1 Department of Laboratory Medicine, The First Affiliated Hospital, Zhejiang University School of Medicine, 310000 Hangzhou, Zhejiang, China
2 Zhejiang Key Laboratory of Clinical In Vitro Diagnostic Techniques, 310000 Hangzhou, Zhejiang, China
3 Institute of Laboratory Medicine, Zhejiang University, 310000 Hangzhou, Zhejiang, China
Abstract
Acute pesticide poisoning frequently results in secondary acute myocardial injury. Identifying predictive biomarkers, such as albumin-corrected anion gap (ACAG), is crucial for early intervention. This study aimed to explore the predictive value of ACAG for secondary acute myocardial injury in patients with acute pesticide poisoning.
A retrospective analysis was conducted on 205 patients with acute pesticide poisoning admitted between March 2017 and December 2024. Patients were classified into a myocardial injury group (n = 81) and a non-myocardial injury group (n = 124) based on the presence of secondary acute myocardial injury. Univariate and binary logistic regression analyses were employed to identify factors influencing secondary acute myocardial injury. Pearson or Spearman correlation analysis was used to analyze the correlation between variables, and receiver operating characteristic (ROC) curves were used to evaluate the predictive value of ACAG.
No statistically significant differences were observed between the two groups in terms of age, gender, body mass index (BMI), pesticide type, admission time, hemoglobin (Hb), white blood cell (WBC) count, platelet count (PLT), renal insufficiency, or admission heart rate (p > 0.05). The myocardial injury group had significantly higher anion gap (AG) and ACAG values, incidence of respiratory failure, Acute Physiology and Chronic Health Evaluation II (APACHE II) scores, and number of severe poisoning cases compared to the non-myocardial injury group (p < 0.05). Albumin levels were significantly lower in the myocardial injury group (p < 0.05). Correlation analysis revealed that ACAG was negatively correlated with albumin (r = –0.528, p < 0.05) and positively correlated with AG and APACHE II scores (r = 0.974, 0.365, p < 0.05). Respiratory failure was negatively correlated with albumin (r = –0.160, p < 0.05) and positively correlated with APACHE II scores (r = 0.206, p < 0.05). Severe poisoning showed a negative correlation with albumin (r = –0.217, p < 0.05) and positive correlations with ACAG and AG (r = 0.230, 0.199, p < 0.05). Binary logistic regression analysis showed that ACAG (odds ratio (OR) = 1.586, p < 0.001), respiratory failure (OR = 4.432, p = 0.001), APACHE II score (OR = 1.242, p < 0.001), and severe poisoning (OR = 3.472, p = 0.008) were significant influencing factors for secondary acute myocardial injury in patients with acute pesticide poisoning. ROC analysis results showed that ACAG had an area under the curve (AUC) of 0.859 (95% confidence interval [CI]: 0.809–0.909, p < 0.001), with a standard error of 0.025, a Youden index of 0.55, sensitivity of 59.26%, and specificity of 95.97%. Patients with ACAG ≤24.54 had significantly higher survival probability and better post-treatment cardiac function indicators than those with ACAG >24.54 (p < 0.05).
ACAG has strong predictive value for secondary acute myocardial injury in patients with acute pesticide poisoning. It is recommended that ACAG be routinely assessed upon admission in these patients.
Keywords
- albumin
- acute myocardial injury
- anion
- pesticide
The frequent occurrence of pesticide poisoning and its associated high fatality rate pose a serious threat to the health of rural residents [1]. Rural hospitals admit hundreds of patients annually for pesticide poisoning, with a mortality rate as high as 15–30% [2, 3]. This alarming situation not only threatens the lives of farmers but also places immense pressure on the rural healthcare system. Patients poisoned with pesticides, especially paraquat and diquat, experience severe pain accompanied by symptoms such as nausea, vomiting, abdominal pain, and diarrhea. Additionally, the respiratory system is severely affected. Following pesticide poisoning, patients may develop progressive respiratory distress and cyanosis due to pesticide-induced damage to the structural and functional integrity of the lungs, leading to pulmonary edema, fibrosis, and respiratory failure. In severe cases, mechanical ventilation may be required within a short period to sustain life [4, 5].
