1 Department of Anesthesiology, The First Affiliated Hospital, Zhejiang University School of Medicine, 311100 Hangzhou, Zhejiang, China
2 Department of Oncology, The First Affiliated Hospital, Zhejiang University School of Medicine, 311100 Hangzhou, Zhejiang, China
Abstract
Studies investigating different classes of vasopressors for septic shock are ongoing, and discrepancies persist among the increasing number of meta-analyses. This umbrella review and evidence map aim to provide a comprehensive overview of the current evidence and to evaluate the highest-quality evidence regarding the efficacy and safety of vasopressors in the treatment of septic shock.
We searched PubMed, Embase, Web of Science, and the Cochrane Database of Systematic Reviews from inception to August 2024. We included meta-analyses of randomized controlled trials that compared vasopressors for the treatment of adult patients with septic shock. The methodological quality of the included meta-analyses was assessed using A MeaSurement Tool to Assess Systematic Reviews 2 (AMSTAR 2). The quality of evidence for each outcome was evaluated using the modified Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach. The best available evidence was identified using the Jadad decision algorithm.
A total of thirty-one eligible meta-analyses were included. The comparison of norepinephrine with vasopressin was the most frequently studied, followed by comparisons of norepinephrine with dopamine. Norepinephrine was found to be superior to dopamine in reducing mortality, heart rate, and the incidence of arrhythmia. Methylene blue demonstrated a reduction in mortality, even though this finding was supported by low GRADE evidence. Meta-analyses comparing norepinephrine with phenylephrine, epinephrine, and angiotensin II showed no significant differences in mortality, also with low GRADE evidence. The addition of vasopressin to norepinephrine was associated with comparable mortality, a lower risk of arrhythmia, and a higher risk of digital ischemia, with moderate GRADE evidence. In contrast, the addition of terlipressin showed no significant differences.
Current evidence fails to demonstrate superior efficacy of alternative vasoactive agents compared to norepinephrine across all evaluated outcome indicators. Considering both the reduced risk of arrhythmias and the increased risk of digital ischemia associated with vasopressin, clinicians should individualize therapy based on patient-specific factors. In addition, our evidence maps identify gaps in the existing literature, highlighting areas for future research.
Keywords
- norepinephrine
- septic shock
- vasoconstrictor agents
- evidence map
- umbrella review
Septic shock, a subset of sepsis characterized by circulatory, cellular, and metabolic dysfunctions, is associated with a mortality rate of up to 50% [1, 2, 3]. Patients with septic shock typically require vasopressor agents to achieve the target mean arterial pressure (MAP) despite adequate fluid resuscitation [4, 5]. Catecholamines, especially norepinephrine, are recommended as the first-line therapy in current clinical practice [6]. However, in the later stages of septic shock, increased doses of catecholamines may not only fail to improve sepsis-related hypotension but also increase the risk of mortality and adverse effects [7]. In recent years, non-catecholamine vasopressors, including vasopressin and analogues (VA), angiotensin II (AT-II), and nitric oxide inhibitors, have emerged as potential alternatives for patients with catecholamine-resistant refractory shock. Nevertheless, their efficacy in reducing mortality and arrhythmias compared to catecholamines remains uncertain, and there is increasing concern regarding their association with digital ischemia [8, 9].
Although numerous meta-analyses on vasopressor use in septic shock have been published, their results are often discordant, and some conclusions are based on low-quality evidence [10, 11, 12, 13]. This heterogeneity in evidence quality presents substantial challenges for translation into clinical practice, as frontline clinicians must make clinical decisions without reliable mechanisms to distinguish robust meta-analyses from those with significant methodological limitations. As a novel tool, evidence map using visual graphs provides clinicians with an intuitive visualization of the available evidence, the quality of that evidence, and critical knowledge gaps that require future research [14]. To date, no published evidence map exists regarding vasoactive agents in sepsis. Therefore, we conducted this umbrella review of meta-analyses of randomized controlled trials (RCTs) and created evidence maps to provide an overview of the current evidence and to identify the best available evidence on the efficacy and safety of vasopressors in the treatment of septic shock.
Our umbrella review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [15, 16] (Supplementary Table 1). A predefined protocol was registered in the International Prospective Register of Systematic Reviews (PROSPERO) (CRD42023420170).
