1 Department of Colorectal Medicine, Zhejiang Cancer Hospital, Hangzhou Institute of Medicine (HIM), Chinese Academy of Sciences, 310022 Hangzhou, Zhejiang, China
2 Department of Radiology, Zhejiang Cancer Hospital, Hangzhou Institute of Medicine (HIM), Chinese Academy of Sciences, 310022 Hangzhou, Zhejiang, China
Abstract
Advanced biliary tract cancer (BTC) is an aggressive malignancy, and often presents with poor prognostic outcomes despite standard chemotherapy. This study aimed to evaluate the preliminary efficacy and safety of toripalimab in combination with gemcitabine + oxaliplatin (GEMOX) as a first-line treatment option for advanced BTC.
This prospective, single-arm, single-center study enrolled 35 patients with advanced BTC. The patients received toripalimab in combination with GEMOX as first-line treatment at Zhejiang Cancer Hospital, China, between February 2021 and April 2023. Each patient received 1 to 11 treatment cycles, and the objective response rate (ORR) was evaluated before and after chemotherapy using the Response Evaluation Criteria for Solid Tumors (RECIST) version 1.1. Progression-free survival (PFS) and overall survival (OS) were assessed using the Kaplan-Meier method. Furthermore, adverse events and their severity were recorded to evaluate the safety profile of the treatment.
A study cohort of 35 patients was enrolled (median age, 57 years), including 15 men and 20 women. Of them, 18 had intrahepatic cholangiocarcinoma, 14 had gallbladder cancer, and 3 had hilar cholangiocarcinoma. Metastatic lesions were most commonly observed in the lymph nodes, liver, and pelvis. The ORR was 20% (95% confidence interval [CI] 8.4%–36.9%), including 2 for complete response (CR) and 5 for partial response (PR). The median PFS and OS were found to be 8.5 and 16.5 months, respectively. All patients experienced adverse events, with 11 (31.42%) of the patients having grade 3 or higher adverse events. The most commonly observed adverse events included increases in alanine aminotransferase (ALT) and aspartate aminotransferase (AST), leukopenia, neutropenia, and thrombocytopenia.
Toripalimab combined with GEMOX showed preliminary antitumor activity and an acceptable safety profile in patients with advanced BTC, warranting further validation in larger cohorts.
Chinese Clinical Trial Registry (ChiCTR2600117154).
Keywords
- biliary tract cancer
- immunotherapy
- chemotherapy
- toripalimab
- GEMOX
Biliary tract cancer (BTC) originates from the epithelial cells of the bile ducts and is predominantly adenocarcinoma, accounting for over 80% of cases [1]. Depending on the anatomic site, BTC is classified into intrahepatic cholangiocarcinoma (ICC), perihilar cholangiocarcinoma (pCCA), gallbladder cancer (GBC), and distal bile duct carcinoma (dCCA) [2]. Overall, BTC constitutes 3% of all gastrointestinal malignancies [3]. Its incidence shows substantial geographic variation, with comparatively higher rates observed in Southeast Asia [4], and China is reported as a high-incidence region. Data from the China National Cancer Center report approximately 52,800 new cases and 40,700 deaths each year [5].
BTC is an aggressive condition that presents with rapid progression, so most patients are diagnosed at an advanced stage and no longer qualify for surgical resection [6]. As a result, prognostic outcomes are poor, with a 5-year survival rate of only 2%–5% in late-stage disease [2]. For advanced BTC, gemcitabine-based chemotherapy remains the standard first-line treatment, commonly administered as gemcitabine + paclitaxel or gemcitabine + cisplatin. Other regimens, including GCS (gemcitabine + paclitaxel + S1) and GEMOX (gemcitabine + oxaliplatin), have shown prognostic improvement in advanced BTC; however, overall therapeutic efficacy remains suboptimal [7].
In a systematic study on chemoresistance in GBC, Lai et al. [8] reported low sensitivity to cisplatin, gemcitabine, and 5-fluorouracil, and suggested that combining additional agents to enhance the efficacy of cisplatin or gemcitabine-based therapy may help reduce chemoresistance. Besides chemotherapy, immune checkpoint inhibitors (ICIs), especially monoclonal antibodies targeting programmed death receptor 1 (PD-1) and programmed death-ligand 1 (PD-L1), have demonstrated promising results across various tumor types [9, 10], including melanoma [11], non-small cell lung cancer [12, 13], renal cancer [14], and hepatocellular carcinoma [15].
