1 Leeds Vascular Institute, Leeds Teaching Hospitals NHS Trust, LS1 3EX Leeds, UK
2 Leeds Institute of Clinical Trials Research, University of Leeds, LS2 9JT Leeds, UK
Abstract
Diabetes-related foot disease represents a growing burden on healthcare services. Peripheral arterial disease (PAD) is a contributing factor in over half of the cases, with treatment of severe PAD through revascularisation being crucial for limb salvage. The Society for Vascular Surgery Wound, Ischaemia, foot Infection (WIfI) classification system enhances diagnosis by providing a framework for identifying high-risk individuals needing intervention. Decision-making regarding which strategy of revascularisation (angioplasty or surgical bypass) is most suitable in these patients is complex, with decisions being primarily guided by the anatomical pattern of the disease, patient fitness, and availability of autologous vein. This review aims to provide a summary of the assessment and management of patients with diabetes-related foot disease and PAD, discussing the factors that influence patient selection for revascularisation. In general, short-segment disease is best treated by an endovascular-first approach, while long-segment occlusions are better managed with open surgery. Ultimately, individuals should be assessed and managed in conjunction with a multi-disciplinary team as part of a holistic management strategy.
Keywords
- diabetes mellitus
- peripheral arterial disease
- angioplasty
- bypass
Diabetes mellitus (DM) represents a significant health burden, affecting over half a billion individuals worldwide and is expected to rise by more than 50% in the next 25 years [1]. It is estimated that 19% to 34% of people with diabetes will develop a diabetic foot ulcer (DFU) during their lifetime [2], with one in five with a DFU requiring either minor (below the ankle) or major (above the ankle) lower limb amputation [3]. Notably, 10% of individuals with DFU will die within the first year of diagnosis, highlighting the substantial mortality risk within this population [4]. The economic impact is equally concerning; in the United Kingdom, approximately 0.9% of the National Health Service (NHS) England budget is spent treating DFU and its associated complications [5].
Peripheral arterial disease (PAD) is a significant contributing factor to morbidity in over 50% of DFU cases [6]. PAD is characterised by partial or complete occlusion of one or more peripheral arteries outside the heart and brain, affecting the upper and lower limbs [7]. In individuals with diabetes, PAD tends to be more diffuse and commonly associated with long-segment arterial occlusions, as opposed to the stenotic lesions typically seen in non-diabetes-related PAD [8]. In addition, individuals without diabetes typically exhibit proximal disease involving the aorto-iliac and superficial femoral arteries, whereas those living with diabetes are more likely to experience infra-geniculate disease. The distal peroneal, dorsalis pedal, and plantar arteries are often spared, making them suitable targets for revascularisation [9].
Identification and treatment of peripheral arterial disease are crucial for limb salvage in diabetes-related foot disease, yet decision-making related to treatment strategies remains complex. This review aims to provide a summary of the assessment and management of such patients, discussing the variety of factors that impact how to select patients for revascularisation, in the form of angioplasty or surgical bypass.
PAD most commonly presents with claudication, described as pain or aching in the calves, thighs, or buttocks during exertion, relieved with rest and reproducible with the same level of exercise. Chronic limb-threatening ischemia (CLTI) represents the end stage of PAD, where resting perfusion is insufficient to meet metabolic demands, manifesting as rest pain and/or tissue loss [8]. However, in patients with DM, the presence of neuropathy can mask the symptoms of both PAD and CLTI, creating significant diagnostic challenges [10]. All patients should be comprehensively examined, including pulse palpation and assessment of neuropathy and tissue loss [8].
