1 Department of Gastroenterology, General Hospital of Northern Theater Command, 110840 Shenyang, Liaoning, China
2 Postgraduate College, China Medical University, 110167 Shenyang, Liaoning, China
3 Department of Gastroenterology, The 963rd Hospital of the Joint Logistics Support Force of the Chinese People’s Liberation Army, 154002 Jiamusi, Heilongjiang, China
Abstract
The detection rate of isolated terminal ileal ulcers (ITIU) is increasing during colonoscopy. However, its clinical significance remains unknown. This study aimed to explore the detection rate and risk factors of ITIU in individuals undergoing colonoscopy and evaluate the outcomes of patients with nonspecific ITIU.
Overall, 11,504 consecutive individuals who underwent colonoscopy at Department of Gastroenterology, General Hospital of Northern Theater Command between 1 July 2021 and 31 December 2022 were retrospectively screened. Among the individuals who completed terminal ileum (TI) intubation, the detection rate of ITIU was calculated. Furthermore, among the individuals with complete baseline data, logistic regression analyses were performed to identify the independent factors associated with ITIU. At least one-year follow-up outcome after the index colonoscopy was evaluated in patients with nonspecific ITIU who received and did not receive empiric therapy.
Overall, 9649 individuals completed TI intubation with an ITIU detection rate of 1.3% (123/9649). Among them, 1709 individuals had complete baseline data and were further selected in the risk factor analysis. Compared with the non-ITIU group, the ITIU group was significantly younger (56 vs. 46, p = 0.010). Multivariate logistic regression analyses also demonstrated that age ≤50 years (adjusted odds ratio [aOR] = 1.947, 95% confidence interval [CI] = 1.056–3.587, p = 0.033) was independently associated with ITIU. There was no significant difference in clinical symptoms (p = 1.000) or colonoscopic findings (p = 0.560) between patients with nonspecific ITIU who received empiric therapy and observational follow-up.
ITIU is not rare during colonoscopy. Young individuals may have a higher probability of suffering from ITIU. Empiric therapy may not be necessary for patients with nonspecific ITIU.
Keywords
- ileal diseases
- Crohn disease
- age
- body mass index
- inflammatory bowel disease
Terminal ileum (TI) refers to the intestinal tract from the ileocecal valve to the proximal end of the ileum, and its length is about 30–40 cm [1]. Feces tend to accumulate at the TI, more probably causing bacterial growth; meanwhile, there is a large amount of intestinal lymphoid tissue and Peyer’s patches within the TI [2, 3]. Therefore, it is more likely to develop ulcers in this region. Notably, if there was no ulcer at any other part of the intestines, isolated terminal ileal ulcers (ITIU) would be defined [1]. It has been reported that the detection rate of ITIU among individuals undergoing colonoscopy screening is 1.45% [4]. Among individuals with gastrointestinal symptoms, the detection rate of ITIU was observed to be 4.90% [5]. Until now, the etiology of ITIU has not been elucidated. It may be associated with infections, drugs, inflammatory bowel disease (IBD), eosinophilic gastroenteritis, ischemic bowel disease, and neoplasms [6, 7, 8, 9]. It has been reported that 1.07–34.5% of individuals with isolated terminal ileitis and ITIU may progress to Crohn disease (CD) during a long-term follow-up period, and the incidence of CD gradually increases over time [5, 8, 10, 11, 12]. Some investigators also suggest that nonspecific ITIU should be considered as an early stage of CD [5, 11, 12, 13], but it usually takes a relatively long time to progress from nonspecific ITIU to CD. Thus, early treatment and close monitoring may be required [5, 6, 8, 12]. However, there are no specific guidelines for the diagnosis and management of ITIU, because it is usually occult, asymptomatic, or nonspecific in symptoms.
Considering the clinical significance of ITIU, it is critical to understand the epidemiology of ITIU and recognize high-risk populations for developing ITIU. It has been reported that body mass index (BMI) may be associated with the number of small intestinal ulcers, but the association of BMI with ITIU was uncertain [14]. Additionally, other potential risk factors associated with ITIU have not been identified. The present study aimed to analyze the detection rate of ITIU and its associated risk factors to strengthen the knowledge of this disease and to explore the outcomes of nonspecific ITIU.
