by subgroup (ICI vs. Chemotherapy). TNM, tumour-node-metastasis; PD-L1, Programmed Cell Death Ligand 1.
Patients were followed for a median duration of 24 months. Overall survival (OS) was estimated using the Kaplan-Meier method, and survival curves for the ICI and chemotherapy groups are presented in Fig. 3. During the 24-month follow-up period, patients in the ICI group consistently demonstrated a higher survival probability compared with those in the chemotherapy group. The 1-year survival rates were 76.8% for the ICI group and 44.6% for the chemotherapy group. At the 24-month cutoff, the 2-year survival rate of 48.2% for the ICI group versus 28.6% for the chemotherapy group.
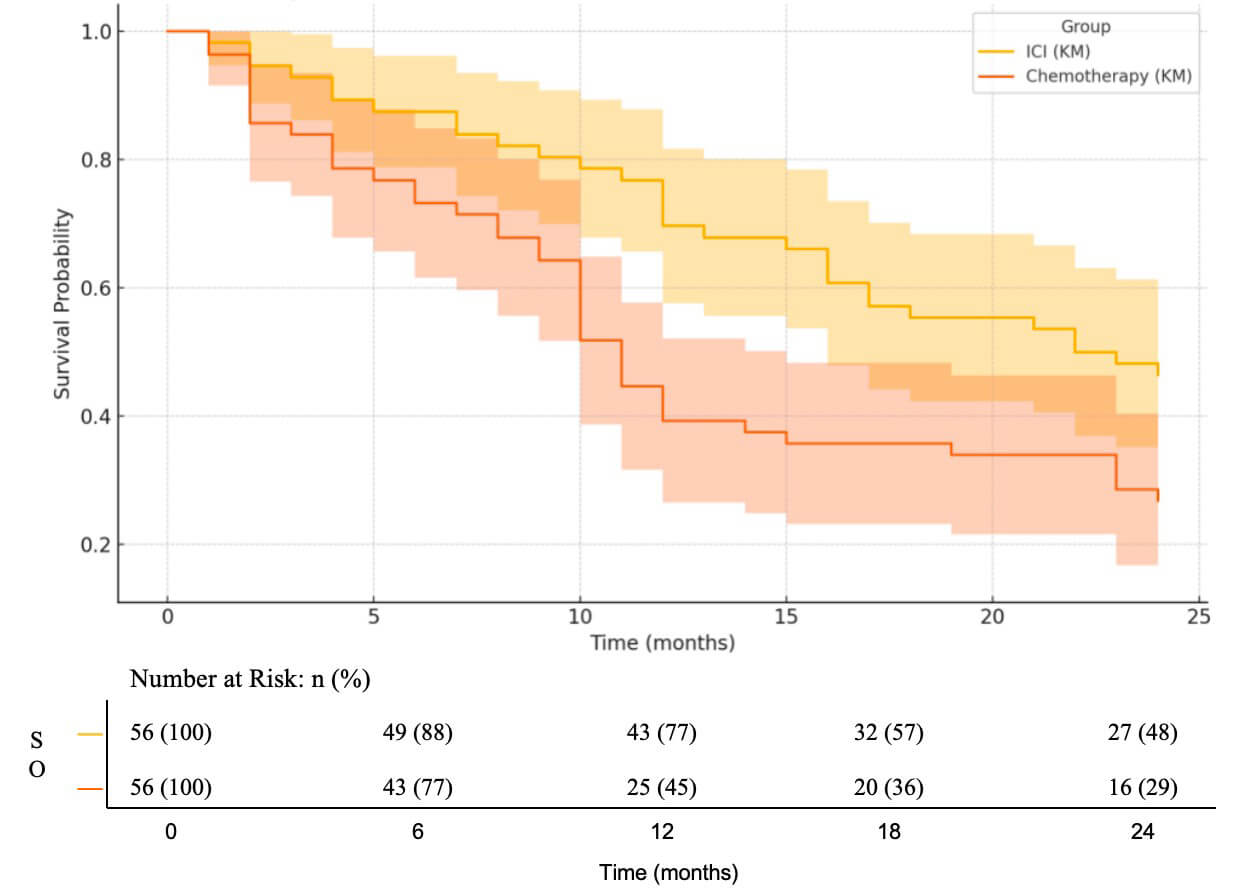 Fig. 3.
Fig. 3.
Kaplan-Meier (KM) analysis of overall survival with integrated number at risk.
The median OS was 22 months in the ICI group compared with 11.5 months in the chemotherapy group. Survival distributions differed significantly between the two groups, as determined by the log-rank test (p = 0.009). These findings indicate that ICI monotherapy significantly prolongs overall survival compared with chemotherapy.
