1 Department of Cardiovascular Medicine, Jinan Third People’s Hospital, 250001 Jinan, Shandong, China
2 Department of Cardiology, Shandong Provincial Hospital Affiliated to Shandong First Medical University, 250000 Jinan, Shandong, China
Abstract
Heart failure with preserved ejection fraction (HFpEF) poses therapeutic challenges, especially among the elderly. Dapagliflozin, a sodium-glucose cotransporter 2 inhibitor, has potential benefits beyond glucose regulation, including possible cardioprotective effects. This study evaluates the efficacy and safety of dapagliflozin in patients over 75 years old with HFpEF.
This retrospective cohort study included 215 patients over 75 years old with HFpEF, divided into a standard treatment group (known as routine group, n = 105) and a dapagliflozin plus standard treatment group (known as dapagliflozin group, n = 110). Key assessments included Kansas City Cardiomyopathy Questionnaire (KCCQ) scores, Six-Minute Walk Test (6MWT), echocardiographic parameters, adverse events, and biochemical markers such as N-terminal pro-B-type natriuretic peptide (NT-proBNP) and B-type natriuretic peptide (BNP), alongside patients’ satisfaction with the treatment administered.
The dapagliflozin group showed significant improvements in KCCQ Overall Summary Score (72.48 ± 12.49 vs 68.59 ± 13.11, p = 0.027) and 6MWT distance (358.47 ± 28.62 m vs 311.69 ± 30.26 m, p < 0.001) compared to the routine group. NT-proBNP and BNP levels showed significantly greater reductions in the dapagliflozin group (p = 0.046 and p = 0.039, respectively). Echocardiographic parameters indicated favorable cardiac remodeling. Incidence of adverse events was similar between groups, with no increase in serious renal or metabolic events in the dapagliflozin group. Dapagliflozin significantly improves patients’ satisfaction in elderly patients with HFpEF (p = 0.041).
Dapagliflozin administration in HFpEF patients over 75 years significantly enhances cardiac function and exercise capacity, as well as improves quality of life markers, without notable safety concerns.
Keywords
- heart failure
- preserved ejection fraction
- aged
- dapagliflozin
- sodium-glucose transporter 2 inhibitors
- heart function tests
- quality of life
Heart failure with preserved ejection fraction (HFpEF) represents a significant and growing public health concern, particularly among the aging population, where it accounts for approximately 50% of all heart failure cases [1]. This condition, characterized by heart failure symptoms despite normal ejection fraction, poses complex diagnostic and therapeutic challenges [2, 3]. Unlike heart failure with reduced ejection fraction (HFrEF), HFpEF lacks established, universally efficacious therapies, making management predominantly reliant on symptomatic relief rather than disease modification [4]. Moreover, patients aged over 75 years old represent a particularly important demographic. This age group often suffers from multiple comorbidities such as hypertension, diabetes, and chronic kidney disease, all of which significantly increase the risk of heart failure. HFpEF is especially prevalent among the elderly, and treatment outcomes tend to be less favorable. Therefore, studying this specific cohort is crucial for optimizing therapeutic strategies and improving patient outcomes [5, 6].
Dapagliflozin, a sodium-glucose cotransporter 2 (SGLT2) inhibitor originally developed for the management of type 2 diabetes mellitus, has emerged as a promising therapeutic agent in the broader context of heart failure [7]. Its mechanisms extend beyond glucose control, offering benefits such as natriuresis, reduced cardiac preload and afterload, and potential anti-inflammatory and antifibrotic effects [8] that can be advantageous in heart failure management [9]. Recent landmark trials, such as Dapagliflozin and Prevention of Adverse-outcomes in Heart Failure (DAPA-HF), have demonstrated dapagliflozin’s efficacy in reducing morbidity and mortality in HFrEF patients, leading to expanded clinical guidelines that advocate its use in the heart failure contexts [10, 11]. However, data specifically addressing its efficacy and safety in older populations with HFpEF remain limited.
The geriatric population, particularly those over 75 years, presents unique physiological challenges [12]. Age-related changes in pharmacokinetics and pharmacodynamics, along with the increased likelihood of frailty, renal impairment, and multiple concomitant medications, complicate heart failure management [13]. Therefore, any additional treatment, such as an SGLT2 inhibitor, must demonstrate substantial efficacy and an acceptable safety profile within this subgroup [14]. Despite the inclusion of older patients in their cohorts, previous studies were not designed to explicitly investigate the interplay between age and HFpEF pathophysiology [15, 16].