We retrospectively screened the medical records of 11,504 consecutive individuals undergoing colonoscopy at the Department of Gastroenterology, General Hospital of Northern Theater Command, between 1 July 2021 and 31 December 2022. The inclusion criteria were the individuals who underwent colonoscopy at our department between 1 July 2021 and 31 December 2022. The exclusion criteria were as follows: (1) incomplete colonoscopy due to endoscopic therapy alone, endoscopic ultrasonography alone, or poor quality of bowel preparation, etc.; (2) history of ileocecal resection; (3) failed cecal intubation due to colonic stenosis from colorectal cancer or severe IBD, patients’ intolerable pain, or colonic redundancy; and (4) successful cecal intubation but without TI intubation. At the Department of Gastroenterology, TI intubation is routinely performed during colonoscopy. However, if the TI intubation is technically difficult or unsafe, endoscopists would not choose to attempt it. The study protocol was approved by the medical ethical committee of General Hospital of Northern Theater Command [Approval number: Y (2023)196]. All of our patients signed their informed consent forms for colonoscopy before undergoing the examination. As mentioned in the ethical approval document of our study protocol, considering the retrospective nature of this study, the use of anonymous data, the lack of direct intervention measures, and the extremely minimal risk to patients, the medical ethics committee of the General Hospital of the Northern Theater Command has waived the requirement for the patients’ written informed consents. This is in accordance with the principles outlined in the Declaration of Helsinki.
Data were collected if they were reported in the medical records [15], including demographics such as gender, age, and BMI; type of patient such as outpatient, inpatient; clinical symptoms such as abdominal pain, diarrhea gastrointestinal bleeding, abdominal distension/discomfort, constipation, formless stool, and screening/surveillance; endoscopic findings such as TI ulcer, ITIU, ileocecal valvulitis/colitis, colonic diverticulosis, and colonic polyp; and diagnoses such as CD, ulcerative colitis (UC), lymphoma, and ankylosing spondylitis. Histology of the ITIU was collected, and information regarding the management of nonspecific ITIU was also collected, mainly including empiric therapy and observational follow-up. Telephone follow-up at least one year after the index colonoscopy was performed in patients with nonspecific ITIU to collect clinical symptoms, colonoscopic findings during re-examination, and follow-up outcomes [1].
ITIU was defined as ulcers occurring only in the terminal ileum during colonoscopy with TI intubation.
The detection rate of ITIU was defined as the percentage of individuals who were diagnosed with ITIU among the individuals with successful TI intubation.
The number of ulcers was defined as single and multiple. Single ulcer was defined as only one ulcerative lesion was found at the TI during colonoscopy. Multiple ulcers were defined as two or more ulcerative lesions were found at the TI during colonoscopy.
Individuals with a BMI of
Normality was evaluated using the Kolmogorov-Smirnov test for continuous
variables, and non-normal distribution data were presented as median
[interquartile range (IQR)], and compared by the Mann-Whitney U test. Categorical
variables were expressed as frequency (percentage), and compared by the
Chi-square test, Chi-squared correction test, or Fisher’s exact test. Among the
individuals with complete baseline data, demographics, BMI, and indications for
colonoscopy were compared between ITIU and non-ITIU groups. Logistic regression
analyses were performed to identify the independent factors associated with ITIU,
and variables with p
A total of 9649 individuals who completed TI intubation were included (Fig. 1). Their characteristics are described in Table 1. The median age was 53 years, 55.8% (5382/9648) of these individuals were older than 50 years, and 55.1% (5315/9649) were male. Among them, 143 individuals had TI ulcer, of whom 20 were also accompanied with ileocecal valve or colon ulcer. Therefore, the detection rate of ITIU was 1.3% (123/9649). Characteristics of patients with ITIU were described in Table 2. The median age was 44 years, 41.5% (51/123) of the patients with ITIU were older than 50 years, and 60.2% (74/123) were male. Among the patients with ITIU, 37.4% (46/123) were detected with a single ulcer. The proportion of patients who were accompanied with ileocecal valvulitis/colitis, colonic diverticula, and colonic polyp was 22.8% (28/123), 5.7% (7/123), and 44.7% (55/123), respectively. Among the 123 patients, 106 had the histology of the ulcers, of whom 99 (93.4%) indicated chronic inflammation and 63 (59.4%) lymphocyte proliferation.
