1 The First Clinical Medical College, Heilongjiang University of Chinese Medicine, 150040 Harbin, Heilongjiang, China
2 The First Clinical Medical College, Harbin Medical University, 150081 Harbin, Heilongjiang, China
3 School of Rehabilitation Medicine and Health Sciences, Hunan University of Medicine, 418000 Huaihua, Hunan, China
4 Department of Respiratory Medicine, The First Affiliated Hospital of Heilongjiang University of Chinese Medicine, 150040 Harbin, Heilongjiang, China
Abstract
Asthma is a common and refractory chronic inflammatory disease of the airways, with its acute episodes governed by a multitude of factors. The postulation that skin barrier damage poses a direct threat to respiratory system health is gaining traction. In fact, asthma is closely associated with various skin barrier dysfunction–associated diseases such as atopic dermatitis and atopic eczema. Patients with atopic eczema frequently exhibit concomitant atopic dermatitis, whereas asthma is often accompanied by allergic rhinitis, constituting the classic pattern of the “allergic march”. Mechanistically, these conditions share key pathological features, including genetic susceptibility (such as filaggrin (FLG) gene mutations), immune dysregulation characterized by a predominantly T helper 2 (Th2) type inflammatory response, and epithelial barrier dysfunction. Environmental factors, such as dust mite allergens, induce systemic sensitization by damaging the skin barrier, subsequently triggering airway inflammation. Targeted therapies have shown significant efficacy in both severe asthma and atopic dermatitis, highlighting a potential pathogenic pathway shared by the two conditions. In this paper, we review the relationships between asthma and skin barrier damage from a mechanistic viewpoint, thereby providing an important theoretical basis for the early prevention and precise treatment strategies of allergic diseases with overlapping pathogenic pathways.
Keywords
- asthma
- skin barrier damage
- correlation
- prevention strategies
- treatment
Bronchial asthma, abbreviated as asthma, is a heterogeneous disease characterized by chronic airway inflammation, reversible airflow obstruction, and hyperreactivity. Its clinical manifestations include respiratory symptoms such as wheezing, shortness of breath, chest tightness, and coughing, which may vary over time [1]. Asthma is a chronic disease that affects both children and adults [2]. According to global statistics regarding asthma, the prevalence rate of asthma among children worldwide is 9.1%, with a rate of 11.0% in teenagers and 6.6% among adults [3]. In 2021, the Global Burden of Disease (GBD) study reported an asthma prevalence of 3340 cases per 100,000 people, with a higher prevalence recorded in males below 20 years old [4]. The prevalence rate is positively correlated with the socio-demographic index (SDI), while the mortality rate is negatively correlated with the socio-demographic index. From 1990 to 2021, the contribution of high body mass index to disability-adjusted life years due to asthma worldwide increased by 4.3%. It has been estimated that from 2022 to 2050, the global age-standardized incidence rate will remain at a high level [4].
The onset of asthma is driven by a multitude of inducers, including infections, allergies, and environmental factors [5]. As the largest organ and primary barrier against external insults, the skin is highly vulnerable to these pathogenic factors, which collectively elicit a series of inflammatory responses that may drive the onset of asthma. In recent years, the association between asthma and skin barrier damage has attracted significant attention. Impaired skin barrier function can lead to the development of inflammatory diseases, including allergic conditions of the skin and lungs [6]. Comorbidities of asthma with atopic dermatitis (AD), allergic sensitization, and atopic eczema (AE) are common in clinical practice [7, 8, 9]. Large-scale epidemiological studies have shown that up to 25% of patients with hand AE also suffer from AD, and about 30% of patients with AD may develop asthma, constituting the classic “atopic march” pattern [10, 11]. This phenomenon highlights a pathophysiological link between the skin and the airway epithelium.
Environmental factors, such as pollutants and allergens, and genetic factors together exacerbate epithelial barrier damage. Individuals who are genetically susceptible to skin-related disorders are at increased risk of more aggressive allergen attacks, which penetrate the epidermis or airway epithelium, thereby activating both innate and adaptive immune responses and ultimately triggering a chronic inflammatory state [12]. This article reviews the relationship between asthma and skin barrier damage, with a focus on their shared pathogenic mechanisms, clinical evidence, and treatment strategies, providing clinicians with a more comprehensive perspective for diagnosis and therapy.
