1 International Nursing School, Hainan Medical University, 571199 Haikou, Hainan, China
2 Department of Nursing, Hainan General Hospital (Hainan Affiliated Hospital of Hainan Medical University), Hainan Medical University, 570311 Haikou, Hainan, China
Abstract
The incidence of major adverse cardiovascular events (MACE) following percutaneous coronary intervention (PCI) remains high among patients with acute ST-segment elevation myocardial infarction (STEMI). The Pan-Immune-Inflammation Value (PIV), an indicator of systemic inflammatory status, has demonstrated prognostic utility across multiple diseases and may serve as an indicator of risk of STEMI patients undergoing PCI. This study aimed to investigate the predictive value of PIV for MACE within one year following PCI in patients with acute STEMI.
A retrospective analysis was conducted on 200 patients diagnosed with acute STEMI who underwent PCI at the Hainan General Hospital between January 2022 and December 2023. Based on the occurrence of MACE within one year post-PCI, patients were categorized into a MACE group (n = 148) and a non-MACE group (n = 52). Univariate and multivariate logistic regression analyses were conducted to identify factors influencing MACE occurrence in STEMI patients following PCI, and the predictive value of PIV was assessed using receiver operating characteristic (ROC) curve analysis.
There were no statistically significant differences between groups in gender, smoking status, body mass index (BMI), red and white blood cell counts, Left Atrium Diameter (LAD), Left Ventricular End-Diastolic Diameter (LVDd), total protein, albumin, triglycerides (TG), and total cholesterol (TC) (p > 0.05). However, significant differences were observed in age, heart rate, left ventricular ejection fraction (LVEF), and PIV (p < 0.05). Multivariate logistic regression analysis identified age, heart rate, PIV, and LVEF as independent predictors of MACE within one year post-PCI in patients with STEMI (p < 0.05). ROC curve analysis showed that the predicted area under the curve (AUC) for PIV in predicting the occurrence of MACE was 0.866 (p < 0.001) (95% confidence interval (CI): 0.809–0.922), with a standard error of 0.029, and a Youden index of 0.62. At this threshold, the sensitivity was 77.70%, and the specificity was 84.60%.
PIV demonstrates high predictive value for MACE within one year after PCI in patients with STEMI. It may serve as a valuable biomarker for assessing the risk of percutaneous coronary atherosclerosis events in this population. Further multi-center studies are warranted to validate its clinical utility.
Keywords
- PIV
- ST elevation myocardial infarction
- percutaneous coronary intervention
Acute ST-segment elevation myocardial infarction (STEMI) is a severe cardiovascular condition characterized by high incidence, high disability rate, and high mortality rate [1]. With the advancement and widespread adoption of percutaneous coronary intervention (PCI) technology, the survival rate of STEMI patients has significantly improved. However, the incidence of postoperative major adverse cardiovascular events (MACE) remains high, seriously affecting patient prognosis and quality of life [2, 3]. Therefore, identifying biomarkers capable of early prediction and evaluation of post-PCI MACE risk in STEMI patients is particularly important.
The Pan-Immune-Inflammation Value (PIV), as an indicator for assessing systemic inflammatory status, has shown significant prognostic value in various diseases in recent years [4]. By integrating neutrophil, platelet, monocyte, and lymphocyte counts, PIV provides a comprehensive reflection of the body’s immune and inflammatory response. It has shown potential in predicting clinical outcomes in patients with advanced cancers and has also been closely associated with the prognosis of conditions such as hypertension [5, 6]. In predicting post-PCI MACE in STEMI patients, PIV may serve an important predictive role.
Monitoring inflammatory markers such as PIV may help in the early identification of high-risk patients for postoperative MACE, thereby enabling the development of more individualized treatment strategies and optimizing patient outcome management [5, 7]. The PIV has demonstrated unique value in predicting MACE within one year after PCI in patients with acute STEMI. Compared to individual inflammatory markers such as the neutrophil-to-lymphocyte ratio (NLR), PIV integrates neutrophil, platelet, monocyte, and lymphocyte counts, offering a more comprehensive assessment of systemic immune-inflammatory status. Zhen et al. [8] found that elevated NLR was associated with poor long-term outcomes in patients with acute coronary syndrome (ACS) and obstructive sleep apnea (OSA). However, PIV, as a comprehensive indicator, may offer a more comprehensive risk assessment.
