1 Department of Control Science and Engineering, Institute of Logistics Science and Engineering, Shanghai Maritime University, 201306 Shanghai, China
2 Department of Respiratory and Critical Care Medicine, Shanghai Sixth People’s Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, 200233 Shanghai, China
Abstract
As the global population ages, age-related chronic obstructive pulmonary disease (COPD) and sarcopenia present significant clinical and economic concerns. Emerging evidence suggests that these conditions are interrelated; however, potential confounding factors may impact observational investigations. Therefore, this study employed a bi-directional Mendelian randomization analysis to systematically investigate the causal relationship—rather than merely conducting a correlation analysis, between sarcopenia-related traits and COPD and hospitalization risk.
Univariate, two-sample, and bi-directional Mendelian randomization (MR) analyses were performed using data from genome-wide association studies. The data on Sarcopenia features, including appendicular lean mass (ALM), hand grip strength (HGS), usual walking pace (UWP), and moderate-to-vigorous physical activity (MVPA) were acquired from the UK Biobank. However, data on COPD and hospitalization risk were sourced from the FinnGen consortium. Inverse-variance weighted (IVW) MR and sensitivity analyses were performed to evaluate causal relationships. Additionally, the observed findings were validated using an independent COPD dataset sourced from the UK Biobank.
The forward MR analysis using IVW revealed a significant negative causality between ALM, HGS, UWP, MVPA, and COPD (all p < 0.05, all pFDR < 0.05) (False Discovery Rate, FDR), as well as with COPD-related hospitalization (all p < 0.05, all pFDR < 0.05). These findings were corroborated by validation analyses. Furthermore, reverse MR assessment demonstrated that COPD alleviates UWP (p = 0.001, pFDR = 0.015); however, validation analysis did not confirm this result. Moreover, additional MR analyses yielded similar trends in causal relationships as evidenced by robust sensitivity tests.
Our study supported a unidirectional, negative causality between sarcopenia-related traits and COPD and hospitalization risk. This provides possible evidence that sarcopenia increases the risk of COPD and hospitalization at the genetic level. Our findings suggest that improving sarcopenia may serve as a promising strategy for minimizing the incidence of COPD and hospitalization risk, thereby reducing the health burden on these patients.
Keywords
- Mendelian randomization
- sarcopenia
- COPD
- hospitalization
Chronic obstructive pulmonary disease (COPD) and sarcopenia, present significant clinical and socioeconomic challenges within geriatric populations [1, 2, 3]. COPD is the third leading cause of death worldwide, with the estimated global financial impact of International Dollars (INT$) 4.3 trillion between 2020 and 2050 [3]. At the population level, a study on the economic burden of sarcopenia assessed its direct healthcare costs in the United States at $18.5 billion for the year 2000 [1].
COPD is a complex and heterogeneous condition characterized by persistent inflammation and various extrapulmonary manifestations [4, 5, 6, 7, 8]. Patients with COPD often exhibit comorbidities, including cardiovascular disease, metabolic dysfunctions, osteoporosis, muscular disorders, cachexia, and mental health conditions, all of which detrimentally affect patient outcomes [5, 7, 8]. Sarcopenia, characterized by age-related reductions in skeletal muscle mass, strength, and function, frequently coexists with COPD [9, 10]. The extrapulmonary manifestations of COPD can severely influence performance by reducing physical activity and diminishing muscle mass and strength, thereby significantly compromising the quality of life [11, 12, 13]. These physical factors are strongly associated with the onset and progression of sarcopenia [14, 15]. A recent systematic meta-analysis reported a higher prevalence of sarcopenia (27.5%) among COPD patients [16]. Furthermore, sarcopenia is related to a range of adverse outcomes, including extended hospitalization, increased risk of fractures, acute exacerbations, falls, disability, and mortality—serving as a strong prognostic indicator for worse disease progression [11, 15]. While observational studies consistently suggest a close association between COPD and sarcopenia, the presence of confounding variables limits the capability to ascertain a direct causal relationship. Therefore, more effective approaches are required to determine their causal relationship, informing targeted management and intervention strategies for these interrelated conditions.