Recent French intergroup guidelines provide an updated, evidence-based framework for the management of BTC, including intrahepatic, perihilar, and distal cholangiocarcinomas, as well as gallbladder carcinomas. Diagnosis and staging primarily rely on contrast-enhanced computed tomography, magnetic resonance imaging, and ultrasound-guided biopsy. For localized disease, surgical intervention followed by adjuvant capecitabine is recommended, whereas the combination of cisplatin and gemcitabine with the anti-PD-L1 inhibitor durvalumab is now established as the standard first-line regimen for advanced disease. The guidelines strongly recommend early comprehensive molecular profiling to identify actionable alterations and guide subsequent treatment decisions [16]. Furthermore, the guidelines emphasize the growing recognition of PD-L1-targeted immunotherapy in BTC and its considerable therapeutic potential, although routine use in China has not yet been established.
Toripalimab is a PD-1 monoclonal antibody developed in China and has received approval for the treatment of several advanced malignancies, including melanoma, nasopharyngeal carcinoma, urothelial carcinoma, non-small cell lung cancer, esophageal cancer, and breast cancer [17, 18, 19, 20]. To date, no clinical evidence supporting its use in BTC has been reported. Given the paucity of evidence on PD-1 inhibition in BTC, investigating toripalimab in combination with standard chemotherapy could provide important insights into its potential role and expanded immunotherapeutic strategies for this difficult-to-treat malignancy. Therefore, this study assessed the preliminary efficacy and safety of toripalimab combined with GEMOX in patients with advanced BTC.
Patients with advanced biliary system tumors receiving first-line therapy at Zhejiang Cancer Hospital, China, between February 2021 and April 2023 were enrolled in this study. This study was approved by the Ethics Committee of Zhejiang Cancer Hospital (Approval No. IRB-2019-91) and was registered under Clinical Trial Registration No. ChiCTR2600117154 (https://www.chictr.org.cn/showproj.html?proj=300563). Before enrollment, all participants were informed in detail about the objectives and procedures, and written informed consent was obtained from each patient or from their legal guardian. All procedures were conducted in accordance with the principles of the Declaration of Helsinki, with due consideration of patients’ rights, safety, and welfare.
Inclusion criteria included (1) histologically confirmed advanced or metastatic
biliary system tumors with no prior treatment; (2) American Joint Committee on
Cancer/Tumor–Node–Metastasis (AJCC/TNM) staging system, stage III–IV; and (3)
Karnofsky Performance Status (KPS) score
Toripalimab (Tuoyi) was obtained from Shanghai Junshi Biosciences Co., Ltd. (Lot. No. c0202302007, Shanghai, China), gemcitabine was purchased from Eli Lilly and Company (Lot. No. 207648A, 207650A, Indianapolis, IN, USA), and oxaliplatin was supplied by Sanofi-Aventis (Lot. No. 210806AM, 210826AN, Paris, France). All patients received gemcitabine 1000 mg/m2 intravenously on day 1 and day 8, oxaliplatin 100 mg/m2 on day 1, and toripalimab 240 mg infused once every 3 weeks. Each treatment cycle was 21 days. Therapy was discontinued in cases of intolerable adverse reactions, withdrawal of informed consent, or death of the patient.
The primary endpoint was objective response rate (ORR). Tumor burden was
assessed using contrast-enhanced computed tomography (CT) or magnetic resonance
imaging (MRI) at baseline and then every 8 weeks until disease progression or
treatment discontinuation. Therapeutic response was evaluated independently by
two experienced radiologists according to the Response Evaluation Criteria for
Solid Tumors (RECIST) version 1.1 [22], In case of discrepancies in the two
assessments, a senior radiologist adjudicated the final results. Response
categories were defined as complete response (CR), disappearance of all target
lesions with pathological lymph nodes reduced to
The secondary endpoints were progression-free survival (PFS) time, overall survival (OS) time, and therapy-related adverse reactions. Adverse events were assessed and graded using the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) version 5.0 [23]. Events were evaluated to determine whether they met the criteria for immune-related adverse events (irAEs). For each patient, the maximum severity grade observed for each adverse event during the study period was recorded.