Management of pesticide poisoning involves the rapid removal of toxic substances, including the removal of contaminated clothing and skin decontamination. In cases of oral ingestion, vomiting and gastric lavage are necessary. Specific antidotes, such as atropine and tripralidoxime, should be administered according to the poisoning agent, for organophosphorus poisoning, while closely monitoring and supporting vital signs. Blood purification therapy should be considered when clinically indicated.
In this study, pesticide poisoning detection and analysis were performed using
Agilent 6495 high-performance liquid chromatography-tandem triple quadrupole mass
spectrometer (Agilent Technologies, Santa Clara, CA, USA). Plasma samples were
pretreated with methanol protein precipitation. An acetonitrile-water gradient
mobile phase was employed with a flow rate of 0.4 mL
The albumin-corrected anion gap (ACAG), a clinical indicator reflecting renal function and acid-base balance status, has garnered increasing attention in recent years. The anion gap, defined as the difference between unmeasured cations and anions in the blood, is widely used to assess acid-base imbalances [6]. As albumin is a negatively charged protein, fluctuations in its concentration can influence the anion gap calculation. Therefore, correcting the anion gap for albumin levels yields more accurate diagnostic insights and better informs clinical management. Several studies have explored the use of ACAG in cardiac-related conditions [7, 8]. However, research on the predictive value of ACAG for myocardial injury due to pesticide poisoning remains limited. This study aimed to investigate the relationship between ACAG and secondary myocardial injury and to evaluate its predictive performance. The goal was to uncover potential associations between ACAG and acute myocardial injury, thereby providing new insights and methods for early intervention and prognostic assessment in patients with pesticide poisoning.
This study included 16 variables. According to the principle that the sample size should be 5–20 times the number of variables, and considering a 20% missing data rate, the required sample size was estimated to range between 96 and 384. Due to the limitations inherent in a retrospective design, achieving complete double-blinding was not feasible. However, data extraction and outcome assessment were independently performed by investigators blinded to group allocation to minimize bias. Screening and enrollment are shown in Fig. 1.
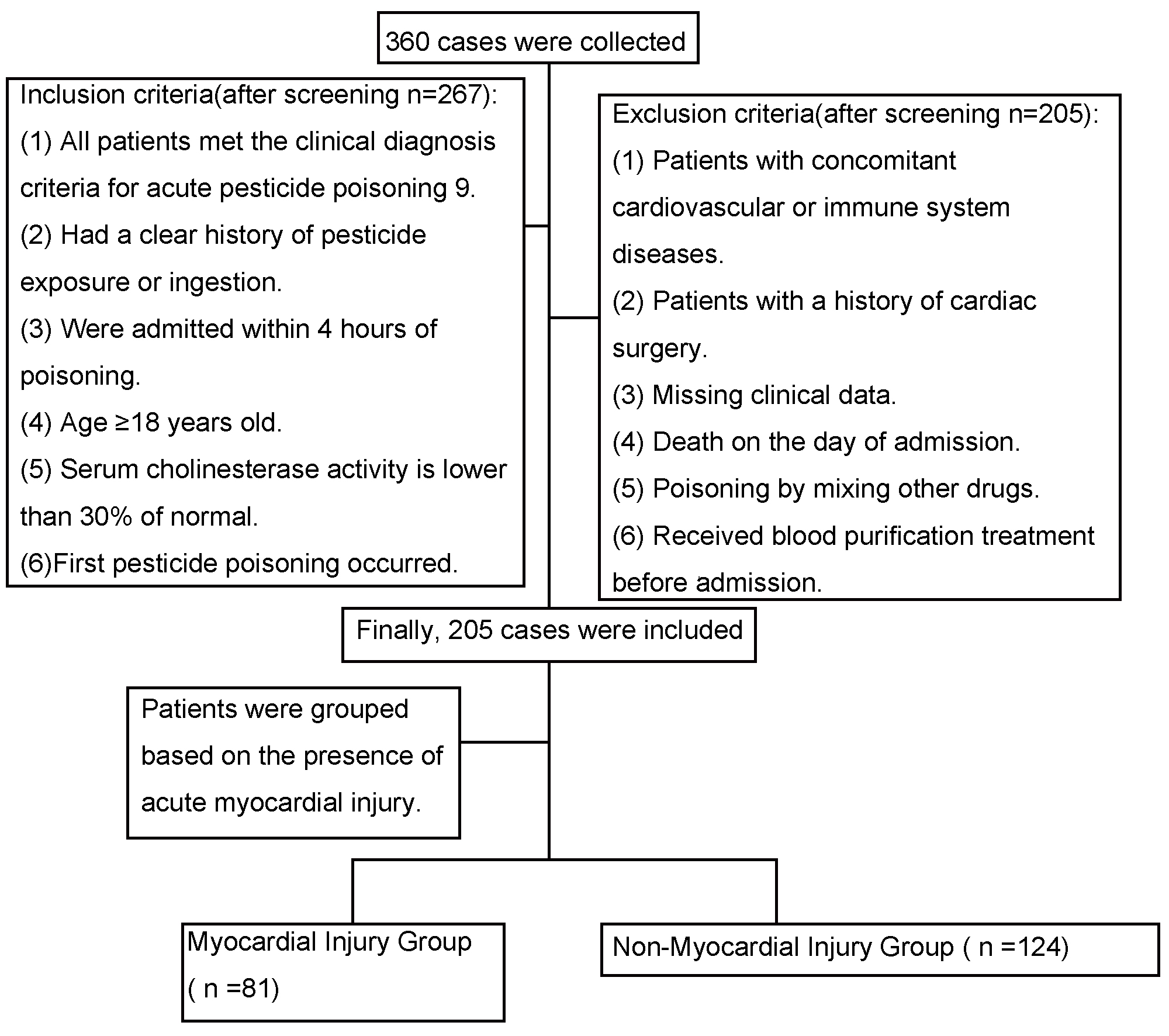 Fig. 1.
Fig. 1.
Study flowchart. The figure was created using Microsoft Word (version 2016, Microsoft Corporation, Redmond, WA, USA).
