1 Department of Pediatrics, The Affiliated Hospital of Xuzhou Medical University, 221000 Xuzhou, Jiangsu, China
Abstract
Lobar pneumonia (LP) remains a significant cause of morbidity in children. Early and accurate assessment of the disease severity is crucial for optimal management, yet readily available and specific biomarkers are lacking. This study aimed to analyze the dynamic changes of aspartate aminotransferase/alanine aminotransferase (De Ritis) ratio in children with LP, and to explore their relationship with the disease severity.
A total of 120 children with LP admitted to the Affiliated Hospital of Xuzhou Medical University from June 2020 to June 2025 were divided into a common pneumonia group (n = 69) and a severe pneumonia group (n = 51) according to the severity of the disease. Hierarchical multiple linear regression was used to analyze the effects of baseline De Ritis ratio, gender, age, body mass index (BMI) and pulmonary lobe lesions on the ΔDe Ritis ratio before treatment and 14 days after treatment. Receiver operating characteristic (ROC) curve analysis was performed to assess the diagnostic efficacy of De Ritis ratio in children with LP before treatment. The dose–response relationship between De Ritis ratio and the condition of LP children was analyzed using a restricted cubic spline. The clinical decision curve was used to analyze the diagnostic efficacy of De Ritis ratio in children with LP.
Repeated measures analysis of variance showed that the De Ritis ratio had significant time effect, between-group effect and time–group interaction effect (all p < 0.001). The De Ritis ratio of the two groups decreased significantly with treatment, with the decline (ΔDe Ritis ratio) being significantly higher in the severe pneumonia group than in the common pneumonia group at each time point (p < 0.05). The results of hierarchical multiple linear regression showed that baseline De Ritis ratio (β < 0), gender, age, BMI and lobar lesions (all β > 0) had a significant effect on De Ritis ratio (p < 0.05). The area under the curve (AUC) for the De Ritis ratio was 0.908, with an optimal cutoff value of 1.52. The restricted cubic spline analysis revealed a non-linear dose-response relationship between De Ritis ratio and LP (pnonlinear < 0.001). According to the clinical decision curve analysis, De Ritis ratio demonstrated substantial clinical utility in the diagnosis of LP.
De Ritis ratio possesses significant clinical value in the evaluation of LP and its dynamic alterations show a consistent relationship with the severity of the condition.
Keywords
- lobar pneumonia
- children
- aspartate aminotransferase
- alanine aminotransferase
- disease severity
Lobar pneumonia (LP) is a common and severe respiratory disease in children, characterized by acute inflammatory consolidation of one or more lobes of the lung. Owing to the widespread inoculation of antibiotics and the advancement of medical technology, the mortality rate attributable to LP has significantly reduced, but severe cases can still lead to serious complications such as empyema, lung abscess, and acute respiratory distress syndrome, posing a major threat to children’s health [1, 2, 3]. Therefore, early and accurate assessment of the severity of the disease and dynamic monitoring of its progress are of great clinical significance for formulating individualized treatment plans and improving the prognosis of affected children. Globally, LP remains a leading infectious cause of pediatric hospitalization, representing a primary contributor to morbidity and causing increased utilization of healthcare resources [4]. The cornerstone of diagnosis for the disease combines clinical assessment with radiological confirmation, typically by chest X-ray, while its management hinges on the timely administration of appropriate antibiotics. The clinical challenge, however, often lies not in diagnosis but in accurately stratifying disease severity at presentation. This stratification is critical, as it dictates management decisions—from outpatient care to hospital admission, and even to the intensive care unit—directly impacting patient outcomes and antibiotic stewardship.
At present, the clinical evaluation of the severity of LP in children depends on imaging examinations (such as chest X-ray, computed tomography [CT]), inflammatory markers (such as C-reactive protein, procalcitonin, white blood cell count) and clinical manifestations [5, 6, 7]. However, these indicators present some limitations in clinical evaluation of the disease. For example, patients are at risk of radiation exposure during imaging examination and achieving high-frequency dynamic monitoring is challenging [8]. Despite the high sensitivity of the conventional set of inflammatory markers, their inadequate specificity is a major concern, as both infectious and non-infectious factors unrelated to LP could contribute to their altered levels [9]. Therefore, current clinical research on pediatric LP has focused on identifying biochemical indicators that are straightforward to measure and provide specific insight into tissue damage and disease progression.