The skin and airway epithelium are embryologically homologous, both originating from the ectoderm, and therefore share a variety of structural proteins and molecules that support barrier function [13]. Mutations of the filaggrin (FLG) gene are among the most characterized genetic susceptibility factors that have an impact on skin barrier function [14]. Individuals carrying the FLG gene mutations experience a reduced secretion of natural moisturizing factors in the epidermis and abnormal stratum corneum structure, which paves the way for allergen penetration into the epidermis to induce sensitization [15]. In the airways, FLG protein is mainly expressed in the nasal epithelium, and its functional deficiency is highly associated with allergic rhinitis [16], which has been confirmed as an important risk factor for the development of asthma [17]. Studies showed that the FLG gene mutations represent a high-risk factor for atopic dermatitis (AD) [18] and are associated with asthma [19]. González-Tarancón et al. [20] confirmed that 12.6% of the 111 AD patients had FLG mutations. Among them, the most common FLG mutation in AD patients was R501X (9.9%), followed by R2447X (2.7%) and 2282del4 (1.8%) [20]. A survey of 261 Han Chinese patients with AD found that among 18 FLG loss-of-function mutations, 10 were newly identified, with K4671X and 3321delA being the most common mutation sites [18]. This demonstrates that there are significant regional or ethnic differences in FLG mutations. FLG deficiency alone can cause impaired skin permeability and disrupt the paracellular barrier function, while abnormalities in the extracellular lamellar bilayers caused by impaired lamellar body secretion can further lead to FLG deficiency. These structural abnormalities can lower the inflammatory threshold of the skin to irritants or haptens, forming an “outside-to-inside” pathogenic mechanism for AD [21]. Notably, a study has discovered the relationship between DNA methylation and AD in the peripheral blood mononuclear cells (PBMCs) of pregnant women exposed to bisphenol A (BPA), as well as its substitutes bisphenol S (BPS) and bisphenol F (BPF). In patients with AD, changes in DNA methylation of skin barrier–related genes (FLG) resulted in damage and inflammation of the skin barrier [22].
In addition to FLG mutations, abnormal expression of the tight junction protein claudin-1 is also associated with AD and asthma [23]. Claudin-1 is a key molecule responsible for maintaining epidermal barrier function, and its downregulation may enhance epidermal permeability [24]. Evidence has demonstrated that multiple tight junction proteins are abnormally expressed in the bronchial epithelium of asthma patients, and this leads to diminished airway barrier function, paving the way for allergen access to antigen-presenting cells, thereby triggering immune responses [25].
T helper 2 (Th2)-type inflammatory responses are central to the link between asthma and diseases related to skin barrier dysfunction [26]. Skin barrier damage induces Langerhans cells and dendritic cells to capture allergens, which subsequently migrate to the local lymph nodes and activate differentiation of naïve T cells into Th2 cells [27, 28]. Activated Th2 cells secrete cytokines such as interleukin (IL)-4, IL-5, and IL-13, initiating an inflammatory cascade, where IL-4 promotes B cells to produce immunoglobulin E (IgE), IL-5 promotes the differentiation and survival of eosinophils, and IL-13 stimulates mucus secretion by epithelial cells and enhances airway hyperresponsiveness (Table 1) [29, 30]. IL-31 is a key driver of pruritus in autoimmune skin diseases [31] and plays a critical role in the development of allergic and inflammatory disorders [32]. IL-31 induces skin itching by stimulating sensory neurons [33, 34]. Elevated IL-33 in the skin activates sensory nerve endings and group 2 innate lymphoid cells (ILC2s), promoting release of histamine and IL-31, which in turn induce scratching behavior. Scratching further disrupts the barrier and exacerbates IL-33 