Data analysis and processing were performed using SPSS version 26.0 software
(IBM Corp., Armonk, NY, USA). As this was a single-arm study, no between-group
comparisons were conducted. Normally distributed continuous variables were
expressed as mean
This study included 35 patients with advanced cholangiocarcinoma, all of whom received the toripalimab plus GEMOX regimen as first-line treatment. Before treatment, 4 patients had a KPS score of 100, 14 patients had a KPS score of 90, and 17 patients had a KPS score of 80. Tumor subtypes included 18 cases of intrahepatic cholangiocarcinoma, 14 cases of GBC, and 3 cases of hilar cholangiocarcinoma. Three patients had a history of hepatitis B virus (HBV) infection, and six had prior surgical treatment history. Metastatic involvement included lung (n = 3), liver (n = 3), peritoneum (n = 4), peritoneal cavity (n = 7), pelvis (n = 5), lymph node (n = 11), and bone (n = 5). The baseline characteristics of patients are shown in Table 1.
| Baseline information | Level/Classification | Value |
| Age | Median (range) | 57 (41–72) |
| Sex | Man | 15 (42.9%) |
| Woman | 20 (57.1%) | |
| Primary lesion | In the liver | 18 (51.4%) |
| Gall | 14 (40.0%) | |
| Fossae transversalis hepatis | 3 (8.6%) | |
| HBV | Yes | 3 (8.6%) |
| Deny | 31 (88.6%) | |
| Not quite clear | 1 (2.9%) | |
| CA242 (U/mL) | Median (range) | 12.25 (6.85–1120.21) |
| CEA (U/mL) | Median (range) | 1.67 (0.57–7.52) |
| CA72-4 (U/mL) | Median (range) | 2.72 (0.62–15.25) |
| CA125 (U/mL) | Median (range) | 39.05 (5.26–106.52) |
| CA19-9 (U/mL) | Median (range) | 362.88 (59.55–15,855.52) |
| CA15-3 (U/mL) | Median (range) | 70.86 (6.85–2500.32) |
| Surgical treatment | Yes | 6 (17.1%) |
| Metastatic involvement | Lung | 3 (8.6%) |
| Liver | 3 (8.6%) | |
| Peritoneal | 4 (11.4%) | |
| Peritoneal cavity | 7 (20.0%) | |
| Pelvic | 5 (14.3%) | |
| Lymph node | 11 (31.4%) | |
| Bone | 5 (14.3%) |
Note: HBV, hepatitis B virus; CA242, carbohydrate antigen 242; CEA, carcinoembryonic antigen; CA72-4, carbohydrate antigen 72-4; CA125, carbohydrate antigen 125; CA19-9, carbohydrate antigen 19-9; CA15-3, carbohydrate antigen 15-3.
After 1 to 11 treatment cycles, tumor response was assessed using RECIST version 1.1. Among the 35 patients, 2 achieved a CR. Both these patients had poorly differentiated intrahepatic cholangiocarcinoma and the following baseline tumor marker levels: carbohydrate antigen 242 (CA242) (20.83 U/mL and 18.52 U/mL), baseline carcinoembryonic antigen (CEA) (1.37 U/mL and 0.85 U/mL), carbohydrate antigen 72-4 (CA72-4) (0.62 U/mL and 0.85 U/mL), carbohydrate antigen 125 (CA125) (60.50 U/mL and 32.20 U/mL), carbohydrate antigen 19-9 (CA19-9) (110.55 U/mL and 81.25 U/mL), and carbohydrate antigen 15-3 (CA15-3) (70.86 U/mL and 19.52 U/mL). Five patients achieved PR, yielding an ORR of 20% (95% confidence interval [CI] 8.4%–36.9%). After treatment, CA242, CA125, CA19-9, and CA15-3 levels decreased significantly, as detailed in Table 2.
| Tumor marker | Pre-treatment level | Post-treatment level |
| CA242 (U/mL) | 12.25 (6.85–1120.21) | 8.52 (6.85–17.75)* |
| CEA (U/mL) | 1.67 (0.57–7.52) | 1.35 (1.06–6.50) |
| CA72-4 (U/mL) | 2.72 (0.62–15.25) | 2.04 (1.05–13.33) |
| CA125 (U/mL) | 39.05 (5.26–106.52) | 12.05 (3.35–56.05)* |
| CA19-9 (U/mL) | 362.88 (59.55–15,855.52) | 88.25 (49.52–185.25)* |
| CA15-3 (U/mL) | 70.86 (6.85–2500.32) | 36.04 (8.25–55.52)* |
Note: Pre-treatment level vs. Post-treatment level, analyzed using paired
Wilcoxon signed-rank test. *p
The median OS time was 16.5 months, and the median PFS time was 8.5 months. The corresponding survival curves are shown in Fig. 1A,B.