The motivation for this study was to investigate the potential advantages of PIV over single inflammatory markers in predicting MACE risk after PCI in STEMI patients and in guiding the optimization of treatment strategies. This study aimed to evaluate the predictive value of PIV for MACE within one year post-PCI in STEMI patients. This provides scientific evidence to support clinicians in developing more precise and individualized treatment plans, and improves patient outcomes and quality of life.
A retrospective cohort of 200 individuals who underwent percutaneous coronary
intervention (PCI) for acute STEMI at the Hainan General Hospital between January
2022 and December 2023 was meticulously selected as the study population. The
inclusion criteria were as follows: (1) All patients met the clinical diagnostic
criteria for acute STEMI [9]; (2) Patients presented with
acute chest pain lasting more than 30 minutes, with symptoms onset occurring
within less than 12 hours; (3) Biochemical markers of myocardial necrosis, such
as serum troponin, showed a rise and/or fall consistent with myocardial ischemia,
along with symptoms of ischemia and electrocardiogram (ECG) findings
demonstrating ST-segment elevation
Exclusion criteria were as follows: (1) Presence of severe organic cardiopulmonary, hepatic, or renal dysfunction, malignant tumor, autoimmune disease, or hematological disorder; (2) Unstable condition or recurrence of chest pain after initial relief; (3) History of previous myocardial infarction or cerebral infarction; (4) Contraindications to anticoagulation or antiplatelet therapy.
By applying these stringent criteria, the study aimed to enhance the accuracy, reliability, and clinical relevance, thereby facilitating a more detailed understanding of PCI outcomes in STEMI patients during the specified timeframe. This study was approved by the Ethics Committee of Hainan General Hospital (EC-YLY-2025-171-01), and the patients provided written informed consent.
A total of 200 patients were categorized into two distinct groups based on postoperative outcomes: the major adverse cardiovascular events (MACE) group (n = 148) and the non-MACE group (n = 52). This classification was based on the presence or absence of major adverse cardiovascular events within the first year following PCI. Patients in the MACE group experienced a range of adverse cardiovascular events, including but not limited to recurrent acute myocardial infarction, malignant arrhythmias, heart failure, cardiogenic shock, and all-cause mortality during the 12-month follow-up period. Patients who did not experience such events were assigned to the non-MACE group as a comparison cohort for statistical analysis.
The PIV value was calculated as follows:
PIV = (neutrophil count
General patient information was collected using an electronic medical record
system and patient interview methods. The collected data were analyzed using SPSS
version 27.0 (International Business Machines Corporation, Armonk, NY, USA). The
Shapiro-Wilk test was used to assess normality. Normally distributed continuous
variables were presented as mean
No statistically significant differences were observed between the MACE and
non-MACE groups in gender, smoking status, body mass index (BMI), red blood cell
count, white blood cell count, Left Atrium Diameter (LAD), Left Ventricular
End-Diastolic Diameter (LVDd), total protein, albumin, triglycerides (TG), and
total cholesterol (TC) (p
| Baseline characteristics | MACE group (n = 148) | Non-MACE group (n = 52) | t/ |
p-value | |
| Age (years) | 61.62 |
59.46 |
5.480 | ||
| Gender | 0.400 | 0.527 | |||
| Male | 75 | 29 | |||
| Female | 73 | 23 | |||
| Smoking | 0.009 | 0.923 | |||
| Yes | 70 | 25 | |||
| No | 78 | 27 | |||
| BMI (kg/m2) | 21.00 |
20.75 |
0.911 | 0.363 | |
| Red blood cell count ( |
4.70 |
4.69 |
0.284 | 0.777 | |
| White blood cell count ( |
8.92 |
8.75 |
1.601 | 0.111 | |
| LAD (mm) | 40.19 |
39.74 |
0.692 | 0.490 | |
| LVDd (mm) | 46.21 |
45.80 |
0.808 | 0.420 | |
| LVEF (%) | 64.09 |
59.17 |
8.133 | ||
| Heart rate (beats/minute) | 83.14 |
73.10 |
9.030 | ||
| PIV | 265.89 |
192.44 |
8.806 | ||
| Total protein (g/L) | 77.97 |
78.28 |
0.328 | 0.743 | |
| Albumin (g/L) | 33.29 |
33.04 |
0.539 | 0.591 | |
| TG (mmol/L) | 1.73 |
1.75 |
–0.892 | 0.373 | |
| TC (mmol/L) | 4.29 |
4.40 |
1.176 | 0.241 | |
Note: BMI, body mass index; LAD, Left Atrium Diameter; LVDd, Left Ventricular End-Diastolic Diameter; LVEF, left ventricular ejection fraction; TG, triglycerides; TC, total cholesterol; PIV, Pan-Immune-Inflammation Value; MACE, major adverse cardiovascular events; PCI, percutaneous coronary intervention; STEMI, ST-segment elevation myocardial infarction.