In this context, the ratio of aspartate aminotransferase (AST) to alanine aminotransferase (ALT)—known as the De Ritis ratio—has received extensive attention in recent years. The ratio was first proposed by Fernando De Ritis in 1957 and was used to assist in the identification of viral hepatitis and alcoholic liver injury. It reflects the pattern of liver cell injury and the degree of mitochondrial dysfunction [10, 11, 12]. Accumulating evidence has proved its broad clinical value beyond the field of liver disease. Multiple studies have shown that AST is widely present in a variety of extrahepatic tissues such as myocardium, skeletal muscle, kidney, red blood cells and lung tissues, and its elevated activity is often closely related to necrosis or severe cell injury [13, 14, 15, 16]. In severe infections such as pneumonia, the release of pathogen toxins, systemic inflammatory response storms, and possible hypoxemia can lead to multiple organ cell damage, especially destruction of cells in the lung tissue and red blood cells, causing significant changes in serum AST levels, thereby affecting the De Ritis ratio [17, 18]. In light of this, we speculate that the De Ritis ratio, a traditional liver function index, has the potential for dynamic monitoring of LP severity in children. The changes may not only indirectly reflect the intensity of systemic inflammatory response caused by pneumonia but may also be related to the degree of damage to the lung tissue itself and other important organs, such as the heart. By dynamically monitoring the trajectory of the ratio, it may be possible to reveal its internal relationship with the changes in pneumonia severity, complications and prognosis, and to provide clinicians with an additional and easy-to-implement evaluation window.
The purpose of this study was to systematically observe the dynamic changes of De Ritis ratio in the course of LP through retrospective analysis, and to explore its correlation with the severity of the disease. It is expected that this study can provide a new laboratory basis for the evaluation system of LP in children and provide a reference for clinical prognosis and intervention timing.
A total of 120 children with LP admitted to the Affiliated Hospital of Xuzhou Medical University from June 2020 to June 2025 were divided into a common pneumonia group (n = 69) and a severe pneumonia group (n = 51) according to the severity criteria outlined in the Chinese national guidelines for the diagnosis and treatment of community-acquired pneumonia in children (2019 edition) [19]. Subjects were included if they: (1) met the diagnostic criteria of LP [20]; (2) had confirmed LP caused by Mycoplasma pneumoniae (confirmed by immunoglobulin M [IgM] serology or polymerase chain reaction [PCR]) or Streptococcus pneumoniae (confirmed by blood culture, sputum culture, or antigen test); (3) aged 5–14 years; and (4) had complete clinical data. Exclusion criteria of this study include: (1) comorbidities with congenital dysplasia and pulmonary dysfunction; (2) previous history of respiratory diseases; (3) comorbidity with malignant tumor; (4) comorbidity with other organ dysfunction; and (5) clinical presentation or laboratory findings highly suggestive of viral pneumonia (e.g., seasonal outbreak, prominent rhinorrhea, wheezing, with negative bacterial workup). The flow of participant selection in the study is illustrated in Fig. 1.
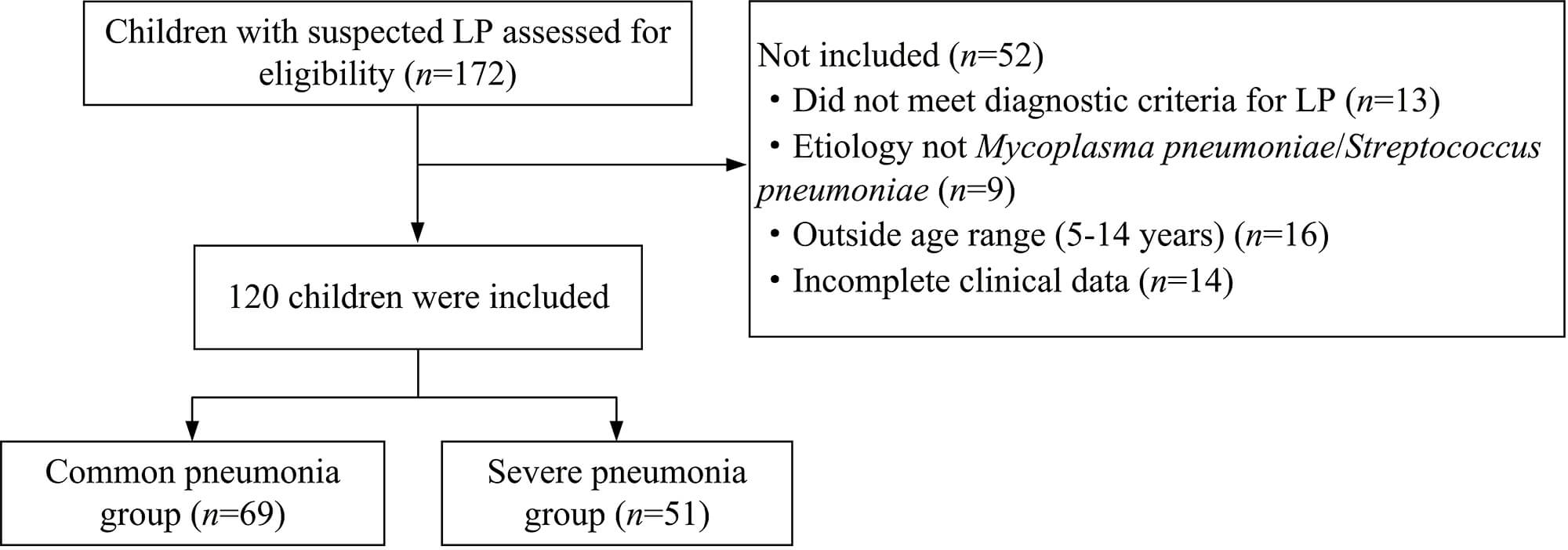 Fig. 1.