Given the current research-based evidence, we hypothesized that sarcopenia has a causal relationship with both COPD and hospitalization risk, and we tested this assumption using a Mendelian randomization (MR) approach. MR analysis includes genetic variants as instrumental variables (IVs) to evaluate causal links between exposures and outcomes, effectively minimizing the risk of confounding factors and reversing causation [17]. Despite the clinical significance causal links between COPD and sarcopenia, limited studies have explored this association using MR approaches. To date, no randomized controlled trials (RCTs) have been performed to evaluate the bidirectional interactions between these two interrelated conditions. Therefore, we conducted a bi-directional two-sample MR analysis to assess causal associations between sarcopenia, COPD, and hospitalization risk, employing appendicular lean mass (ALM), hand grip strength (HGS), usual walking pace (UWP), and moderate-to-vigorous physical activity (MVPA).
As shown in Fig. 1, a two-sample bidirectional MR analysis was conducted to examine the likelihood of sarcopenia-related traits causally impacting COPD and hospitalization risk, and vice versa. Subsequently, replication analyses were performed to validate the results from the initial MR analysis. The MR analysis is based on 3 key assumptions: (1) Independence, where the IVs must not be linked to any known confounders; (2) Relevance, where IVs must be associated with the exposures; (3) Exclusivity, where IVs must influence the outcomes exclusively through the exposures [18]. Reporting procedures adhered strictly to Strengthening the Reporting of Observational Studies in Epidemiology Using Mendelian Randomization (STROBE-MR) guidelines [19]. Since this study reanalyzed previously published data, no additional ethical approval was required.
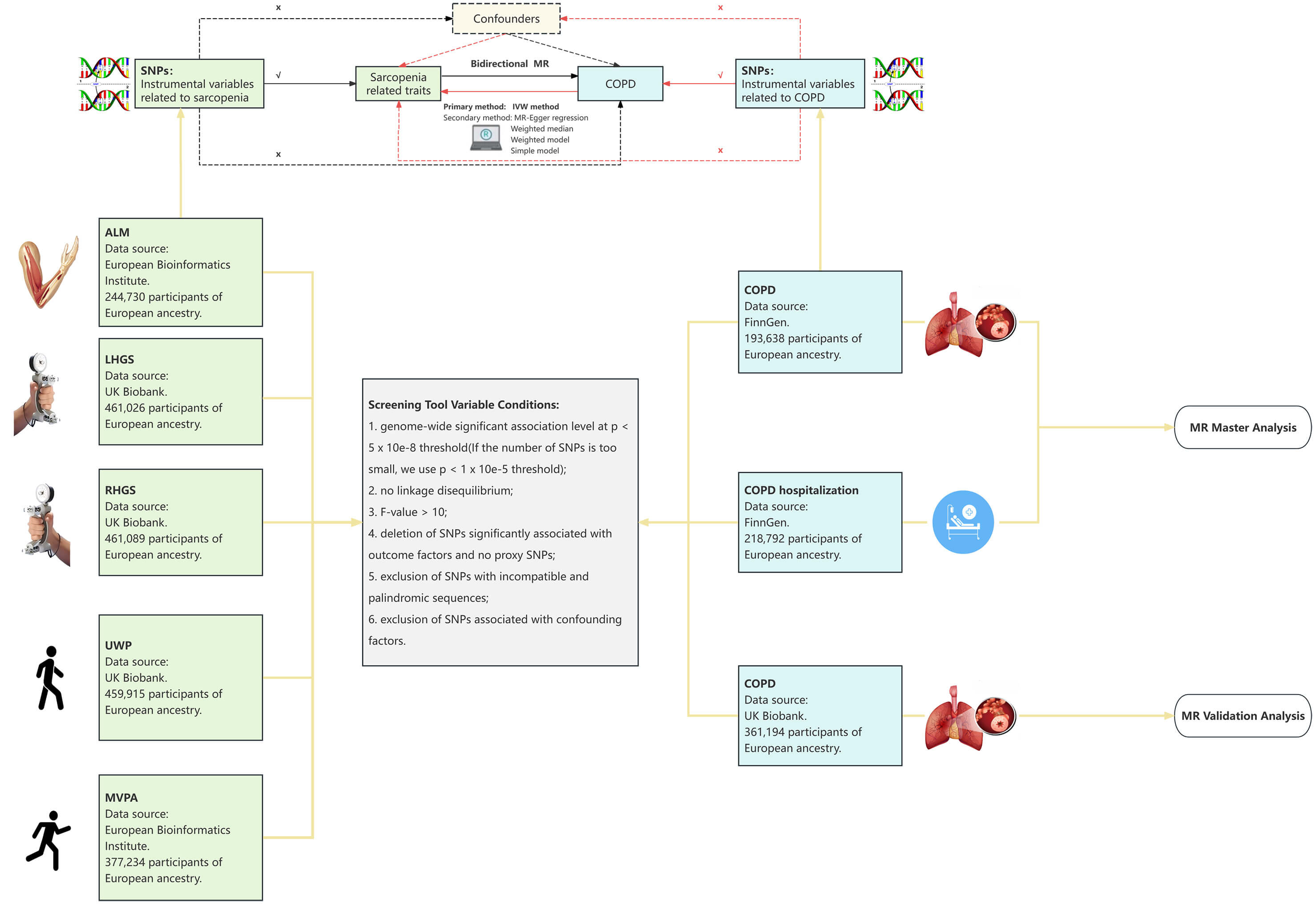 Fig. 1.
Fig. 1.