Pulse palpation is a poor discriminator of PAD, therefore, other tools to assess
ischaemia should be implemented [11]. Currently, there is
no single bedside test, or combination of tests, with proven superior diagnostic
accuracy for PAD in people with diabetes. In patients with diabetes-related
neuropathy, medial arterial calcification can render the ankle-brachial
index (ABI) unreliable due to non-compressible vessels [12]. Toe
pressures and toe-brachial index (TBI) measurements are less affected by
incompressibility and therefore may be more sensitive in diagnosing severe
ischaemia, and are better predictors of amputation risk [13]. However, the ability of bedside tests to exclude disease is limited, and
normal results should be interpreted with caution. PAD is less likely in the
presence of ABI 0.9–1.3; TBI
The ischaemia threshold required for healing is not constant, varying with the extent of tissue loss and infection, prompting the development of the Wound, Ischaemia, foot Infection (WIfI) score using three key factors: wound characteristics (W), the degree of ischaemia (I), and the presence and severity of foot infection (fI) (Table 1; Ref. [14]). There are 4 grades of severity (0–3; none, mild, moderate, severe) for each component. Each component score is combined to grade the patient from clinical stages 1 to 4, with each successive stage correlating to an increased risk of major lower limb amputation and increased benefit of revascularisation. The WIfI classification is an objective, reproducible system used to stratify at-risk limbs across various populations, with high interobserver reliability and successful applications across different ethnicities [15]. WIfI has also been validated for use in patients with DFU, providing clinicians with a tool to estimate the potential benefit of revascularisation in patients with foot ulcers or gangrene, as well as predicting wound healing and the risk of amputation [16]. Its use in PAD and DFU is recommended by the 2019 Global Vascular Guidelines [8] and the 2023 Intersocietal Guidelines on PAD in patients with diabetes [17].
| Grade | Wound (W) | Ischaemia (I) | Foot infection (fI) | ||
| ABI | Ankle pressure (mmHg) | Toe pressure (mmHg) | |||
| 0 | Ischaemic rest pain; without frank ulcer or gangrene | Wound without purulence or manifestations of uninfected 1 infection | |||
| 1 | Minor tissue loss: small shallow ulceration |
0.60–0.79 | 70–99 | 40–59 | |
| 2 | Major tissue loss: deeper ulceration(s) with exposed bone, joint or tendon, ulcer 5–10 cm2 not involving calcaneus; gangrenous changes limited to digits. Salvageable with extensive forefoot surgery | 0.40–0.59 | 50–69 | 30–39 | Infection in patient who is systemically stable but has |
| 3 | Extensive ulcer/gangrene |
Infection in patient with systemic or metabolic toxicity (SIRS/sepsis) | |||
SIRS, systemic inflammatory response syndrome; ABI, ankle-brachial index. Adapted from Mills et al. [14], available under the Creative Commons CC-BY-NC-ND license.
In patients with ischaemia being considered for revascularisation, appropriate anatomical imaging is crucial to determine the presence and extent of arterial disease. Detailed imaging of tibial and pedal vessels is needed for intervention planning. Common modalities include arterial colour duplex ultrasound (CDUS), computed tomographic angiography (CTA), magnetic resonance angiography (MRA), and intra-arterial digital subtraction angiography (DSA). While these techniques enhance diagnostic accuracy, each has limitations: CDUS and CTA are often affected by severe medial arterial calcification, and MRA may not fully capture the calcification extent, which is critical for revascularisation planning. DSA remains the gold standard for imaging, particularly for below-knee disease, although it is generally reserved for cases where MRA or CTA is unavailable or inadequate for defining anatomy. Additionally, DSA is frequently used in patients likely to undergo endovascular interventions, providing precise imaging that aids in procedural planning, potentially allowing diagnostic imaging and intervention in the same sitting [8, 17].
The decision to revascularise patients with PAD and diabetes is complex. The current intersocietal guidelines broadly categorise recommendations to treat based on the severity of ischemia and the presence of gangrene into three groups: (1) DFU with clinical findings of non-severe ischaemia; (2) DFU with clinical findings of severe ischaemia; and (3) DFU with infection or gangrene involving any portion of the foot [17].
Non-severe ischaemia is characterised by findings indicative of PAD, such as
absent pulses, monophasic or absent pedal Doppler waveforms, ankle pressure
Delaying revascularisation for more than two weeks in DM increases the risk of
limb loss [19], and a shorter time to revascularisation
(
In patients who present with foot infection or gangrene, the priority is urgent assessment and consideration of immediate drainage of infection, since a delay in treatment can result in rapid tissue destruction and life-threatening sepsis [17]. For individuals with an abscess or infection of a deep foot compartment requiring urgent drainage, or where gangrene must be removed to control infection, immediate surgical intervention should be prioritised [23], in conjunction with broad-spectrum antibiotic therapy, tailored to tissue culture results. Once sepsis is controlled and the patient stabilises, prompt revascularisation should then be considered within a few days.