In recent times, the Sotagliflozin in Patients with Diabetes and Recent Worsening Heart Failure (SOLOIST-WHF) trial and other similar studies have indicated potential cardioprotective benefits of SGLT2 inhibitors in HFpEF, but specific treatments for HFpEF remain limited, especially for patients over 75 years old, who often have multiple comorbidities and are underrepresented in clinical trials [14, 17]. Given that older adults were underrepresented in clinical trials, retrospective studies can bridge this knowledge gap by focusing on real-world evidence to better understand the efficacy and safety of dapagliflozin in older adults suffering from HFpEF. This retrospective study aims to evaluate the efficacy and safety of dapagliflozin in patients over 75 years old with HFpEF.
This research was conducted in accordance with the ethical principles set forth in the Declaration of Helsinki. This research employed a retrospective cohort study design, focusing on patients with HFpEF who received treatment at Shandong Provincial Hospital Affiliated to Shandong First Medical University between June 2022 and April 2024. Of these, 105 patients underwent standard treatment and were classified into the routine group, while 110 patients were treated with a combination of standard therapy and dapagliflozin and were classified into the dapagliflozin group. Data on patient demographics, such as gender, age, and underlying conditions, as well as medication usage and adverse reactions, were extracted from the hospital’s medical record system (Fig. 1).
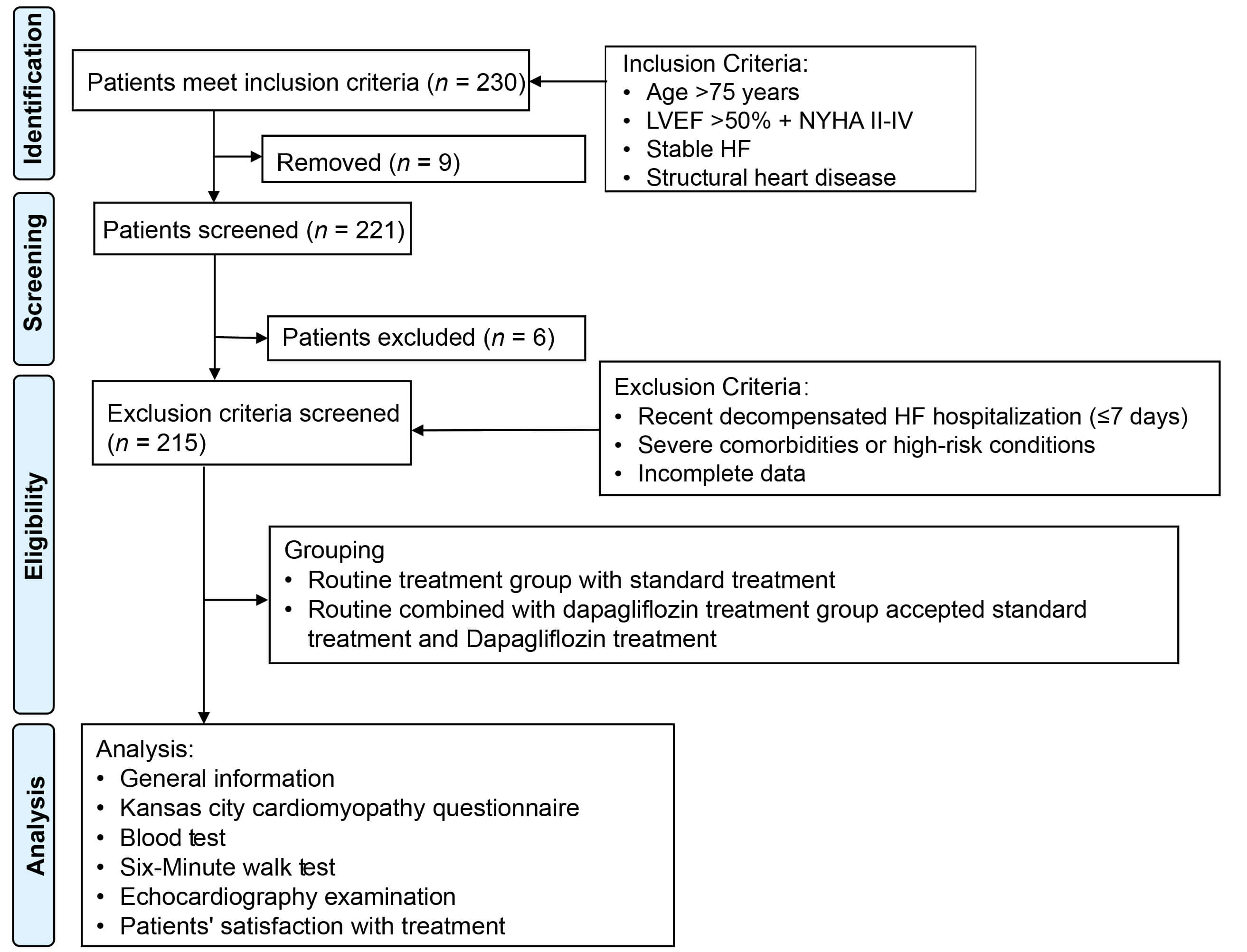 Fig. 1.