A flow chart of study design. The lines and arrows indicate that instrumental variables (IVs) are linked to exposure and can influence the outcome only through this exposure. The dashed lines represent that the IVs are not influenced by any confounding variables. COPD, chronic obstructive pulmonary disease; MR, Mendelian randomization; IVW, inverse-variance weighted; ALM, appendicular lean mass; LHGS, left-hand grip strength; RHGS, right-hand grip strength; SNPs, single nucleotide polymorphisms; UWP, usual walking pace; MVPA, moderate-to-vigorous physical activity.
Since published genome-wide association studies (GWAS) data (https://www.ebi.ac.uk/gwas/) targeting sarcopenia are lacking, we explored causal relationships using sarcopenia-related traits. Based on the European Working Group on Sarcopenia in Older People 2 (EWGSOP2) consensus, sarcopenia diagnosis involves three key domains: muscle quantity/quality, physical performance, and muscle strength [20]. Therefore, in this MR analysis, we selected sarcopenia-related traits that reflect these three domains: ALM representing low muscle quantity, HGS indicating low muscle strength, and UWP and MVPA denoting low physical performance. These criteria were used as proxies for sarcopenia in this study. This approach enhances the accuracy and comprehensiveness of MR analyses assessing sarcopenia [21].
For this analysis, IVs were identified using publicly available GWAS summary statistics: ALM (n = 244,730) and MVPA (n = 377,234) were obtained from European Bioinformatics Institute, while right HGS (n = 461,089), left HGS (n = 461,026) and UWP (n = 459,915) were from UK Biobank,
a prospective cohort comprising
Additionally, another GWAS dataset for COPD (n = 361,194) sourced from the UK Biobank was utilized for MR validation analysis. As the exposure and outcome datasets in the primary MR analysis were sourced from different sample repositories (with no sample geographic or temporal overlap between UK Biobank and FinnGen, and FinnGen officially confirming that its samples are independent of other international cohorts), there was no sample overlap, ensuring the independence of our analyses. All GWAS data were based on populations of European descent. The sources of the specific phenotypic data are summarized in Table 1.