Modalities for revascularisation can be broadly classified into open, endovascular or hybrid procedures. Collectively, the goal should be the restoration of durable, in-line, pulsatile flow to the foot. An open procedure (surgical bypass) is where blood flow is re-routed through a conduit that connects a suitable proximal and distal vessel, thus bypassing the diseased segment of the artery. An endovascular procedure (angioplasty) is a minimally invasive procedure in which catheters, wires and stents/balloons are used to improve the flow through a vessel, without the need for multiple large incisions. A hybrid procedure combines both open and endovascular options.
The vascular surgeon assesses multiple core components which impact the decision to proceed and the modality of revascularisation: (1) the assessment of fitness and perceived life expectancy; (2) the anatomy of the PAD; (3) the presence of a suitable autologous vein.
This population is at higher risk of poor outcomes due to the combined effects of diabetes-related vascular complications and progressive PAD. Three prognostic models have been proposed for predicting survival benefits after revascularisation in those with CLTI: Bypass versus Angioplasty in Severe Ischaemia of the Leg (BASIL) Weibull [24]; PREVENT-III [25]; and FinVasc [26] (Table 2). However, their performance across the majority of validation studies suggests these models are less appropriate on an individual level, which is reflected in the Global Vascular Guidelines, which recommend that no specific tool and model can be recommended in preference to others [8].
| BASIL Weibull | PREVENT-III | FinVasc | |
| Variables used in model | Age | Age | Foot gangrene |
| Tissue loss | Tissue loss | Urgency of procedure | |
| Serum creatinine | Dialysis | Diabetes | |
| Ankle pressure | Coronary artery disease | Coronary artery disease | |
| History of ischaemic heart disease | Haematocrit value | ||
| History of cerebrovascular accident | |||
| Below-knee bollinger angiogram score | |||
| Body mass index | |||
| Smoking status | |||
| Outcome | Survival at 2-year post-procedure | Amputation-free survival at 1 year | Amputation-free survival at 30 days |
BASIL, Bypass versus Angioplasty in Severe Ischaemia of the Leg.
Ultimately, patients may not be suitable for revascularisation when there is no realistic prospect of wound healing, when major amputation is unavoidable, or if achieving a functional foot is unlikely due to limited life expectancy. In these cases, revascularisation may offer no benefit and could cause harm, especially in patients with high peri-procedural risk due to advanced disease and multiple co-morbidities. Specific groups that may be unsuitable include those who are frail, have poor functional status, are bed-bound, or have extensive tissue loss that makes the foot unsalvageable. Decisions should be personalised, considering patient preferences and involving the multidisciplinary team [17].
The Global Limb Anatomic Staging System (GLASS) provides a mechanism through which disease severity can be stratified to describe the likelihood of success of revascularisation (Fig. 1; Ref. [8, 27]). In general, disease patterns with a low GLASS score, usually short-segment stenosis or occlusions with otherwise patent vessels, can be managed with endovascular solutions. On the other hand, a higher GLASS score representing anatomically complex disease, such as multiple or long-segment occlusions, requires open or hybrid procedures to achieve in-line flow with acceptable rates of patency [8]. Unfortunately, disease patterns frequently fall into an indeterminate range in which decision-making is more challenging and may be influenced by locally available expertise.