release, creating a localized inflammatory cycle. Skin-derived ILC2s migrate via circulation to the lungs, where it is amplified by IL-33, exacerbating airway inflammation [35]. Activation of dendritic cells by thymic stromal lymphopoietin (TSLP) of human epithelial origin triggers the conversion of cluster of differentiation 4 (CD4)+ T cells into IL-4, IL-13 and IL-5 secreting Th2 cells that mediate allergic sensitization of the lungs and the skin (Table 1) [36, 37]. In animal models, dendritic cells derived from mouse bone marrow produced TSLP signals to promote type 2 immune responses at the barrier surface [38, 39].
| Immune mediator | Source | Main functions | Pathological roles |
| IL-4 | Th2 cells | Promotes Th2 differentiation and IgE class switching | Triggers an allergic reaction and stimulates B cells to produce IgE |
| IL-5 | Th2 cells | Promote the proliferation of activated B cells and indirectly regulate the production of IgE | Stimulate the sensory nerves and trigger intense itching; inhibit barrier repair; work in synergy with IL-4 and IL-13 to promote the production of specific IgE by B cells, leading to inflammation |
| IL-13 | Th2 cells | Induces mucus secretion and promotes fibrosis | Skin fibrosis |
| IL-31 | Th2 cells | Stimulates sensory neurons | Induces itch |
| IL-33 | Epithelial cells | Activates sensory nerve endings and ILC2s to release histamine and IL-31; scratching disrupts barrier, aggravating IL-33 release | Triggers scratching and activates inflammatory cycle |
| IgE | B cells | Binds to the FcεRI receptor on the surface of mast cells | Induces an allergic reaction |
| Histamine | Mast cells, basophilic granulocytes | Expansion; increases permeability | Causes hives, rashes and bronchial constriction |
| TSLP | Epithelial cells | Activates dendritic cells | Induces Th2-type inflammation |
Abbreviations: Ig, immunoglobulin; IL, interleukin; ILC2s, group 2 innate
lymphoid cells; TSLP, thymic stromal lymphopoietin; Fc
Mast cells play a key role in both skin and airway inflammation [40, 41]. In
patients with chronic spontaneous urticaria, mast cells can be activated through
the crosslinking mechanism of the Fc epsilon RI (Fc
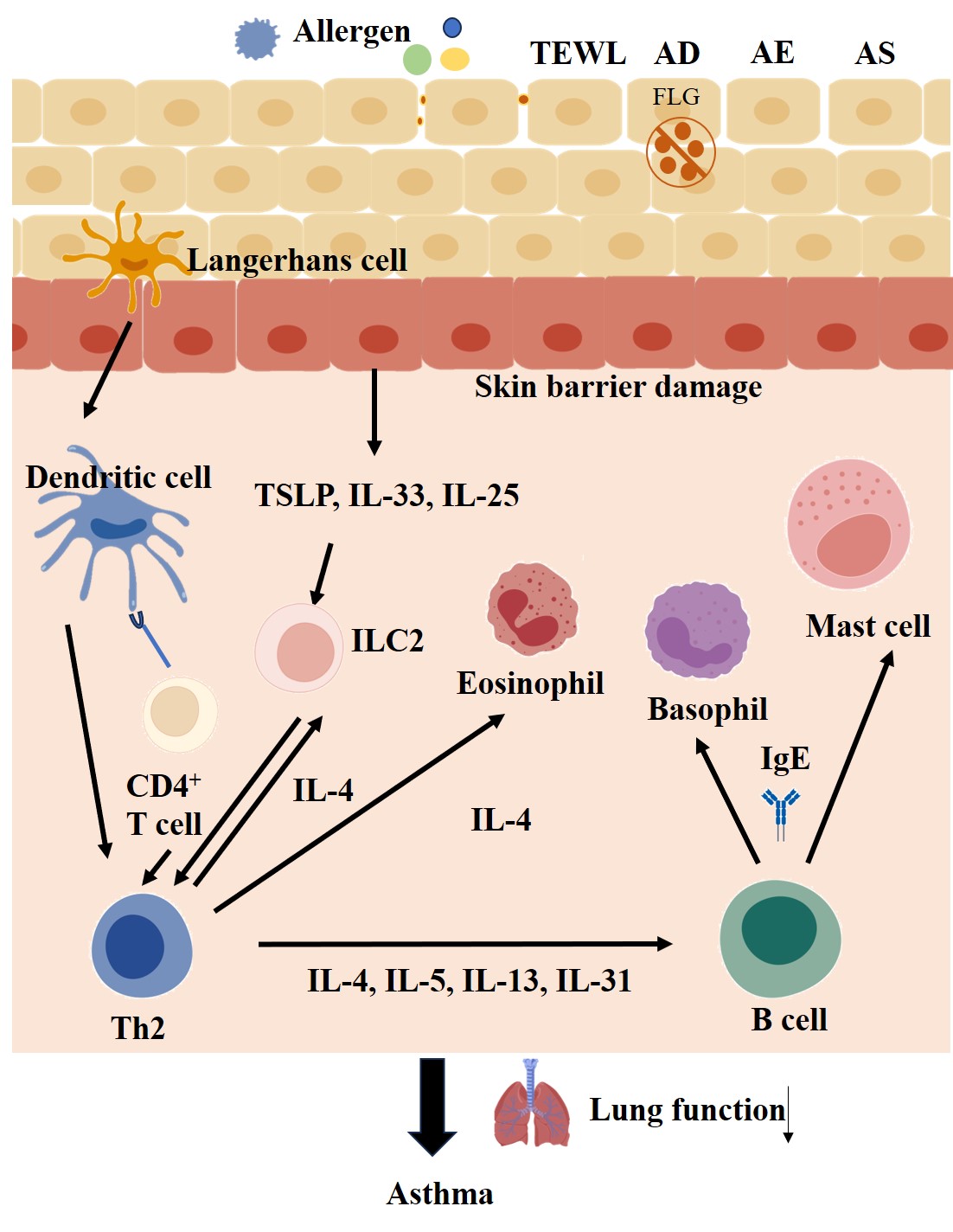 Fig. 1.
Fig. 1.
Molecular mechanism of asthma development as a result of impaired skin barrier function. The image was drawn through BioGDP (https://biogdp.com/) and reprinted from Färdig et al. [45], available under the CC BY-NC license (https://creativecommons.org/licenses/by-nc/4.0/). Abbreviations: AD, atopic dermatitis; AE, atopic eczema; AS, allergic sensitization; FLG, filaggrin; IgE, immunoglobulin E; IL, interleukin; ILC2, group 2 innate lymphoid cell; TEWL, trans-epidermal water loss; Th2, T helper 2; TSLP, thymic stromal lymphopoietin; CD4, cluster of differentiation 4.
As an important characteristic of impaired skin barrier function, itching is a core symptom of AD [47]. This sensation triggers the urge to scratch, exerting a severe impact on quality of life (QoL) and psychosocial well-being [48, 49]. Intense itching leads to scratching, which further damages the skin barrier, promotes the release of inflammatory mediators, and facilitates the invasion of allergens [48]. Transient Receptor Potential (TRP) channels play an important role in pruritus. Transient Receptor Potential Vanilloid-1 (TRPV1) has been reported to be upregulated in the skin of patients with AD [50]. TRPV1 activity is increased in allergic rhinitis [51], characterized by itchy eyes and sneezing. Another Trp channel that plays a major role in the propagation of itch, Transient Receptor Potential Ankyrin 1 (TRPA1), acts downstream of skin neuropeptide substance P, triggering itch sensation [52]. TRPA1 is increased in nerve fibers, keratinized cells, and trypsin-like positive mast cells from the diseased skin of patients with AD. Notably, the TRPA1 expression in dermal cells of healthy skin remains minimal [53]. In addition, Transient Receptor Potential Vanilloid-3 (TRPV3) expression is increased in AD-injured skin [54].