| Exposure/Ending | GWAS ID | Year | Sample size | Number of SNPs | PMID |
| ALM | ebi-a-GCST90000027 | 2020 | 244,730 | 18,164,071 | 33097823 |
| HGS (left) | ukb-b-7478 | 2018 | 461,026 | 9,851,867 | NA |
| HGS (right) | ukb-b-10215 | 2018 | 461,089 | 9,851,867 | NA |
| UWP | ukb-b-4711 | 2018 | 459,915 | 9,851,867 | NA |
| MVPA | ebi-a-GCST006097 | 2018 | 377,234 | 11,808,007 | 29899525 |
| COPD | finn-b-J10_COPD | 2021 | 193,638 | 16,380,382 | NA |
| COPD hospitalization | finn-b-COPD_HOSPITAL | 2021 | 218,792 | 16,380,466 | NA |
| COPD | ukb-d-COPD_EARLYANDLATER | 2018 | 361,194 | 10,360,720 | NA |
Note: GWAS, genome-wide association studies; NA, not available; ALM, appendicular lean mass; HGS, hand grip strength; UWP, usual walking pace; MVPA, moderate-to-vigorous physical activity; COPD, chronic obstructive pulmonary disease.
The IVs were selected following the three core assumptions of MR analysis [18]: the IV must be substantially related to exposure, must be independent of confounding factors, and should exclusively affect the outcome through exposure, thereby minimizing horizontal pleiotropy. The criteria used for IV selection were as follows:
(1) Single nucleotide polymorphisms (SNPs) exhibiting genome-wide significant
association (p
(2) A linkage disequilibrium correlation coefficient of r2
(3) The strength of each IV was evaluated using an F statistic
(4) SNPs linked to outcome factors were excluded, and proxy SNPs were absent.
(5) Harmonization of SNPs between outcome and exposure datasets was conducted, removing palindromic SNPs and incompatible alleles.
(6) IVs associated with known confounding factors, including smoking, air pollution, blood lipid levels, alcohol consumption, body mass index, were excluded using PhenoScanner v2. Comprehensive data on selected IVs are given in Supplementary Table 1. All of our analyses in PhenoScanner v2 were completed by March 8, 2024, after which date the site was discontinued, but its data source (GWAS Catalog) is still separately accessible even when PhenoScanner is not available. In this case, we directly used the GWAS Catalog (https://www.ebi.ac.uk/gwas/home) as an alternative.
A random effects inverse-variance weighted (IVW) analysis was used to examine causal links between the exposures and outcomes, as this approach assumes that the selected SNPs represent valid IVs and offer greater precision under these conditions [26]. Supplementary analysis includes the weighted mode, simple mode, weighted median, and MR-Egger approaches [27]. Using multiple MR approaches improves the robustness of the causal estimates across various analytical scenarios.
Three approaches were used for sensitivity analysis to ensure the robustness of
our results: evaluation of heterogeneity, assessment of horizontal pleiotropy,
and “leave-one-out (LOO)” analysis. Heterogeneity was evaluated by applying
Cochran’s Q test; significant heterogeneity (p
Two-sample MR analysis was performed through R (version 4.4.1; R Foundation for
Statistical Computing, Vienna, Austria), using the ‘TwoSampleMR’ [32] and ‘MR-PRESSO’ [30] packages. These packages
are accessible through the Comprehensive R Archive Network (CRAN). For
dichotomous variable outcome, results were presented as odds ratios (ORs) with
95% confidence intervals (CIs) to elucidate the causal links between
sarcopenia-related traits and both COPD and hospitalization risk. A significance
threshold was set at a p-value
The screening process yielded 27–356 IVs that achieved genome-wide significance. All selected SNPs exhibited F statistics exceeding 10, ranging from 19.5 to 772.9, indicating the strength of genetic instruments. Through PhenoScanner, 108 SNPs were identified as potentially related to confounding factors. Specific details on these IVs are provided in Supplementary Table 1.