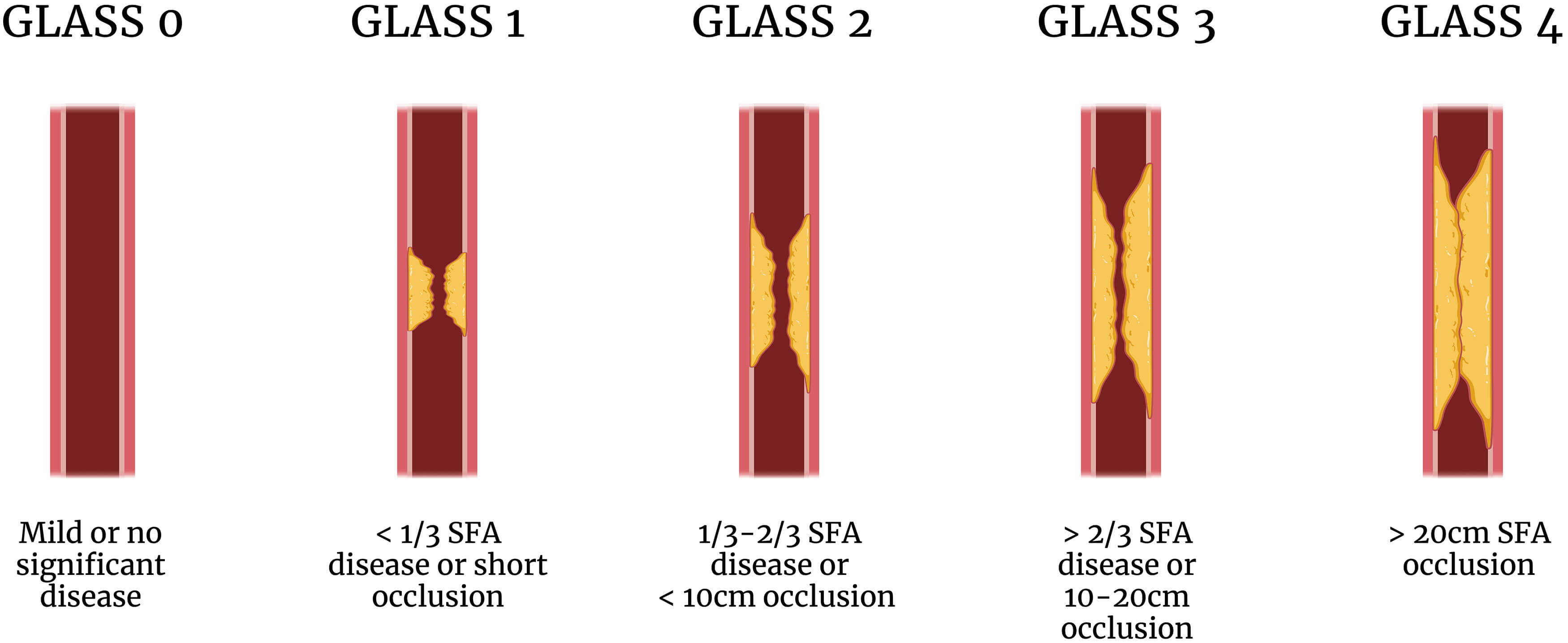 Fig. 1.
Fig. 1.
Simplified illustrative example of the GLASS classification system [8] using disease of the superficial femoral artery (SFA). GLASS, Global Limb Anatomic Staging System. Created in BioRender ([27]; https://BioRender.com/t95n377).
An alternative suggestion for using the anatomy of the PAD to guide the treatment modality is the angiosome theory. Angiosomes are an anatomical unit of tissue supplied by a specific source artery and drained by corresponding veins. In the ankle and foot, angiosomes can be topographically mapped into six territories [28] (Fig. 2; Ref. [29]), acting as anatomical targets for revascularisation. Direct revascularisation, implying revascularisation into an artery supplying the angiosomal area of tissue loss, may provide some benefit over indirect revascularisation (targeting an artery not directly supplying areas of tissue loss). A systematic review has found mixed results in this domain, with improved one-year healing rates for direct revascularisation through endovascular interventions (66.9–96.4% vs 54.6–85.7%) and open bypass (81–90.9% vs 41–68.5%). However, while most studies reported improved outcomes with direct revascularisation, results are heterogeneous, and the certainty of the evidence was found to be very low. Similarly, inconclusive results were found for amputation-free survival, overall survival, and limb salvage [30].