Fig. 1.
Flowchart of patient selection and group allocation. LP, lobar pneumonia.
This study did not perform an a priori sample size calculation. Therefore, a
post-hoc power analysis was conducted to verify the statistical power of our
findings. Based on the observed De Ritis ratio difference between groups (1.08 vs
1.85, standard deviation [SD] = 0.47) from our final cohort, with a significance
level (
All enrolled children received management according to the standardized guidelines for pediatric community-acquired pneumonia [20]. The treatment regimens were consistent between the common and severe pneumonia groups in principle, with adjustments based on disease severity.
Antimicrobial Therapy: Empirical antibiotic therapy was initiated promptly upon admission. The primary regimen consisted of a macrolide (e.g., azithromycin 10 mg/kg/day intravenously) targeting Mycoplasma pneumoniae, often in combination with a third-generation cephalosporin (e.g., ceftriaxone 50–80 mg/kg/day intravenously) against Streptococcus pneumoniae and other potential bacterial pathogens. The antibiotic therapy was later de-escalated or adjusted based on clinical response and microbiological results.
Supportive Care: Supportive care consisting of antipyretics (e.g., acetaminophen) for fever control, oxygen therapy to maintain oxygen saturation above 92%, and bronchial hygiene techniques was provided as needed.
Severe Case Management: Children in the severe pneumonia group received more intensive supportive care, which includes higher-flow oxygen, closer monitoring in a specialized ward, and more frequent assessment of potential complications. The core antimicrobial principles remained the same for both groups.
Several observation indicators were assessed in this study:
(1) General information: gender, age, body mass index (BMI), duration of fever, the length of hospital stay, lung lobe lesion side.
(2) Laboratory indicators: white blood cell (WBC) count, neutrophil (NEU) count,
lymphocyte (LYM) count, platelet (PLT) count, red blood cell (RBC) count,
neutrophil-to-lymphocyte ratio (NLR), erythrocyte sedimentation rate (ESR),
procalcitonin (PCT), C-reactive protein (CRP), immunoglobulin A (IgA),
immunoglobulin G (IgG), immunoglobulin M (IgM), the percentage of Cluster of
Differentiation 4 (CD4)+ T cells, Cluster of Differentiation 8 (CD8)+ T cells and
CD4+/CD8+ ratio, AST, and ALT. The ratio of AST/ALT (De Ritis ratio)
was determined before treatment and on the third, seventh and 14th day of
treatment. The absolute change in the De Ritis ratio over the treatment course
was calculated as follows:
(3) Medication use: The use of key therapeutic medications during hospitalization was recorded, including azithromycin and acetaminophen.
Data analysis was performed using SPSS software, version 22.0 (IBM Corp.,
Armonk, NY, USA). Normality of continuous variables were tested using
Shapiro–Wilk test. Normally distributed are expressed as mean
The children in the severe pneumonia group exhibited significantly longer fever
duration and length of hospital stay than those in the common pneumonia group.