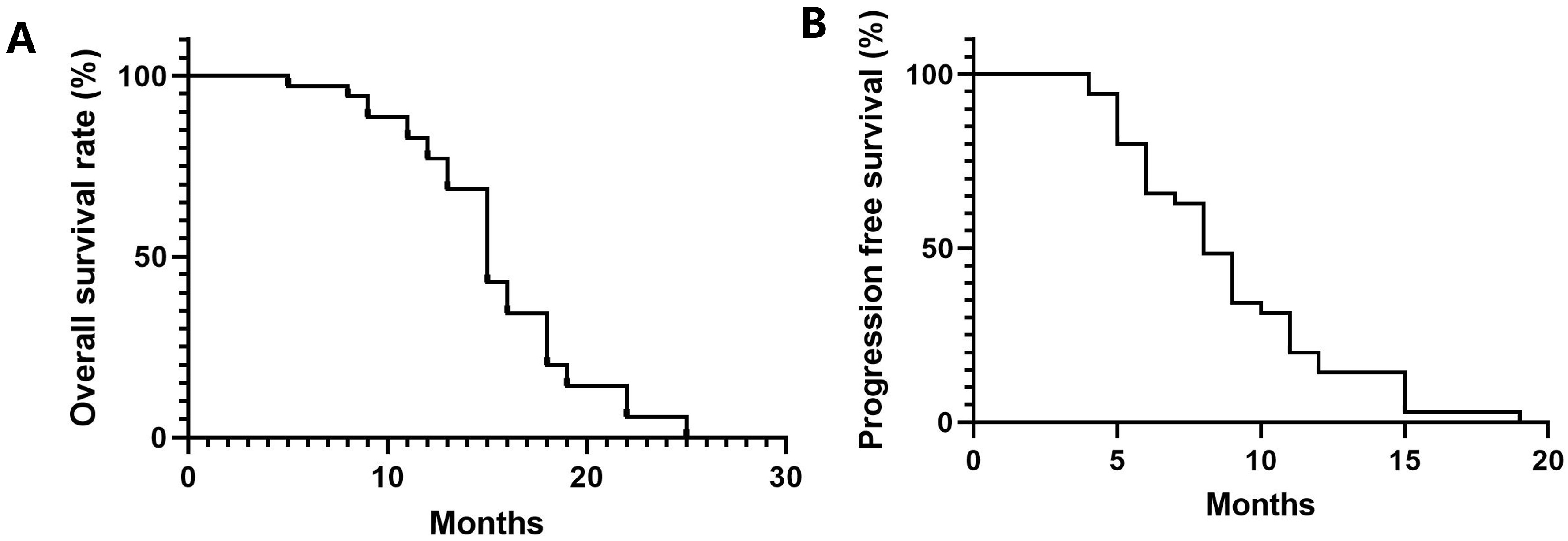 Fig. 1.
Fig. 1.
Overall survival (OS) and progression-free survival (PFS) curves of patients with advanced biliary tract cancers. (A) OS curves, median OS time was 16.5 months. (B) PFS curves, median PFS time was 8.5 months.
All patients experienced adverse events, with 11 (31.42%) of them developing
grade 3 or higher adverse events. These events included elevated alanine
aminotransferase (ALT), increased aspartate aminotransferase (AST), decreased
white blood cell count, granulocytopenia, cholecystitis, bowel obstruction,
decreased platelet count, intra-abdominal infection, and liver injury. These
adverse events were all possibly treatment-related responses. Increases in
ALT/AST and liver injury were considered potential immune-related adverse events
(irAEs) associated with toripalimab, whereas hematologic toxicities were
consistent with GEMOX-related myelosuppression. Other complications, such as
cholecystitis, bowel obstruction, gastrointestinal symptoms, and renal injury,
may be attributable to either immunotherapy or chemotherapy components. No
treatment-related deaths were reported. Most grade
| Adverse events | ||
| Elevated ALT | 11 | 0 |
| Elevated AST | 15 | 0 |
| Decreased white blood cell count | 21 | 5 |
| Granulocytopenia | 20 | 3 |
| Cholecystitis | 11 | 1 |
| Bowel obstruction | 9 | 0 |
| Erythropenia | 9 | 0 |
| Intra-abdominal infection | 3 | 1 |
| Liver injury | 11 | 2 |
| Thrombocytopenia | 15 | 1 |
| Renal injury | 3 | 1 |
| Gastrointestinal symptom | 17 | 0 |
| Sum (number of patients) | 35 | 11 |
Note: ALT, alanine aminotransferase; AST, aspartate aminotransferase. Each adverse event type is listed separately, and one patient may experience multiple adverse events; therefore, the total number of events exceeds the number of patients affected.