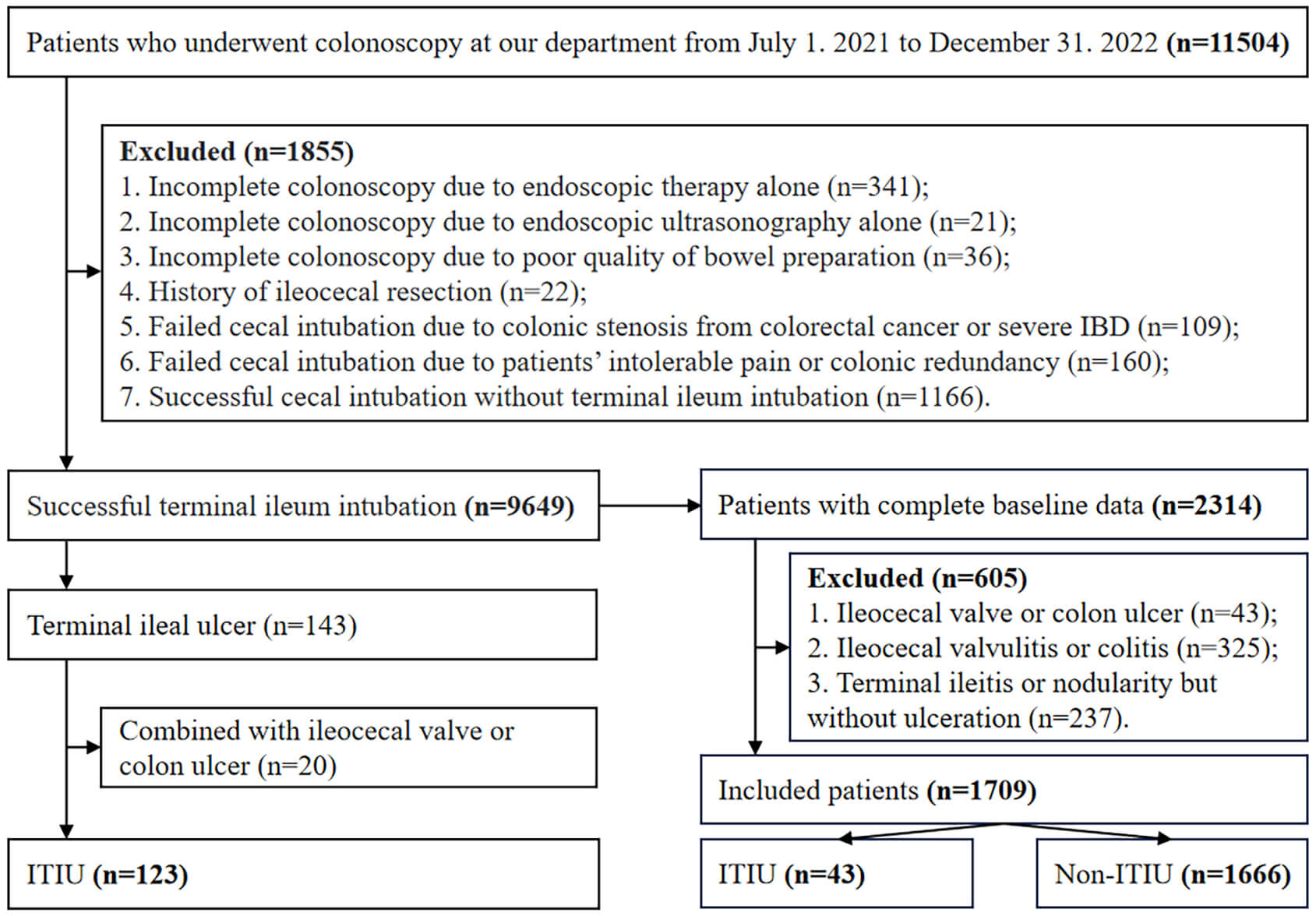 Fig. 1.
Fig. 1.
Flowchart of patients’ enrollment. ITIU, isolated terminal ileal ulcers; IBD, inflammatory bowel disease.
| Variables | No. Pts. | Median (IQR) or Frequency (percentage) | |
| Gender | |||
| Male (%) | 9649 | 5315 (55.1%) | |
| Female (%) | 9649 | 4334 (44.9%) | |
| Age (years) | 9648 | 53 (40, 62) | |
| 9648 | 4266 (44.2%) | ||
| 9648 | 5382 (55.8%) | ||
| Outpatient (%) | 9649 | 7084 (73.4%) | |
| Major colonoscopic findings | |||
| TI ulcer (%) | 9649 | 143 (1.5%) | |
| ITIU (%) | 9649 | 123 (1.3%) | |
| Terminal ileitis or nodularity but without ulceration (%) | 9649 | 582 (6.0%) | |
| Colitis (%) | 9646 | 937 (9.7%) | |
| Colonic diverticulosis (%) | 9646 | 490 (5.1%) | |
| Colonic polyp (%) | 9649 | 5530 (57.3%) | |
Abbreviations: IQR, interquartile range; No. Pts., numbers of patients; TI, terminal ileum; ITIU, isolated terminal ileal ulcers. The age was missing in one included patient, and the information regarding colitis and colonic diverticulosis were missing in three included patients based on our database.