Other indicators, such as NEU, PLT, NLR, ESR, PCT, CRP, AST, De Ritis ratio, IgA,
IgG, IgM, CD8+ T cells, were significantly higher in the severe pneumonia
group than in the common pneumonia group, except for CD4+ T cells and
CD4+/CD8+ ratio, which were comparatively lower in the severe
pneumonia group (p
| Group | Common pneumonia group (n = 69) | Severe pneumonia group (n = 51) | t/ |
p | |
| Gender, n (%) | 0.197 | 0.657 | |||
| Male | 38 (55.07) | 26 (50.98) | |||
| Female | 31 (44.93) | 25 (49.02) | |||
| Age (years) | 5.23 |
5.41 |
0.639 | 0.524 | |
| BMI (kg/m2) | 16.65 |
16.63 |
0.050 | 0.960 | |
| Fever duration (days) | 7.12 |
9.67 |
13.181 | ||
| Length of hospital stay (days) | 11.45 |
15.83 |
8.134 | ||
| Pulmonary lobe lesions, n (%) | 2.604 | 0.107 | |||
| Unilateral | 55 (79.71) | 34 (66.67) | |||
| Bilateral | 14 (20.29) | 17 (33.33) | |||
| WBC count( |
11.23 |
12.18 |
1.904 | 0.059 | |
| NEU count ( |
6.54 |
9.87 |
10.858 | ||
| LYM count ( |
3.89 |
3.75 |
0.760 | 0.449 | |
| PLT count ( |
285.34 |
320.56 |
3.904 | ||
| RBC count ( |
4.56 |
4.62 |
0.888 | 0.377 | |
| NLR | 1.68 |
2.63 |
8.407 | ||
| ESR (mm/h) | 28.45 |
45.67 |
11.787 | ||
| PCT (µg/L) | 1.35 |
1.89 |
6.844 | ||
| CRP (mg/L) | 38.76 |
45.43 |
4.129 | ||
| AST (U/L) | 28.34 |
48.92 |
11.861 | ||
| ALT (U/L) | 26.55 |
27.83 |
1.150 | 0.253 | |
| De Ritis ratio | 1.08 |
1.85 |
9.738 | ||
| IgA (g/L) | 1.23 |
1.59 |
6.170 | ||
| IgG (g/L) | 10.45 |
13.78 |
13.582 | ||
| IgM (g/L) | 1.65 |
1.82 |
3.083 | 0.003 | |
| CD4+ T cells (%) | 38.45 |
32.12 |
5.514 | ||
| CD8+ T cells (%) | 24.56 |
28.91 |
5.090 | ||
| CD4+/CD8+ ratio | 1.57 |
1.11 |
7.790 | ||
| Macrolide use, n (%) | 62 (89.86) | 45 (88.24) | 0.080 | 0.778 | |
| Antipyretic use, n (%) | 58 (84.06) | 42 (82.35) | 0.061 | 0.804 | |
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; BMI, body mass index; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; LYM, lymphocyte; NEU, neutrophil; NLR, neutrophil-to-lymphocyte ratio; PLT, platelet; RBC, red blood cell; PCT, procalcitonin; WBC, white blood cell; CD4, Cluster of Differentiation 4; CD8, Cluster of Differentiation 8; IgA, immunoglobulin A; IgG, immunoglobulin G; IgM, immunoglobulin M.
The results of the repeated measures ANOVA showed that the De Ritis ratio in
both groups showed a downward trend during the course of treatment (Ftime =
85.42, ptime
| Group | Common pneumonia group (n = 69) | Severe pneumonia group (n = 51) | Repeated measures ANOVA | ||
| Effect | F-value | p-value | |||
| Before treatment | 1.08 |
1.85 |
Time | 85.42 | |
| Treatment for 3 days | 0.98 |
1.62 |
Group | 62.14 | |
| Treatment for 7 days | 0.89 |
1.32 |
Time |
18.36 | |
| Treatment for 14 days | 0.82 |
0.95 |
|||
Note: *p
The
| Variable | p | F | R2 | Adjusted R2 | p for | |||
| Layer 1 | 50.450 | 0.310 | 0.304 | 0.310 | 50.450 | |||
| Baseline De Ritis ratio | −0.557 | |||||||
| Layer 2 | 30.112 | 0.410 | 0.398 | 0.100 | 18.842 | |||
| Baseline De Ritis ratio | −0.540 | |||||||
| Gender | 0.275 | 0.003 | ||||||
| Layer 3 | 25.367 | 0.458 | 0.441 | 0.048 | 9.115 | 0.003 | ||
| Baseline De Ritis ratio | −0.520 | |||||||
| Gender | 0.268 | 0.005 | ||||||
| Age | 0.220 | 0.002 | ||||||
| Layer 4 | 21.225 | 0.488 | 0.466 | 0.030 | 6.210 | 0.014 | ||
| Baseline De Ritis ratio | −0.505 | |||||||
| Gender | 0.255 | 0.008 | ||||||
| Age | 0.208 | 0.004 | ||||||
| BMI | 0.185 | 0.015 | ||||||
| Layer 5 | 19.225 | 0.518 | 0.490 | 0.028 | 6.510 | 0.012 | ||
| Baseline De Ritis ratio | −0.495 | |||||||
| Gender | 0.248 | 0.009 | ||||||
| Age | 0.201 | 0.005 | ||||||
| BMI | 0.178 | 0.018 | ||||||
| Pulmonary lobe lesions | 0.175 | 0.012 |
Notes: Coding of categorical variables are as follows: Gender (Male = 1, Female = 0); Pulmonary lobe lesions (Bilateral = 1, Unilateral = 0).
Abbreviations: BMI, body mass index; LP, lobar pneumonia.
