1 University College London Hospitals NHS Foundation Trust, NW1 2PG London, UK
2 Royal Free London NHS Foundation Trust, NW3 2QG London, UK
3 Chelsea and Westminster Hospital NHS Foundation Trust, SW10 9NH London, UK
Abstract
Inferior vena cava (IVC) anomalies are uncommon, with absent infrarenal IVC reported as one of the rarest congenital vascular anomalies. They are associated with deep vein thrombosis (DVT) particularly in younger patients, which can have serious complications if not recognised and treated appropriately.
We describe the case of a 35-year-old male with absent infrarenal IVC presenting with acute DVT.
Radiologists and clinicians should understand the relationship between IVC anomalies and DVT, particularly in younger patients.
This case highlights the importance of multi-modality diagnostic and interventional imaging techniques in assessing and treating patients effectively.
Keywords
- thrombosis
- vascular
- radiology
- interventional radiology
- congenital
- inferior vena cava
- case report
Absent infrarenal inferior vena cava (IVC), also known as infrarenal IVC agenesis is an extremely rare congenital vascular anomaly with an estimated prevalence of 0.0005–1% [1]. It is one of several types of IVC anomaly, the most common of which include but not limited to IVC duplication, left-sided IVC, interruption of the IVC, and circumaortic or retroaortic left renal vein. IVC anomalies are estimated to occur in 0.3–0.5% of the population, and whilst in most cases these do not result in pathology, some of these result in lower limb venous stasis and thus cause an increased risk of deep vein thrombosis (DVT) particularly in the young adult cohort [1, 2]. We report a case of an adult male who presented with unprovoked DVT with extensive clot burden who on further investigation had complete absence of the infrarenal IVC. We aim to widen clinical awareness of this rare anomaly as a risk factor for thrombosis and encourage the consideration of multimodality diagnostic imaging in select individual cases of unprovoked bilateral DVT.
A 35-year-old male presented to a London district general hospital with a one-day history of left thigh pain and swelling. He works a sedentary desk job, is independent in activities of daily living, moderately active, and is a non-smoker. He has no relevant past medical history, with no personal or family history of DVT. He weighed 60.5 kg and had a normal body mass index (BMI), and his only regular medication is escitalopram (20 mg once daily) prescribed for anxiety. There were no identifiable provoking factors for DVT, such as recent travel, infection, or trauma.
On examination the patient exhibited unilateral swelling of the left thigh and calf. There were no overlying skin changes, and the remaining clinical examination findings were normal. He underwent a Doppler ultrasound of the left leg which demonstrated a proximal common femoral vein thrombus (Fig. 1).
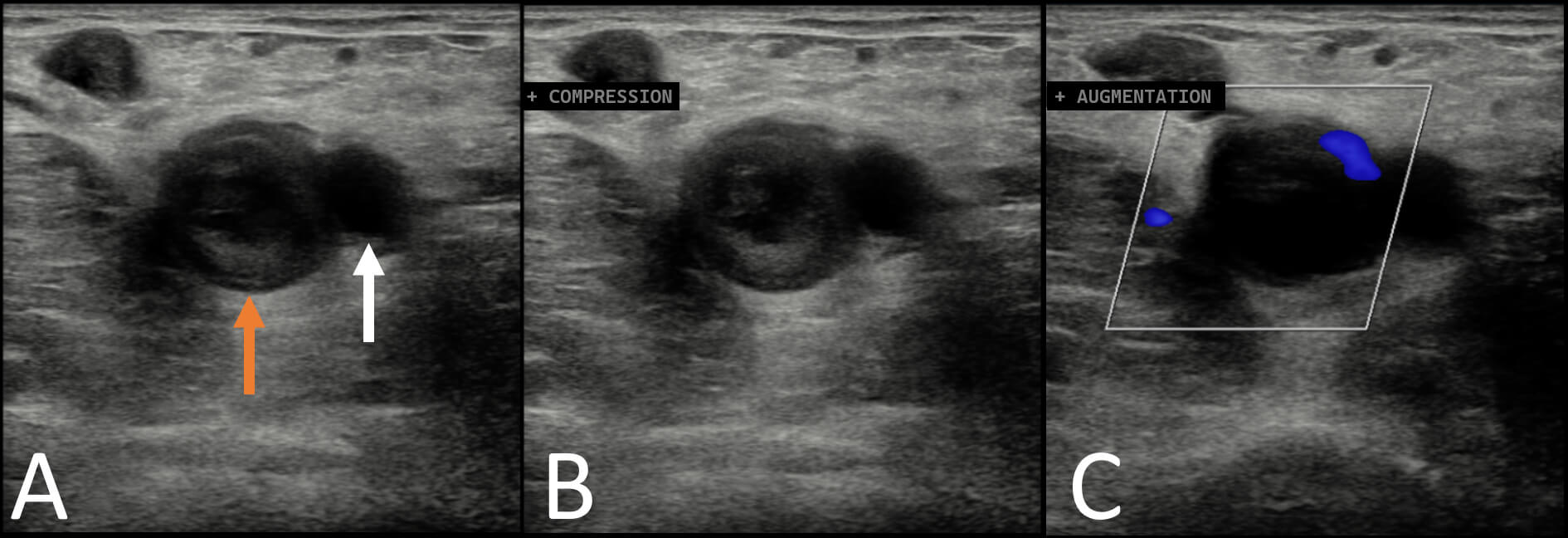 Fig. 1.