In the forward MR analysis, sarcopenia-associated traits were used as exposure
variables, while COPD and hospitalization risk were outcome variables. The IVW
results indicated a causal influence of sarcopenia-associated traits on COPD and
hospitalization risk (all p
| Exposures | Outcomes | OR | 95% CI | p-value | p FDR |
| ALM | COPD | 0.904 | 0.824–0.992 | 0.033 | 0.034 |
| LHGS | COPD | 0.542 | 0.375–0.784 | 0.001 | 0.003 |
| RHGS | COPD | 0.613 | 0.440–0.855 | 0.004 | 0.006 |
| UWP | COPD | 0.255 | 0.127–0.515 | 1 |
0.001 |
| MVPA | COPD | 0.650 | 0.459–0.920 | 0.015 | 0.020 |
| ALM | Hospitalization | 0.898 | 0.816–0.988 | 0.027 | 0.028 |
| LHGS | Hospitalization | 0.539 | 0.373–0.779 | 0.001 | 0.003 |
| RHGS | Hospitalization | 0.635 | 0.457–0.884 | 0.007 | 0.012 |
| UWP | Hospitalization | 0.290 | 0.142–0.592 | 0.001 | 0.003 |
| MVPA | Hospitalization | 0.659 | 0.455–0.956 | 0.028 | 0.028 |
Note: IVW, inverse-variance weighted; MR, Mendelian randomization; OR, odds ratio; CI, confidence interval; FDR, False Discovery Rate.
 Fig. 2.
Fig. 2.
Forest plots of the causal relationship between sarcopenia-related traits on COPD and hospitalization risk in the forward MR analysis. (A) The causal relationship between ALM and COPD. (B) The causal relationship between ALM and hospitalization. (C) The causal relationship between LHGS and COPD. (D) The causal relationship between LHGS and hospitalization. (E) The causal relationship between RHGS and COPD. (F) The causal relationship between RHGS and hospitalization. (G) The causal relationship between UWP and COPD. (H) The causal relationship between UWP and hospitalization. (I) The causal relationship between MVPA and COPD. (J) The causal relationship between MVPA and hospitalization.
In the reverse MR analysis, genetically predicted COPD and hospitalization were
examined as exposure variables, with sarcopenia-associated traits as outcomes to
elucidate potential reverse causal effects. The IVW analysis suggested a
significant negative correlation, indicating that COPD was associated with
diminished UWP (ORUWP = 0.993, 95% CI: 0.989–0.997, p = 0.001,
p FDR = 0.015) (Table 3, Fig. 3). However, no causal effects of COPD
or hospitalization on ALM, HGS, and MVPA were observed (p FDR
 Fig. 3.
Fig. 3.
Forest plots of the causal relationship between COPD and hospitalization risk on sarcopenia-related traits in the reverse MR analysis. (A) The causal relationship between COPD and ALM. (B) The causal relationship between hospitalization and ALM. (C) The causal relationship between COPD and LHGS. (D) The causal relationship between hospitalization and LHGS. (E) The causal relationship between COPD and RHGS. (F) The causal relationship between hospitalization and RHGS. (G) The causal relationship between COPD and UWP. (H) The causal relationship between hospitalization and UWP. (I) The causal relationship between COPD and MVPA. (J) The causal relationship between hospitalization and MVPA.