To evaluate the diagnostic efficacy of De Ritis ratio in children with LP before
treatment, an ROC curve was plotted. The AUC was 0.908 (95% CI 0.845–0.972,
p
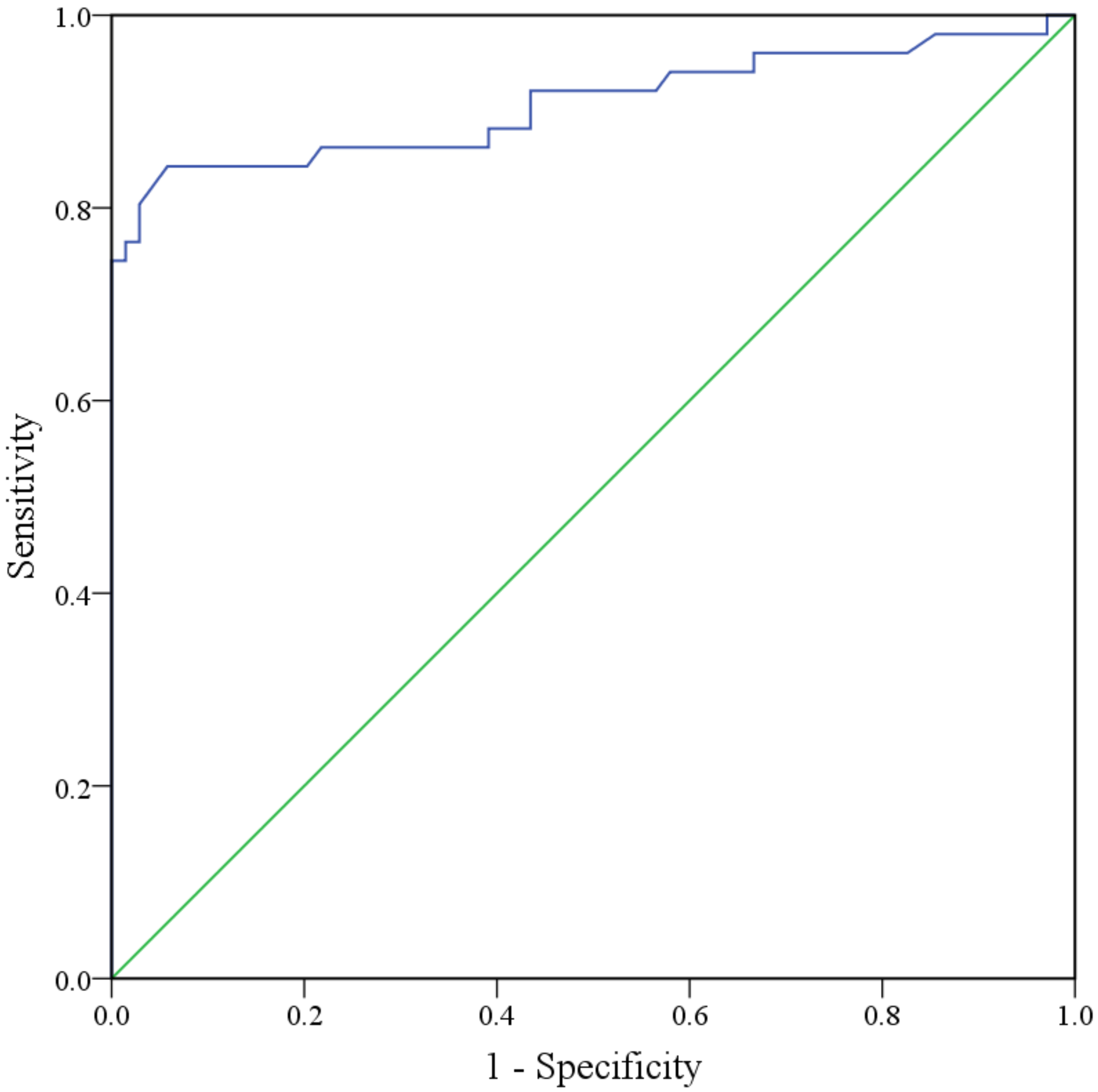 Fig. 2.
Fig. 2.
Receiver operating characteristic (ROC) curve analysis of diagnostic efficacy of De Ritis ratio in predicting the severity of pediatric lobar pneumonia.