Fig. 1.
Flowchart depicting participant selection and inclusion in this study. Abbreviations: HF, heart failure; LVEF, left ventricular ejection fraction; NYHA, New York Heart Association.
The study received approval from the Ethics Committee of Shandong Provincial Hospital Affiliated to Shandong First Medical University (Approval No. 2024-427). Due to the study’s reliance solely on de-identified patient data, informed consent was waived, as no potential harm or impact on patient care was anticipated.
Participants were eligible for the study if they were over 75 years old, had a
clinical diagnosis of HFpEF [18] with a left ventricular
ejection fraction (LVEF) greater than 50%, and were classified under the New
York Heart Association (NYHA) functional classes II to IV. Additional inclusion
criteria include stable history of heart failure (defined as no hospitalization
for heart failure within the last three months prior to enrollment), evidence of
structural heart disease, and elevated levels of natriuretic peptides,
specifically N-terminal pro-B-type natriuretic peptide (NT-proBNP)
Participants were excluded if they had been hospitalized for decompensated heart failure within the past seven days; had an estimated glomerular filtration rate (eGFR) of less than 20 mL/min/1.73 m2 at admission; had type 1 diabetes, multiple organ dysfunction, or allergies to any medications used in the study; or had malignant tumors. Additionally, patients with incomplete clinical records or missing key data points were also excluded.
Standard care was given to each patient of both groups to address their
coexisting conditions. Treatment regimens included: 5 mg once daily enalapril
tablets (angiotensin-converting enzyme [ACE] inhibitor; China Resources Shuanghe
Limin Pharmaceutical Co., Ltd., Jinan, China, Approval No. H20066730), 100 mg
once daily losartan potassium tablets (angiotensin II receptor blocker [ARB];
Ningbo Korcomer Novartis Pharmaceutical Co., Ltd., Ningbo, China, Approval No.
H20213818), 50 mg once daily Entresto (angiotensin receptor-neprilysin inhibitor
[ARNI]; Beijing Novartis Pharmaceutical Co., Ltd., Beijing, China, Approval No.
J20190001), 10 mg once daily propranolol (
For the group receiving routine treatment plus dapagliflozin, dapagliflozin tablets (10 mg per tablet, packaged as 14 tablets per pack, AstraZeneca Pharmaceuticals Co., Ltd., Shanghai, China, Approval No. H20234463) were additionally administered. The dosage was set at 10 mg once daily over the 12-week treatment period [20].
The Kansas City Cardiomyopathy Questionnaire (KCCQ) was employed to assess heart failure-related symptoms, specifically their frequency, severity, and recent changes, along with evaluations of physical function, quality of life, and social function, both before and after treatment [21]. Within the KCCQ, the Total Symptom Score (TSS) measures the frequency and severity of symptoms, while the Physical Limitation Score (PLS) assesses physical function. The Clinical Summary Score (CSS) encompasses both the symptom and physical function domains. Meanwhile, the Overall Summary Score (OSS) integrates all major domains, including symptom frequency and severity, physical function, quality of life, and social function. Each domain’s validity, reproducibility, responsiveness, and interpretability have been independently confirmed. Scores were scaled from 0 to 100, with higher scores indicating better health status. The instrument demonstrates high reliability, with a Cronbach’s alpha of 0.97 and an intraclass correlation coefficient of 0.95 [22].
Fasting venous blood samples (5 mL) were collected from patients both before and after treatment. These samples were treated with ethylenediaminetetraacetic acid (EDTA) as an anticoagulant and centrifuged at 2800 rpm for 15 minutes to separate the serum. The serum was then analyzed for NT-proBNP, BNP, eGFR, and glycated hemoglobin (HbA1c) using an automated biochemical analyzer (BS-280, Mindray, Shenzhen, China).