Variables found to be significant in the univariate analysis, namely age, heart
rate, PIV, and LVEF, were included as independent variables in the multivariate
logistic regression analysis. After value assignment (original values were used),
the occurrence of MACE was treated as the dependent variable (MACE = 1, no MACE =
0). The multivariate logistic regression results showed that age, heart rate,
PIV, and LVEF were independent predictors of MACE within one year following PCI
in patients with STEMI (p
| Variable | SE | Wald | p-value | Exp ( |
95% CI | ||
| Lower limit | Upper limit | ||||||
| Age | 0.619 | 0.171 | 13.183 | 1.858 | 1.330 | 2.595 | |
| Heart rate | 0.340 | 0.099 | 11.686 | 0.001 | 1.405 | 1.156 | 1.707 |
| LVEF | 0.282 | 0.065 | 18.884 | 1.325 | 1.167 | 1.505 | |
| PIV | 0.033 | 0.008 | 15.709 | 1.034 | 1.017 | 1.051 | |
| Constant | –86.719 | 17.468 | 24.645 | 0.000 | - | - | |
Note: LVEF, left ventricular ejection fraction; PIV, Pan-Immune-Inflammation Value; CI, confidence interval; SE, standard error.
Receiver operating characteristic (ROC) analysis was performed to assess the
predictive value of PIV for MACE within one year after PCI in patients with
STEMI. The results showed that the area under the curve (AUC) for PIV was 0.866
(p
| Factor | AUC | Standard error | 95% CI | Youden index | Sensitivity (%) | Specificity (%) | p-value | Cut-off value |
| Age | 0.716 | 0.040 | 0.638–0.793 | 0.28 | 41.90 | 86.50 | 0.001 | 62.50 |
| Heart rate | 0.819 | 0.031 | 0.758–0.880 | 0.48 | 79.10 | 69.20 | 75.50 | |
| LVEF | 0.851 | 0.029 | 0.795–0.907 | 0.52 | 81.11 | 69.20 | 60.27 | |
| PIV | 0.866 | 0.029 | 0.809–0.922 | 0.62 | 77.70 | 84.60 | 223.04 |
Note: LVEF, left ventricular ejection fraction; PIV, Pan-Immune-Inflammation Value; ROC, receiver operating characteristic; AUC, area under the curve.
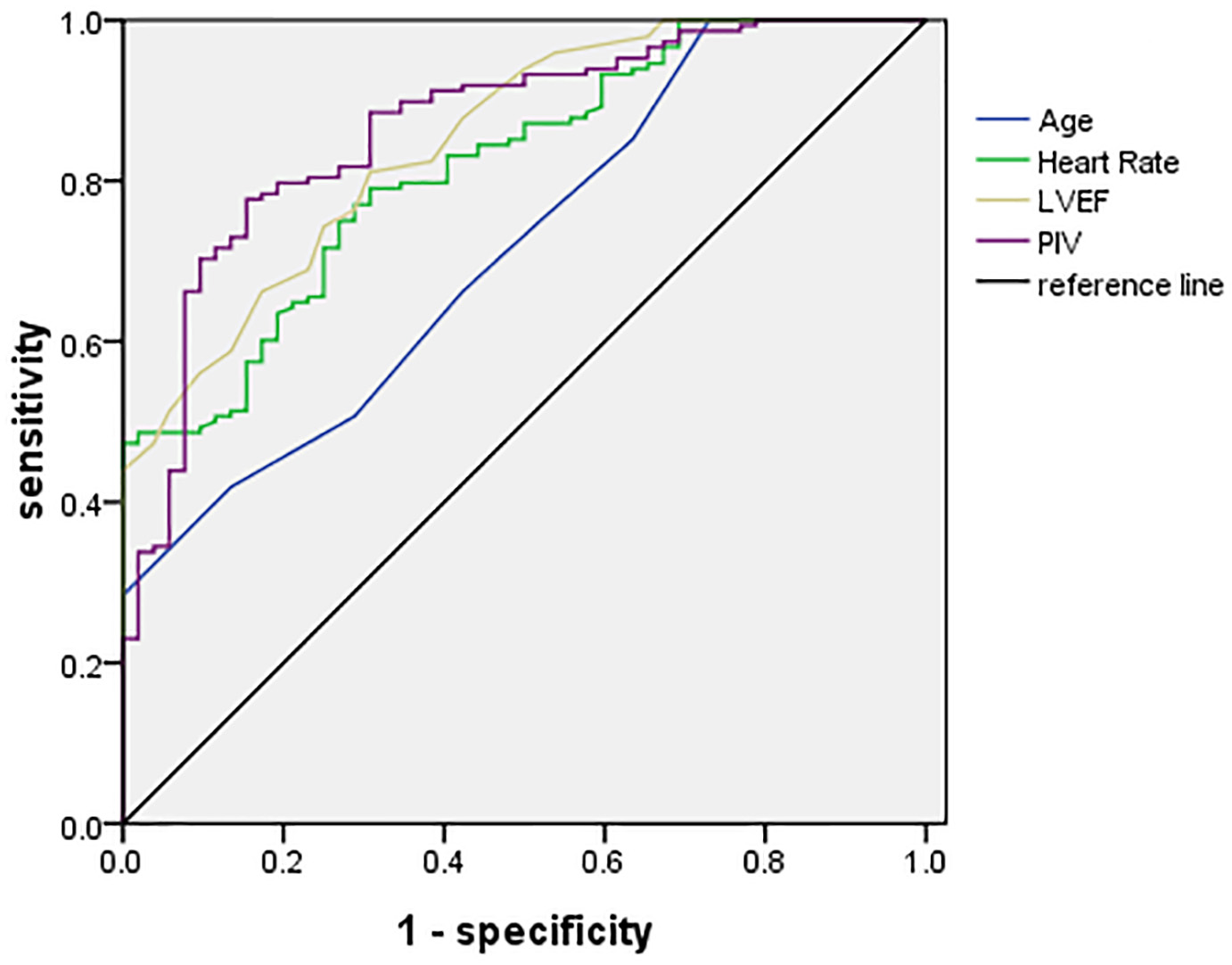 Fig. 1.
Fig. 1.
ROC curve analysis. The ROC curve illustrates the diagnostic performance of each predictor for MACE following PCI in STEMI patients. Among the variables analyzed, PIV demonstrated the highest AUC, indicating superior predictive accuracy.