The neuropeptide substance P is expressed in both the skin and the airways, and participates in the process of neurogenic inflammation [55, 56]. Also, mast cells and sensory nerves are involved in neurogenic inflammation and pruritus associated with chronic skin inflammation [44]. Substance P promotes the release of histamine from human skin mast cells through a pertussis toxin-sensitive and protein kinase C-dependent mechanism [57]. In the asthmatic airway, substance P is involved in bronchoconstriction and mucus secretion [58]. This neuro-immune interaction may be an important mechanism linking pathological changes in the skin and the airways (Fig. 1).
Environmental factors play a key role in the pathogenesis of asthma and skin
barrier damage. House dust mites (HDM) are the primary indoor allergens
responsible for allergic rhinitis and asthma [59]. HDM sensitization is present
in asthmatic adults, whose severity is associated with exposure to the HDM
allergens [60]. The Der p 2 protein is one of the main dust mite excreted
allergens that play a key role in the pathogenesis of asthma [61]. Wan et
al. [62] discovered that higher concentrations of HDM were associated with a
higher prevalence of asthma and allergies in the children investigated. A Der f 1
concentration exceeding 2000 ng/g is of clinical significance since it provides
useful insights into the diagnosis of eczema. This indicates a dose–response
trend in the prevalence of asthma and eczema with increasing concentrations of
HDM (threshold
AD is a recurrent, inflammatory, and pruritic skin disease that is often accompanied by respiratory symptoms [67, 68]. Thirteen prospective cohort studies (including four birth cohort studies and nine atopic eczema (AE) cohort studies) have shown that infants with extrinsic AD are at risk of developing asthma [69]. Through a systematic review and meta-analysis of 39 publications, including 458,810 participants, Li et al. [70] found that the relative risk (RR) of asthma in AD was 2.16 (95% confidence interval [CI]: 1.88–2.48), with higher risk noted in severe AD cases (RR: 2.40; 95% CI: 1.96–2.94) compared to mild AD (RR: 1.82; 95% CI: 1.03–3.23) or moderate AD cases (RR: 1.51; 95% CI: 1.30–1.75). The risk of developing asthma was slightly higher in early-onset AD than in late-onset AD and was higher in boys than in girls. Epidemiological surveys indicate that the incidence of asthma continues to rise among both children and adults with AD, suggesting that this phenomenon is attributed to overlapping genetic risk variations and environmental triggers [71]. According to research data from China, 16.7% of patients with AD also suffer from asthma, and 33.7% have allergic rhino-conjunctivitis. The prevalence of these extracutaneous comorbidities increases with age [72]. Early abnormalities in skin barrier function can be detected in AD patients, manifested as reduced stratum corneum hydration, increased trans-epidermal water loss (TEWL), and altered pH values [73]. Mechanistically, under the influence of genetic factors, the innate skin barrier dysfunction allows allergens and other factors to penetrate the body through the skin barrier, triggering type 2 inflammation and the production of inflammatory mediators that cause allergy. These activated cells and inflammatory mediators can further act on the airways. In addition, upon inhalation by patients with AD, these allergens can directly affect the airway epithelium, leading to activation of sensitized inflammatory cells and synthesis of inflammatory mediators, which in turn cause airway smooth muscle spasm, mucosal edema, and increased mucus secretion—all of which precipitate the development of asthma [74]. It has been reported that asthmatic patients have reduced levels of antiviral interferons (IFNs). Rhinovirus or respiratory syncytial virus released by the virus-infected nasal epithelial cells spreads to the lower airways, leading to epithelial cell death and the release of active IL-33, thereby triggering type 2 inflammation. Meanwhile, antiviral IFNs can directly inhibit the proliferation of Th2 cells and ILC2s, as well as the production of type 2 cytokines [75]. In addition, Staphylococcus aureus, a common infection in the AD patients, releases superantigens that further amplify the inflammatory response; this mechanism further plays a role in acute asthma exacerbations [76]. AD is categorized according to its severity (Scoring Atopic Dermatitis (SCORAD) score) as mild, moderate, and severe. As the severity of AD increases, the incidence of asthma also gradually rises. Growing evidence demonstrates that patients with AD are prone to developing asthma since this skin disorder is substantially associated with the severity of asthma and the duration of skin damage [77].
Among the affected children, 45% developed symptoms in the first six months of life, 60% in the first year of life, and up to 85% developed AD by age 5 [78]. The severity of AD was associated with the risk of developing rhinitis and elevated levels of total and specific immunoglobulin E (IgE) antibodies [79]. While investigating the prevalence of allergic rhinitis and asthma among 2270 children with AD, Kapoor et al. [80] found that nearly 66% of those aged 3 years suffered from allergic rhinitis or asthma, and that the presence of these disorders was associated with poor control of AD. These processes are consistently characteristic of the “atopic march”. Repeated sensitization to cutaneous allergens in animal models leads to epidermal barrier dysfunction, which results in AD phenotypes and systemic sensitization, and increases the risk of allergic rhinitis, lung inflammation, and airway hyperresponsiveness [81]. Skin exposure to aeroallergens in mouse models induces systemic Th2 immunity, which leads to nasal allergic reactions, suggesting that the skin is an effective site for antigenic sensitization during the development of experimental allergic rhinitis [82]. Indeed, murine models have shown that exposure of the epidermis to ovalbumen after removal of the stratum corneum induces a strong systemic Th2 immune response characterized by an increase in IL-4 secretion by T cells from the draining lymph nodes and high levels of allergen-specific IgE and immunoglobulin G1 (IgG1) [83]. Findings from these animal experiments lend further credence to their interplay, which constitutes the “atopic march”.