| Variables | No. Pts. | Median (IQR) or Frequency (percentage) | |
| Gender | |||
| Male (%) | 123 | 74 (60.2%) | |
| Female (%) | 123 | 49 (39.8%) | |
| Age (years) | 123 | 44 (34, 57) | |
| 123 | 72 (58.5%) | ||
| 123 | 51 (41.5%) | ||
| Outpatient (%) | 123 | 87 (70.7%) | |
| Major colonoscopic findings | |||
| Ileocecal valvulitis/colitis (%) | 123 | 28 (22.8%) | |
| Colonic diverticulosis (%) | 123 | 7 (5.7%) | |
| Colonic polyp (%) | 123 | 55 (44.7%) | |
| Ulcer number | |||
| Single (%) | 123 | 46 (37.4%) | |
| Multiple (%) | 123 | 77 (62.6%) | |
| Histopathologic characteristics | |||
| Chronic inflammation (including acute phase) | 106 | 99 (93.4%) | |
| Lymphocyte proliferation | 106 | 63 (59.4%) | |
| Dysplasia | 106 | 15 (14.2%) | |
| Ulcer | 106 | 11 (10.4%) | |
| Mucosal muscle hyperplasia | 106 | 5 (4.7%) | |
| Eosinophilic infiltration | 106 | 3 (2.8%) | |
| Atypical cells | 106 | 1 (0.9%) | |
Abbreviations: IQR, interquartile range; No. Pts., numbers of patients; ITIU, isolated terminal ileal ulcers.
A total of 1709 individuals had completed baseline data, and their characteristics were described in Table 3. The median age was 56 years, 51.3% (876/1709) were male, and 63.4% (1084/1709) were older than 50 years. The median BMI was 23.90 kg/m2. Colonoscopy was performed for formless stool in 34.5%, for screening/surveillance in 31.6%, and for abdominal pain in 21.0%. The proportion of individuals with ITIU was 2.5% (43/1709).
| Variables | No. Pts. | Median (IQR) or Frequency (percentage) | |
| Gender | |||
| Male (%) | 1709 | 876 (51.3%) | |
| Female (%) | 1709 | 833 (48.7%) | |
| Age (years) | 1709 | 56 (43, 64) | |
| 1709 | 625 (36.6%) | ||
| 1709 | 1084 (63.4%) | ||
| BMI (kg/m2) | 1709 | 23.90 (21.63, 26.12) | |
| BMI |
1709 | 82 (4.8%) | |
| BMI = 18.5–23.9 kg/m2 (%) | 1709 | 787 (46.1%) | |
| BMI = 24.0–27.9 kg/m2 (%) | 1709 | 638 (37.3%) | |
| BMI |
1709 | 202 (11.8%) | |
| Sedated colonoscopy (%) | 1709 | 218 (12.8%) | |
| Outpatient (%) | 1709 | 1131 (66.2%) | |
| Indications for colonoscopy | |||
| Abdominal pain (%) | 1709 | 359 (21.0%) | |
| Diarrhea (%) | 1709 | 141 (8.3%) | |
| Gastrointestinal bleeding (%) | 1709 | 86 (5.0%) | |
| Abdominal distension/discomfort (%) | 1709 | 172 (10.1%) | |
| Constipation (%) | 1709 | 199 (11.6%) | |
| Formless stool (%) | 1709 | 590 (34.5%) | |
| Screening/surveillance (%) | 1709 | 540 (31.6%) | |
| Major colonoscopic findings | |||
| ITIU (%) | 1709 | 43 (2.5%) | |
Abbreviations: IQR, interquartile range; No. Pts., numbers of patients; BMI, body mass index; ITIU, isolated terminal ileal ulcers.