| Exposures | Outcomes | OR | 95% CI | p-value | p FDR |
| COPD | ALM | 0.988 | 0.977–0.999 | 0.039 | 0.097 |
| COPD | LHGS | 0.994 | 0.988–1.000 | 0.048 | 0.097 |
| COPD | RHGS | 0.995 | 0.990–1.000 | 0.048 | 0.097 |
| COPD | UWP | 0.993 | 0.989–0.997 | 0.001 | 0.015 |
| COPD | MVPA | 0.994 | 0.985–1.004 | 0.233 | 0.290 |
| Hospitalization | ALM | 0.992 | 0.979–1.004 | 0.176 | 0.252 |
| Hospitalization | LHGS | 0.997 | 0.990–1.004 | 0.377 | 0.377 |
| Hospitalization | RHGS | 0.997 | 0.991–1.003 | 0.261 | 0.290 |
| Hospitalization | UWP | 0.994 | 0.990–0.999 | 0.011 | 0.053 |
| Hospitalization | MVPA | 0.991 | 0.982–1.001 | 0.065 | 0.109 |
Following several sensitivity analyses, heterogeneity was assessed using
Cochran’s Q-test based on the MR-Egger and IVW frameworks, while MR-PRESSO and
MR-Egger assessments evaluated horizontal pleiotropy. Heterogeneity was observed
in the MR analysis assessing the effects of COPD and hospitalization on ALM and
MVPA, as well as the impact of COPD on left HGS, requiring the utilization of
random-effects IVW for these associations (Table 4). No significant heterogeneity
was detected in the other two-sample MR analyses (p
| Exposures | Outcomes | Horizontal pleiotropy test | Horizontal pleiotropy test | Heterogeneity-test | Heterogeneity-test |
| MR-Egger | MR-PRESSO | IVW | MR-Egger | ||
| Intercept (p) | p | Cochran’s Q (p) | Cochran’s Q (p) | ||
| ALM | COPD | –0.005 (0.086) | 0.086 | 391.1 (0.084) | 387.8 (0.097) |
| ALM | Hospitalization | –0.004 (0.151) | 0.089 | 399.2 (0.052) | 396.9 (0.057) |
| LHGS | COPD | –0.007 (0.413) | 0.199 | 123.55 (0.195) | 122.79 (0.191) |
| LHGS | Hospitalization | –0.008 (0.346) | 0.312 | 116.80 (0.335) | 115.86 (0.332) |
| RHGS | COPD | 0.004 (0.585) | 0.315 | 133.67 (0.303) | 133.35 (0.288) |
| RHGS | Hospitalization | 0.002 (0.775) | 0.729 | 117.02 (0.705) | 116.93 (0.684) |
| UWP | COPD | –0.006 (0.661) | 0.471 | 43.02 (0.427) | 42.81 (0.393) |
| UWP | Hospitalization | –0.011 (0.448) | 0.474 | 41.87 (0.433) | 42.47 (0.451) |
| MVPA | COPD | –0.005 (0.540) | 0.263 | 109.43 (0.223) | 109.0 (0.210) |
| MVPA | Hospitalization | –0.005 (0.558) | 0.088 | 119.11 (0.082) | 118.69 (0.076) |
| COPD | ALM | –0.003 (0.119) | 0.675 | 51.7 (0.008) | 47.5 (0.016) |
| Hospitalization | ALM | –0.003 (0.121) | 0.504 | 50.66 (0.003) | 45.92 (0.007) |
| COPD | LHGS | 3 |
0.058 | 38.9 (0.082) | 38.7 (0.066) |
| Hospitalization | LHGS | 4 |
0.407 | 58.56 (0.001) | 58.32 (4.4 |
| COPD | RHGS | –3 |
0.346 | 29.5 (0.387) | 29.3 (0.342) |
| Hospitalization | RHGS | 1 |
0.085 | 38.77 (0.085) | 38.76 (0.067) |
| COPD | UWP | –6 |
0.438 | 28.4 (0.441) | 28.4 (0.387) |
| Hospitalization | UWP | 2.7 |
0.287 | 31.75 (0.285) | 31.60 (0.247) |
| COPD | MVPA | –0.002 (0.362) | 0.246 | 64.07 (0.001) | 62.46 (0.001) |
| Hospitalization | MVPA | –0.001 (0.474) | 0.509 | 52.64 (0.009) | 51.74 (0.008) |
MR-PRESSO, The Mendelian Randomization Pleiotropy RESidual Sum and Outlier.
Additionally, no evidence of horizontal pleiotropy was found across the
two-sample MR analyses (p
In the MR validation analysis, the causal influence of ALM, HGS, UWP, and MVPA on COPD was successfully replicated, with effect directions consistent with those observed in the primary MR analysis (Supplementary Tables 2,3). No causal impacts of COPD on sarcopenia-associated traits were observed, and the observed association between COPD and UWP was not replicated in the MR validation analysis (Supplementary Tables 2,3). However, sensitivity analyses validated the robustness of these outcomes (Supplementary Tables 2,3 and Supplementary Fig. 2). According to these findings, it was concluded that sarcopenia negatively impacts COPD causally.