There is a moderate association between AE in infancy and asthma in childhood [84], which is demonstrated in a prospective, population-based study investigating the association between AE occurrence at age 2 and asthma development at age 6 (odds ratio [OR]: 1.80; 95% CI: 1.10–2.96) [85]. Eczema patients who carry specific IgE antibodies against common environmental allergens at 2 to 4 years of age are at a higher risk of developing allergic rhinitis and asthma than those with eczema without IgE sensitization [86]. The Tucson Children’s Respiratory Study found that eczema in infancy was an independent risk factor for persistent wheezing, and that 18% of children with wheezing at age 6 had eczema before age 2 [87]. A prospective study examining the development of allergy and asthma in infants with eczema who were followed up to 7 years of age showed that eczema improved in 82 of 94 children, but 43% of them developed asthma and 45% developed allergic rhinitis [79]. Children with eczema are at higher risk of developing asthma, and early exacerbations of eczema are associated with an increased risk of sensitization to inhalant allergens. These studies suggest that IgE sensitization to environmental allergens in patients with eczema is an important factor in the progression to the allergic phenotype of asthma. A study conducted in Tasmania on the impact of eczema on the development of asthma from childhood to adulthood found that childhood eczema was significantly associated with new-onset asthma in three separate life stages, namely preadolescence, adolescence, and adult life [88]. Collectively, these studies illustrate the concept of “atopic march”. Children with AE or those who are sensitized to allergens in early childhood are more likely to develop asthma and allergic reaction syndrome [69]. A prospective study of 10-year-old children showed that the progression from AE in infancy to early allergic reaction syndrome and subsequently to asthma in childhood supports the concept of the “atopic march” [89].
Chronic urticaria (CU) is characterized by intensely itchy wheals that rapidly
develop central edema surrounded by erythematous areas [90, 91]. In addition,
mast cell mediators induce local vasodilatation, which increases capillary
permeability and plasma leakage, thereby intensifying erythema [92]. The link
between chronic urticaria and asthma may reflect aberrant crosstalk between
helper T cells (Th2 type) and mast cells. For example, Shefler et al.
[93] reported that mast cell activity may be indirectly influenced by distal T
cells. Perhaps this subtle abnormality of crosstalk may lead to the co-occurrence
of chronic urticaria and asthma in the same patient. In a cross-sectional study
of 11,271 patients with chronic urticaria 1215 (10.8%) were found to have asthma
compared with 4342 (6.5%) of 67,216 controls, suggesting that patients with
chronic urticaria are at significantly higher risk of developing asthma than the
general population [94]. Mast cells act as common effector cells in both
urticaria and asthma, and their activation mechanisms are common to both
diseases. IgE sensitization is higher in patients with chronic urticaria, similar
to atopic diseases [95]. In patients with chronic urticaria, allergens act by
stimulating the production of IgE, which binds to Fc
Characterized by erythema and silvery scales on the trunk and extremities, psoriasis is a chronic inflammatory disease of the skin, with a prevalence ranging up to 11.43% [97]. Psoriasis is considered an immune-mediated disease associated with IL-23 and IL-17 [98]. Both psoriasis and asthma share common inflammatory cytokine–mediated mechanisms. IL-17 may serve as a biomarker for this phenotype, and recent studies have shown that asthma-associated genes with high IL-17 expression overlap with those altered in psoriasis [99]. This suggests that psoriasis and asthma have similar immunophenotypes. Abnormal activation of T helper 17 (Th17) cells in patients with psoriasis leads to high levels of IL-17 expression, a proinflammatory factor that not only drives the hyperproliferation of keratin-forming cells to form characteristic scaly plaques but also migrates to respiratory tissues through bloodstream. Approximately 24% of asthmatics present with a “psoriasis-like immunophenotype” characterized by frequent acute exacerbations, significant sputum and mucosal neutrophilia, reduced lung microbiota diversity, and elevated urinary thromboxane B2 levels [99]. Of note, there is a two-way link between psoriasis and asthma. Cohort [100] and cross-sectional [101, 102, 103] studies have demonstrated the risk of asthma in patients with psoriasis. Two cohort studies have examined the risk of psoriasis in patients with asthma [104, 105]. A meta-analysis revealed that patients with psoriasis had a higher risk of developing asthma (OR: 1.48; 95% CI: 1.28–1.68). Meanwhile, the overall risk of psoriasis was higher in patients with asthma (OR: 1.33; 95% CI: 1.23–1.44) [106].
