1 Department of Otolaryngology, The Second Affiliated Hospital of Heilongjiang University of Chinese Medicine, 150000 Harbin, Heilongjiang, China
2 Outpatient Department, The Second Affiliated Hospital of Heilongjiang University of Chinese Medicine, 150000 Harbin, Heilongjiang, China
3 Department of Otolaryngology, The First Affiliated Hospital of Harbin Medical University, 150001 Harbin, Heilongjiang, China
Abstract
Allergic rhinitis (AR) is an upper respiratory disease that affects inflammation levels, nasal function, and mental health in patients. However, the effect of AR severity on these indicators remains obscure. This study aimed to explore the impacts of AR severity on levels of inflammatory factors, nasal function, anxiety and depression.
The clinical data of 188 patients with AR from January 2022 to January 2025 were collected and retrospectively analyzed. The patients were divided into mild group (n = 90) and moderate/severe group (n = 98) based on the severity of AR. Meanwhile, 79 healthy individuals matched in age, gender, and body mass index (BMI) with the AR patients were included in the control group. Nasal airway resistance (NAR) and nasal mucociliary clearance time (NMCT) were detected. Hospital Anxiety and Depression (HAD) scale was applied for the assessment of anxiety and depression. Serum level of C-reactive protein (CRP) was measured using an automatic biochemical analyzer. Serum procalcitonin (PCT) and nasal lavage fluid levels of interleukin-1β (IL-1β) and tumor necrosis factor-α (TNF-α) were measured using commercial assay kits.
Compared with the control group, the CRP, PCT, IL-1β, TNF-α, NAR, NMCT, and HAD anxiety and depression scores in AR patients were significantly increased (both p < 0.05). Compared with the mild group, the moderate/severe group exhibited increased levels of inflammatory biomarkers, NAR, NMCT, and HAD anxiety and depression scores (p < 0.05). In the mild group, anxiety and depression were correlated with the NAR, CRP, PCT, IL-1β, and TNF-α (p < 0.05); NMCT was correlated with the depression (p < 0.05). In moderate/severe group, anxiety and depression were correlated with the NAR, NMCT, CRP, PCT, IL-1β, and TNF-α (p < 0.05). The correlation between anxiety and depression and nasal function and inflammatory factors in moderate/severe group were stronger than those in mild group.
The anxiety/depression and inflammation levels in AR patients increase, while the nasal function decreases, with the deteriorating severity of the disease. Anxiety and depression are correlated with nasal function and inflammation levels, with a more prominent correlation detected in patients with moderate/severe AR than those with mild disease.
Keywords
- allergic rhinitis
- inflammatory
- nasal function
- anxiety
- depression
Allergic rhinitis (AR) is a common chronic upper respiratory disease characterized by chronic inflammation of the nasal mucosa [1]. In China, approximately 250 million people suffer from AR, with an incidence rate in adults of approximately 18.6%–52.9% in northern China [2]. The clinical features of AR include sneezing, watery nasal discharge, nasal congestion, and nasal itching. The diagnosis of AR is based on the clinical symptoms, medical history, and results of allergen testing (such as serum-specific immunoglobulin E [sIgE] measurements and skin prick tests) [1]. The existing diagnostic methods for AR are limited by their invasiveness, relatively high false-positive rates in the outcomes, and high costs, especially in sIgE detection. The clinical managements of AR mainly lie in preventive (e.g., blocking exposure to allergens) and pharmacological approaches [3, 4].
Inflammation is a key mechanism underlying the pathogenesis of AR. Allergens stimulate the nasal mucosa, activate inflammatory cells, inducing the generation and release of inflammatory mediators, such as histamine and leukotrienes. These inflammatory mediators act on the blood vessels of the nasal mucosa, causing vasodilation and increased permeability, which lead to congestion and swelling of the nasal mucosa. They also stimulate sensory nerve endings in the nasal mucosa, causing nasal discomfort [5, 6, 7]. Previous study reported that the serum level of interleukin-17A (IL-17A) was significantly correlated with severity of AR [8]. In addition, the neutrophil-to-lymphocyte ratio in patients with moderate/severe AR was higher compared to those with mild AR [9]. However, these studies examined a limited number of inflammatory markers, leaving the changes in other inflammatory indicators across different severities of AR insufficiently delineated.