A total of 205 patients with acute pesticide poisoning (paraquat or diquat)
admitted to the First Affiliated Hospital, Zhejiang University School of
Medicine, between March 2017 and December 2024 were enrolled. Patients were
classified into the myocardial injury group (n = 81) and the non-myocardial
injury group (n = 124) based on the occurrence of secondary acute myocardial
injury. The inclusion criteria were as follows: (1) Patients met the clinical
diagnostic criteria for acute pesticide poisoning [9]; (2) A clear history of pesticide exposure or ingestion; (3) Hospital
admission within 4 hours of poisoning; (4) Age
Two days after admission, if the following two criteria are met, secondary myocardial injury was diagnosed two days after admission if two or more of the following criteria were: (1) Clinical symptoms such as chest tightness, palpitations, or precordial discomfort; (2) Cardiac enzyme levels at least two times higher above the normal reference range; (3) New electrocardiographic abnormalities, including ST-segment and T-wave (ST-T) segment alterations, frequent premature ventricular contractions, or conduction blocks [10].
The Poison Severity Score (PSS) was developed between 1990 and 1994 by the
European Association of Poison Centers and Clinical Toxicologists
(EAPCCT) [11], in collaboration with the International
Program on Chemical Safety (IPCS) and the European Commission, as a standardized
and reliable scoring system to describe the severity of poisoning. The score does
not provide prognostic information. PSS grades poisoning severity as follows: 0 =
no symptoms; 1 = mild; 2 = moderate; 3 = severe; and 4 = fatal. Therefore, all
deceased patients are assigned a score of 4. In this study, patients with a PSS
score
Within 24 hours of hospitalization, blood gas analysis, complete blood count, and biochemical parameters were measured. The albumin-corrected anion gap (ACAG) and anion gap (AG) were calculated using the following formulas [12]:
The Acute Physiology and Chronic Health Evaluation II (APACHE II) [13] comprises three components: the acute physiology score, age score, and chronic health score. A higher total score indicates a more severe condition and an increased risk of mortality.
Respiratory failure [14] is defined as Partial Pressure
of Oxygen in Arterial blood (PaO2)
Patients’ echocardiograms before treatment and after treatment and survival within 6 months after discharge were collected.
General patient information was retrieved from the hospital’s electronic medical record system.
The collected experimental data were analyzed using SPSS version 27.0 (IBM
Corporation, Armonk, NY, USA). Normality was tested using the Shapiro-Wilk test.