STEMI, a severe cardiovascular emergency, demands rapid coronary artery blood flow restoration to limit myocardial injury. While PCI is the standard treatment for STEMI, MACE post-PCI still greatly influences patient prognosis. Therefore, identifying effective predictive markers to recognize high-risk patients and enable proactive interventions is essential for improving outcomes in STEMI patients [10]. This study focused on investigating the utility of the Pan-Immune-Inflammation Value (PIV) in predicting the occurrence of MACE among patients with STEMI following PCI.
PIV is a composite index derived from routine blood tests. These cellular components are crucial in mediating inflammation and immune responses, and variations in their levels may reflect the systemic inflammatory status and immune function [11, 12, 13]. Lymphocytes regulate the intensity and direction of immune responses, thereby maintaining immune homeostasis. An elevated PIV may reflect a heightened inflammatory response and immune activation, closely associated with myocardial damage, reperfusion injury, and the subsequent tissue repair processes following PCI in STEMI patients [14, 15]. Therefore, PIV holds promise as a valuable biomarker for predicting the risk of MACE after PCI in STEMI patients [16].
Age emerges as a significant risk factor for cardiovascular complications. In this study, age was confirmed as an independent predictor of MACE post-PCI in STEMI patients. An elevated heart rate may reflect enhanced cardiac sympathetic nervous system activity and increased myocardial oxygen demand [17]. In STEMI patients, an accelerated heart rate could exacerbate the severity of myocardial ischemia and injury, thereby raising the risk of cardiovascular events. The identification of heart rate as an independent predictor in this study further underscores the clinical importance of heart rate management in the management of STEMI patients.