We systematically searched PubMed (https://pubmed.ncbi.nlm.nih.gov), Embase (https://www.embase.com), Web of Science (https://www.webofscience.com), and the Cochrane Database of Systematic Reviews (https://www.cochranelibrary.com) from database inception to August 2024. To ensure a sensitive search, we combined exploded MeSH terms with relevant text words, using the keywords “vasopressor”, “septic shock”, and “meta-analysis”. The detailed search strategy is provided in Supplementary Table 2. In addition, we screened the reference lists of all included reviews and pertinent clinical guidelines for additional literature.
Studies were included based on the following criteria: meta-analyses of RCTs that compared one vasopressor with another vasopressor or placebo in the treatment of adult patients with septic shock. We summarized outcomes including mortality, length of Intensive Care Unit (ICU) stay, length of hospital stay, adverse events, kidney function, and hemodynamic and metabolic parameters. Studies were excluded if they: (1) were conducted in participants with vasodilatory shock without subgroup analysis for septic shock, (2) aimed to evaluate medication discontinuation or withdrawal timing, or (3) were published as abstracts or protocols only. Studies published in English and Chinese were considered for inclusion. Two reviewers independently screened studies by title and abstract, followed by full-text review. Discrepancies were resolved through discussion.
One reviewer initially extracted data using a standardized Excel form, after which a second reviewer independently verified the extracted data. Any disagreements between reviewers were resolved through discussion. From each meta-analyses, we extracted information including the number of component RCTs, interventions, comparators, outcomes, risk of bias, and methodological details for each outcome (such as the type of effect model, effect size, and degree of heterogeneity as measured by I2). For studies on vasodilatory shock, we extracted subgroup data for septic shock when available.