Compared with the non-ITIU group, the ITIU group was significantly younger (46 vs. 56, p = 0.010). In the ITIU group, the common indications for colonoscopy were formless stool (34.9%), screening/surveillance (32.6%), and abdominal pain (23.3%) (Table 4).
| Variable | N (“Non-ITIU, n = 1666”) | N (“ITIU, n = 43”) | p-value | ||
| Median (IQR) or Frequency (percentage) | Median (IQR) or Frequency (percentage) | ||||
| Age (years) | 56 (43, 64) | 46 (33, 59) | –2.567 | 0.010 | |
| Female (%) | 813 (48.8%) | 20 (46.5%) | 0.088 | 0.767 | |
| BMI (kg/m2) | 23.91 (21.64, 26.12) | 23.15 (20.20, 25.71) | –0.897 | 0.370 | |
| BMI |
78 (4.7%) | 4 (9.3%) | 1.078 | 0.299* | |
| BMI = 18.5–23.9 kg/m2 (%) | 769 (46.2%) | 18 (41.9%) | 0.312 | 0.577 | |
| BMI = 24.0–27.9 kg/m2 (%) | 626 (37.6%) | 12 (27.9%) | 1.675 | 0.196 | |
| BMI |
193 (11.6%) | 9 (20.9%) | 3.513 | 0.061 | |
| Indications for colonoscopy | |||||
| Abdominal pain (%) | 349 (20.9%) | 10 (23.3%) | 0.134 | 0.714 | |
| Diarrhea (%) | 139 (8.3%) | 2 (4.7%) | 0.346 | 0.556* | |
| Gastrointestinal bleeding (%) | 84 (5.0%) | 2 (4.7%) | 0.000 | 1.000* | |
| Abdominal distension/discomfort (%) | 167 (10.0%) | 5 (11.6%) | 0.008 | 0.930* | |
| Constipation (%) | 194 (11.6%) | 5 (11.6%) | 0.000 | 0.997 | |
| Formless stool (%) | 575 (34.5%) | 15 (34.9%) | 0.003 | 0.960 | |
| Screening/surveillance (%) | 526 (31.6%) | 14 (32.6%) | 0.019 | 0.891 | |
*Chi-squared correction test. Abbreviations: IQR, interquartile range; BMI, body mass index; ITIU, isolated terminal ileal ulcers.
No significant multicollinearity was detected among the variables included in
the multivariate logistic regression model (all VIF
| Variable | No. Pts. | Univariate analyses | Multivariate analyses | |||||||||
| SE | Wald | OR (95% CI) | p-value | SE | Wald | aOR (95% CI) | p-value | |||||
| Age |
1709 | 0.709 | 0.310 | 5.236 | 2.033 (1.107–3.732) | 0.022 | 0.666 | 0.312 | 4.562 | 1.947 (1.056–3.587) | 0.033 | |
| Female vs. male | 1709 | –0.092 | 0.310 | 0.088 | 0.912 (0.497–1.674) | 0.767 | ||||||
| BMI (kg/m2) | ||||||||||||
| BMI |
1709 | 0.736 | 0.538 | 1.875 | 2.088 (0.728–5.990) | 0.171 | ||||||
| BMI = 18.5–23.9 kg/m2 (yes vs. no) | 1709 | –0.175 | 0.313 | 0.311 | 0.840 (0.455–1.551) | 0.577 | ||||||
| BMI = 24.0–27.9 kg/m2 (yes vs. no) | 1709 | –0.441 | 0.344 | 1.649 | 0.643 (0.328–1.261) | 0.199 | ||||||
| BMI |
1709 | 0.703 | 0.383 | 3.378 | 2.020 (0.954–4.276) | 0.066 | 0.617 | 0.385 | 2.564 | 1.854 (0.871–3.945) | 0.109 | |
| Indications for colonoscopy | ||||||||||||
| Abdominal pain (yes vs. no) | 1709 | 0.134 | 0.366 | 0.134 | 1.144 (0.558–2.343) | 0.714 | ||||||
| Diarrhea (yes vs. no) | 1709 | –0.624 | 0.730 | 0.731 | 0.536 (0.128–2.239) | 0.392 | ||||||
| Gastrointestinal bleeding (yes vs. no) | 1709 | –0.085 | 0.733 | 0.013 | 0.919 (0.219–3.863) | 0.908 | ||||||
| Abdominal distension/discomfort (yes vs. no) | 1709 | 0.166 | 0.483 | 0.119 | 1.181 (0.459–3.042) | 0.730 | ||||||
| Constipation (yes vs. no) | 1709 | –0.002 | 0.482 | 0.000 | 0.998 (0.388–2.567) | 0.997 | ||||||
| Formless stool (yes vs. no) | 1709 | 0.016 | 0.324 | 0.003 | 1.016 (0.539–1.918) | 0.960 | ||||||
| Screening/surveillance (yes vs. no) | 1709 | 0.045 | 0.330 | 0.019 | 1.046 (0.548–1.997) | 0.891 | ||||||
Abbreviations: No. Pts., numbers of patients; OR, odds ratio; aOR, adjusted odds ratio; CI, confidence interval; BMI, body mass index; ITIU, isolated terminal ileal ulcers; SE, standard error.