The nasal cavity is a key part of the respiratory tract. Patients with AR face impaired nasal function and experience complete or partial nasal obstruction, which reduces airflow through the nasal passages. This obstruction necessitates compensatory mouth breathing, either partially or completely, which may further augment the risk of bronchospasm [10]. In addition, the nasal cilia play an important role in dust and pathogen clearance, serving as a critical defense mechanism for maintaining respiratory health. In AR patients, however, nasal ciliary clearance function is reduced due to a decrease in ciliary number, structural defects, among other reasons [11]. It has been reported that children with moderate/severe AR had much lower nasal mucociliary clearance compared with their mild disease counterparts [12]. Nevertheless, it is still unclear how nasal function changes across varying levels of AR severity in adult patients.
The clinical manifestations of AR include sneezing, watery nasal discharge, nasal congestion, and nasal itching. These symptoms have a profound impact on the patients’ daily functioning, social interactions, and sleep, markedly diminishing their quality of life and influencing their psychological emotions [13]. A study has reported that the severity and duration of AR could affect mental health and were positively correlated with anxiety and depression occurring in the patients [14]. While previous studies have assessed alterations in anxiety, depression, inflammation levels, and nasal function in AR of varying severity, most studies focused on investigating each of the aforementioned aspects in isolation, without attempting to explore the relationship between physiological abnormalities and psychological health. Interestingly, the association of anxiety and depression with inflammation is well-recognized [15, 16]. Inflammation has been shown to influence anxiety and depression in patients with asthma [17]. However, it remains unclear whether anxiety and depression are affected by inflammation and nasal function in AR patients, and whether these effects vary with disease severity.
Thus, addressing these knowledge gaps may help enhance clinical screening and enable early mental health intervention for AR patients. This study aimed to explore the effects of varying AR severity levels on inflammatory markers, nasal function, anxiety and depression. In this retrospective study, changes in inflammatory markers, nasal function, anxiety and depression in both mild and moderate/severe AR patients were investigated. In addition, the correlation of anxiety and depression with inflammatory markers and nasal function in AR patients was explored. The findings of the current study will facilitate the development of personalized treatment and enhance the diagnosis and treatment of psychological conditions in AR.
The information of patients with AR who visited The Second Affiliated Hospital
of Heilongjiang University of Chinese Medicine from January 2022 to January 2025
was collected for retrospective analysis. A total of 188 patients were included.
The inclusion criteria are as follows: (1) patients meeting the diagnostic
criteria for AR [18], including presentation of clinical symptoms (sneezing,
watery nasal discharge, nasal congestion, and nasal itching), and positive skin
prick test or positive sIgE result; (2) patients with persistent AR, marked by
presentation of clinical symptoms lasting for
The severity of AR was assessed according to the total nasal symptom score
(TNSS). Specifically, the nasal symptoms (sneezing, watery nasal discharge, nasal
congestion, and nasal itching) were scored using a 4-point Likert scale: 0 (no
symptoms); 1 (mild symptoms, easy to tolerate); 2 (obvious symptoms, but still
tolerable); 3 (unbearable symptoms, affecting daily activities or sleep). The
total scores of four nasal symptoms were calculated. A total TNSS score
In addition, healthy individuals who attended the same hospital for routine physical examination were included in the control group (n = 79). The age, gender and body mass index (BMI) of these healthy individuals were matched with those of the AR patients. All subjects in the control groups were confirmed negative for AR based on their medical history and clinical examination findings. A set of exclusion criteria were applied in the selection of subjects assigned to the control group: (1) presence of other allergic diseases and immunodeficiency diseases; (2) presence of diabetes and thyroid disease; (3) presence of other respiratory inflammatory diseases, such as asthma and tracheitis; and (4) a history of upper and lower respiratory tract surgery. Fig. 1 shows the flowchart of patient selection.