The methodological quality of the included meta-analyses was assessed using the validated A MeaSurement Tool to Assess Systematic Reviews 2 (AMSTAR 2) [17]. The quality of evidence for each outcome was evaluated using a modified Grading of Recommendations Assessment, Development, and Evaluation (GRADE) tool, classifying evidence as high, moderate, low, or very low (Supplementary Table 3) [18, 19]. The best available evidence for each outcome was identified using the Jadad decision algorithm [20]. Quality assessment and evidence grading were performed independently by two reviewers, with disagreements resolved by discussion. No further meta-analyses were conducted. Details of the included meta-analyses were summarized in tables. Evidence maps were created to provide an overview of the findings from the meta-analyses, as well as the best evidence for each outcome, including the corresponding GRADE ratings. When direct comparisons were available, indirect data from network meta-analyses were not summarized.
We identified 1116 citations in our initial search and 837 citations remained after removing duplicates. After browsing titles, abstracts, and full-text reading, 31 meta-analyses [9, 10, 11, 12, 13, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46] were ultimately included in our review (Fig. 1) (Ref. [16]). No additional studies were found through citation checking. Details of the excluded papers following full-text review are provided in Supplementary Table 4.
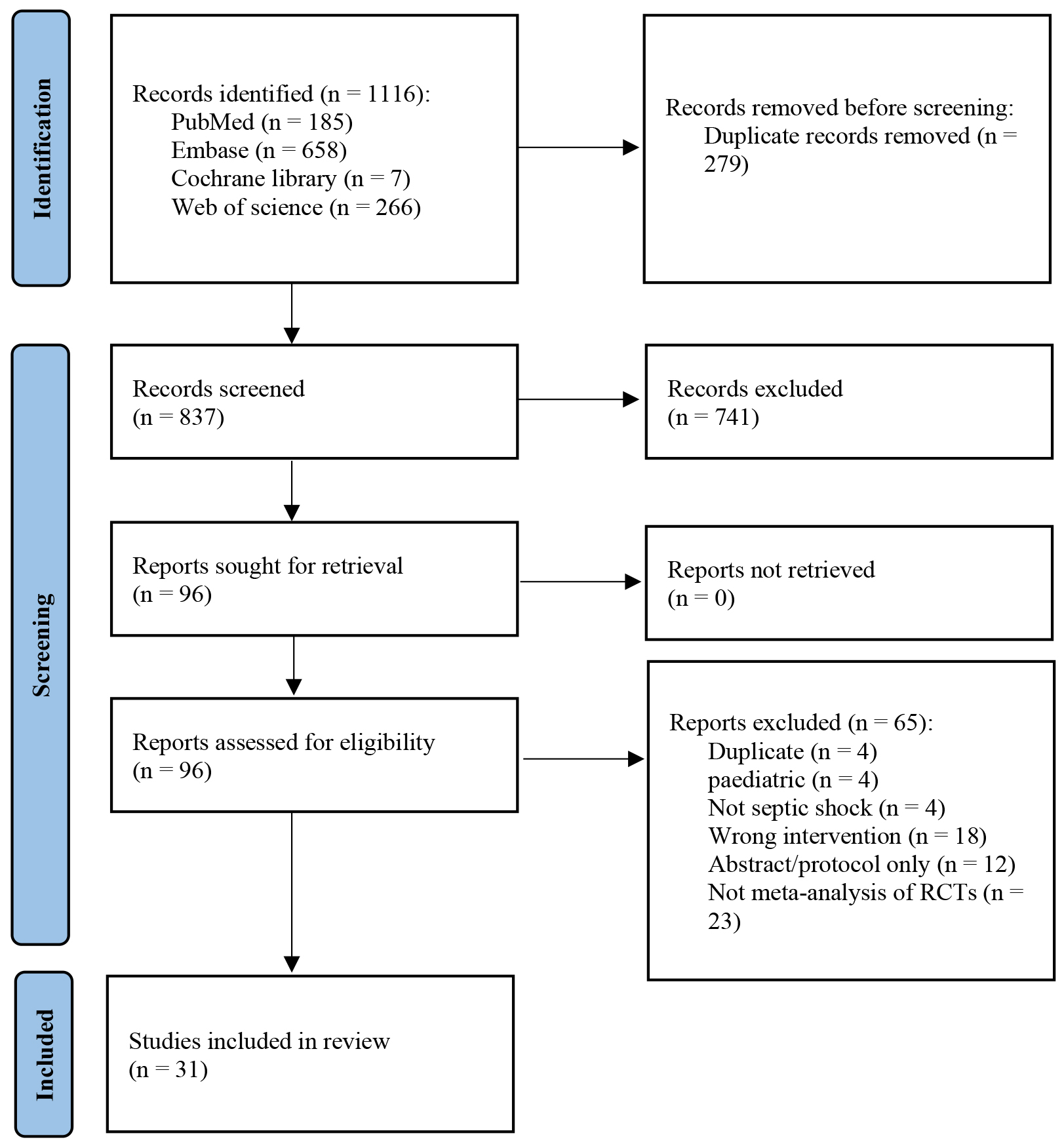 Fig. 1.
Fig. 1.
Study flow diagram. Adapted from Page et al. [16] [BMJ Publishing Group Ltd], available under the Creative Commons License (https://creativecommons.org/licenses/by/4.0/). RCT, randomized controlled trial.
The characteristics of the 31 meta-analyses are presented in Table 1 (Ref. [9, 10, 11, 12, 13, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46]). Of these, 24 were pairwise meta-analyses, six were network meta-analyses, and one was an Individual Participant Data (IPD) meta-analysis. Given the available direct comparisons, we did not summarize indirect data from the network meta-analyses. The publication years of these meta-analyses ranged from 2012 to 2024. The vasopressors evaluated included norepinephrine, dopamine, phenylephrine, epinephrine, vasopressin, terlipressin, methylene blue (MtB), and AT-II. Among these, norepinephrine versus VA was the most frequently studied comparison, followed by norepinephrine versus dopamine. The primary outcomes assessed in the meta-analyses included mortality, ICU or hospital length of stay, renal function, hemodynamic and metabolic parameter changes, and adverse events. All direct comparisons and outcomes are summarized in the evidence maps (Supplementary Table 5).