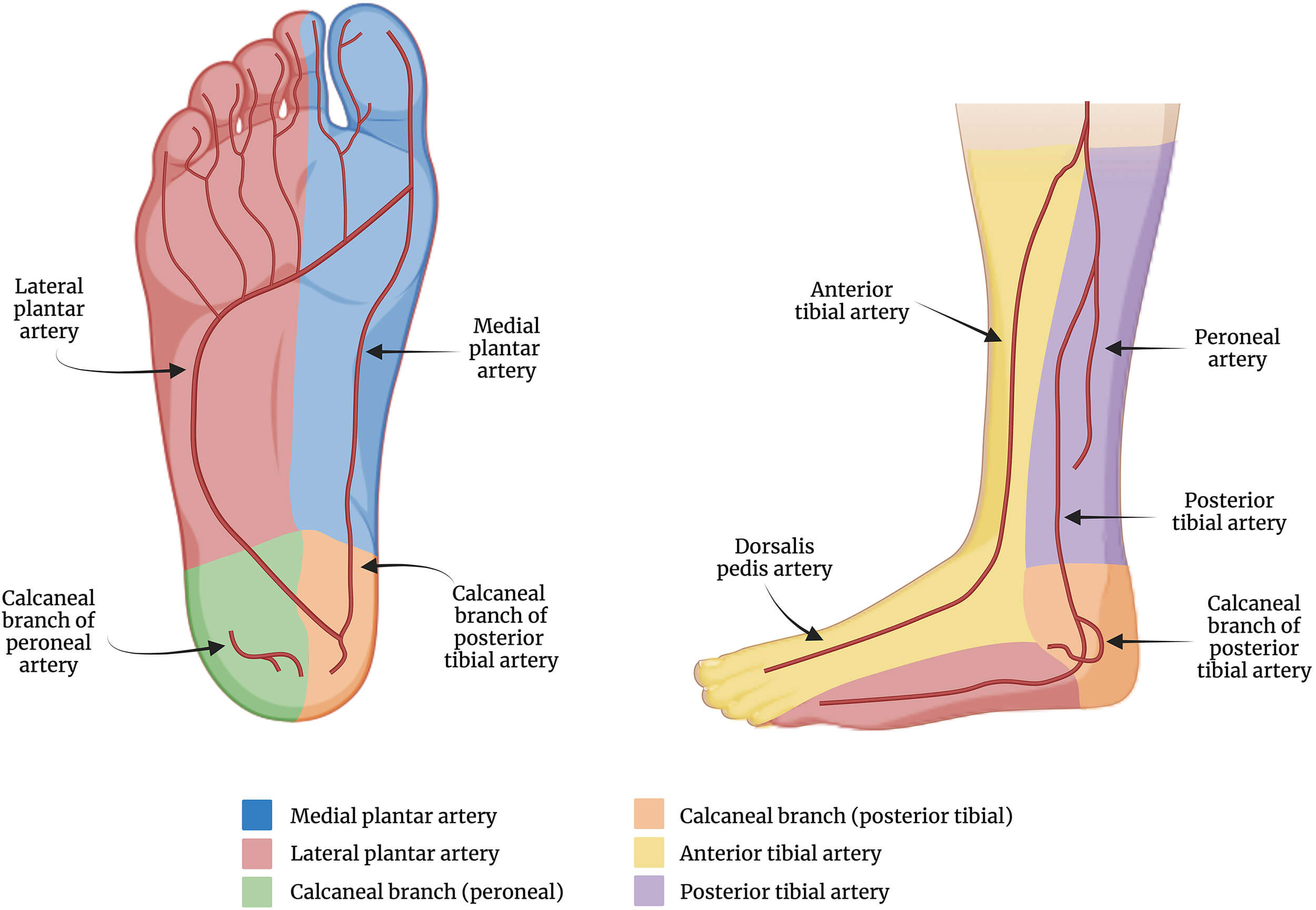 Fig. 2.
Fig. 2.
Anatomical distribution of the angiosomes of the foot and lower leg. Created in BioRender ([29]; https://BioRender.com/l73h427).
Surgical bypass can be accomplished by using an autologous vein or prosthetic material. The presence of a suitable venous conduit is a favourable factor when planning to undertake a bypass over an endovascular approach. The ipsilateral great saphenous vein (GSV) is most commonly used, with studies highlighting patency up to 80% at 5 years [31]. Other native vessels may be considered, including contralateral GSV, short saphenous vein, or upper limb veins. A Cochrane review found moderate-quality evidence favouring an improved long-term (5-year) primary patency for autologous vein grafts when compared to prosthetic materials for above-knee bypasses. However, for below-knee bypasses, there remains limited evidence that directly compares these conduit choices [32].