This study utilized an univariable two-sample bi-directional MR to systematically elucidate the causal links between sarcopenia-associated traits, including ALM, HGS, UWP, and MVPA and the risk of COPD and hospitalization. The findings revealed a significant negative causal relationship, demonstrating that genetically determined reduction in ALM, HGS, UWP, and MVPA increases the risk of COPD and hospitalization. Furthermore, these associations were successfully validated through replication analyses. Although the primary MR analysis suggested a negative correlation between COPD and diminished UWP, this finding was not replicated in the MR validation analysis. To our knowledge, this is the first bi-directional MR study to comprehensively evaluate the causal link between sarcopenia-associated traits and COPD and hospitalization risk, while accounting for potential confounders.
Previous observational studies have indicated that sarcopenia elevates the risk of COPD. A prospective, population-based cohort investigation undertaken in Rotterdam, The Netherlands, revealed a higher incidence of COPD among sarcopenic (26.9%) and pre-sarcopenic (29.1%) patients relative to the nonsarcopenic (13.4%) individuals [34]. Similarly, another study involving 469,830 participants from the UK Biobank indicated that gait-muscle group individuals had a 4.16-fold higher risk of COPD than those with the normal physical ability (HR: 4.16, 95% CI: 2.59–6.70), followed by those with severe sarcopenia (HR: 3.85, 95% CI: 2.24–6.62). Sarcopenia in this study was described based on the combination of 3 indicators of physical function: muscle mass, grip strength, and gait speed [35]. The Invecchiare in Chianti (InCHIANTI, Aging in the Chianti Region) study, which longitudinally analyzed 538 participants, revealed that sarcopenia was correlated with a higher hospitalization rate (60% vs. 48%, p = 0.087). Even after adjusting for potential confounders, sarcopenia remained notably associated with hospital admission (HR: 1.57; 95% CI: 1.03–2.41) [36]. Furthermore, a retrospective analysis of 174,808 COPD patients in the United States in 2011 reported associations between muscle loss and increased morbidity and mortality, with sarcopenic patients experiencing longer hospital stays and higher associated costs compared to non-sarcopenic patients. These observations are consistent with our results, indicating a causal association between sarcopenia and increased risks of COPD and hospitalization. According to EWGSOP definitions, the diagnosis of sarcopenia requires meeting both low grip strength and low muscle mass, with severe sarcopenia characterized by reduced gait speed. Our findings demonstrate the substantial negative causal association between ALM, MVPA, UWP, and HGS with COPD and hospitalization, supporting the conclusion that sarcopenia increases the risk of COPD and related hospitalizations.
Several systematic reviews and meta-analyses of prior observational studies have suggested an association between COPD and an elevated risk of sarcopenia [11, 16, 37]. While epidemiological surveys across varied populations have shown different prevalence rates of sarcopenia among COPD patients, meta-analyses consistently underscore its frequent co-occurrence. However, in our study, no genetic evidence supports the causal impact of COPD and hospitalization on sarcopenia-related traits. While the primary MR analysis suggested a negative correlation between COPD and diminished UWP, this finding was not replicated in the MR validation analysis. Given our study’s rigor in applying FDR correction for multiple comparisons and considering the complex interrelations between COPD and sarcopenia, these results warrant cautious interpretation. Further investigation is needed to assess these associations more thoroughly in future studies. Sarcopenia-related traits, like ALM and HGS, serve as key predictors of muscle mass, and low muscle mass has been linked to poor pulmonary function [38, 39]. A cross-sectional study analyzing 452 COPD patients reported a significant association between reduced lean mass and elevated risk of emphysema, a crucial phenotype of COPD [40]. Meanwhile, UWP and MVPA are used as a measure of physical activity performance levels. Evidence suggests that prolonged sedentary behavior may lead to an inadequate physical exercise, adversely affecting both muscle and lung functions, thereby increasing the risk of COPD [41, 42, 43]. Conversely, higher physical activity has been associated with a lower risk of COPD, likely through preserving lean mass and reducing oxidative stress and minimizing chronic airway inflammation [44, 45, 46, 47, 48]. Moreover, MVPA has been reported to have positive effects on lung function at the population level [49]. Taken together, these interrelated clinical factors provided a considerable basis for the observed association between sarcopenia and the higher incidence of COPD and hospitalization risk.