Fig. 1.
Ultrasound images of the left common femoral vein. (A) Echogenic thrombus within the proximal femoral vein (orange arrow). Femoral artery denoted by white arrow. (B) Thrombus was not compressible. (C) Limited to no venous flow was demonstrated on Doppler imaging on augmentation by calf compression.
The upper extent of the common femoral vein thrombus was not determined so the patient underwent a computed tomography (CT) venogram of the chest and lower limbs. This revealed the absence of the infrarenal IVC (Fig. 2A), with external iliac veins draining into separate left and right lumbar veins (Fig. 2C). As a result, the calibre of the lumbar, azygous, and hemi-azygous veins was larger than normal (Fig. 2B). The thrombus in the left leg extended from the popliteal vein into the left lumbar vein where its upper extent lay at the level of the fourth lumbar vertebral body (Fig. 2D). Additionally, the left internal iliac vein also contained thrombus. The remainder of the CT chest, abdomen, and pelvis examination was normal.
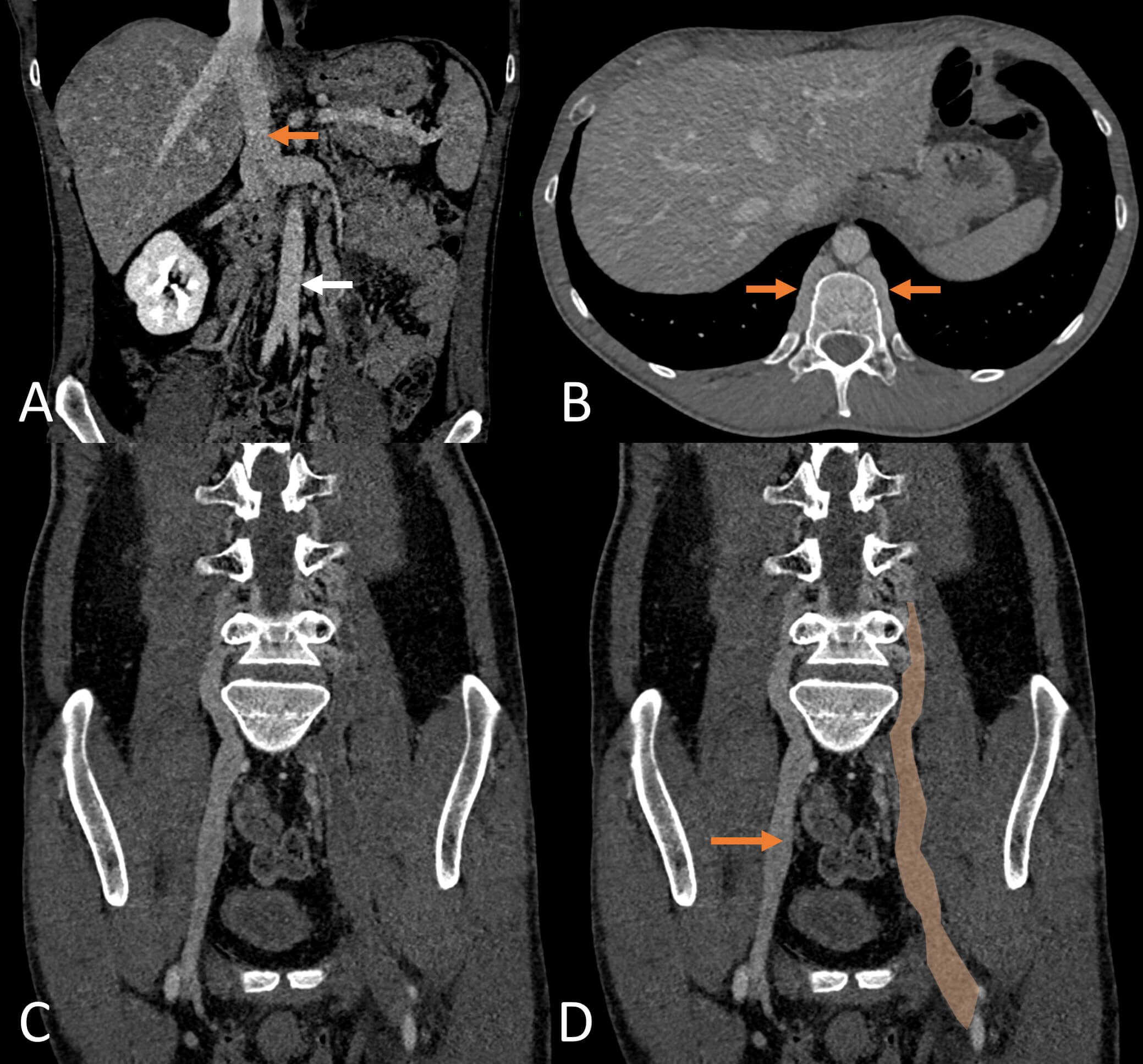 Fig. 2.
Fig. 2.
Computed tomography (CT) venogram. (A) Coronal view of the upper abdomen shows that the inferior vena cava (orange arrow) prematurely terminates with a right and left renal vein. The infrarenal inferior vena cava (IVC) is absent. Abdominal aorta (white arrow). (B) Unusually prominent lumbar veins (arrows) on axial view. (C,D) Modified coronal views of the lower abdomen and pelvis. The right external iliac vein (arrow) drains into the lumbar vein replacing the typical IVC pathway for blood flow. No IV contrast is seen within the left external iliac and lumbar veins superiorly to the level of L4 (orange box) indicative of extensive thrombus.
Following this diagnosis, the patient was administered low molecular weight heparin (11,000 units subcutaneous Tinzaparin) and transferred to a tertiary centre. Five days later direct venography was performed (Fig. 3A), and an EKOS™ catheter was deployed under the interventional radiology team. EKOS™ technology combines ultrasonic waves with endoluminal administration of tissue-type plasminogen activator (tPA). The endoluminal ultrasound creates micro-perforations within the clot, increasing the surface area available for tPA activity and thereby accelerating thrombolysis.