A total of 17 patients with ITIU had their specific causes, including 9 with drug-induced ulcer, 2 with CD, 3 with UC, 2 with lymphoma, and 1 with ankylosing spondylitis (Fig. 2).
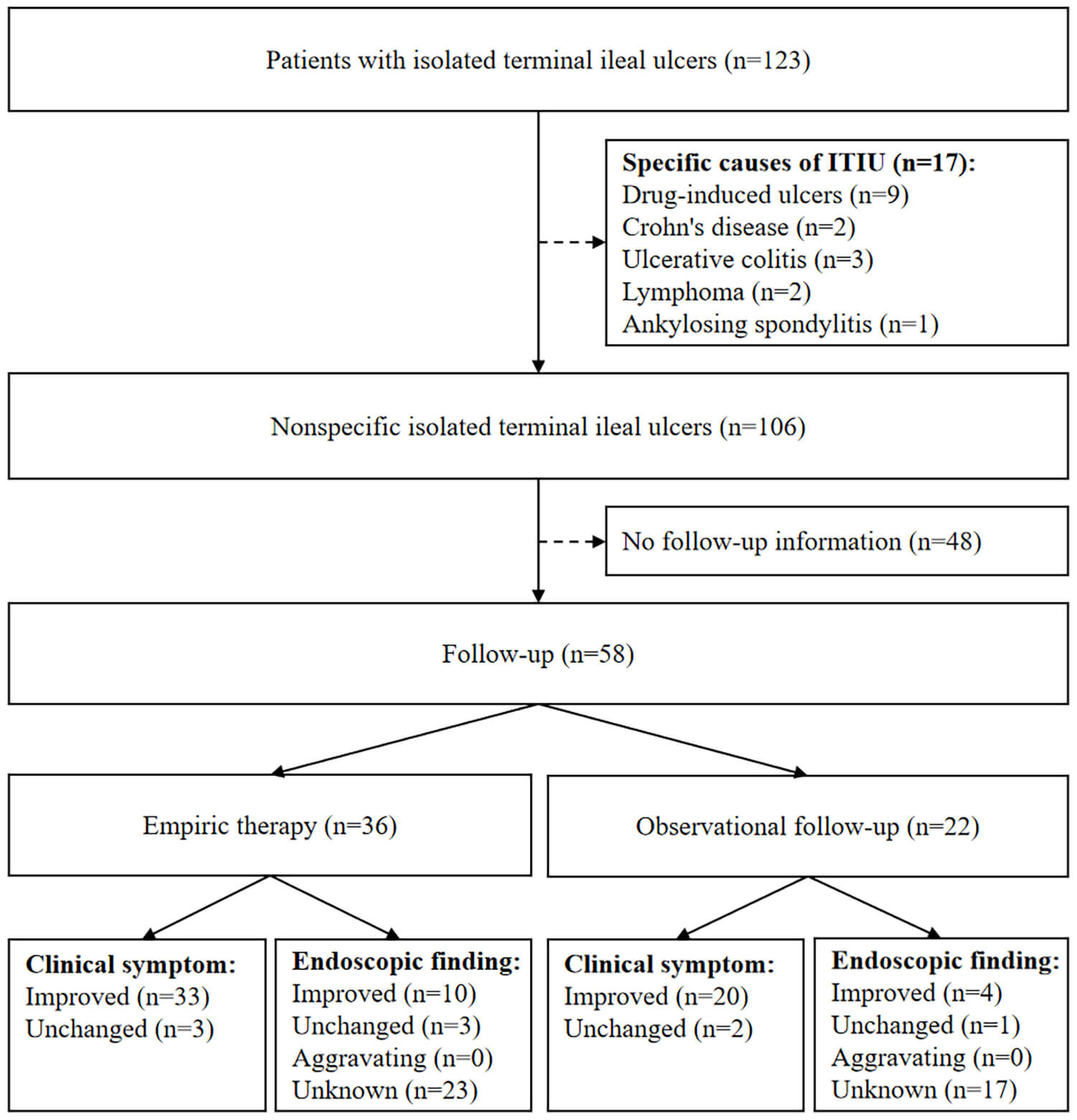 Fig. 2.
Fig. 2.
Flowchart of follow-up. ITIU, isolated terminal ileal ulcers.