Advanced BTC is highly aggressive and invasive, and for those with unresectable advanced diseases, systemic chemotherapy has been the primary treatment option. Recently, the treatment landscape for advanced BTC has entered an era in which immunotherapy is integrated with chemotherapy [24, 25, 26, 27]. During the 2022 American Society of Clinical Oncology-Gastrointestinal Cancer Symposium, outcomes from the Phase III TOPAZ-1 trial were unveiled [28]. In patients with advanced BTC, the combination of durvalumab with gemcitabine + cisplatin (GC) significantly improved outcomes compared with GC alone, increasing median OS to 12.8 months versus 11.5 months, and prolonging median PFS to 7.2 months versus 5.7 months. Subsequently, updated TOPAZ-1 results were presented at the European Society for Medical Oncology Asia Conference 2024 [29]. Following a median follow-up period of 41.3 months, median overall survival was 12.9 months in the durvalumab plus GC treatment group compared with 11.3 months in the placebo plus GC treatment group (hazard ratio [HR]: 0.74). The 3-year overall survival rate was 14.6% with durvalumab plus GC treatment, nearly double that observed with placebo plus GC (6.9%). Among patients with disease control, the 3-year OS rate was 17.0% in the durvalumab arm versus 7.6% in the placebo arm. The safety profile of chemotherapy plus durvalimab remains acceptable, with comparable rates of grade 3/4 treatment-related adverse events (TRAEs) between groups (62.7% vs. 64.9%), and common immune-mediated adverse events (imAEs) included dermatitis (0.9% vs. 0) and liver injury (0.6% vs. 0.3%) [29].
An additional Phase III clinical trial has confirmed the effectiveness of combining immunotherapy with chemotherapy in advanced BTC. Phase KEYNOTE-966 (NCT04003636) demonstrated that pembrolizumab plus GC regimen significantly improved median overall survival compared with placebo plus GC (12.7 months vs. 10.9 months), corresponding to a 17% decrease in the risk of death (HR = 0.83, 95% CI: 0.72–0.95) [30]. This treatment regimen also showed a comparable safety profile, with similar rates of grade 3/4 treatment-related adverse events (TRAEs) (70% vs. 69%). The incidence of imAEs and infusion reactions was 22%, with hypothyroidism (9% vs. 3%) and pneumonia (5% vs. 2%) being the most commonly observed imAEs. The phase III evidence supports chemo-immunotherapy as a first-line standard, and combinations of durvalumab or pembrolizumab with GC regimen are now established first-line treatment options for advanced BTC.
In China, GEMOX is widely used as a first-line chemotherapy regimen for advanced BTC, particularly due to its lower renal toxicity profile compared with cisplatin-based protocols and requires less intensive hydration. Furthermore, oxaliplatin may enhance tumor immunogenicity, which could potentially augment responses to PD-1 inhibition. Toripalimab, a widely used domestic PD-1 antibody approved for multiple solid tumors, represents a crucial immunotherapeutic option in the Chinese setting. The current findings provide preliminary clinical evidence supporting the potential therapeutic synergy between toripalimab and GEMOX in BTC.