For normally distributed continuous variables, data were presented as mean
Comparisons of age, gender, body mass index (BMI), pesticide type, time to
admission, hemoglobin (Hb), white blood cell (WBC) count, platelet count (PLT),
renal dysfunction, and admission heart rate revealed no statistically significant
differences between groups (p
| Variable | Myocardial injury group (n = 81) | Non-myocardial injury group (n = 124) | t/ |
p-value | |
| Age (years) [ |
45.85 |
43.85 |
1.660 | 0.098 | |
| Gender [n (%)] | 0.009 | 0.926 | |||
| Male | 38 | 59 | |||
| Female | 43 | 65 | |||
| BMI (kg/m2) [n (%)] | 0.273 | 0.601 | |||
| 50 | 72 | ||||
| 31 | 52 | ||||
| Pesticide type [n (%)] | 0.012 | 0.914 | |||
| Paraquat | 30 | 45 | |||
| Diquat | 51 | 79 | |||
| Time to admission (hours) [n (%)] | 3.462 | 0.063 | |||
| 33 | 35 | ||||
| 48 | 89 | ||||
| APACHE II score [ |
29.03 |
24.07 |
11.162 | ||
| Severe poisoning [n (%)] | 16.156 | ||||
| Yes | 41 | 29 | |||
| No | 40 | 95 | |||
| ACAG (mmol/L) [ |
25.14 |
20.12 |
11.407 | ||
| AG (mmol/L) [ |
20.72 |
16.70 |
9.572 | ||
| Albumin (g/L) [ |
26.33 |
30.33 |
8.724 | ||
| Hb (g/L) [ |
110.73 |
111.35 |
0.422 | 0.674 | |
| WBC count ( |
13.11 |
13.40 |
0.906 | 0.366 | |
| PLT ( |
192.20 |
193.94 |
0.682 | 0.496 | |
| Respiratory failure [n (%)] | 6.780 | 0.009 | |||
| Yes | 32 | 28 | |||
| No | 49 | 96 | |||
| Renal dysfunction [n (%)] | 0.628 | 0.428 | |||
| Yes | 11 | 22 | |||
| No | 70 | 102 | |||
| Admission heart rate [n (%)] | 0.647 | 0.421 | |||
| 20 | 37 | ||||
| 61 | 87 | ||||
Note: BMI, body mass index; APACHE II, Acute Physiology and Chronic Health Evaluation II; Hb, Hemoglobin; WBC, white blood cell; PLT, platelet count; AG, anion gap; ACAG, albumin-corrected anion gap.
According to correlation analysis, ACAG was negatively correlated with albumin
(r = –0.528, p
| Variable | APACHE II | ACAG | AG | Albumin | Severe poisoning | Respiratory failure | |
| APACHE II | r | 1 | 0.365 | 0.334 | –0.273 | 0.067 | 0.206 |
| p | - | 0.339 | 0.003 | ||||
| ACAG | r | 0.365 | 1 | 0.974 | –0.528 | 0.230 | 0.124 |
| p | - | 0.001 | 0.076 | ||||
| AG | r | 0.334 | 0.974 | 1 | –0.323 | 0.199 | 0.096 |
| p | - | 0.004 | 0.172 | ||||
| Albumin | r | –0.273 | –0.528 | –0.323 | 1 | –0.217 | –0.160 |
| p | - | 0.002 | 0.022 | ||||
| Severe poisoning | r | 0.067 | 0.230 | 0.199 | –0.217 | 1 | 0.029 |