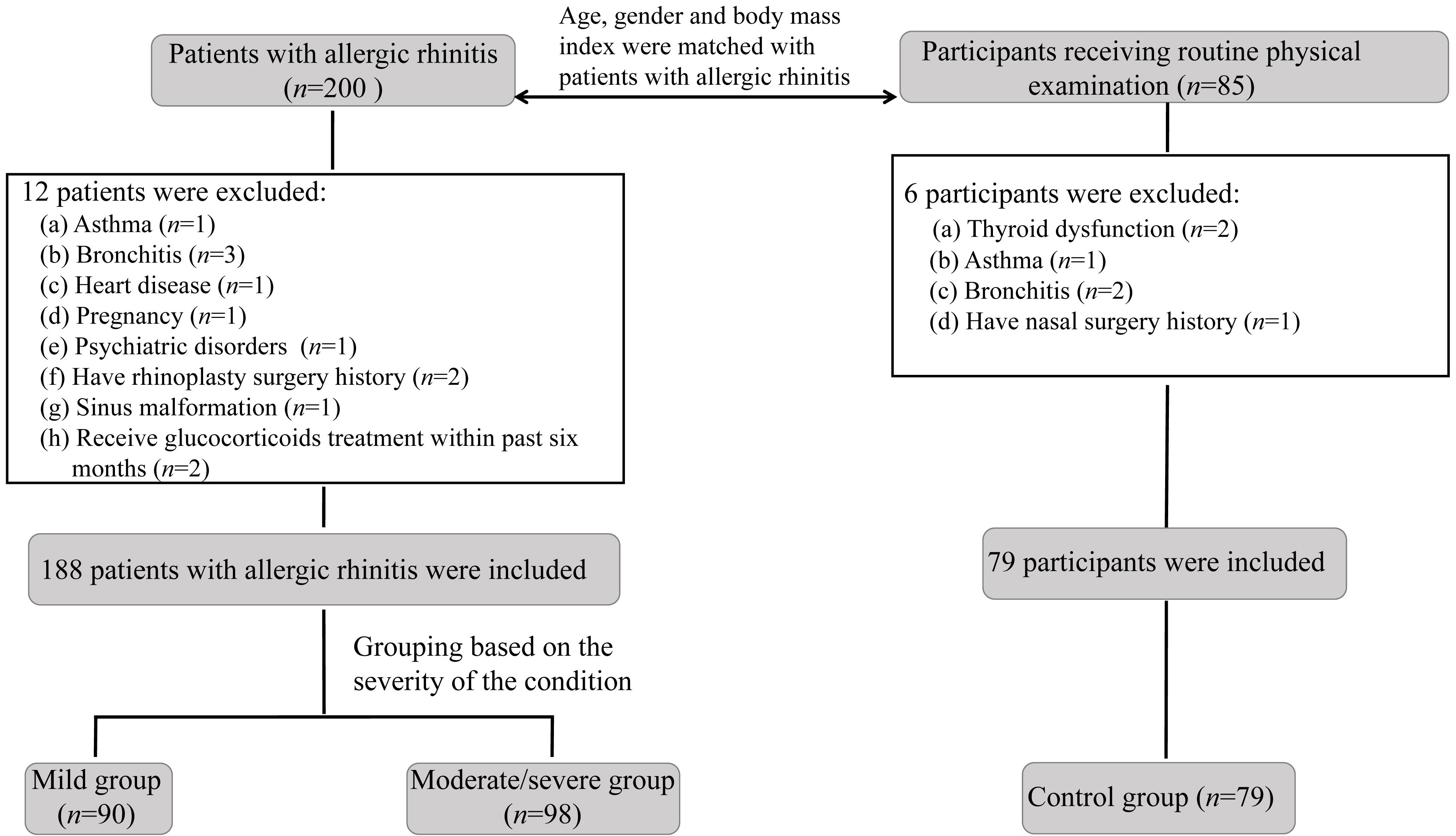 Fig. 1.