A total of 106 patients who had nonspecific ITIU received empiric therapy, including 5-Aminosalicylic Acid (5-ASA), probiotics, and 5-ASA plus probiotics, and observational follow-up. Among them, 48 patients could not be further contacted during the follow-up period, and 58 could be followed by telephone at least one year after the index colonoscopy. Furthermore, 36 of the 58 patients received empiric therapy during their median follow-up period of 25 months, and another 22 did not receive further therapy during their median follow-up period of 23 months. Neither clinical symptoms (p = 1.000) nor colonoscopic findings (p = 0.560) were significantly different between patients who received empiric therapy and those who received observational follow-up assessment (Table 6).
| Outcomes | Empiric therapy* (n = 36) | Observational follow-up (n = 22) | p-value | ||
| Duration of follow-up months [Median (IQR)] | 25 (20, 28.75) | 23 (16.75, 27.50) | –1.293 | 0.196 | |
| Clinical symptom | Not available | 1.000a | |||
| Improved | 33 (91.7%) | 20 (90.9%) | |||
| Unchanged | 3 (8.3%) | 2 (9.1%) | |||
| Endoscopic finding | 1.16 | 0.560 | |||
| Improved | 10 (27.8%) | 4 (18.2%) | |||
| Unchanged | 3 (8.3%) | 1 (4.5%) | |||
| Aggravating | 0 (0%) | 0 (0%) | |||
| Unknown | 23 (63.9%) | 17 (77.3%) | |||
*Empiric therapy includes 5-Aminosalicylic Acid (5-ASA), probiotics, and 5-ASA plus probiotics; a Fisher’s exact test; ITIU, isolated terminal ileal ulcers; IQR, interquartile range.
Our study showed that the detection rate of ITIU in individuals undergoing
colonoscopy was 1.3% (123/9649). Among them, 4.1% (5/123) were diagnosed with
IBD (2 with CD and 3 with UC). Common clinical manifestations of ITIU were
formless stool and abdominal pain. In addition, age
As well known, IBD often occurs in young adults, and the peak age of its onset is 18 to 49 years [17]. Considering a potential correlation between ITIU and IBD [5, 6], it is reasonable to believe that the age of individuals also influences the occurrence of ITIU, which is consistent with the findings of our current study. There are some possible explanations for this association, as follows. First, ITIU alone, without any involvement of the rest of the intestines, can be observed at the early stage of IBD [6, 18]. Specifically, nearly 30% of patients with CD have ulcers confined to the TI alone [19], and nearly 17% of patients with UC can be accompanied by ulcers in the TI [20]. Our study found that 10.7% (3/28) of patients with ITIU and concurrent ileocecal valvulitis/colitis were finally diagnosed with UC, probably because backwash ileitis resulted in ileal ulcers. Therefore, a higher risk of ITIU in individuals younger than 50 years old can be explained by a higher incidence of IBD in younger individuals [17]. Second, TI is a special part of the intestinal tract where feces tend to accumulate, and bacteria thrive. It contains a large amount of Peyer’s patches, which are responsible for the intestinal immune system or intestinal immunity. Notably, the number of Peyer’s patches gradually decreases with age, and peaks at the age of 15–25 years old. Once the bacteria thrive, the immune response would be strong at the TI owing to the presence of Peyer’s patches. Taken together, it is more likely to develop ulcers in the TI in young individuals [2, 3, 21, 22].
There seems to be a positive correlation between the number of small intestinal
ulcers and BMI [14]. Previous studies suggest that the diversity of gut
microbiota is lower in obese people [23, 24, 25]. Especially, most obese people
have diets that are low in fiber but high in protein and fat. Notably, these
diets can alter the ratios of microbes and metabolites that modulate inflammation
[26, 27, 28]. Therefore, intestinal inflammation can be more easily increased in
obese people. Moreover, the presence of certain bacteria in obese people can
impair the function of intestinal tight junctions, allowing bacterial products to
pass through the intestinal wall easily and then induce an inflammatory response
[14, 23, 29]. By comparison, our study demonstrated only a marginally positive
association between ITIU and BMI
At present, there is no consensus on routine TI intubation during screening colonoscopy. Traditionally, considering the time-consuming nature of TI intubation and a low detection rate of TI lesions, routine TI intubation is not recommended, and TI intubation is only recommended for individuals with gastrointestinal symptoms or suspected IBD [4, 30, 31]. However, our study showed that the detection rate of ITIU was 1.3% and that approximately 90% of individuals undergoing colonoscopy completed the TI intubation. Interestingly, we found that a relatively high proportion of ITIU individuals underwent screening colonoscopy, who did not have any obvious gastrointestinal symptoms and were asymptomatic. Thus, it seems that clinical manifestations are not associated with ITIU, and the necessity of TI intubation should be recognized in all individuals undergoing screening colonoscopy.