| Study | Journal title | No. of RCTs included | Intervention | Comparator | Outcome indicators | Last search date | Synthesis |
| Belletti [22] | Journal of Critical Care | 33 | Vasopressor | The other vasopressor | Mortality | Jun 2015 | Network meta-analysis |
| Belletti [21] | British Journal of Anaesthesia | 7 | Vasopressor | Placebo | Mortality | Jan 2014 | Pairwise meta-analysis |
| Belletti [9] | PLoS ONE | 10 | Catecholamine | Non-catecholamine | Mortality | Dec 2014 | Pairwise meta-analysis |
| Cheng [10] | Critical Care | 43 | Vasopressor | The other vasopressor | Mortality; ICU-Los; hospital-Los; AE | Feb 2018 | Network meta-analysis |
| Chidambaram [23] | Journal of Critical Care | 4 | Norepinephrine | Vasopressin | Mortality; ICU-Los; AE | Apr 2018 | Pairwise meta-analysis |
| De Backer [24] | Critical Care Medicine | 6 | Dopamine | Norepinephrine | Mortality; ICU-Los; hospital-Los; ARR | Jun 2011 | Pairwise meta-analysis |
| Huang [26] | BMC Anesthesiology | 9 | Catecholamine | Terlipressin | Mortality; ICU-Los; DO2; VO2; Lac; HR; CI; MAP; AE; ARR; PI; Scr; UO | July 2018 | Pairwise meta-analysis |
| Huang [25] | Frontiers in Pharmacology | 6 | Norepinephrine | Terlipressin | Mortality; AE; ARR; DI; MAP; HR; UO; Scr; OI | Mar 2019 | Pairwise meta-analysis |
| Jia [27] | Shock | 47 | Vasopressor | The other vasopressor | Mortality; ICU-Los; AE; MI; PI; ARR | May 2022 | Network meta-analysis |
| Jiang [28] | Critical Care | 20 | Catecholamine | Vasopressin and/or analogues | Mortality; ICU-Los; AE; ARR; DI | Jul 2018 | Pairwise meta-analysis |
| Li [29] | Zhonghua Wei Zhong Bing Ji Jiu Yi Xue | 8 | Catecholamine | Terlipressin | Mortality; AE; DI | Dec 2019 | Pairwise meta-analysis |
| Lu [30] | Pteridines | 13 | Norepinephrine | Dopamine | Mortality; DO2; VO2; Lac; HR; CI; MAP; SVRI | / | Pairwise meta-analysis |
| McIntyre [11] | JAMA | 22 | Catecholamine | Vasopressin and analogues | Mortality; AF; MI | Feb 2018 | Pairwise meta-analysis |
| Nagendran [31] | Journal of the Intensive Care Society | 13 | Vasopressor | The other vasopressor | Mortality; ARR | Sep 2014 | Network meta-analysis |
| Nagendran [12] | Intensive Care Medicine | 4 | Norepinephrine | Vasopressin | Mortality; ICU-Los; hospital-Los AE, ARR; DI; MI; RRT requirement; RRT duration | Jan 2019 | IPD meta-analysis |
| Nedel [13] | Critical Care Medicine | 14 | Catecholamine | Vasopressin and analogues | RRT requirement; AKI | Jun 2017 | Pairwise meta-analysis |
| Neto [32] | Critical Care | 7 | Norepinephrine | Vasopressin or terlipressin | Mortality; DO2; VO2; Lac; CI; AE; NE reduction | 2011 | Pairwise meta-analysis |
| Oba [33] | Journal of Critical Care | 9 | Vasopressor | The other vasopressor | Mortality | Sep 2013 | Network meta-analysis |
| Polito [34] | Intensive Care Medicine | 7 | Vasopressin and analogues | Catecholamine | Mortality | Mar 2011 | Pairwise meta-analysis |
| Pruna [35] | Journal of Cardiothoracic and Vascular Anesthesia | 5 | Methylene blue | Placebo | Mortality | Mar 2023 | Pairwise meta-analysis |
| Ruslan [36] | The Western Journal of Emergency Medicine | 11 | Norepinephrine | Other vasopressors | Mortality; ARR; MI; the number of participants who achieved the target MAP, time to achieve the target MAP | Aug 2019 | Pairwise meta-analysis |
| Tan [37] | International Journal of Clinical and Experimental Medicine | 11 | Norepinephrine | Vasopressin and/or terlipressin | Mortality; ICU-Los; DO2; VO2; Lac; HR; CI; SVRI; MAP; UO | Oct 2015 | Pairwise meta-analysis |
| Vasu [38] | Journal of Intensive Care Medicine | 6 | Norepinephrine | Dopamine | Mortality; ARR | May 2010 | Pairwise meta-analysis |
| Yao [39] | Frontiers in Pharmacology | 23 | Vasopressin or/and analogues | Catecholamine | Mortality; ICU-Los; hospital-Los; AE; ARR; MI; DI | Oct 2019 | Pairwise meta-analysis |
| Yin [40] | International Journal of Clinical and Experimental Medicine | 8 | Norepinephrine | Dopamine or vasopressin | Mortality; DO2; VO2; CI; HR; CI; MAP; SVRI | Jan 2017 | Pairwise meta-analysis |
| Zhao [41] | Chinese Journal of Evidence-Based Medicine | 9 | Norepinephrine | Dopamine | Mortality; HR; MAP; CI; ARR | Jun 2011 | Pairwise meta-analysis |