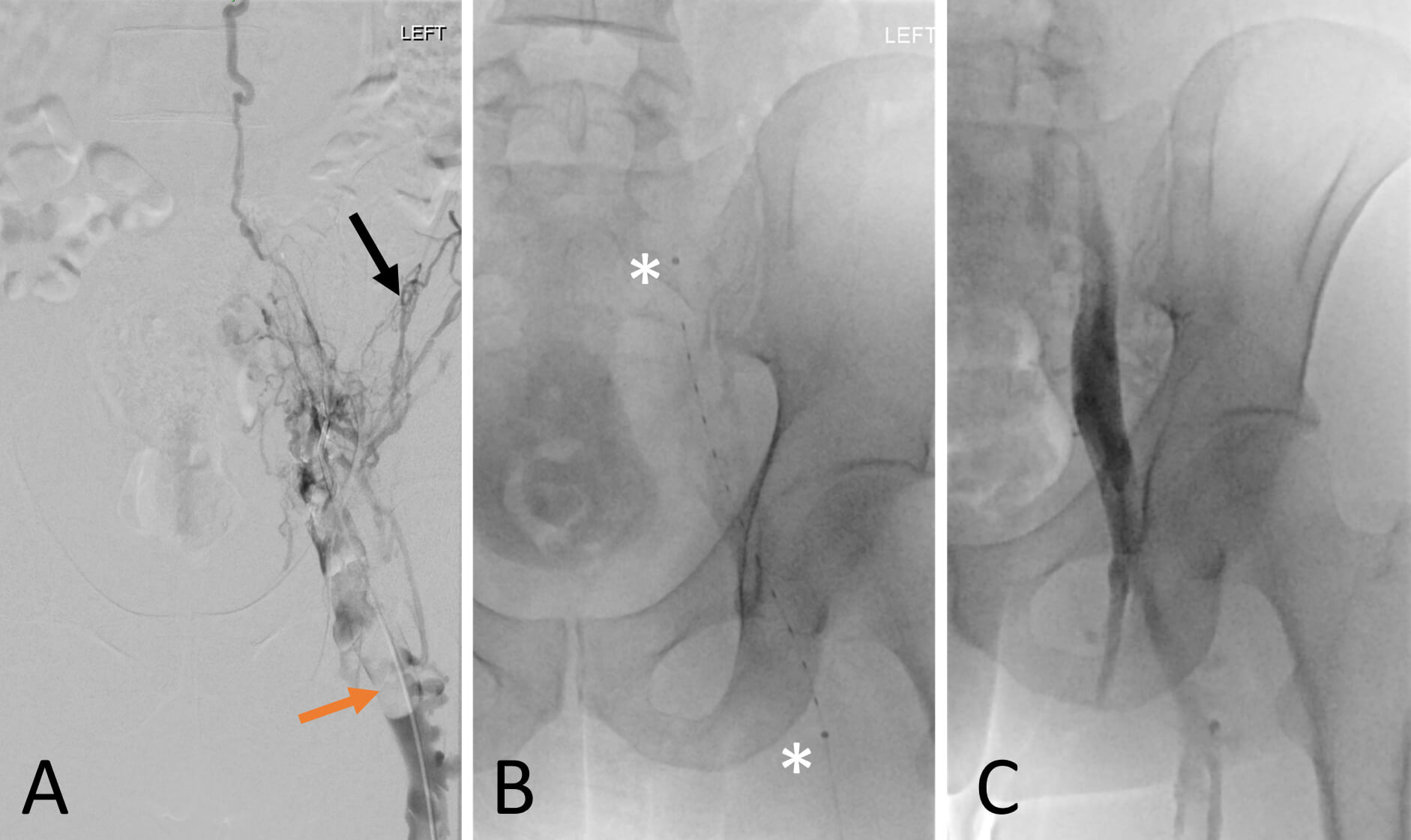 Fig. 3.
Fig. 3.
Left leg venography and catheter directed thrombolysis with EKOS™ endovascular system. (A) Left leg venogram demonstrates poor contrast opacification of the left common femoral vein (orange arrow) and contrast filling multiple small collateral veins (black arrow). (B) EKOS™ catheter in situ with asterisks denoting the 12 cm treatment zone within the proximal femoral vein and external iliac vein. (C) Repeat venogram demonstrates previously occluded common femoral vein was patent with flow up into the external iliac vein after 24 hours of treatment. The IVC was not demonstrable on venography.
The left popliteal vein was punctured, and the catheter and wire were
manipulated to the occluded left lumbar vein. A 12 cm treatment zone was deployed
(Fig. 3B) using the EKOS™ endovascular system (5.4 Fr infusion catheter,
product number: H74939590106120, Boston Scientific, Marlborough, MA, USA). Due to
the unusual variant anatomy, the stent position was definitively confirmed using
dynaCT. Endoluminal tPA infusion was administered for a total of 18 hours
(alteplase 12 mL/h, and heparin 20 mL/h). Fibrinogen blood levels were monitored
every six hours, and fibrinogen blood products were transfused when levels fell
to
He improved clinically and repeat venography performed the following day demonstrated improved blood flow and a reduced clot burden (Fig. 3C). Consequently, the endoluminal catheter was removed after a total of 24 hours. He recovered with no postoperative complications and was discharged on a six-month regimen of oral rivaroxaban (15 mg twice daily for 3 weeks, followed by 20 mg once daily).