In 2022, Professor Li and colleagues [31] reported favorable survival outcomes with the toripalimab plus gemcitabine and S-1 (GS) combination regimen, with a median PFS of 7.0 months and a median OS of 15.0 months. The disease response rate was 30.6%, and the disease control rate was 87.8%. Adverse reactions to toripalimab combined with chemotherapy were generally manageable. Moreover, a recently reported domestic Phase II trialinvestigated gemcitabine plus cisplatin combined with sintilimab in malignant biliary tumors. Among 30 included patients, the study showed an objective response rate of 36.7%, a median PFS of 15.9 months, and a median OS of 5.1 months [32]. Consistent with these findings, a retrospective study assessing the efficacy of camrelizumab plus GEMOX as first-line treatment for advanced biliary cancer across 30 patients with advanced intrahepatic cholangiocarcinoma and a median follow-up time of 24.0 (21.5–26.5) months reported that ORR and DCR were 40% and 73.3%, respectively, while median PFS and OS were 7.5 months and 17.0 months, respectively [33].
Immunotherapy combined with chemotherapy has become the standard first-line treatment of BTC. However, current clinical evidence indicates that the survival outcomes achieved with this strategy remain modest. Therefore, there is a crucial clinical need to further optimize immunotherapy in BTC. Key research domains include the identification of reliable biomarkers to select patients most likely to benefit from immunotherapy and the development of treatment approaches tailored to various anatomical subtypes of BTC.
In this study, we investigated the preliminary efficacy and safety of toripalimab combined with GEMOX in patients with advanced BTC. Despite the small sample size, the regimen demonstrated significant antitumor activity: 2 patients achieved a complete response, and 5 achieved a partial response, yielding an ORR of 20% (7/35; 95% CI: 8.4%–36.9%). Although this response rate appears lower than that reported in some previous studies of PD-1 inhibitor-based chemo-immunotherapy [31, 32, 33], our study cohort consisted predominantly of stage IV patients with distant metastases and significant tumor burden, a population that typically shows less sensitivity to systemic therapy. Therefore, even a modest response may translate into a clinically meaningful benefit in this difficult-to-treat population. These findings remain consistent with the therapeutic trends observed in prior studies evaluating immunotherapy-based combinations in BTC.
Notably, early progression was observed in a proportion of our cohort, indicating that the treatment regimen is not uniformly effective across all advanced BTC cases. Therefore, larger sample sizes and more comprehensive investigations are needed to elucidate the exact efficacy of toripalimab combined with GEMOX and to clarify which patient subgroups are most likely to benefit. In terms of safety assessment, adverse events observed in this study were generally controllable. The rate of grade 3–4 adverse events was 31.42%, comparable to those observed in KEYNOTE-966 and TOPAZ-1, and the overall complication pattern was similar [28, 30].
We acknowledge several limitations to this study. First, it was a single-arm, single-center exploratory study without a comparator arm (control group); therefore, the results should be viewed as hypothesis-generating rather than definitive. Second, the relatively small sample size and the inclusion of patients with multiple anatomical BTC subtypes introduced clinical heterogeneity, which limits the reliability of subgroup findings. Moreover, treatment responses were assessed using RECIST 1.1 without blinded independent central review, which may have introduced assessment bias. Therefore, these findings should be interpreted with caution and warrant confirmation in larger, multicenter randomized controlled trials.
In conclusion, toripalimab combined with GEMOX demonstrated anti-tumor activity and an acceptable safety profile in a subset of patients with advanced BTC. However, the overall therapeutic benefit was limited and heterogeneous, underscoring the need for further large-scale, multicenter studies to validate these findings and to identify the patient population that is most likely to benefit from PD-1-based therapeutic approaches.
• Toripalimab in combination with GEMOX demonstrated significant anti-tumor activity in patients with advanced BTC.
• Toripalimab in combination with GEMOX showed an acceptable safety
profile, with grade
• This prospective study provides preliminary clinical evidence for the feasibility of toripalimab plus GEMOX in advanced BTC.
The data underlying this study are available from the corresponding authors upon reasonable request.
MQY, HJZ and WXL conceived and designed the study. WXF, BXL, YHT and JCC collected and analyzed data and interpreted the results. MQY wrote the original draft, and the other authors conducted a thorough review and editing of the manuscript and made significant revisions. All authors have read and approved the final version, participated sufficiently in the work, and agreed to be accountable for all aspects of the study.
This study was approved by the Ethics Committee of Zhejiang Cancer Hospital (Approval No. IRB-2019-91). Before enrollment, all participants were fully informed in detail about study objectives and procedures, and written informed consent was obtained from each patient or from their legal guardian. All procedures were conducted in accordance with the principles of the Declaration of Helsinki, with due consideration of patients’ rights, safety, and welfare.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