| p | 0.339 | 0.001 | 0.004 | 0.002 | - | 0.678 | |
| Respiratory failure | r | 0.206 | 0.124 | 0.096 | –0.160 | 0.029 | 1 |
| p | 0.003 | 0.076 | 0.172 | 0.022 | 0.678 | - |
Multicollinearity analysis was performed on significant variables identified in
univariate analysis, leading to the exclusion of AG and albumin. ACAG,
respiratory failure, APACHE II score, and severe poisoning were used as
independent variables in a binary logistic regression model, with the occurrence
of secondary acute myocardial injury in patients with acute pesticide poisoning
as the dependent variable (myocardial-injury = 1; non-myocardial-injury = 0). The
results of the binary Logistics regression analysis indicated that ACAG
(OR = 1.586 (1.358, 1.853)), respiratory failure (OR = 4.432 (1.838, 10.685)), APACHE II
(OR = 1.242 (1.118, 1.380)), and severe poisoning (OR = 3.472 (1.392, 8.664)) were
influencing factors for the occurrence of secondary acute myocardial injury in
patients with acute pesticide poisoning (p
| Influencing variable | Assignment |
| ACAG | Continuous (original value) |
| Respiratory failure | No = 0, Yes = 1 |
| APACHE II | Continuous (original value) |
| Severe poisoning | No = 0, Yes = 1 |
| Variable | SE | Wald | p-value | Exp (β) | 95% CI | ||
| Lower limit | Upper limit | ||||||
| APACHE II | 0.217 | 0.054 | 16.278 | 1.242 | 1.118 | 1.380 | |
| ACAG | 0.462 | 0.079 | 33.830 | 1.586 | 1.358 | 1.853 | |
| Severe poisoning (Yes) | 1.245 | 0.467 | 7.120 | 0.008 | 3.472 | 1.392 | 8.664 |
| Respiratory failure (Yes) | 1.489 | 0.449 | 10.996 | 0.001 | 4.432 | 1.838 | 10.685 |
| Constant | –17.747 | 2.430 | 53.344 | - | - | ||
CI, confidence interval; SE, standard error.
The ROC analysis showed that the area under the curve (AUC) for ACAG was 0.859
(p
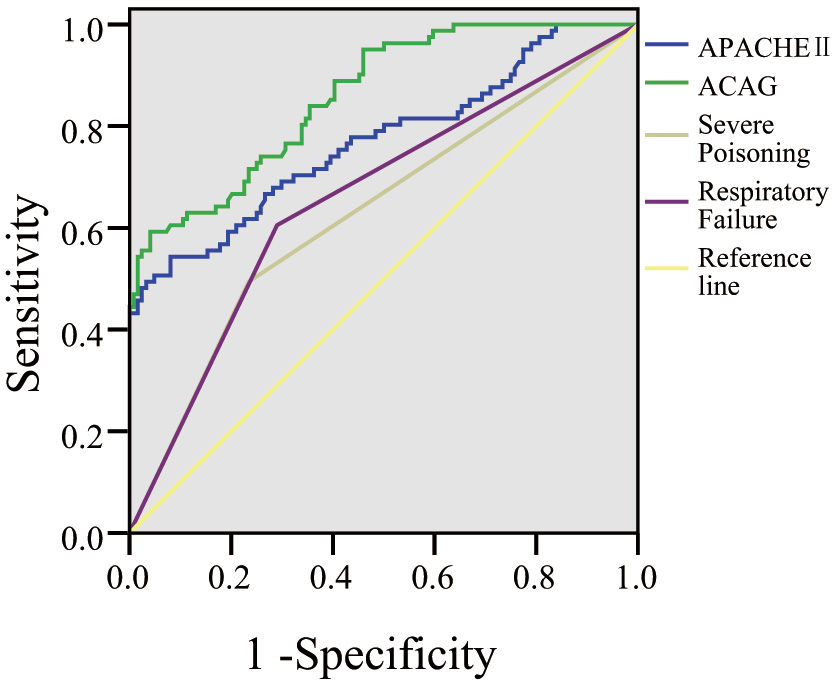 Fig. 2.
Fig. 2.
Receiver operating characteristic (ROC) curve analysis.
| Variable | AUC | Standard error | 95% CI | Youden index | Sensitivity (%) | Specificity (%) | p-value | Cutoff value |
| APACHE II | 0.770 | 0.035 | 0.701–0.839 | 0.46 | 54.32 | 91.94 | 29.12 | |
| ACAG | 0.859 | 0.025 | 0.809–0.909 | 0.55 | 59.26 | 95.97 | 24.54 | |
| Severe poisoning | 0.630 | 0.041 | 0.551–0.709 | 0.26 | 49.38 | 76.61 | 0.002 | - |
| Respiratory failure | 0.657 | 0.040 | 0.580–0.735 | 0.31 | 60.49 | 70.97 | - |
AUC, area under the curve; ROC, receiver operating characteristic.