At a six-month follow-up in the haematology clinic, he had made a good clinical recovery. He had returned to his normal activity levels and reported no residual symptoms in the left leg. A repeat lower leg ultrasound performed six months post-treatment was normal (Fig. 4), and blood screening for clotting disorders was also normal. The long-term management plan includes lifelong anticoagulation therapy with annual clinical reviews under the haematology team. The CARE checklist has been attached as Supplementary Material associated with this article.
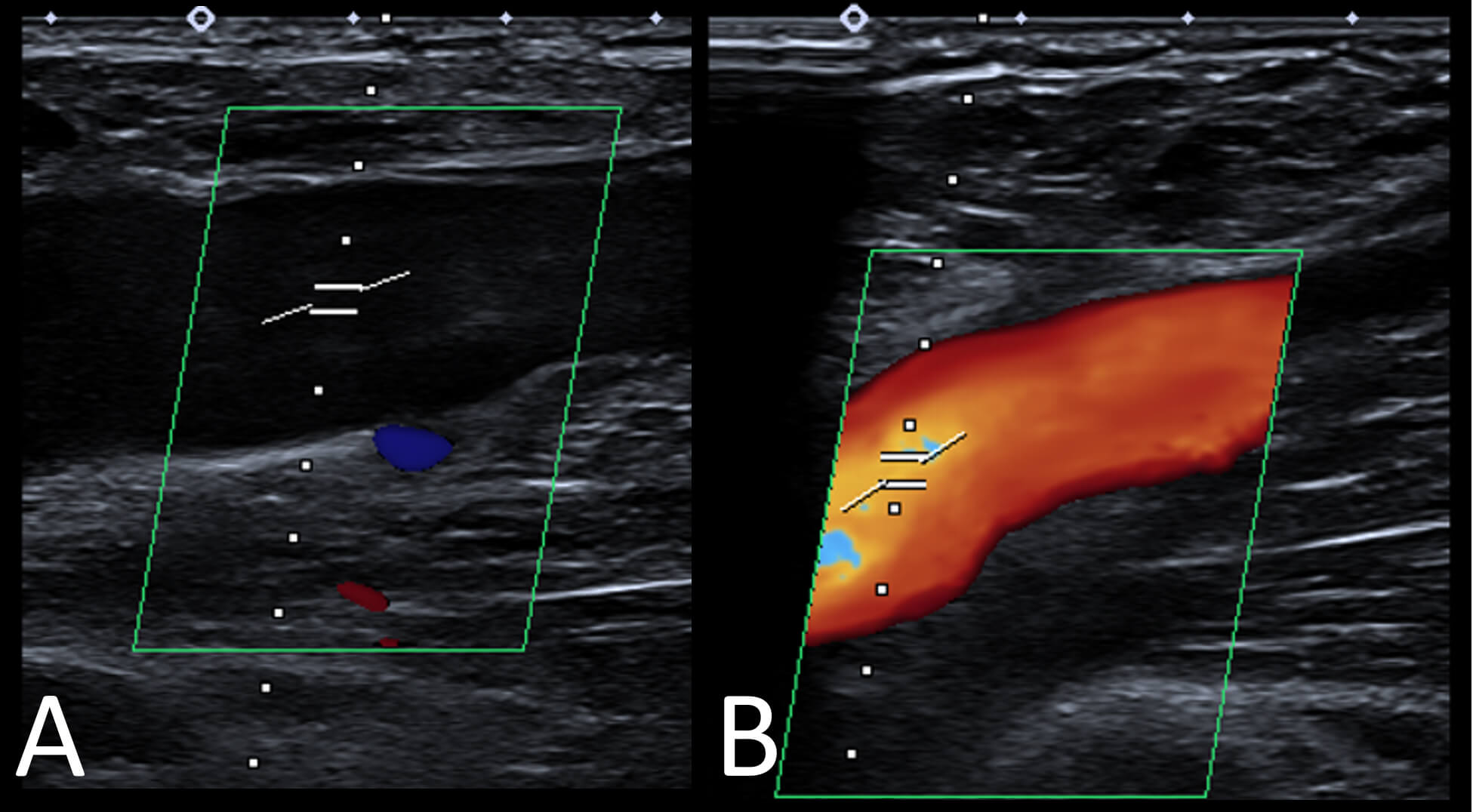 Fig. 4.