| Zhong [42] | Journal of Intensive Care | 23 | Non-catecholamine | Norepinephrine | Mortality; ICU-Los; hospital-Los; DO2; VO2; Lac; HR; CI; SVRI; MAP; AE; ARR; DI; MI; AKI; Scr; RRT requirement; RRT duration | Apr 2020 | Pairwise meta-analysis |
| Vasopressin subgroup | |||||||
| Angiotension II subgroup | |||||||
| Zhou [45] | Therapeutics and Clinical Risk Management | 21 | Vasopressor | The other vasopressor | Mortality; DO2; VO2; Lac; HR; CI; SVRI; MAP; cardiac AE | Dec 2014 | Network meta-analysis |
| Zhou [43] | Zhonghua Wei Zhong Bing Ji Jiu Yi Xue | 11 | Norepinephrine | Dopamine | Mortality; DO2; VO2; Lac; HR; CI; SVRI; MAP | Dec 2012 | Pairwise meta-analysis |
| Zhou [44] | Military Medical Research | 7 | Norepinephrine | Vasopressin | Mortality; DO2; VO2; CI; SVRI | Dec 2013 | Pairwise meta-analysis |
| Zhu [46] | Journal of Intensive Care | 9 | Telipressin | Catecholamine norepinephrine subgroup | Mortality; ARR; hospital-Los | Jul 2018 | Pairwise meta-analysis |
AE, adverse events; AF, atrial fibrillation; AKI, Acute Kidney Injury; ARR, arrhythmia; CI, cardiac index; DI, digital ischemia; DO2, oxygen delivery; HR, heart rate; ICU, Intensive Care Unit; IPD, Individual Participant Data; Lac, Lactic acid; Los, Length of stay; MAP, mean arterial pressure; MI, myocardial injury; NE, norepinephrine; OI, Oxygenation Index; PI, Peripheral Ischemia; RCT, randomized controlled trial; RRT, renal replacement therapy; Scr, serum creatinine; SVRI, systemic vascular resistance index; UO, urinary output; VO2, oxygen consumption.
AMSTAR 2 quality assessment (Supplementary Table 6) showed that only one meta-analysis (3.2%) was rated as high methodological quality, two (6.5%) as moderate quality, five (16.1%) as low quality, and 23 (74.2%) as critically low quality. The most frequently omitted items were: failure to explain the study design criteria for inclusion (Item 3), failure to report the funding sources of included trials (Item 10), and failure to provide a list of excluded studies (Item 7).
The best available evidence of each outcome, based on the Jadad decision algorithm, is summarized in Table 2a and Table 2b and stratified by intervention type.
| Intervention vs. Comparator | Mortality | DO2 | VO2 | Lac | HR | CI | SVRI | MAP | ICU-Los | Hospital-Los |
| NE vs. DA | 11 studies, n = 1718 •• | 4 studies, n = 105 • | 4 studies, n = 105 •• | 4 studies, n = 105 •• | 5 studies, n = 195 • | 5 studies, n = 195 •• | 5 studies, n = 195 • | 4 studies, n = 94 • | 2 studies, n = 1296 •• | 2 studies, n = 1296 •• |
| NE vs. PE | 2 studies, n = 86 •• | |||||||||
| NE vs. EP | 2 studies, n = 218 •• | |||||||||
| NE vs. other vasopressors | 7 studies, n = 4139 ••• | |||||||||
| Catecholamines vs. non-catecholamines | 23 studies, n = 4225 ••• | 12 studies, n = 3203 ••• | 6 studies, n = 2188 ••• | |||||||
| Catecholamines vs. VP | 10 studies, n = 2256 ••• | 4 studies, n = 1466 •• | ||||||||
| Catecholamines vs. TP | 11 studies, n = 1093 ••• | 2 studies, n = 50 • | 2 studies, n = 69 • | 2 studies, n = 62 • | 4 studies, n = 137 •• | 3 studies, n = 107 •• | 5 studies, n = 176 •• | 6 studies, n = 783 •• | 3 studies, n = 173 ••• | |
| NE vs. non-catecholamines | 18 studies, n = 4217 •• | 5 studies, n = 151 •• | 5 studies, n = 170 •• | 4 studies, n = 324 •• | 10 studies, n = 889 •• | 7 studies, n = 416 •• | 5 studies, n = 151 •• | 9 studies, n |
12 studies, n = 3253 •• | 8 studies, n = 2519 •• |
| NE vs. VP | 4 studies, n = 1451 ••• | 2 studies, n = 53 • | 2 studies, n = 53 • | 2 studies, n = 53 • | 3 studies, n = 831 •• | 3 studies, n = 294 •• | 2 studies, n = 53 • | 3 studies, n = 831 •• | 3 studies, n = 672 •• | 2 studies, n = 655 ••• |
| NE vs. TP | 8 studies, n = 838 ••• | 3 studies, n = 89 • | 3 studies, n = 89 • | 3 studies, n = 89 • | 2 studies, n = 87 •• | 3 studies, n = 89 • | 2 studies, n = 87 •• | 4 studies, n = 210 ••• | ||
| NE vs. AT-II | 2 studies, n = 341 ••• | |||||||||
| Placebo vs. MtB | 5 studies, n = 262 •• | |||||||||
| Placebo vs. vasopressors | 5 studies, n = 132 •• |
GRADE rating: ••• moderate; •• low; • very low.