The Six-Minute Walk Test (6MWT) was administered indoors on a flat 30-meter corridor, with patients instructed to walk as far as possible within 6 minutes, in accordance with the American Thoracic Society guidelines [23]. The total distance covered during the 6-minute period was recorded in meters.
Color Doppler echocardiography (Vivid E95, GE HealthCare, Chicago, IL USA) was employed to assess cardiac function using a 3.0 MHz probe frequency both before and after treatment. Subjects were positioned in the left lateral decubitus position, and the apical four-chamber view was used for pulsed Doppler measurements. The left ventricular end-diastolic dimension (LVEDD), left ventricular end-systolic dimension (LVESD), and left ventricular end-diastolic volume (LVEDV) were measured from the parasternal long-axis view. The left ventricular mass (LVM) was calculated using the area-length method, from which the left ventricular mass index (LVMI) was derived. Additionally, the left ventricular remodeling index (LVRI) was determined by dividing LVM by LVEDV.
Adverse events were defined as any untoward medical occurrence in a patient receiving treatment, temporally associated with the use of the study medication, whether or not considered related to the medication. Adverse events were monitored and recorded throughout the 12-week treatment period through regular clinical follow-up visits and review of medical records. All adverse events were documented in the electronic medical record system, including their severity, duration, and any actions taken. Serious adverse events were defined as those resulting in death, life-threatening conditions, hospitalization, or significant disability. The primary adverse events of interest included hypoglycemia, diabetic ketoacidosis, acute kidney injury, myocardial infarction, stroke, and lower limb amputations.
A hospital-developed questionnaire was utilized to evaluate patients’ satisfaction with the treatments administered in both groups. The questionnaire was scored on a scale from 0 to 100 points. The total scores obtained were categorized as follows: very satisfied (90–100 points), satisfied (60–89 points), and dissatisfied (less than 60 points). Higher scores indicated greater levels of satisfaction among participants.
Data were analyzed using the SPSS version 29.0 statistical software (SPSS Inc.,
Chicago, IL, USA). Categorical data are presented as frequencies (n) and
percentages (%). The chi-square test was applied using the basic formula when
the sample size was
The groups were well-matched in terms of age (80.15
| Parameters | Routine group (n = 105) | Dapagliflozin group (n = 110) | t/ |
p | |
| Age (years) | 80.15 |
80.34 |
0.913 | 0.362 | |
| Male | 54 (51.43%) | 62 (56.36%) | 0.527 | 0.468 | |
| BMI (kg/m2) | 29.78 |
29.27 |
1.674 | 0.096 | |
| Hypertension | 92 (87.62%) | 94 (85.45%) | 0.216 | 0.642 | |
| Type 2 diabetes mellitus | 47 (44.76%) | 55 (50.00%) | 0.591 | 0.442 | |
| Dyslipidemia | 67 (63.81%) | 68 (61.82%) | 0.091 | 0.763 | |
| Ischemic heart disease | 21 (20.00%) | 21 (19.09%) | 0.028 | 0.867 | |
| Stroke | 11 (10.48%) | 9 (8.18%) | 0.335 | 0.563 | |
| Atrial fibrillation | 68 (64.76%) | 69 (62.73%) | 0.096 | 0.756 | |
| Previous myocardial infarction | 22 (20.95%) | 22 (20.00%) | 0.030 | 0.863 | |
| Chronic obstructive pulmonary disease | 12 (11.43%) | 10 (9.09%) | 0.320 | 0.572 | |
| Atherosclerotic cardiovascular disease | 57 (54.29%) | 57 (51.82%) | 0.131 | 0.717 | |
| Smoking history | 25 (23.81%) | 31 (28.18%) | 0.533 | 0.465 | |
| Drinking history | 34 (32.38%) | 33 (30.00%) | 0.142 | 0.706 | |
| LVEF (%) | 55.83 |
56.16 |
1.095 | 0.275 | |
| Duration of HF (years) | 3.11 |
3.24 |
0.897 | 0.371 | |
| Previous hospitalization for HF | 42 (40.00%) | 48 (43.64%) | 0.292 | 0.589 | |
| Systolic blood pressure (mmHg) | 128.16 |
129.28 |
0.571 | 0.569 | |
| Diastolic blood pressure (mmHg) | 72.24 |
72.95 |
0.510 | 0.611 | |
| Heart rate (beats/min) | 70.94 |
70.39 |
0.396 | 0.692 | |
| NYHA classification | 1.509 | 0.470 | |||
| II | 77 (73.33%) | 84 (76.36%) | |||
| III | 27 (25.71%) | 23 (20.91%) | |||
| IV | 1 (0.95%) | 3 (2.73%) | |||
Abbreviations: BMI, body mass index; HF, heart failure; LVEF, left ventricular ejection fraction; NYHA, New York Heart Association.