Fig. 1.
Flowchart of patient selection for this study.
A nasal resistance measurement device (zk-nr-100c, Anhui Zhongke Medical Devices Co., Ltd., Hefei, China) was applied to detect nasal airway resistance (NAR). Nasal mucociliary clearance time (NMCT) was assessed using saccharin test. Specifically, after clearing the nasal secretions, a saccharin particle was placed on the posterior surface of the nasal turbinates of each subject. The subjects were instructed to swallow once every 30 s. The time needed for the subject to perceive a sweet taste was recorded. NAR and NMCT measurements were obtained from routine clinical records and were performed by trained physicians according to a standardized protocol at our hospital.
Hospital Anxiety and Depression (HAD) scale was applied for the assessment of anxiety and depression [20]. The HAD scale contains 14 items. Each item has a score range from 0 (no symptomatic) to 3 (severe symptoms). Seven items of the scale are used to measure anxiety severity (0–21 points), whereas the remaining seven items are oriented for measurement of depression severity (0–21 points). The total scores for anxiety and depression were calculated, with 0–7 indicating normal, 8–10 possible anxiety or depression, and 11–21 clinical levels of anxiety and depression.
Fasting venous blood was collected from every patient in the morning after
having fasted for 12 hours. The serum C-reactive protein (CRP) level was detected
using an automatic biochemical analyzer (BC-5390CRP, Mindray Medical, Shenzhen,
China). Serum procalcitonin (PCT) levels were detected using commercial kits (H151-1-2, Nanjing Jiancheng Bioengineering Institute, Nanjing, China). Given the alternation of inflammatory factors were sensitive in nasal mucosa of patients with allergic rhinitis, the levels of interleukin-1β (IL-1
SPSS 27.0 (IBM Corp., Armonk, NY, USA) was applied for statistical analysis. The
Shapiro–Wilk test was employed to assess the normality of data distribution.
One-way analysis of variance (ANOVA) with Tukey’s post hoc test was used for the
inter-group comparisons of normally distributed data, which are expressed as mean
The general information of AR patients is displayed in Table 1. There was no
significant difference in general information among three groups (p
| General information | Control group (n = 79) | Mild group (n = 90) | Moderate/severe group (n = 98) | F/H/ |
p | |
| Age (years) | 45.05 |
44.43 |
44.97 |
0.153 | 0.858 | |
| Gender (n, %) | 1.798 | 0.408 | ||||
| Female | 41 (51.90%) | 38 (42.22%) | 43 (43.88%) | |||
| Male | 38 (48.10%) | 52 (57.78%) | 55 (56.12%) | |||
| BMI (kg/m2) | 23.75 (21.14, 26.07) | 22.85 (20.90, 25.40) | 22.68 (20.19, 24.74) | 3.549 | 0.170 | |
| Smoking history (n, %) | 3.592 | 0.166 | ||||
| Yes | 27 (34.18%) | 25 (27.78%) | 21 (21.43%) | |||
| No | 52 (65.82%) | 65 (72.22%) | 77 (78.57%) | |||
| Drinking history (n, %) | 2.415 | 0.299 | ||||
| Yes | 28 (35.44%) | 24 (26.67%) | 25 (25.51%) | |||
| No | 51 (64.56%) | 66 (73.33%) | 73 (74.49%) | |||
Abbreviation: BMI, body mass index.
There were significant differences in inflammatory indicators among the three
subject groups (p
| Inflammatory indicators | Control group (n = 79) | Mild group (n = 90) | Moderate/severe group (n = 98) | F/H | p |
| CRP (mg/L) | 6.25 |
7.92 |
10.41 |
116.596 | |
| PCT (ng/mL) | 0.03 (0.02, 0.04) | 0.04 (0.03, 0.05)* | 0.06 (0.05, 0.07)*& | 104.052 | |
| IL-1 |
4.45 |
12.17 |
29.34 |
1370.216 | |
| TNF- |
34.33 |
74.55 |
113.10 |
328.979 |
Note: *p
Abbreviations: CRP, C-reactive protein; IL-1
The NAR and NMCT in the control group were lower than those in the AR patients
(p
| Group | NAR (Pa/cm3/s) | NMCT (min) |
| Control group (n = 79) | 0.30 |
8.50 |
| Mild group (n = 90) | 0.66 |
11.02 |
| Moderate/severe group (n = 98) | 0.79 |
14.57 |
| F | 268.867 | 260.500 |
| p |
Note: *p
Abbreviations: NAR, nasal airway resistance; NMCT, nasal mucociliary clearance time.