AT-II, angiotensin II; CI, cardiac index; DA, dopamine; DO2, oxygen delivery; EP, epinephrine; GRADE, Grading of Recommendations Assessment, Development, and Evaluation; HR, heart rate; ICU, Intensive Care Unit; Lac, Lactic acid; Los, Length of stay; MAP, mean arterial pressure; MtB, methylene blue; NE, norepinephrine; PE, phenylephrine; SVRI, systemic vascular resistance index; TP, terlipressin; VO2, oxygen consumption; VP, vasopressin.
| Intervention vs. Comparator | AE | ARR | DI | MI | AKI incidence | Scr | RRT requirement | UO | RRT duration |
| NE vs. DA | 3 studies, n = 1997 •• | ||||||||
| NE vs. other vasopressors | 6 studies, n = 3974 ••• | 3 studies, n = 2983 ••• | |||||||
| Catecholamines vs. non-catecholamines | 14 studies, n = 3206 ••• | 12 studies, n = 1143 ••• | 9 studies, n = 2929 ••• | 10 studies, n = 1609 ••• | 7 studies, n = 1366 ••• | 4 studies, n = 1263 ••• | |||
| Catecholamines vs. VP | 7 studies, n = 1382 •• | 4 studies, n = 1319 •• | 4 studies, n = 1267 •• | ||||||
| Catecholamines vs. TP | 4 studies, n = 672 ••• | 3 studies, n = 640 •• | 2 studies, n = 610 •• | 4 studies, n = 156 •• | |||||
| NE vs. non-catecholamines | 14 studies, n |
3 studies, n = 846 •• | 11 studies, n |
6 studies, n |
4 studies, n |
6 studies, n |
8 studies, n |
3 studies, n | |
| NE vs. VP | 4 studies, n = 1453 ••• | 4 studies, n = 1453 ••• | 4 studies, n = 1453 ••• | 3 studies, n = 1438 •• | 4 studies, n = 1452 •• * | 2 studies, n = 53 •• | 2 studies, n = 151 •• | ||
| NE vs. TP | 2 studies, n = 546 •• | 2 studies, n = 546 •• | 2 studies, n = 546 •• | 2 studies, n = 87 •• | 4 studies, n = 146 •• |
GRADE rating: ••• moderate; •• low.
* non-significant in random-effects specification, downgrade one level.
AE, adverse events; AKI, Acute Kidney Injury; ARR, arrhythmia; DI, digital ischemia; GRADE, Grading of Recommendations Assessment, Development, and Evaluation; MI, myocardial injury; RRT, renal replacement therapy; Scr, serum creatinine; UO, urinary output.
All included meta-analyses, except Nedel et al. [13], reported mortality as an outcome (Supplementary Table 5a). Ten meta-analyses compared norepinephrine with dopamine. According to the Jadad decision algorithm, the evidence indicates that dopamine is associated with higher mortality (low GRADE evidence; [43]) (Table 2a). Meta-analyses comparing norepinephrine with phenylephrine (low GRADE evidence) or epinephrine (low GRADE evidence) showed similar effects [27, 45]. For comparisons between catecholamines and non-catecholamines, five meta-analyses reported inconsistent findings [9, 11, 28, 34, 39]. However, the most recent meta-analysis with the largest number of RCTs found no significant difference (moderate GRADE evidence; [39]). Meta-analyses focusing exclusively on norepinephrine versus vasopressin [12], terlipressin [46], or AT-II [42] also showed no significant differences in mortality (moderate GRADE evidence; Table 2a). Interestingly, MtB seemed to improve survival in the most recent meta-analysis [35].
Dopamine, vasopressin, and terlipressin did not reduce the length of stay compared to norepinephrine [12, 24, 27]. The GRADE quality of evidence for these findings ranged from low to moderate (Table 2a). For phenylephrine, epinephrine, AT-II, and MtB, no meta-analyses reported their effects on length of stay.