The results of restricted cubic spline analysis showed that the De Ritis ratio
increased with the risk of severe pneumonia in children with LP. The correlation
between the De Ritis ratio and LP severity showed a non-linear dose-response
relationship (
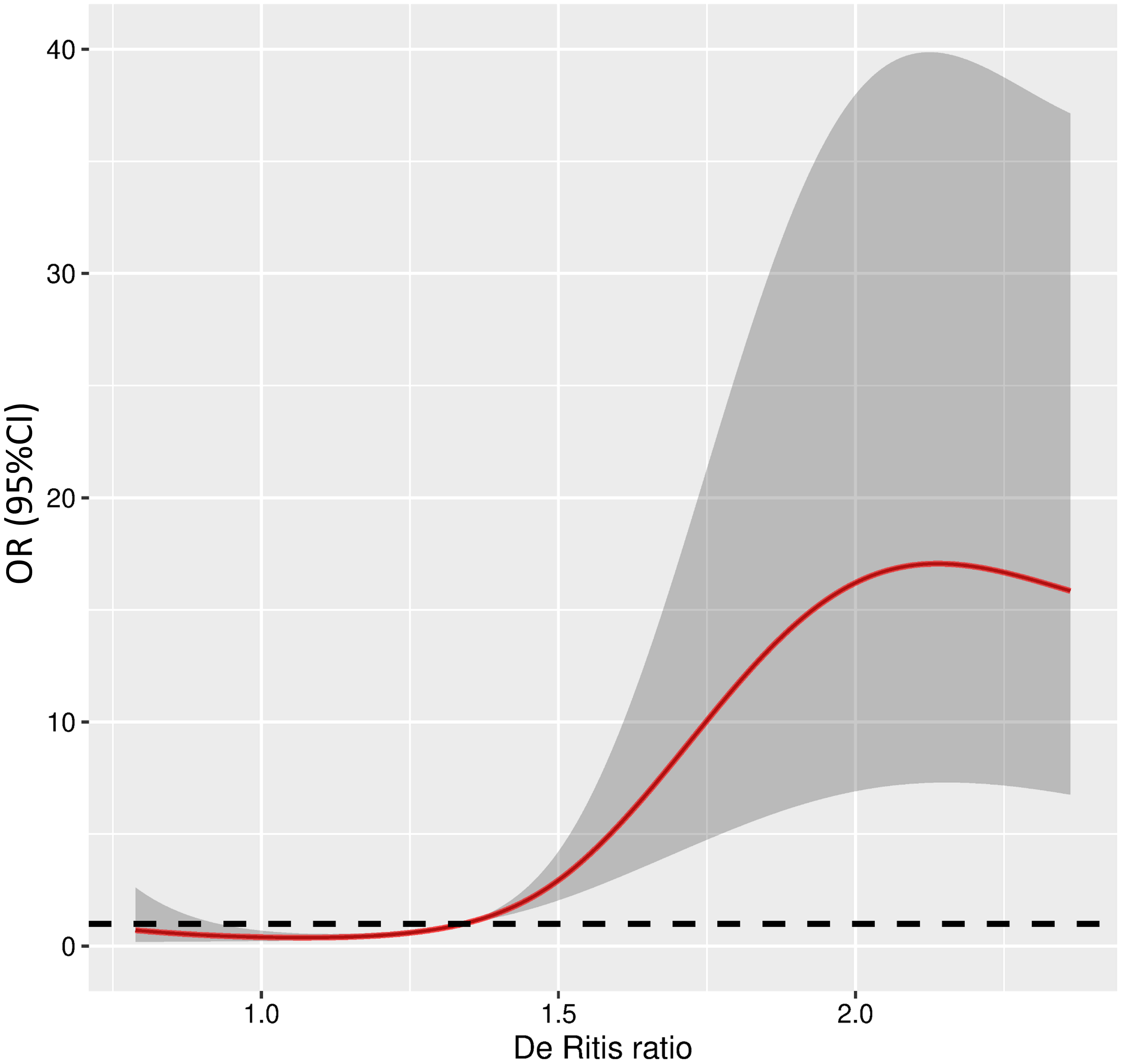 Fig. 3.
Fig. 3.
Restricted cubic spline plot of dose–response relationship between De Ritis ratio and severity of pediatric lobar pneumonia. OR, odds,ratio; CI, confidence interval.
In the clinical decision curve analysis, the decision curve of De Ritis ratio was higher than that of full intervention curve and full non-intervention curve when the threshold probability was in the range of 0.05–0.9, that is, the net benefit of the De Ritis ratio was higher, suggesting that the De Ritis ratio has significant clinical value in the identification of children with severe LP and implementation of clinical intervention may lead to better clinical outcomes, as shown in Fig. 4.