Fig. 4.
Six-month post-treatment ultrasound images of the left common femoral vein. (A) Normal appearance of the common femoral vein with no evidence of residual thrombus. (B) Normal Doppler flow is demonstrated.
The IVC embryologically originates from the cardinal, subcardinal, supracardinal, and vitelline veins. IVC anomalies typically result from one or more of these vessels failing to regress or anastomose and the type of abnormality depends on which vessels persist and or regress. For example, duplicated IVCs result from the persistence of the left supracardinal vein, left sided IVCs occur because of abnormal right supracardinal vein regression, and an interrupted IVC with azygos continuation results from a failure to form the anastomosis between the right subcardinal vein and the vitelline vein [3]. While the above examples represent the more common IVC anomalies, estimates of the prevalence of IVC anomalies amongst the general population vary widely in the literature ranging from 0.3% to 8.7% [1, 4].
Congenital absence of the infrarenal IVC is an extremely rare IVC anomaly, with prevalence reported to be 0.0005–1% [1]. Unlike the other IVC variants, an absent infrarenal IVC is not thought to be a true embryonic anomaly, but rather a consequence of in utero peri-natal IVC thrombosis, which causes regression and involution of the once present IVC. Indeed, there have been cases of image-proven perinatal IVC thrombus where subsequent imaging demonstrates an absent IVC [5]. Anatomically, as in the case we describe the renal, suprarenal, and hepatic segments of the IVC are preserved, with the common iliac or the internal/external iliac veins draining into the azygous venous system via the ascending lumbar veins. Despite the ascending lumbar veins expanding to accommodate the larger-than-usual blood flow, patients can still suffer from venous insufficiency and pelvic congestion syndrome [6].
IVC anomalies are an established risk factor for deep vein thrombosis (DVT) particularly in younger patients and are found in up to 5% of under 30s with a confirmed DVT diagnosis [1]. Congenital absence of the infrarenal IVC often results in venous insufficiency and the pooling of venous blood in the lower limbs. This venous stasis is one of the key risk factors in Virchow’s triad of factors that contribute to thrombosis. There are multiple case reports in the literature of patients with otherwise asymptomatic absent infrarenal IVC who present with DVT, oftentimes recurrent [7]. The development of guidelines recommending investigations for malignancy in unprovoked DVT, as well as the ever-increasing provision of cross-sectional imaging, has led to more cases of absent infrarenal IVC being identified. Clinicians should be suspicious of absent IVC in cases where patients are under 30 years old and/or present with proximal or bilateral thrombus [8].
There is yet to be consensus on the long-term management of these patients, and this single case study is limited in its ability to produce any large-scale conclusions [6]. We are also limited by our 6-month follow-up interval which restricts our ability to assess longer-term outcomes. In the future, a case series or systemic review would be able to more comprehensively compare different approaches to managing the risks that comes with this anomaly. Although absent infrarenal IVC is generally considered a benign variant, the resultant venous stasis and DVT risk could result in life-threatening pulmonary embolus. It has also been shown that these patients are at a higher risk of developing post-thrombotic syndrome than DVT patients without absent IVC [8]. In most cases, patients are placed on lifelong anticoagulation and advised to use compression stockings [6, 9]. In contrast, some authors have found that patients who were taken off anticoagulation after a mean treatment period of 35 months had no more risk of DVT than the general population [10]. If these findings could be replicated in larger patient groups (a challenge in such a rare variant), then the risk of haemorrhage that comes with lifelong anticoagulation could potentially be avoided. Until then, a patient-centred mutualistic approach seems most prudent. After an initial period of anticoagulation, patients ought to be consulted and advised by a specialist clinician familiar with the literature. Only after a comprehensive risk-benefit analysis has been done, considering the individual patient characteristics and comorbidities, should a shared decision regarding long term anticoagulation be made.
Any data included in this study is available upon request by contacting the corresponding author.
GA, JC, RC and AS made significant contributions to the design, planning and write-up of the manuscript. GA, JC, RC and AS contributed to important editorial changes in the manuscript and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Written informed consent for the publication of this case report was obtained from the patient. This manuscript complies with the principles of the Declaration of Helsinki.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/BJHM50375.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