Table 3 presents the results of the univariate and multivariate Cox
proportional hazards analyses for OS. In the univariate analysis, the treatment
group emerged as the only significant predictor of OS. Compared with the ICI
group, patients receiving chemotherapy had a significantly increased risk of
death (hazard ratio [HR] = 1.836; 95% CI: 1.144–2.948; p = 0.012). No
other baseline characteristics were identified as significant predictors in the
univariate analysis (all p
| Variable | Univariate analysis | Multivariate analysis | |||
| HR (95% CI) | p-value | aHR (95% CI)1 | p-value | ||
| Treatment group (Chemo vs. ICI) | 1.836 (1.144–2.948) | 0.012 | 1.796 (1.112–2.900) | 0.017 | |
| Age ( |
1.085 (0.676–1.742) | 0.736 | 1.022 (0.625–1.671) | 0.930 | |
| Gender (Male vs. Female) | 1.108 (0.693–1.771) | 0.668 | - | - | |
| Histology (Squamous vs. Non-squamous) | 0.878 (0.551–1.400) | 0.585 | - | - | |
| ECOG score (1 vs. 0) | 1.307 (0.790–2.163) | 0.297 | 1.308 (0.782–2.188) | 0.307 | |
| PD-L1 expression ( |
1.280 (0.802–2.040) | 0.300 | 1.245 (0.778–1.995) | 0.361 | |
| Smoking history | - | 0.763 | - | - | |
| Past smoker vs. Never smoked | 0.932 (0.516–1.684) | 0.816 | - | - | |
| Current smoker vs. Never smoked | 1.147 (0.657–2.000) | 0.630 | - | - | |
| Previous systemic treatments ( |
1.296 (0.805–2.087) | 0.285 | - | - | |
| N stage | - | 0.055 | - | - | |
| N2 vs. N1 | 1.009 (0.452–2.247) | 0.983 | - | - | |
| N3 vs. N1 | 1.869 (0.955–3.650) | 0.068 | - | - | |
| EGFR mutation (Yes vs. No) | 0.951 (0.521–1.735) | 0.869 | - | - | |
| ALK rearrangement (Yes vs. No) | 1.738 (0.241–12.517) | 0.583 | - | - | |
Notes: HR, hazard ratio; aHR, adjusted hazard ratio; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; ICI, immune checkpoint inhibitor. 1Multivariate model was adjusted for treatment group, age, ECOG score, and PD-L1 expression.
For a robust multivariate analysis, treatment group and other clinically relevant variables (age, ECOG performance status, and PD-L1 expression) were included to adjust for potential confounders. After adjustment, the treatment group remained an independent and significant prognostic factor for OS. Chemotherapy was associated with a significantly higher risk of mortality relative to ICI therapy (adjusted hazard ratio [aHR] = 1.796; 95% CI: 1.112–2.900; p = 0.017).
Adverse events (AEs) observed in this study are summarised in Table 4. Clear differences in safety profiles were noted between the two groups, with the most prominent disparities observed in hematologic toxicity. The incidence of leukopenia was significantly higher in the chemotherapy group (p = 0.039). Similarly, anaemia (any grade) was significantly more frequent in the chemotherapy group compared to the ICI group (p = 0.013). irAEs were predominantly observed in the ICI group. Compared with the chemotherapy group, patients receiving ICI therapy experienced significantly higher rates of hypothyroidism (p = 0.027) and rash/pruritus (p = 0.008), both at any grade level.
| Adverse event | Severity | ICI group (n = 56) | Chemotherapy group (n = 56) | Chi-square value | p-value* |
| n (%) | n (%) | ||||
| Leukopenia | All grades | 3 (5.4%) | 10 (17.9%) | 4.264 | 0.039 |
| Grade |
1 (1.8%) | 5 (8.9%) | 1.585 | 0.208* | |
| Anaemia | All grades | 7 (12.5%) | 18 (32.1%) | 6.231 | 0.013 |
| Grade |
1 (1.8%) | 4 (7.1%) | 0.837 | 0.360* | |
| Thrombocytopenia | All grades | 4 (7.1%) | 10 (17.9%) | 2.939 | 0.086 |
| Grade |
0 (0.0) | 2 (3.6%) | 0.509 | 0.476* | |
| Nausea | All grades | 12 (21.4%) | 15 (26.8%) | 0.439 | 0.508 |
| Grade |
0 (0.0) | 1 (1.8%) | - | 1.000** | |
| Hypothyroidism | All grades | 6 (10.7%) | 0 (0.0) | 4.403 | 0.027** |
| Grade |
0 (0.0) | 0 (0.0) | - | - | |
| Rash/Pruritus | All grades | 11 (19.6%) | 2 (3.6%) | 7.049 | 0.008 |
| Grade |
1 (1.8%) | 0 (0.0) | - | 1.000** | |
| Pneumonitis | All grades | 1 (1.8%) | 0 (0.0) | - | 1.000** |
| Grade |
0 (0.0) | 0 (0.0) | - | - |
Notes: ICI, immune checkpoint inhibitor. *p-values were calculated using the chi-square corrected test; **p-values were calculated using Fisher’s exact test.