The use of ACE inhibitors was similar between the two groups, with 33.33% in
the routine group and 31.82% in the dapagliflozin group (p = 0.813)
(Table 2). ARBs were utilized by 37.14% of the routine group and 35.45% of the
dapagliflozin group (p = 0.797). The use of ARNIs was low in both
groups, at 3.81% and 2.73%, respectively (p = 0.950).
| Parameters | Routine group (n = 105) | Dapagliflozin group (n = 110) | p | |
| ACE inhibitor | 35 (33.33%) | 35 (31.82%) | 0.056 | 0.813 |
| ARB | 39 (37.14%) | 39 (35.45%) | 0.066 | 0.797 |
| ARNI | 4 (3.81%) | 3 (2.73%) | 0.004 | 0.950 |
| 82 (78.10%) | 83 (75.45%) | 0.210 | 0.647 | |
| MRA | 39 (37.14%) | 46 (41.82%) | 0.491 | 0.483 |
| Loop diuretics | 83 (79.05%) | 91 (82.73%) | 0.471 | 0.492 |
| Pacemaker | 16 (15.24%) | 14 (12.73%) | 0.282 | 0.595 |
| ICD | 1 (0.95%) | 2 (1.82%) | 0.000 | 1.000 |
Abbreviations: ACE, angiotensin-converting enzyme; ARB, angiotensin II receptor blocker; ARNI, angiotensin receptor-neprilysin inhibitor; MRA, mineralocorticoid receptor antagonist; ICD, internal cardiac defibrillator.
Both groups had similar baseline levels of NT-proBNP and BNP, with no
significant differences prior to treatment (p
| Parameters | Time points | Routine group (n = 105) | Dapagliflozin group (n = 110) | t | p |
| NT-proBNP (pg/mL) | Before treatment | 734.45 |
733.15 |
0.154 | 0.878 |
| After treatment | 659.34 |
619.04 |
2.008 | 0.046 | |
| BNP (pg/mL) | Before treatment | 147.64 |
148.75 |
0.300 | 0.764 |
| After treatment | 138.29 |
132.36 |
2.079 | 0.039 | |
| eGFR (mL/min/1.73 m2) | Before treatment | 54.78 |
55.59 |
0.364 | 0.716 |
| After treatment | 53.11 |
50.13 |
2.122 | 0.035 | |
| HbA1c (%) | Before treatment | 6.31 |
6.23 |
0.576 | 0.565 |
| After treatment | 6.12 |
5.75 |
2.623 | 0.009 |
Notes: ** p
Abbreviations: BNP, B-type natriuretic peptide; eGFR, estimated glomerular filtration rate; HbA1c, glycated hemoglobin; NT-proBNP, N-terminal pro-B-type natriuretic peptide.
Baseline scores were similar across all KCCQ subscales, with no significant
differences observed before treatment (p
| KCCQ subscales | Time points | Routine group (n = 105) | Dapagliflozin group (n = 110) | t | p |
| OSS | Before treatment | 66.35 |
66.49 |
0.051 | 0.959 |
| After treatment | 68.59 |
72.48 |
2.228 | 0.027 | |
| CSS | Before treatment | 68.48 |
68.85 |
0.154 | 0.878 |
| After treatment | 71.61 |
74.87 |
2.152 | 0.033 | |
| TSS | Before treatment | 70.15 |
70.34 |
0.061 | 0.951 |
| After treatment | 74.28 |
80.06 |
2.097 | 0.037 | |
| PLS | Before treatment | 64.38 |
64.15 |
0.650 | 0.517 |
| After treatment | 66.39 |
67.67 |
2.799 | 0.006 |
Notes: ** p
Abbreviations: CSS, Clinical Summary Score; KCCQ, Kansas City Cardiomyopathy Questionnaire; OSS, Overall Summary Score; PLS, Physical Limitation Score; TSS, Total Symptom Score.