There were significant differences in anxiety, depression, the number of
clinical anxiety and depression among three groups (p
| HAD | Control group (n = 79) | Mild group (n = 90) | Moderate/severe group (n = 98) | H/ |
p |
| Anxiety | 3 (2, 4) | 7 (5, 8)* | 8 (6, 10)*& | 125.899 | |
| Depression | 4 (2, 5) | 6 (3, 9)* | 7 (6, 9)*& | 78.994 | |
| Number of clinical anxiety cases (n, %) | 0 (0) | 12 (13.33%) | 16 (16.33%) | / | |
| Number of clinical depression cases (n, %) | 0 (0) | 10 (11.11%) | 12 (12.24%) | / | 0.001 |
Note: Clinical depression is defined as a HAD depression score
Abbreviation: HAD, Hospital Anxiety and Depression.
In the mild group, anxiety and depression were correlated with the NAR (r =
0.334 and 0.442; p = 0.001 and
| Anxiety | Depression | |||
| r | p | r | p | |
| NAR | 0.334 | 0.001 | 0.442 | |
| NMCT | 0.119 | 0.264 | 0.242 | 0.022 |
| CRP | 0.476 | 0.458 | ||
| PCT | 0.234 | 0.027 | 0.234 | 0.026 |
| IL-1 |
0.356 | 0.001 | 0.449 | |
| TNF- |
0.493 | 0.395 | ||
Abbreviations: CRP, C-reactive protein; IL-1
In the moderate/severe group, anxiety and depression were correlated with the
NAR (r = 0.699 and 0.566; both p
| Anxiety | Depression | |||
| r | p | r | p | |
| NAR | 0.699 | 0.566 | ||
| NMCT | 0.490 | 0.528 | ||
| CRP | 0.699 | 0.540 | ||
| PCT | 0.351 | 0.298 | 0.003 | |
| IL-1 |
0.619 | 0.578 | ||
| TNF- |
0.546 | 0.459 | ||
On a global scale, AR contributes to considerable medical and health burdens.
Primarily caused by the interaction between allergens and immunoglobulin E (IgE)
antibodies on the surface of airway cells [21], AR is clinically manifested by
sneezing, runny nose, nasal congestion, and itching. Continuous exposure to
allergens precipitates the development of chronic inflammation, which leads to
nasal mucosa damage—a trigger of various nasal symptoms. AR not only causes
breathing challenges, but may also lead to teary eyes, loss of taste and smell.