According to the literature, there is a close and multifaceted link between skin barrier dysfunction and the clinical manifestations of asthma: (1) AD and asthma: Genetically and environmentally driven skin barrier defects enhance allergen entry into the skin, thereby activating a systemic type 2 (Th2) immune response. Activated inflammatory cells and mediators then act on the airways, causing airway inflammation and asthma. (2) AE and asthma: IgE sensitization to common allergens is a key risk factor of asthma development in eczema patients. Poor control of early eczema is closely associated with subsequent development for allergic airway disease. (3) CU and asthma: In both of these conditions, mast cells act as the central effector cells. Abnormal Th2-type immune responses and IgE‑mediated mast cell activation and degranulation are common pathological mechanisms linking the two conditions. (4) Psoriasis and asthma: The association between psoriasis and asthma lies in the abnormal activation of the IL‑23/Th17 immune axis, which is different from the Th2 pathway. Elevated proinflammatory factors such as IL‑17 not only drive the development of psoriatic skin lesions but also affect the airways via the circulation and lead to an asthma phenotype characterized by neutrophilic inflammation. Different types of skin diseases trigger local and systemic inflammation through distinct immune pathways, key factors, and effector cells. In the event of an impaired skin barrier or a dysregulated immune system, this inflammatory response can “remotely” affect the airways, indirectly contributing to different aspects of asthma development, such as its clinical manifestations, severity, and risk augmentation.
A three-tiered approach to curtailing the risk of asthma induced by skin barrier damage has been proved to be effective in reducing morbidity and improving prognosis:
Primary prevention (risk avoidance): This preventive strategy centers on blocking the penetration of allergens into damaged skin. This approach entails allergen profiling by means of patch testing and serum lgE measurements, as well as the implementation of targeted protection [107, 108]. For those who are allergic to dust mites, anti-mite bedspreads and regular (weekly) washing of bedding in hot water are required. Further steps and requirements include installation of air purifier (High Efficiency Particulate Air (HEPA) filter) and maintenance of humidity at 40–50% to prevent mold growth. Additionally, microbial exposure intervention and early childhood exposure to farm environments have been found to increase microbial diversity and reduce asthma risk [109].
Secondary prevention (early intervention): This form of preventive strategy focuses on skin barrier repair. Barrier repair is assessed by TEWL testing every three months. Targeted prophylaxis of the skin barrier should be introduced early in life before the clinical manifestations of AD appear. This is important because in newborns and infants, the skin barrier is immature at birth and typically achieves full functionality by 6 months of age [110]. Two randomized controlled pilot studies showed that daily application of a standard petroleum jelly moisturizer to the whole body, starting within the first week of life, is effective in preventing the later onset of AD [111, 112]. In addition, daily treatment of infants with moisturizers reduces the risk of dry skin and dermatitis, which is associated with lower TEWL, lower pH, and increased stratum corneum hydration, demonstrating that the protective effect of moisturizers is attributed to improved skin function [113]. Similarly, a randomized controlled trial observed that moisturizers containing ceramides and amino acids may help reduce AD in high-risk neonates [114].
Tertiary prevention (immunomodulation): Tertiary prevention entails remodeling of immune homeostasis through lifestyle interventions. This strategy requires 7–8 hours of deep sleep to maintain the physiological rhythm of melatonin for inhibiting mast cell activation [115]. For genetically susceptible individuals, supplementation with specific probiotic strains (e.g., Lactobacillus rhamnosus strain GG) should be considered during pregnancy and infancy [116]. Furthermore, skin barrier disruption triggers Th2 inflammation by upregulating proinflammatory cytokine expression, and conversely, Th2 immune cytokines impair skin barrier function through reduced expression of stratum corneum proteins, which enhances epidermal allergen activation, thereby creating a vicious cycle [117, 118, 119]. Active anti-inflammatory therapy can be deployed to achieve effective control of AD and mitigation of Th2 inflammation. Experimental studies on the epidermis of ovalbumin (OVA)-sensitized mice showed that topical corticosteroid pretreatment inhibited the expansion of eosinophils in the skin and intestinal mucosa, while oral administration suppressed the induction of allergic symptoms [120]. Through a retrospective cohort study, Fukuie et al. [121] found a significant decrease in serum total IgE levels in patients with moderate to severe AD after receiving continuous active topical corticosteroid treatment for 2 years.
Omalizumab, by binding to free IgE, blocks its interaction with the Fc epsilon
RI (Fc
| Medicine | Clinical trial number | Target site of action | Mechanism of action |
| Omalizumab | NCT00314574 | Anti-IgE monoclonal antibody | Neutralizes free IgE and reduces mast cell activation |
| Tezepelumab | NCT02525094 | TSLP inhibitor | Blocks the epithelial alarm protein TSLP |
| Dupilumab | NCT02948959 | IL-4R |
Blocks the IL-4/IL-13 signaling pathway |
| IBI3002 | NCT06213844 | IL-4R |
Blocks the IL-4R |
| Lebrikizumab | NCT05916365 | Anti-IL-13 | Suppresses the effect induced by IL-13 |
| Tralokinumab | |||
| Mepolizumab | NCT05598814 | Anti-IL-5 | Inhibits the IL-5 signaling pathway |
| Reslizumab | |||
| Benralizumab | NCT03401229 | Anti-IL-5Ra | Inhibits the signal transduction induced by the IL-5Ra ligand |
Abbreviations: Ig, immunoglobulin; IL, interleukin; TSLP, thymic stromal lymphopoietin.