Before treatment, the distances were similar between the routine group and the
dapagliflozin group (244.58
| Time points | Routine group (n = 105) | Dapagliflozin group (n = 110) | t | p |
| Before treatment (m) | 244.58 |
244.26 |
0.037 | 0.970 |
| After treatment (m) | 311.69 |
358.47 |
11.651 |
Notes: *** p
Abbreviation: 6MWT, Six-Minute Walk Test.
Baseline measurements of LVESD, LVEDD, LVMI and LVRI were similar between the
routine group and the dapagliflozin group (p
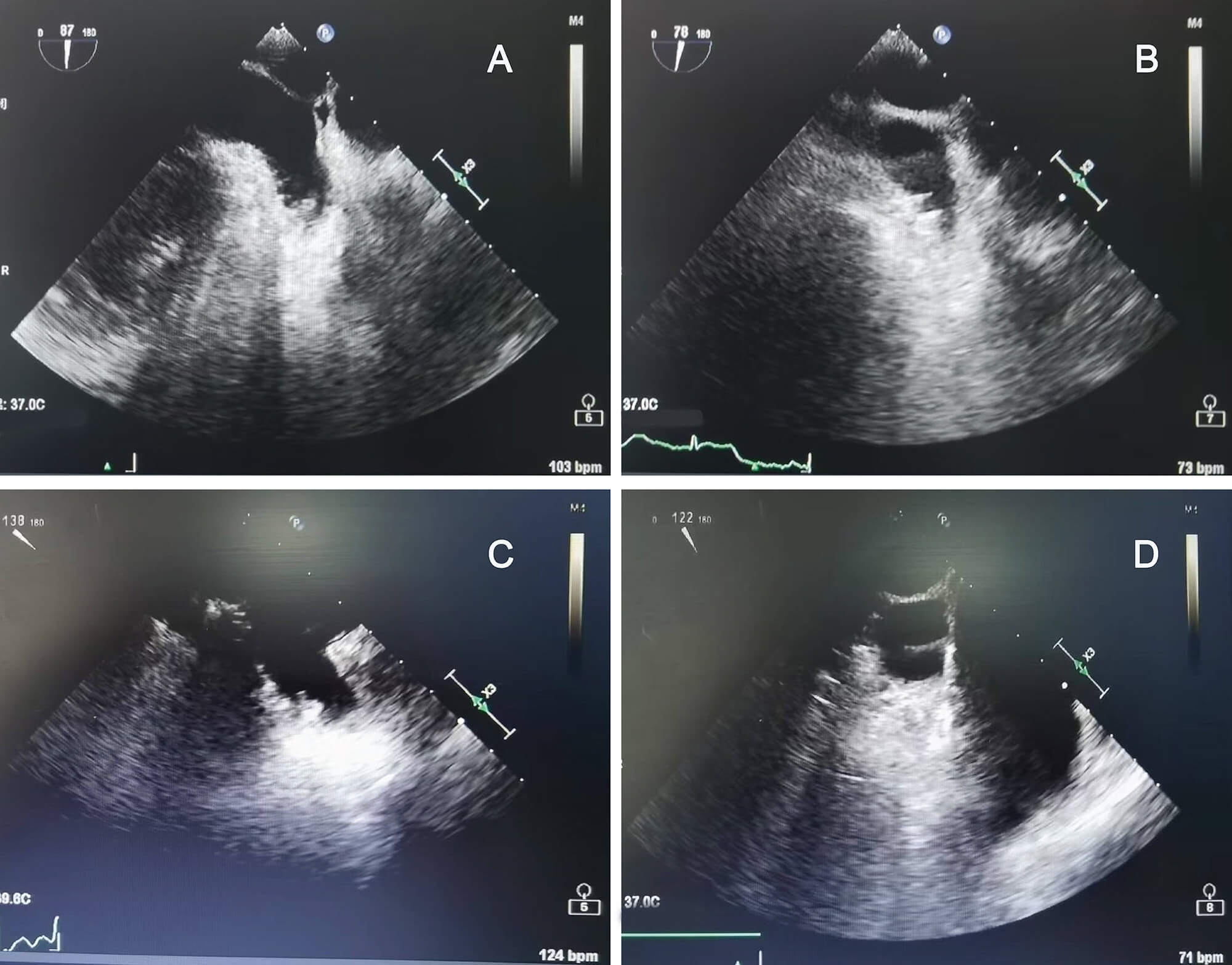 Fig. 2.
Fig. 2.