Patients with AR typically experience sleep disorders, fatigue, and irritability,
which markedly diminish their quality of life, causing significant disruptions to
their social and daily life [14, 22]. In this study, compared with healthy
individuals, AR patients scored highly for depression and anxiety, and a
significantly greater portion of these patients presented with clinical symptoms
of depression and anxiety. Compared with the mild AR patients, the patients with
moderate/severe AR had significantly higher anxiety and depression scores, and
more subjects from this group reported manifestations of clinical
anxiety and depression symptoms. These findings indicate that throughout the
course of the disease, AR promotes negative emotions of depression and anxiety,
with patients affected by severe conditions showing more pronounced impacts. A
previous cross-sectional study conducted in Portugal also reported similar
results, showing that patients with moderate/severe AR exhibited higher scores
for anxiety and depression [14]. But the proportion of anxiety and depression in
AR patients (27.8% and 36.5%) was higher than that in our study, probably due
to the different statistical methods utilized in analyses. In their research,
patients with a HAD score
The mucociliary system is an important defense mechanism that protects the nasal mucosa from allergens. Through the coordinated, directional movement of cilia, mucus and the trapped foreign particles (such as dust, bacteria, and allergens) from the nasal cavity can be moved toward the nasopharynx, where they will be ultimately swallowed or expelled [23]. The damage of nasal mucociliary clearance (NMC) can lead to recurrent episodes of AR [12, 24]. The saccharin test is commonly used to evaluate NMC, with the advantages of ease of operation, noninvasiveness, and low cost. It measures the time required for saccharin granules to be transported from the back of the nasal turbinates to the pharynx, until the subject perceives a sweet taste [25]. The NAR measurement reflects the degree of nasal ventilation in an individual. In the AR contexts, allergens stimulate the nasal mucosa, causing blood vessel wall thickening and expansion of fibrous tissue, which are the contributory factors of increased nasal resistance [26]. In this study, both NAR and NMCT were significantly higher in AR patients compared to healthy controls. The NAR and NMCT of moderate/severe AR patients were higher than those of mild AR patients, indicating the possibility of nasal dysfunction exacerbation with AR severity. The ciliary structure in the nasal epithelium of AR patients is generally damaged, characterized by sparse cilia, disorganized microtubule arrangement, and loss of motor arms [27]. AR is also accompanied by allergic reaction resulting in increased mucus secretion and the alteration in composition and properties of mucus [28, 29]. These changes contribute to longer NMCT in AR patients. Batmaz and Alicura Tokgöz [12] reported an increasing trend of NMCT in children with AR as the disease worsened in severity. We also observed a similar trend in adult patients with AR in the present study.
Chronic inflammation of the nasal mucosa is the primary factor underlying
persistent AR. Exposure to allergens stimulates the nasal mucosa, leading to the
accumulation of inflammatory cells and the release of various inflammatory
mediators, which in turn cause damage to the nasal mucosa [5, 6]. Among these
inflammatory mediators is CRP, which rapidly augments the response to infection
or tissue damage and can activate the complement system as well as regulate
phagocytosis. PCT serves as a direct marker of the intensity of systemic
inflammation [30]. An increase in IL-1
Several limitations of this research should be acknowledged. Owing to the retrospective design of this study, the results may be subject to potential bias. Therefore, a clinical prospective study is warranted to validate our findings in the future. In addition, this study only included patients with persistent AR and did not consider those with intermittent AR. Future studies could take disease persistence into account to provide a more comprehensive analysis.
As the severity of AR increases, patients exhibit higher levels of anxiety and depression, elevated inflammation levels, and impaired nasal function. Anxiety and depression are correlated with both nasal function and inflammation levels, with these correlations being more pronounced in patients with moderate/severe AR than in those with mild disease.
• Patients with moderate/severe allergic rhinitis (AR) exhibit
higher levels of C-reactive protein (CRP), procalcitonin (PCT), interleukin-1
beta (IL-1
• The nasal mucociliary clearance time (NMCT) and nasal airway resistance (NAR) in moderate/severe AR patients were higher than those in mild AR patients.
• Moderate/severe AR patients experienced worse anxiety and depression compared to mild AR patients.
• In patients with mild AR, anxiety and depression were correlated
with NAR, CRP, PCT, IL-1
• Compared with those with mild AR, patients with moderate/severe AR
exhibited stronger correlations of anxiety and depression with NAR, NMCT, CRP,
PCT, IL-1
The data analyzed in this study are available from the corresponding author upon reasonable request.
WH and LJ designed the research study. WH and XMP performed the research. WH and XPY analyzed the data. WH and LJ wrote the initial draft. All authors contributed to the important editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was approved by the ethics committee of The Second Affiliated Hospital of Heilongjiang University of Chinese Medicine (No. L2024-C01-05). All procedures were carried out in compliance with the Declaration of Helsinki, and informed consent was obtained from every participant.
Not applicable.
This study is supported by Project of Heilongjiang Provincial Administration of Traditional Chinese Medicine (2HY2024-007).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