Lebrikizumab and tralokinumab are biologic agents used to treat asthma, and both
of them act on the IL-13 target (Table 2) [133, 134, 135, 136]. Mepolizumab and reslizumab
exert inhibitory effects on asthma by targeting IL-5 (Table 2) [137, 138].
Featuring enhanced antibody-dependent cell-mediated cytotoxicity,
benralizumab—a humanized monoclonal antibody targeting IL-5R
Proactively repairing the skin has become a new strategy for the prevention and
treatment of allergic diseases. The results of the Protocol for a Randomized,
Blinded, Parallel, Three-Group, Phase II Trial (PAF Study) showed that emollients
effectively prevent AD in high-risk newborns [141]. For patients with AD,
emollients containing ceramides can repair the lamellar structure of the stratum
corneum and reduce trans-epidermal water loss [142]. A prospective randomized
controlled clinical trial showed that treatment with barrier-enhancing
moisturizers can delay the relapse of AD [143]. The optimized emollient mixture
has been demonstrated to possess an ability to maintain a low pH level (
Research has indicated that the dysfunction of the skin barrier caused by
microbial imbalance may increase the risk of systemic allergic inflammation,
including respiratory inflammation [148]. Targeted therapies against Staphylococcus aureus (such as fusidic acid cream) have shown effectiveness in managing AE
[149]. Microbiota transplantation therapies, such as topical application of
Roseomonas mucosa derived from healthy donors, have demonstrated
therapeutic improvements in skin barrier function in patients with AD [150]. The
microbial community in the nasopharynx of children with asthma exhibited a higher
Shannon diversity index (combined intensive care unit (ICU) and medium care unit
(MCU), p = 0.002), and there were significant differences in the
composition of the microbial community compared to the healthy control group
(R2 = 1.9%, p
Table 3 offers detailed comparisons regarding efficacy, advantages and disadvantages, cost-effectiveness, indications, and contraindications across three therapeutic modes: targeted biological agents, skin barrier repair therapies, and microbial regulation.
| Dimension | Targeted biological agents | Skin barrier repair therapies | Microbial regulation |
| Efficacy | Excellent | Good | General |
| Advantages | 1. Targeted precise treatment, systemic control of inflammation | 1. Convenient for oral/topical use, low cost | 1. Regulation of immune homeostasis |
| 2. Long-term control | 2. High safety | 2. Reduced recurrence rate | |
| 3. Wide applicability | |||
| Disadvantages | 1. High cost | 1. Long-term safety risks, due to blood clots, and infections | 1. Unstable efficacy |
| 2. Increased infection risk (reoccurrence of tuberculosis, reactivation of hepatitis B) | 2. Consistent applications of barrier repair agents are required to achieve effectiveness | 2. High strain specificity | |
| 3. Requires regular injections | 3. Slow start | ||
| Cost-effectiveness | Low cost-effectiveness ratio | Excellent cost-effectiveness ratio | 1. Low cost of probiotics and oligogalactans |
| 2. High cost in research and development of microbial transplantation or engineered bacteria therapy | |||
| Indications | Moderate to severe disease: | Mild to moderate disease or combination therapy: | Early/chronic diseases: |
| 1. Eosinophilic asthma | 1. Atopic dermatitis | 1. Staphylococcus aureus-related AD | |
| 2. AD | 2. Dry skin/barrier damage | 2. Intermission asthma | |
| 3. Psoriasis | 3. Individuals with allergy | ||
| 4. Chronic urticaria | |||
| Contraindications | 1. Active infections (e.g., tuberculosis, hepatitis) | 1. Progressive lesions | 1. Immunocompromised individuals (risk of bacterial transplantation) |
| 2. Allergy to biologic ingredients | 2. Individuals at high risk of blood clots | 2. Open wounds (topical application contraindicated) | |
| 3. Widespread infections |
Abbreviation: AD, atopic dermatitis.
Asthma is a chronic inflammatory disease of the respiratory tract and poses an adverse impact on patients’ quality of life [157]. Currently, the global incidence of asthma continues to show an increasing trend, and its prevalence has been projected to increase from a global population of nearly 300 million patients to 400 million by 2025 [158]. Skin barrier damage has a direct or indirect impact on respiratory health, causing severe medical conditions such as asthma [159]. Multifaceted evidence, spanning the genetic, immune, and environmental aspects, suggests that the skin barrier is a prerequisite for the development of asthma, highlighting the impairment of epidermal barrier as a shared pathogenic mechanism of these diseases [45, 160].