Echocardiographic images before and after treatment. (A,B) Representative images of patients from the routine group before (A) and after treatment (B). (C,D) Representative images of patients from the dapagliflozin group before (C) and after treatment (D).
| Parameters | Time points | Routine group (n = 105) | Dapagliflozin group (n = 110) | t | p |
| LVESD (mm) | Before treatment | 44.16 |
44.24 |
0.190 | 0.849 |
| After treatment | 38.51 |
36.95 |
3.111 | 0.002 | |
| LVEDD (mm) | Before treatment | 58.08 |
58.24 |
0.200 | 0.842 |
| After treatment | 54.12 |
52.32 |
2.768 | 0.006 | |
| LVMI (g/m2) | Before treatment | 120.27 |
122.36 |
1.074 | 0.284 |
| After treatment | 108.34 |
104.23 |
2.372 | 0.019 | |
| LVRI (g/mL) | Before treatment | 1.66 |
1.63 |
1.322 | 0.188 |
| After treatment | 1.43 |
1.37 |
3.469 |
Notes: *** p
Abbreviations: LVEDD, left ventricular end-diastolic dimension; LVESD, left ventricular end-systolic dimension; LVMI, left ventricular mass index; LVRI, left ventricular remodeling index.
There were no occurrences of lower limb amputations, diabetic ketoacidosis (DKA), or severe hypoglycemia in either group (p = 1.000) (Table 7). A single instance of nonfatal myocardial infarction (MI) and stroke was reported in the routine treatment group, while there were no cases in the dapagliflozin group, but these differences were not significant (p = 0.488 for both). Acute kidney injury occurred in 4.76% of the routine group and 4.55% of the dapagliflozin group, with no statistically significant difference (p = 1.000). These findings suggest that adding dapagliflozin to the treatment regimen does not increase the risk of these adverse events in the studied population.
| Parameters | Routine group (n = 105) | Dapagliflozin group (n = 110) | p | |
| Lower limb amputations | 0 (0.00%) | 0 (0.00%) | None | 1.000 |
| DKA | 0 (0.00%) | 0 (0.00%) | None | 1.000 |
| Severe hypoglycemia | 0 (0.00%) | 0 (0.00%) | None | 1.000 |
| Nonfatal MI | 1 (0.95%) | 0 (0.00%) | None | 0.488 |
| Stroke | 1 (0.95%) | 0 (0.00%) | None | 0.488 |
| Acute kidney injury | 5 (4.76%) | 5 (4.55%) | 0.000 | 1.000 |
Abbreviations: DKA, diabetic ketoacidosis; MI, myocardial infarction.
In the dapagliflozin group, 30.91% of patients expressed high level of treatment satisfaction (labeled as “very satisfied”) compared to 19.05% of patients in the routine group (Table 8). The proportion of patients who were “satisfied” with the treatments administered was similar across the two groups, with 64.76% in the routine group and 61.82% in the dapagliflozin group. However, the percentage of “dissatisfied” patients was lower in the dapagliflozin group (7.27%) compared to the routine group (16.19%). Overall, the satisfaction rate was significantly higher in the dapagliflozin group than in the routine group (92.73% vs 83.81%, p = 0.041). These findings suggest that elderly HFpEF patients over 75 years old are generally satisfied with the addition of dapagliflozin to the standard treatment regimen.
| Level of satisfaction | Routine group (n = 105) | Dapagliflozin group (n = 110) | p | |
| Very satisfied | 20 (19.05%) | 34 (30.91%) | ||
| Satisfied | 68 (64.76%) | 68 (61.82%) | ||
| Dissatisfied | 17 (16.19%) | 8 (7.27%) | ||
| Satisfaction rate | 88 (83.81%) | 102 (92.73%) | 4.158 | 0.041 |
Dapagliflozin has primarily gained attention as an SGLT2 inhibitor used in diabetic populations [24]. However, recent investigations have demonstrated its potential benefits beyond glycemic control, particularly in patients with heart failure [25, 26]. The mechanisms through which dapagliflozin ameliorates HFpEF symptoms can be attributed to its multifaceted physiological effects. These include its ability to promote osmotic diuresis and natriuresis, which alleviate cardiac preload and afterload, potentially translating to improved cardiac efficiency and reduced myocardial stress [27]. This effect likely explains the observed reductions in both NT-proBNP and BNP levels in our dapagliflozin-treated cohort, as evidenced by a significant decrease post-treatment compared to the control group. The mechanisms through which dapagliflozin ameliorates HFpEF symptoms can be attributed to its multifaceted physiological effects. For instance, a recent study demonstrated that dapagliflozin treatment was associated with a notable reduction in LVMI in patients with HFpEF [28].