PIV, the focal point of this study, demonstrated excellent predictive accuracy for MACE following PCI in STEMI patients. ROC analysis revealed an AUC of 0.866 for PIV, indicating its high sensitivity and specificity. This observation suggests that PIV could serve as a simple yet robust biomarker for risk assessment following PCI in STEMI patients. In the study of coronary artery disease and atherosclerosis, investigating the biological mechanisms of inflammation and immunity is crucial, although many aspects remain unresolved. The recent study [18] has shown that immune infiltration in atherosclerosis may be mediated by the copper tumor-associated iron droop gene, offering a new perspective for understanding the pathogenesis of the disease. This gene may influence the progression of atherosclerosis by regulating the infiltration and function of immune cells. Moreover, the interaction between inflammation and immunity in atherosclerosis is complex and refined, involving the activation and regulation of multiple cell types and signaling pathways. As a comprehensive immune-inflammation marker, PIV integrates data from multiple immune cell types and more comprehensively reflects the systemic immune-inflammatory status. Therefore, it may offer unique advantages in elucidating these biological mechanisms. Future research should aim to further analyze these mechanisms and evaluate the value of biomarkers such as PIV in disease prediction, therapeutic decision-making, and prognosis assessment.
Stratifying STEMI patients into distinct risk categories based on PIV levels assists clinicians in formulating more personalized treatment strategies. For individuals with elevated PIV, proactive interventions such as intensified pharmacological regimens, lifestyle modifications, or consideration of more aggressive revascularization procedures may be employed to reduce the likelihood of MACE occurrence [19]. Regular monitoring of PIV changes provides timely insights into a patient’s inflammatory status and immune response, supporting appropriate adjustments to therapeutic strategies for optimal prognostic management.
Utilizing the predictive capacity of PIV allows for the early identification of high-risk individuals, enabling the focused deployment of healthcare resources for close monitoring and timely intervention. This targeted approach enhances overall healthcare efficiency and promotes more rational allocation of limited clinical resources. These strategies underscore the importance of PIV in post-PCI risk stratification and management among STEMI patients, highlighting its potential to improve clinical outcomes and optimize the use of healthcare resources.
Although the results of this study are significant, there are still limitations, such as potential selection and information bias inherent in retrospective studies, relatively small sample sizes, single-center sources, and focusing only on PIV in patients with STEMI. In order to verify the predictive power and clinical applicability of PIV, prospective studies are needed to collect more comprehensive and accurate data to reduce retrospective study deviations and improve the reliability and universality of results. The single-center retrospective design of this study or the introduction of selection bias highlights the need for future multi-center prospective study verification. Expanding the sample size and integrating multi-center data can further improve the reliability and versatility of the results. Multi-center cooperation can integrate clinical and data resources in multiple places, laying a solid foundation for in-depth research. Future research can also explore the joint prediction performance of PIV and other biomarkers (such as C-reactive protein (CRP), interleukin-6 (IL-6)). The integration of multiple markers can more comprehensively evaluate the pathophysiological status of the body and provide more adequate information for clinical decision-making. In addition, this study did not adjust for multiple comparisons (e.g., Bonferroni correction), which simplified statistics but increased the risk of Type I errors and affected the reliability of the results. Future research should examine the predictive value of PIV on different populations (such as the elderly, diabetics, and hypertensive patients) and evaluate its application potential.
In conclusion, PIV is a simple and reliable predictor. This study is the first to confirm the independent predictive value of PIV for MACE in STEMI patients following PCI, with its AUC (0.866) significantly surpassing traditional markers such as the neutrophil-to-lymphocyte ratio (NLR). It demonstrates significant value in predicting MACE in patients with STEMI after PCI. Monitoring changes in PIV can facilitate early identification of high-risk patients and optimize prognostic management to improve clinical outcomes.
The data used to support the findings of this study are available from the corresponding author upon request.
JYQ and NZ designed the research study. HZ, HMZ and JYQ performed the research. CL and XDL analyzed the data. JYQ wrote the first draft. All authors contributed to revising the manuscript critically for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was approved by the Ethics Committee of Hainan General Hospital (EC-YLY-2025-171-01). The entire experimental procedure was carried out in accordance with the Declaration of Helsinki. The patients provided written informed consent.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