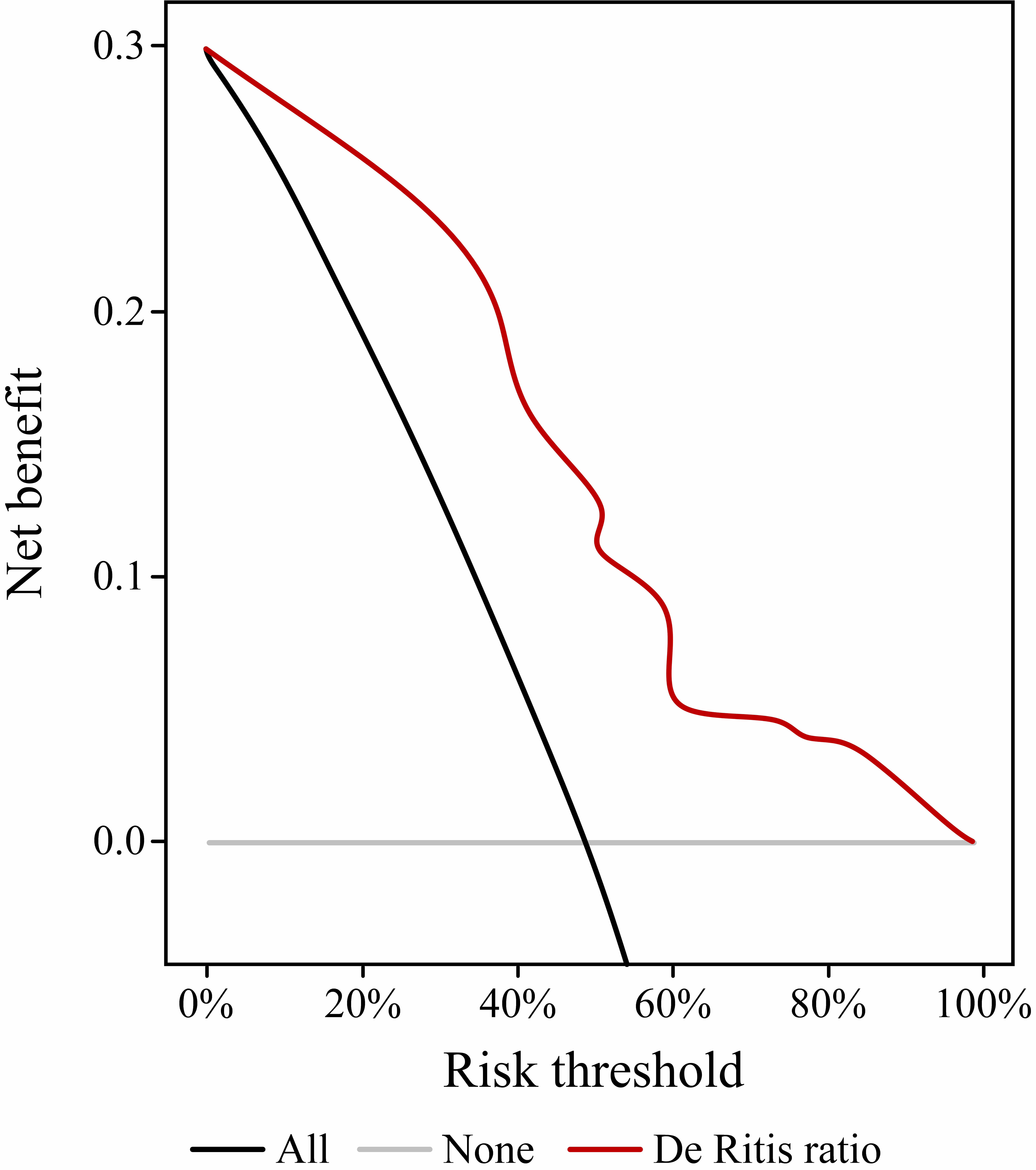 Fig. 4.
Fig. 4.
Decision curve analysis of De Ritis ratio in predicting the severity of pediatric lobar pneumonia.
In this study, the clinical data of 120 children with LP were systematically
analyzed, and the dynamic changes of De Ritis ratio in the course of the disease
and their correlation with the disease severity were discussed. The results
showed that the De Ritis ratio of children with severe LP was significantly
higher than that of children with ordinary-severity LP at all time points—both
before and after treatment, accompanied by a greater alteration (
Traditionally, the De Ritis ratio is mainly used for the differential diagnosis of hepatobiliary diseases. However, building on existing literature, the present study provides additional evidence that the clinical significance of the De Ritis ratio extends beyond liver disease. AST is not only present in the cytoplasm of liver cells, but also in mitochondria [21]. It is also widely distributed in various extrahepatic tissues such as myocardium, skeletal muscle, kidney, red blood cells, and lung tissues. ALT is mainly localized in the cytoplasm of hepatocytes [22]. In the pathological process of LP, especially severe LP, the massive release of bacterial toxins, strong systemic inflammatory response, and possible hypoxemia can lead to direct cell damage and necrosis in multiple organs [23, 24]. We speculate that extensive inflammation and destruction of parenchymal cells in lung tissue, hemolysis (leading to destruction of red blood cells and release of AST), and potential myocardial injury (such as toxic myocarditis) may be accountable for the significant elevation of serum AST. In contrast, ALT levels did not exhibit significant changes in the contexts of pulmonary inflammation. Therefore, the increase in the AST/ALT ratio may essentially reflect the extensive tissue and cell damage caused by LP, especially the severe damage or necrosis of mitochondria-rich cells. In this study, the higher levels of NEU, NLR, PCT, CRP and other inflammatory indicators in the severe pneumonia group indicate the presence of a more severe inflammatory response, which may underlie the observed increase in the De Ritis ratio. The process of its dynamic decline may be closely related to the control of inflammation and the gradual repair of tissue damage.