To establish a causal relationship between skin barrier damage and asthma,
multi-omics and immunological evidence is necessary to explain the association
between the skin condition and respiratory decline. Additionally, randomized
controlled trials are required to validate the efficacy of skin barrier damage
interventions in reducing asthma incidence. It is equally important to identify
infants at risk of lifelong chronic atopic diseases, in order to facilitate the
implementation of early therapeutic interventions. Treatments aimed at
maintaining and repairing the epidermal barrier in infants may prevent the
subsequent development of asthma. Screening potential therapeutic candidates
based on biomarkers (such as eosinophils, IgE, TSLP, TEWL) is crucial for
advancing the development of personalized treatments for pathologies linked to
both skin and respiratory conditions. Trans-epidermal water loss (TEWL) is an
important biomarker for evaluating the skin barrier function [161]. Infants aged
3 months experiencing impaired skin barrier have a TEWL value
Current research landscape in this field is constrained by several limitations. First, the standardization of assessment methods is lacking. Existing studies adopted various TEWL measurement devices (open chamber vs. closed chamber), employed different environmental temperature and humidity conditions, and relied on different skin areas for measurement, posing a challenge to directly compare their results [166, 167]. Furthermore, most studies conducted evaluations at only one time point, failing to reflect the dynamic changes in skin barrier function and delineate their temporal relationship with asthma activity. Secondly, despite a clear association between FLG mutations and an increased risk of asthma, approximately 40% of carriers do not develop asthma [168], implying that epigenetic modifications or protective effects from environmental factors could be at play. For instance, early exposure to a farm environment may enhance immune tolerance through microbial exposure, thereby counteracting pathological risks arising from genetic factors. Conversely, abnormal skin barrier function in a significant number of asthmatic children with non-FLG mutations indicates that other genes or environmental exposures, such as excessive use of detergents and changes in humidity, could independently contribute to the risks for these conditions [169]. Thirdly, animal models are unable to accurately depict the chronic process of atopy observed in humans. Of note, despite the epidemiological association between skin barrier damage and the risk of asthma, it has been reported that the incidence of asthma in some children with severe eczema did not show a corresponding reduction even after the patients had achieved significant amelioration of skin symptoms as a result of active barrier repair treatments using moisturizers [170]. These conflicting results may be attributed to several factors: (1) Missed treatment window period: The optimal window for preventing immune deviation and promote skin barrier repair is typically between 3 and 6 months after birth, commonly known as the “sensitization critical period”; (2) Insufficient protection of distant organs: It is challenging to improve integrity of the airway barrier if localized skin treatment is utilized; (3) Persistent dysbiosis: Microbial imbalance may continue to drive systemic inflammation even after skin symptoms improve. Notably, some patients with severe asthma have normal skin barrier function, while others with generalized AD display no respiratory symptoms [169]. This discrepancy likely stems from several aspects: (a) Tissue-specific regulatory mechanisms: For instance, ILC2s have different differentiation states in different organs; (b) Differences in local environment: The microbial composition, pH value, and humidity could vary between the skin and the airway; (c) Differences in neural innervation: The neural circuitry underlying the itch–scratch response in the skin is far more pronounced than that of the airways. Asthma and skin barrier dysfunction are closely linked through shared genetic backgrounds, immune mechanisms, and environmental influences. Elucidating these relationships not only deepens our understanding of the overall pathogenesis of allergic diseases, but also provides a theoretical basis for treatment strategies.
Owing to advances in molecular technologies, our understanding of the association between asthma and skin barrier damage has expanded substantially, establishing a robust and systematic theoretical framework commonly referred to as the “skin-lung axis”. Future research should focus on the following aspects:
(i) To effectively control the exacerbation of skin barrier dysfunction and asthma, it is crucial to conduct early prevention strategies that have been validated through multicenter randomized controlled trials. Implementing preventive measures for skin barrier care, applying skin barrier repair agents consistently on infant patients, controlling the risk of atopic disease progression, and preventing asthma occurrence.
(ii) Future investigations should also focus on formulating strategies for precision-guided interventions. This entails designing individualized approaches tailored to patients’ biological and clinical profiles such as their barrier-dominant, immune-dysregulation, and neuro-immune subtypes. For example, patients classified as having immune dysregulation may be better candidates for targeted biological therapies.
(iii) By combining genomic, epigenomic, transcriptomic, and proteomic data, future studies should conduct comprehensive analyses to delineate the shared pathogenic mechanisms underlying asthma and skin barrier diseases. Particular attention should be given to the contribution of non-Th2 inflammatory pathways (e.g., Th17/T helper 22 (Th22)) in non-hypereosinophilic asthma and in specific skin phenotypes.
(iv) Conceptualization efforts should be made to establish interdisciplinary teams to deliver efficient therapies for patients affected by asthma and skin barrier diseases. For instance, a joint outpatient clinic tackling both dermatologic and respiratory pathologies can be set up to provide diagnostic assessments and treatments to a niched patient group.
In summary, asthma and skin barrier damage arise from complex interactions among genetic, environmental, and immune factors. Future research should prioritize the development of integrated management strategies targeting both the skin and the respiratory tract. By combining early barrier protection with precise targeted therapies, it may be possible to achieve the goal of “one intervention for multiple diseases”, ultimately improving the overall quality of life for patients with allergic conditions.
• Asthma and skin barrier damage share overlapping pathogenic mechanisms, including genetic mutations, immune and inflammatory abnormalities, neuroimmune and itch–scratch circuitry, and environmental factors that compromise barrier integrity.
• Asthma is associated with various skin barrier diseases, such as atopic dermatitis and atopic eczema.
• Targeted biologics, barrier repair therapies, and microbiome modulation represent treatment strategies that address the shared pathogenic mechanisms underlying both skin barrier dysfunction and asthma.
• Given the limited efficacy of current early prevention and treatment strategies for asthma, more comprehensive therapeutic approaches are needed to enhance patient quality of life.
The data used to support the findings of this study are available from the corresponding author upon request.
JBY and ZYL designed the research study and wrote the first draft. ZY, XW, LF and JZT performed the research. All authors contributed to the important editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