The survival probability in the group with ACAG
| Group | ACAG |
ACAG |
p-value | |
| Survival rate (%) | 70.39 | 39.62 | 5.452 | 0.020 |
| Survived (n) | 107 | 21 | - | - |
| Deceased (n) | 45 | 32 | - | - |
Before treatment, no statistically significant differences were observed in
cardiac function indicators between the groups (p
| Group | Count (n) | LVEDD (mm) | LVESD (mm) | LVEF (%) | |||
| Before | After | Before | After | Before | After | ||
| ACAG |
152 | 50.32 |
43.85 |
38.58 |
32.12 |
43.85 |
62.85 |
| ACAG |
53 | 50.55 |
47.88 |
37.95 |
35.77 |
43.28 |
57.55 |
| t-value | 0.270 | 5.838 | 1.047 | 7.055 | 0.788 | 4.708 | |
| p-value | 0.787 | 0.296 | 0.431 | ||||
Note: *p
LVEDD, left ventricular end diastolic diameter; LVESD, left ventricular end systolic diameter; LVEF, left ventricular ejection fraction.
In this study, clinical data from 205 patients with acute pesticide poisoning were analyzed to investigate the predictive value of ACAG for secondary acute myocardial injury. The results indicate that ACAG is a valuable indicator for predicting secondary acute myocardial injury in such patients, demonstrating high sensitivity and specificity.
Significant differences were observed between the two groups in ACAG, respiratory failure, APACHE II score, and poisoning severity. Binary logistic regression analysis confirmed that ACAG, respiratory failure, APACHE II score, and severity of poisoning are independent influencing factors for the development of secondary acute myocardial injury in patients with acute pesticide poisoning. A previous study by Aydın and Aksakal [16] also indicated a significant association between elevated ACAG and increased mortality rate in hospitalized heart failure patients. These findings are consistent with the current study, providing additional validation for the current research. These findings suggest that ACAG, respiratory failure, APACHE II score, and poisoning severity are key factors contributing to the risk of secondary acute myocardial injury following pesticide poisoning. Several potential mechanisms may explain these associations. First, ACAG reflects electrolyte balance. Pesticide poisoning often leads to electrolyte imbalances, particularly acid-base disturbances. As a sensitive indicator of these imbalances, changes in ACAG may reflect variations in intracellular and extracellular ion concentrations in myocardial cells, contributing to myocardial injury.
Second, ACAG is related to inflammatory responses. Pesticide exposure triggers systemic inflammation, and changes in albumin levels may indicate the severity and duration of this response [17]. Since ACAG adjusts the anion gap corrected for albumin, it may indirectly reflect the inflammatory response status of the body, thereby affecting myocardial outcomes [18]. Third, ACAG is linked to renal function. Pesticide-induced nephrotoxicity may impair acid-base balance. By correcting for albumin levels, ACAG offers a more accurate reflection of the renal function status on the anion gap, which is closely associated with myocardial injury [19, 20]. Lastly, ACAG directly correlates with the severity of poisoning. Patients with severe poisoning show significantly higher ACAG values than those with mild poisoning, indicating that ACAG may serve as a sensitive marker for identifying patients at higher risk of myocardial injury.
Further correlation analysis revealed that ACAG is negatively correlated with albumin (r = –0.528) and positively correlated with anion gap (AG), APACHE II score and severe Poisoning (r = 0.974, 0.365, 0.230). These correlation results suggest that variations in ACAG are closely related to the overall severity of the patient’s condition, with a particularly strong correlation with the APACHE II score, which reflects the clinical severity. This further supports the feasibility of using ACAG as a predictive indicator for secondary acute myocardial injury in patients with acute pesticide poisoning. Although ACAG demonstrates high specificity and may help reduce unnecessary medical interventions, its sensitivity is relatively low. This may be due to its stronger emphasis on accurately identifying patients without acute myocardial injury, which may limit its potential to capture certain characteristics of patients with acute myocardial injury. Consequently, some cases may be missed during screening, which limits its effectiveness as a universal clinical screening tool.