The available evidence indicates that norepinephrine, compared to dopamine, reduces heart rate (very low GRADE evidence) and cardiac index (low GRADE evidence), while increasing systemic vascular resistance index (very low GRADE evidence) ([43]; Table 2a; Supplementary Table 5b). Vasopressin, when compared with norepinephrine, did not show significant differences in hemodynamic or metabolic variables, except for a reduction in heart rate. The GRADE quality of evidence for these findings ranged from very low to low quality (Table 2a). Terlipressin was associated with reductions in heart rate, oxygen delivery (DO2), and cardiac index compared to norepinephrine, with GRADE evidence ranging from very low to low quality (Table 2a).
Four meta-analyses comparing norepinephrine with dopamine consistently demonstrated a benefit of norepinephrine in reducing arrhythmias (low GRADE quality; Supplementary Table 5c). Meta-analyses comparing catecholamines with non-catecholamines indicated that non-catecholamines did not confer advantages in reducing total adverse events, arrhythmia events, or myocardial injury. Moreover, they were associated with a higher incidence of severe digital ischemia compared to catecholamines (moderate GRADE evidence; Table 2b). In direct comparisons of norepinephrine with vasopressin or terlipressin [12, 23, 25, 32, 45], vasopressin was associated with fewer arrhythmia events but a higher risk of digital ischemia (moderate GRADE quality). However, these differences were not observed for terlipressin compared to norepinephrine (low GRADE evidence; Table 2b).
Six meta-analyses reporting renal outcomes were included [12, 13, 25, 26, 37, 42] (Supplementary Table 5c). None of these studies compared norepinephrine with dopamine. Only one meta-analysis compared catecholamines with non-catecholamines and found no significant differences in the incidence of Acute Kidney Injury (AKI) or the need for renal replacement therapy (RRT) (moderate GRADE evidence) [13]. Four studies focused exclusively on comparing norepinephrine with vasopressin or terlipressin [12, 25, 26, 37]. Only Nagendran et al. [12] reported that vasopressin reduced the need for RRT (low GRADE evidence). However, for outcomes such as serum creatinine level, urinary output, and RRT duration, neither vasopressin nor terlipressin showed additional benefits over norepinephrine (low GRADE evidence; [12, 25, 37]) (Table 2b).
This umbrella review and evidence map evaluated and summarized the current evidence regarding vasopressor administration in patients with septic shock. The most studied vasopressor agents were norepinephrine, dopamine, and VA. Dopamine was associated with a higher risk of mortality and a greater incidence of arrhythmias. VA demonstrated similar mortality, metabolic parameters, renal function, and total adverse effects when compared with norepinephrine. Notably, only vasopressin was associated with fewer arrhythmias but a higher incidence of digital ischemia than norepinephrine. The evidence concerning MtB and AT-II was limited and primarily focused on mortality outcome indicators. Emerging data suggest that MtB may confer a survival benefit.
Although the administration of mixed non-catecholamines showed lower mortality than norepinephrine, subgroup analyses comparing norepinephrine with vasopressin, terlipressin, or AT-II did not show significant differences. This may be attributable to the confounding effects of different drugs with varying mechanisms of action and side effects, or more likely, to the few studies included in these subgroup analyses. In addition, studies on non-catecholamines frequently involved concomitant norepinephrine administration in clinical practice, which differs from studies evaluating dopamine. MtB has been proposed as a vasoactive agent for refractory septic shock due to its ability to reverse nitric oxide overproduction [47]. A recent meta-analysis reported that MtB treatment may be superior in the treatment of septic shock; however, the evidence is limited by the small number of trials and patients included [35]. Therefore, further RCTs are required to confirm these considerations.
In addition to their vasoconstrictive effects, catecholamines have adverse effects including arrhythmias and myocardial ischemia. Several studies have demonstrated a significantly increased risk of death associated with arrhythmias [48, 49, 50]. The use of non-catecholamines as adjuvant agents to catecholamines may reduce the adverse effects associated with catecholamines [51]. Only one IPD meta-analysis suggested that vasopressin could prevent norepinephrine-associated arrhythmias [12]. One meta-analysis showed that adding vasopressin to catecholamines decreased the risk of atrial fibrillation compared to catecholamines in post-cardiac surgery patients, but this effect did not reach statistical significance in septic patients, although no significant interaction effect was found between the subgroups [11]. This discrepancy may be due to the higher incidence of atrial fibrillation in post-cardiac surgery patients (73%) compared to those with septic shock (13%), indicating that larger sample sizes are needed to detect statistically significant differences in the latter group. The adverse effects of terlipressin compromise its clinical application. The Surviving Sepsis Campaign guidelines recommend against the use of terlipressin due to a higher incidence of digital ischemia compared to norepinephrine. This recommendation is based on a single RCT that reported digital ischemia in 12.6% of patients receiving terlipressin versus 0.35% in the norepinephrine group [52]. Another RCT conducted in cirrhotic patients with septic shock reported peripheral cyanosis in 28.6% of patients treated with terlipressin compared to 9.5% in those receiving norepinephrine [53]. A meta-analysis pooling these two RCTs found no significant difference, but substantial heterogeneity was present [25].