In a randomized controlled trial (RCT) enrolling 82 patients with ITIU, there was no significant difference in clinical efficacy or endoscopic outcome during short-term and 1-year follow-up periods between patients who received experimental treatment with mesalazine (orally, 4.0 g, quaque die [qd]) for 3 months and those who received observational follow-up [32]. Our current study also showed no significant difference in the efficacy between patients receiving empiric therapy and observational follow-up after 1-year follow-up. Recently, another RCT enrolling 60 patients with isolated terminal ileum abnormalities demonstrated no significant difference in clinical or endoscopic efficacy between patients who received combination antimicrobial therapy (rifaximin, orally, 550 mg, bid, for two weeks; albendazole, orally, 400 mg, as a single dose; tinidazole, orally, 1 g, bis in die [bid], for three days) and those who received symptomatic treatment (probiotics; proton pump inhibitors; prokinetic) during short-term and 3-month follow-up periods [33]. It has been reported that the rate of progression from ITIU to IBD is high, ranging from 29% to 54%, during a follow-up period of 4 to 7 years. Therefore, nonspecific ITIU should be considered as an early stage of IBD and treated early and monitored closely [13, 18, 34, 35, 36]. Considering that it is difficult to distinguish between IBD and ITIU, especially in asymptomatic individuals [10, 31], long-term follow-up and an adaptable treatment strategy seem to be necessary for patients with ITIU.
Our study has a few limitations. First, owing to the retrospective nature of this study, some baseline data, such as height, weight, and indications for colonoscopy, and follow-up information, were unavailable for all individuals. Second, in our study, some patients received probiotics or 5-ASA as empiric therapy for ileal ulcers. However, a systematic review and meta-analysis regarding the efficacy of aminosalicylic acid preparations for the treatment of CD revealed that the effectiveness of sulfasalazine for treating mild-to-moderate active CD was marginal. Additionally, olsalazine and mesalazine, at a low (1–2 g/d) or high (3.2–4.0 g/d) dosage, did not significantly differ from placebo in inducing remission of CD [37]. Furthermore, the current European Crohn’s and Colitis Organization guidelines do not recommend the use of oral 5-ASA for the treatment of ileal CD [38]. Finally, in our study, the BMI classifications are based on the China-specific criteria, which differ from the World Health Organization standards, particularly in establishing thresholds for overweight and obesity. This is primarily because the risk stratification of comorbidities and mortality in the Chinese population is dependent upon lower BMI cut-off values [39, 40, 41]. Further studies should validate this conclusion in population outside the China according to the World Health Organization standards of BMI.
In conclusion, ITIU is not rare during colonoscopy, and predominates in younger
individuals with an age
• ITIU had a detection rate of 1.3% and is predominantly observed
in younger individuals with an age
• The necessity of TI intubation should be recognized in all individuals undergoing screening colonoscopy.
• Younger age is an independent risk factor for ITIU.
• Empiric therapy does not improve the outcomes of patients with nonspecific ITIU.
All data included in this study are available from the corresponding authors upon reasonable request.
Conceptualization: XS, XQ; Formal analysis: DS, XS and XQ; Data curation: DS, RC, CL, YL, JH, SW, YS, KW, CG and XQ; Writing—original draft: DS, RC, JH, CL, CG, and XQ; Writing—review and editing: DS, RC, JH, CL, CG, YL, XS, XQ; Supervision: XS, XQ. All authors contributed to the important editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This work was approved by the medical ethical committee of General Hospital of Northern Theater Command [Approval number: Y (2023)196]. All of our patients signed their informed consent forms for colonoscopy before undergoing the examination. Considering the retrospective nature of this study, the use of anonymous data, the lack of direct intervention measures, and the extremely minimal risk to patients, the medical ethics committee of the General Hospital of the Northern Theater Command has waived the requirement for the patients’ written informed consents. This is in accordance with the principles outlined in the Declaration of Helsinki.
Not applicable.
This work was supported by the Independent Research Funding of General Hospital of Northern Theater Command (ZZKY2024018).
Given his role as Editorial Board member, Xingshun Qi had no involvement in the peer-review of this article and has no access to information regarding its peer-review. Full responsibility for the editorial process for this article was delegated to John Alcolado. Other authors declared no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