The occurrence of comorbidities in sarcopenia and COPD involves a
multidimensional biological interaction, with systemic inflammation playing a
crucial role. Increased levels of serum pro-inflammatory factors, such as
C-reactive protein (CRP), Interleukin-6 (IL-6), and tumor necrosis factor-alpha
(TNF-
The growing understanding of the biological mechanisms linking sarcopenia and COPD carries significant therapeutic implications. Evidence suggested that managing sarcopenia in COPD patients is both feasible and effective through well-structured exercise programs supported by a balanced nutritional intake. Various other events, such as resistance training, aerobic exercise, and pulmonary rehabilitation have been found to improve muscle strength and endurance, while adequate protein intake, vitamin D, omega-3s, and calories help preserve muscle mass [54, 55, 56, 57]. Given the possible negative causal association observed in our study, combining these interventions may maximize therapeutic outcomes and reduce the risk of sarcopenia-related COPD exacerbation and hospitalization. These evidence-based strategies, based on the latest research findings, provide clinicians with a crucial framework for public health decision-making aimed at managing sarcopenia to reduce its impact on COPD patients.
The current study has several significant strengths. Firstly, it comprehensively evaluated critical features related to muscle quality, muscle strength, and physical performance, while also including hospitalization phenotypes alongside COPD, thereby enhancing the rigor and representativeness of the study. Secondly, MR studies, often considered natural randomized controlled trials, offer more robust findings than traditional observational studies. Furthermore, we executed extensive validation and sensitivity analyses to confirm the result’s robustness, especially when heterogeneity was observed: a random-effects IVW was used to minimize its effect. Finally, we effectively excluded confounding factors such as air pollution, smoking, alcohol consumption, body mass index (BMI), and blood lipid levels, mitigating potential biases.
Nonetheless, we acknowledge certain limitations in our study. Specifically, our study population was limited to participants of European ancestry. Due to significant genetic differentiation between East Asian and European populations, with about 12% of SNPs exhibiting significant Fixation Index (FST) differences (indicative of considerable allele frequency differences), which may limit the generalizability of the results across different ethnic groups [58]. Additionally, stratification based on common factors such as age and sex were unattainable due to constraints in the available summarized GWAS data. Moreover, the causal links between COPD-related hospitalization and sarcopenia-associated traits could not be validated due to a lack of relevant data. Lastly, there are inherent limitations in the MR approach. However, the use of MR-PRESSO helped to reduce the risk of horizontal pleiotropy by identifying and excluding outlier SNPs, its validity relies on the assumption of linearity and cannot entirely exclude the possibility of residual pleiotropy arising from nonlinear causation or epigenetic modulation [30].
It should also be noted that, in the partial MR analysis, we employed a
relatively lenient threshold (p
In conclusion, this study indicates a causal association between sarcopenia and elevated risks of COPD and hospitalization, providing reliable genetic evidence for the adverse impacts of sarcopenia on both outcomes within the European population. These findings underscore the clinical and public health importance of targeting sarcopenia improvement as a promising strategy to reduce the incidence of COPD and minimize hospitalization risk.
All data generated or analyzed during this study are available from the corresponding author on reasonable request.
ZK designed the study. SN acquired the data. ZK and SN interpreted the data. SN and XL drafted the manuscript. XL was involved in the analysis and interpretation of data in the clinical part of medicine. XL focused on participating in the writing of the medical clinical section. All authors contributed to the critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/BJHM50164.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