Clinicians routinely aim to maintain adequate blood perfusion by achieving the target MAP; however, a higher MAP does not always avoid cellular oxygen delivery and utilization disorders in patients with septic shock. Indicators such as hypoxia-induced lactic acid and the oxygen consumption (VO2): lactate ratio are associated with prognosis in septic shock [54, 55]. Nevertheless, current meta-analyses have not found significant differences in VO2 or lactate levels between different vasoactive agents.
Norepinephrine reduces renal perfusion by inducing vasoconstriction of the renal afferent arterioles, whereas vasopressin increases glomerular filtration by promoting vasoconstriction of the renal efferent arterioles [56]. As such, vasopressin is considered promising agent for renal protection. One meta-analysis that focused on renal outcomes reported that vasopressin reduced the requirement of RRT and the incidence of AKI in patients with distributive shock, but not in those with septic shock [13]. Another meta-analysis found only weak evidence supporting vasopressin in reducing RRT requirement (relative risk 0.86, 95% confidence interval 0.74–0.99) and using a random-effects model, this reduction was not statistically significant [12]. These inconclusive results may reflect the complexity of sepsis-induced renal injury, which extends beyond hemodynamic factors [56, 57].
Although non-catecholamine vasopressors appear to be safe, they have not demonstrated significant improvements in the prognosis of septic shock patients. One hypothesis suggests that the timing of vasopressin initiation may influence outcomes in septic shock. For instance, one study found that adding vasopressin to catecholamine vasopressors at higher norepinephrine-equivalent dose or lactate concentrations was associated with higher mortality [58]. Several retrospective cohort studies that compared early versus late vasopressin administration suggest that early administration is associated with a shorter time to shock reversal [59, 60, 61]. However, these findings are limited by the retrospective and observational study design; multicenter RCTs are required for further validation. Our umbrella review and evidence map have several strengths, including the use of a comprehensive search strategy and the provision of detailed information regarding vasopressors use in septic shock. In addition, we used the Jadad decision algorithm to identify the best available evidence for each outcome. Nevertheless, our results should be considered in light of several limitations. First, only three meta-analyses achieved a moderate to high AMSTAR score, and most of the evidence, based on meta-analyses, was rated as low to moderate GRADE quality. Second, the study patients were included according to previous criteria for sepsis instead of the updated sepsis-3 definition [2]. Third, most included meta-analyses focused on mortality outcome, and some outcomes lacked pooled analysis. Fourth, there was substantial clinical heterogeneity in study design (including varying shock severity, type and doses of vasopressors, resuscitation strategies, clinical endpoints, and therapeutic escalation strategies) as well as in endpoints. Subgroup analyses and meta-regression were limited by the lack of eligible studies.
There is low-quality evidence suggesting that patients with septic shock benefit more from noradrenaline treatment than from dopamine. Although the addition of non-catecholamine vasopressors has been shown to reduce heart rate, there is insufficient high-quality evidence to demonstrate improvements in mortality, metabolic variables, renal injury, or overall adverse events. Therefore, clinicians should individualize the choice of vasopressors in the treatment of septic shock and remain vigilant for the occurrence of arrhythmias and digital ischemia when making treatment decisions. Furthermore, there is a need for prospective, large-scale, high-quality RCTs focusing on specific subgroups of septic shock patients and the optimal timing of non-catecholamine administration to provide new insights.
AKI, Acute Kidney Injury; AMSTAR 2, A MeaSurement Tool to Assess Systematic Reviews 2; AT-II, angiotensin II; DO2, oxygen delivery; GRADE, Grading of Recommendations Assessment, Development, and Evaluation; ICU, Intensive Care Unit; IPD, Individual Participant Data; MAP, mean arterial pressure; MtB, methylene blue; PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses; RCTs, randomized controlled trials; RRT, renal replacement therapy.
The data used to support the findings of this study are available from the corresponding author upon request.
FS and SWS conceived and designed this study. FS and CX searched the literature, collected data, evaluated evidence quality and drafted the manuscript. All authors contributed to revising the manuscript critically for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/BJHM50162.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