To evaluate the predictive value of ACAG for acute myocardial injury, this study employed ROC curve analysis. The findings indicate that ACAG has the highest predictive value, with an AUC of 0.859 (95% CI: 0.809–0.909), a standard error of 0.025, and a Youden index of 0.55. At this threshold, the sensitivity was 59.26%, and the specificity was 95.97%. A previous study by Lu et al. [21] also demonstrated that ACAG performs well in predicting in-hospital mortality in patients with acute myocardial infarction, which is consistent with the results of this study. These findings suggest that ACAG not only exhibits high predictive accuracy but also excels in specificity. High specificity indicates that, in clinical practice, ACAG can accurately identify patients without acute myocardial injury, thereby helping avoid unnecessary medical interventions and optimizing the use of medical resources.
Furthermore, this study compared the survival probability and post-treatment
cardiac function indicators of patients across different ACAG levels. The results
showed that the survival probability in the ACAG
Based on the findings of this study, several clinical implications can be proposed. First, in patients with elevated ACAG, more intensive cardioprotective interventions, such as optimizing electrolyte balance or administering antioxidant therapy, may yield significant benefits. For example, dynamic monitoring of ACAG levels may guide the targeted use of antioxidants (e.g., N-acetylcysteine) or inform the application of mechanical ventilation to improve oxygenation in patients with respiratory failure, thereby reducing the risk of myocardial injury. Moreover, emerging therapeutic approaches such as exome-delivered circular RNAs (circRNAs) offer promising new directions for myocardial protection. Hu et al. [23] reported that adipose-derived exosomal circ-0008302 could mitigate oxidative stress-induced myocardial damage by targeting the Mir-466i-5p/MStrA pathway, suggesting that RNA-based exosome therapy may represent a novel treatment strategy for patients with elevated ACAG.
Current standard treatments for acute myocardial injury, such as beta blockers
and Angiotensin Converting Enzyme (ACE) inhibitors, are not yet stratified based
on ACAG levels. Future studies should explore the potential value of ACAG as a
treatment stratification biomarker. For instance, whether patients with ACAG
The results of this study suggest that ACAG holds promising potential for predicting secondary acute myocardial injury in patients with acute pesticide poisoning. However, certain limitations should be acknowledged. First, the sample size was relatively small, which may impact the accuracy and generalizability of the results. Future research should include larger-scale, multicenter studies for further validation. Second, this study did not assess the dynamic changes in ACAG over time. Continuous monitoring may better capture disease progression and treatment outcomes. Additionally, this study did not employ statistical methods such as the DeLong test to compare the AUC values of ACAG with other predictors (e.g., APACHE II score). Without such comparisons, it is challenging to determine whether the differences are statistically significant. The current results only suggest a certain degree of predictive value, and more conclusive evidence is needed. Future studies should focus on expanding the sample size and incorporating dynamic monitoring to enhance the predictive accuracy and clinical utility of ACAG in the management of patients with acute pesticide poisoning.
In conclusion, this study confirms the predictive value of ACAG for secondary acute myocardial injury in patients with acute pesticide poisoning through in-depth analysis of clinical data. ACAG is closely associated with the overall severity of the patient’s condition and demonstrates excellent specificity. Additionally, ACAG levels are strongly linked to patient prognosis. With continued research and advances in clinical practice, broader applications of ACAG in the management of acute pesticide poisoning are anticipated. Simultaneously, the ongoing development and refinement of other relevant indicators and diagnostic methods will be essential to further improve patient outcomes and enhance the overall quality of care for patients with acute pesticide poisoning.
The data used to support the findings of this study are available from the corresponding author upon request.
JW: conceptualization, methodology, validation, formal analysis, investigation, data curation, writing—original draft, writing—review & editing, visualization; BL: methodology, formal analysis. Both authors contributed to revising the manuscript critically for important intellectual content. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was approved by the Clinical Research Ethics Committee of the First Affiliated Hospital, Zhejiang University School of Medicine (approval number: IIT20230383B-R1), which also granted a waiver of written informed consent. A retrospective analysis was conducted on the cases of paraquat and diquat poisoning from March 2017 to December 2024. The risk to the subjects in this study is no greater than the minimum risk. Waiving informed consent will not have an adverse impact on the rights and health of the subjects. All procedures were conducted following the principles outlined in the Declaration of Helsinki.
We thank the Department of Laboratory Medicine, the First Affiliated Hospital, Zhejiang University School of Medicine, and Zhejiang Key Laboratory of Clinical In Vitro Diagnostic Techniques for technical assistance.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