In this retrospective study, we analysed pretreated patients with NSCLC and lymph node metastasis. Pembrolizumab monotherapy demonstrated significantly superior efficacy over docetaxel chemotherapy, with significant improvements in ORR and OS. Although not statistically significant, a trend toward a higher DCR was observed in the ICI group. Furthermore, the safety profile of the ICI treatment was more favourable, showing fewer severe hematologic toxicities.
Our findings on the efficacy of ICI monotherapy are consistent with results from landmark clinical trials that established the utility of ICIs in second-line therapy for advanced NSCLC [6]. For instance, Zhao et al. [6] demonstrated that pembrolizumab, an anti-Programmed Cell Death Protein 1 (anti-PD-1) monoclonal antibody, demonstrated significant antitumour efficacy and favourable safety in patients with advanced NSCLC expressing PD-L1 in at least 50% of tumour cells. Interestingly, the ORR observed in our study (50.0% in the ICI group vs. 26.8% in the chemotherapy group) was higher than that reported in multiple large randomised controlled trials [4, 12, 13]. This discrepancy may be attributed to the single-center, retrospective design, which could introduce selection bias. Although PSM helped balance known baseline variables, residual confounding from unmeasured variables cannot be fully excluded.
Notably, the benefits of ICI therapy were markedly pronounced in
specific subgroups. Compared to chemotherapy, ICI treatment significantly
improved ORR in patients with PD-L1 expression
In terms of safety, our study revealed distinct toxicity profiles between the chemotherapy and ICI groups. These results are consistent with previous key clinical trials [4, 13, 16] and underscore the fundamentally different mechanisms of action of the two treatment strategies. In the chemotherapy group, adverse events were dominated by bone marrow suppression, particularly leukopenia, consistent with the classical toxicity pattern of cytotoxic agents. Chemotherapy exerts antitumour effects by targeting rapidly dividing cells but also inevitably damages proliferating normal cells, such as hematopoietic stem cells, resulting in predictable hematologic toxicities. In contrast, the ICI group exhibited a lower overall incidence of severe adverse events, highlighting their advantage in reducing conventional cytotoxicity. However, immune-related adverse events (irAEs), including hypothyroidism and rash, were unique to the ICI group, consistent with previous study [14]. Unlike chemotherapy, ICI toxicities stem not from direct cytotoxic damage but from immune system hyperactivation, which may induce autoimmunity and injury to healthy tissues. These distinct toxicity profiles require different clinical management strategies. For chemotherapy, supportive care and organ function recovery are critical, whereas ICI management relies on immunosuppression, requiring a careful balance between controlling irAEs and maintaining antitumour immunity. Future research should prioritise early detection systems and refined grading strategies to optimise irAE management.
This study has several limitations. First, as a single-center retrospective analysis, its generalizability is restricted, and selection bias may exist. Second, although data collection was comprehensive, rare irAEs such as colitis and hepatitis may have been underreported. Third, while PSM reduced baseline imbalances between groups, it cannot substitute for randomisation. Finally, the relatively small sample size may have constrained the statistical power of subgroup analyses. Future multicenter, prospective studies with larger cohorts are necessary to validate these findings.
In summary, this real-world study provides evidence that pembrolizumab monotherapy offers superior tumour response rates and survival benefits compared with docetaxel monotherapy in patients with unresectable NSCLC with lymph node metastasis following failure of prior platinum-based chemotherapy. Moreover, pembrolizumab demonstrated a distinct safety profile, with a notably lower incidence of severe hematologic toxicities. These findings support the role of ICI monotherapy as a viable and potentially advantageous treatment option for this challenging and clinically significant patient population.
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
LX, GW and WL designed the study. All authors conducted the study. RM and KM collected and analysed the data. LX drafted the manuscript. All authors contributed to the critical revision of the manuscript for important intellectual content. All authors gave final approval of the version to be published. All authors participated fully in the work, took public responsibility for appropriate portions of the content, and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or completeness of any part of the work were appropriately investigated and resolved.
This study was approved by the Ethics Committee of Xi’an International Medical Center Hospital (approval number: 2024034) and was performed in accordance with the principles of the Declaration of Helsinki. Informed consent has been obtained from all participants involved in the study.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