Another vital mechanism by which dapagliflozin may exert its positive effects on heart function is through the improvement of energy metabolism within the myocardium [29]. Dapagliflozin has been suggested to shift myocardial substrate utilization from fatty acids to more efficient ketone bodies, which may enhance cardiac energetics and contribute to improved cardiac output and function [30]. This shift in substrate utilization could partly account for the significant improvements observed in the echocardiographic parameters, specifically concerning left ventricular dimensions and remodeling indices.
Furthermore, this study’s data on improved physical performance, as indicated by the 6MWT, further support the hypothesis that dapagliflozin enhances not just cardiac function but also overall physical capacity. Such improvements were likely linked to suppression of heart failure symptoms and enhanced diastolic function, which allows for patients’ engagement in physical activities [31].
The propensity of dapagliflozin to improve quality-of-life metrics, as shown by significant enhancements in KCCQ scores, reflects the broader impact of the medication beyond merely symptomatic relief. The observed improvements in scores related to physical limitation, symptom burden, and overall health perception emphasize the holistic benefits of this treatment. By significantly alleviating the symptomatic burden of heart failure, dapagliflozin appears to empower patients with better physical functioning and satisfaction with daily activities, possibly due to its role in fluid balance and energy optimization [32].
From a safety perspective, this study presents an encouraging profile for dapagliflozin in elderly patients, a group often laden with comorbidities and increased vulnerability to drug-related adverse effects [33]. The absence of increased incidents of serious adverse events such as DKA, severe hypoglycemia, and acute kidney injuries was particularly noteworthy, suggesting that dapagliflozin can be safely integrated into the therapeutic regimen for elderly patients with HFpEF.
The impact of dapagliflozin on renal function deserves special attention. The study noted a mild reduction in eGFR in the dapagliflozin group compared to the routine group. However, this reduction, while statistically significant, was not accompanied by a rise in clinically significant renal adverse events. This suggests a hemodynamic rather than structural basis for the renal changes observed with the dapagliflozin treatment, aligning with its pharmacodynamic effects on improved cardiac output and suppression of renal hyperfiltration.
The dapagliflozin treatment was met with higher satisfaction among our study participants, as noted by the increased satisfaction metrics. The high level of satisfaction could be reflective not only of the symptomatic relief experienced by patients but also their perception of safety and efficacy, underscored by the lack of serious side effects. Dapagliflozin’s influence on patient satisfaction also reinforces the importance of tailoring therapies that resonate with patient-centered care goals [26].
It was important to discuss the limitations inherent in our study. The retrospective nature of this analysis constrains our ability to establish firm causative conclusions. While we employed methods to balance baseline characteristics between groups, the inherent biases and confounding factors typical in non-randomized studies cannot be entirely ruled out. Furthermore, the study focused on an elderly cohort in a single hospital, thereby limiting generalizability to broader populations and settings.
Future research directions include randomized controlled trials specifically aimed at uncovering the long-term benefits and potential risks of dapagliflozin in diverse populations with HFpEF. Such studies could provide more robust evidence on the drug’s efficacy and safety, potentially guiding the implementation of therapy and standard care practices.
This study corroborates the growing evidence base supporting the use of dapagliflozin in HFpEF patients over 75 years of age, demonstrating significant improvements in cardiac function, exercise capacity, biochemical markers, and quality of life without introducing notable safety concerns. The multifactorial benefits of dapagliflozin, as evidenced by our findings, emphasize its potential as an integral component of heart failure management strategies, particularly in the complex and vulnerable elderly patient population.
All data included in this study are available from the corresponding author upon reasonable request.
JW conceived and designed the study, analyzed and interpreted the data, and drafted the manuscript. ZLL jointly completed the design of the study and contributed to data collection. GYZ supervised the project, analyzed the data, and finalized the manuscript. All authors contributed to revising the manuscript critically for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of it.
This study was approved by the Ethics Committee of Shandong Provincial Hospital Affiliated to Shandong First Medical University (Approval No. 2024-427) and informed consent was waived for this retrospective study because only de-identified patient data were utilized, and no risk or impact on patient care was anticipated. All procedures utilized in this study adhered to the ethical principles outlined in the Declaration of Helsinki.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