The core finding of this study is the strong correlation between the De Ritis
ratio and LP severity. The severe pneumonia group not only had a significantly
higher baseline De Ritis ratio before treatment, but its ratio also persistently
exceeded that of the common pneumonia group at every evaluated time point during
the treatment process. More importantly, the magnitude of its decline
(
Hierarchical multiple linear regression further revealed that gender, age, BMI,
and the range of lung lesions were independent influencing factors of
It is noteworthy that our analysis yielded two complementary De Ritis ratio values for clinical assessment, both derived from the ROC analysis framework. The primary analysis provided an optimal diagnostic cutoff of 1.52, which offers the best balance between sensitivity (83.42%) and specificity (94.76%) for discriminating between common and severe pneumonia at presentation. Furthermore, internal validation via bootstrapping refined our understanding of this threshold. The bootstrapping procedure, which assesses the stability of the ROC results, generated a 95% confidence interval for the optimal cutoff ranging from 1.28 to 1.41, with a point estimate of 1.34. The lower boundary and point estimate are clinically informative. A ratio above 1.52 offers strong diagnostic confidence for severe pneumonia, while values between 1.34 and 1.52 indicate a high-risk or early warning zone. Patients with De Ritis ratios within the 1.34–1.52 interval may not meet the definitive diagnostic threshold but are likely at significantly increased risk for severe disease, warranting closer clinical monitoring and a lower threshold for escalating care.
Application of the De Ritis ratio in LP severity stratification has significant clinical advantages: First, it is economical and efficient. As both the AST and ALT are conventional biochemical indicators, their detection is relatively inexpensive and rapid owing to the existing assays. Second, the De Ritis ratio is conducive to dynamic monitoring, without causing radiation exposure, and can be repeatedly detected to evaluate the disease progression and monitor treatment efficacy. Third, the De Ritis ratio has higher specificity than the conventional indicator. Compared with the traditional inflammatory markers such as CRP and PCT, it provides a direct assessment of the degree of tissue damage or the severity of pneumonia. Based on our clinical decision curve analysis, the De Ritis ratio can inform clinical decision-making, offering higher net benefit to patients within a wide range of threshold probabilities. This shows that incorporating this indicator into the clinical decision-making system (such as whether to upgrade treatment or whether to transfer patients to pediatric intensive care unit [PICU]) has practical clinical application value, which can facilitate early and accurate identification of high-risk children, as well as optimization of treatment strategies, thereby improving patient prognosis.
Several limitations of this study should be highlighted. Firstly, this is a
single-center retrospective study, which may be subject to selection bias.
Secondly, the sample size adopted was relatively limited, necessitating more
multicenter studies with larger samples to validate our findings in the future.
Thirdly, despite the implementation of a comprehensive set of microbiological
tests to exclude viral pneumonia (using PCR for common respiratory viruses), the
possibility of undetected or emerging viral pathogens cannot be entirely ruled
out, which might represent a potential source of misclassification bias. Fourth,
the lack of a prospective a priori sample size calculation weakens the rigor of
the study design to some extent. Although this study demonstrates a correlation,
the specific mechanisms underlying the increased De Ritis ratio—such as the
relative contributions from lung tissue, myocardium, or red blood cells—require
further in-depth investigation through basic research using animal models and
targeted marker analyses. Finally, this study did not analyze pathogen-specific
In summary, the De Ritis ratio was significantly increased in children with LP, especially in cases of severe pneumonia, and its dynamic changes were closely associated with the disease severity. The ratio is a readily obtainable, low-cost biomarker with high diagnostic efficiency and substantial clinical utility. When combined with existing clinical indicators, it has the potential to contribute to a comprehensive system for early assessment and dynamic monitoring of LP severity in children, provide robust support for accurate clinical decision-making, and ultimately improve patient outcomes.
• The De Ritis ratio is significantly elevated in children with severe lobar pneumonia and demonstrates distinct dynamic changes throughout the disease course, with a more pronounced decrease observed in severe cases.
• This ratio exhibits high diagnostic accuracy for severe lobar pneumonia (AUC = 0.908), with an optimal cutoff value of 1.52.
• A nonlinear dose–response relationship exists between the De Ritis ratio and severe lobar pneumonia.
• The
• As an economical and readily available biomarker, the De Ritis ratio shows substantial clinical utility for severity assessment, dynamic monitoring, and clinical decision-making in the contexts of pediatric lobar pneumonia.
All experimental data included in this study can be obtained by contacting the corresponding author if needed.
RL designed the research study, analyzed the data and drafted the manuscript. SSC performed the research. QQY collected the data. All authors contributed to the important editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was conducted following the principles of the Declaration of Helsinki and was approved by the Ethics Committee of the Affiliated Hospital of Xuzhou Medical University (Approval No.: XYFY2025-KL475-01). Written informed consent was obtained from the parents or legal guardians of all participating children.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