Multiple studies have attempted to address whether surgical bypass or
angioplasty is better. The BASIL trial randomised 452 participants with CLTI to
either a surgery-first approach using vein, or an endovascular-first
revascularisation strategy, for infrainguinal disease. 58% of participants had
DM. While 30-day outcomes were similar, surgery-first was associated with
improved amputation-free survival beyond 2 years [33]. This was reflected in a cost-effectiveness analysis, demonstrating an
increased cost of a surgery-first strategy (
The BEST-CLI trial explored whether angioplasty or open surgery was superior in
managing infrainguinal disease, measured by a composite primary outcome of major
adverse limb events or death. There was a high proportion of participants with
diabetes (69.3% of 1830). Participants with and without suitable autologous
veins were in separate cohorts. There was diversity of anatomical levels in which
bypasses were performed, including above-knee popliteal (15.3%), below-knee
popliteal (24.7%), and tibial and pedal vessel (51.4%) anastomoses, reflecting
the diversity of disease patterns often seen in DM. This identified a reduction
in the composite outcome in the surgical group in the presence of a suitable vein
(42.6% vs 57.4%; hazard ratio [HR] 0.68; p
Infrapopliteal disease represents a challenging pattern of disease strongly associated with comorbid DM. The BASIL-2 trial compared a bypass-first strategy against a balloon angioplasty-first strategy for the management of infrapopliteal disease in 345 patients, of which almost 70% had DM. This identified a significant improvement in amputation-free survival (47% vs 37%; HR 1.35; p = 0.037), largely driven by reduced mortality, in the endovascular-first group. However, challenges in patient recruitment and the prolongation of follow-up time led to the capture of mortality that was possibly not related to the intervention. Indeed, amputation rates and quality of life scores were similar. Further, TBI, WIfI scores and re-intervention rates were lower in the bypass-first cohort, suggesting that surgical intervention may provide improvements in peripheral perfusion and reduction in the need for subsequent admission or intervention [36]. While this trial generally supports an endovascular-first strategy, there may be a role for a bypass-first strategy in a carefully selected, low-risk subset of patients with severe ischaemia.
Whilst an aortoiliac pattern of disease is less associated with DM, many individuals also smoke tobacco and suffer from hyperlipidaemia, resulting in a less common but important cohort whose ischaemia is compounded by aortoiliac disease. The anatomic distribution of such disease is usually described using the TransAtlantic Inter-Society Consensus (TASC) classification. Traditionally, TASC C and D patterns were managed by open surgery. However, technological advances mean that many individuals are now treated with endovascular techniques. While open surgery has been associated with improved patency rates and limb salvage, the morbidity associated with open revascularisation of these lesions is high, leading many to prefer an endovascular first strategy [37]. No randomised control studies have yet been published in this domain; the Open revascularisation in severe oCClusive aorto-iliac disease (EVOCC) trial is recruiting participants to address this knowledge gap [38].
In the absence of conclusive randomised trials for either a surgery-first or an endovascular-first strategy, the ultimate decision to revascularise, and thus which method to use, relies upon a multi-disciplinary team approach with input from surgeons and interventionalists who can offer either treatment modality. Moreover, the ability to offer the optimal current method of patient selection, local expertise, availability of resources, and patient preference will also influence this decision-making.
The identification and treatment of peripheral artery disease is paramount for limb salvage in people with diabetes. Following clinical examination, non-invasive vascular assessment forms a critical step in the screening and prognostication of diabetic foot disease. The WIfI classification system provides a framework through which high-risk individuals in need of intervention can be easily identified. Revascularisation is an important determinant in the successful healing of those with significant lower limb ischaemia. The anatomic pattern of disease, presence of suitable venous conduit, and relative physiologic fitness of the individual are the chief determinants in the choice of revascularisation strategy. However, patient preference and technological availability also play critical roles. In general, short-segment disease should be managed with an endovascular-first strategy, whereas long-segment occlusions are better managed with open surgery. Where both modalities could be considered suitable for an individual, the presence of a suitable autologous vein should be considered, with discussion by a multi-disciplinary team including surgeons/interventionalists who can offer both options, to select the optimal treatment based upon local expertise and available resources.
All the data of this study are included in this article.
JG, CA and DR contributed equally to the design and content of this review. JG drafted the manuscript. All authors contributed to the important editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Christopher Ashmore and David Russell are supported in part by the National Institute for Health and Care Research (NIHR) Leeds Biomedical Research Centre (BRC) (NIHR203331). David Russell is also funded through an NIHR Advanced Fellowship (NIHR300633). The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health and Social Care.
This research received no external funding.
The Fig. 1 and Fig. 2 were created using BioRender. The authors have no financial or personal relationship with https://www.biorender.com, and the use of this tool does not imply any endorsement. The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


