1 Department of Human Movement Science, Incheon National University, 22012 Incheon, Republic of Korea
2 Division of Sport Science, Sport Science Institute & Health Promotion Center, Incheon National University, 22012 Incheon, Republic of Korea
†These authors contributed equally.
Abstract
Aging can cause degenerative changes in motor and cognition-related brain areas, presumably by interfering with gait performance in healthy aging populations. We aimed to assess the effects of transcranial direct current stimulation (tDCS) on single- and dual-task walking performances in healthy older adults using meta-analytic approaches.
Eleven studies were qualified based on the inclusion criteria: (a) healthy older adults, (b) treatment = tDCS protocols, (c) control = sham stimulation, (d) gait performance outcomes, and (e) randomized controlled trials using parallel or crossover designs. Effect sizes were estimated using standardized mean difference (SMD) to examine gait performances between active tDCS and sham stimulation. A separate random-effect meta-analysis was performed to determine the effects of tDCS protocols on gait performance during single- and dual-task walking tasks.
During single-task walking, the random-effects meta-analysis showed improvements in stride time variability (SMD = 0.203; p = 0.005) and functional mobility (SMD = 0.595; p < 0.001). Moreover, single-task walking performances were improved when the tDCS protocols targeted the primary motor cortex (SMD = 0.424; p = 0.005) and used off-line stimulation (SMD = 0.168; p = 0.008). During dual-task walking, tDCS improved gait speed (SMD = 0.177; p = 0.025) and dual-task cost for gait speed (SMD = 0.548; p < 0.001). Dual-task walking performances were advanced when the tDCS protocols targeted the dorsolateral prefrontal cortex (SMD = 0.231; p = 0.029) and multiple areas including prefrontal cortex (SMD = 0.382; p = 0.001), and applied off-line stimulation (SMD = 0.249; p < 0.001).
These findings indicate that the tDCS protocols may be a promising tool to support mobility and reduce gait-related challenges in the healthy aging population.
Keywords
- aging
- gait
- transcranial direct current stimulation
- meta-analysis
- lower limb
Aging can cause degenerative changes in the central and peripheral nervous systems presumably leading to impaired motor and cognitive functions [1, 2, 3]. Specifically, structural and functional changes in the motor and premotor cortical regions often occur in older adults and interfere with motor actions and executive function [4, 5]. Moreover, muscle mass and strength tend to decrease with age, and this age-related muscle atrophy leads to impaired activities of daily living [6, 7]. These age-related changes also affect gait performance, as indicated by the decreased gait speed and increased gait variability [8, 9, 10, 11]. Given that single-task walking performance (i.e., locomotion without executing any other task) is normally related to a greater risk of falls [12, 13], identifying an effective way to improve gait performances is necessary for advancing the independent life of a healthy aging population.
Beyond single-task walking, successful dual-task walking performances (i.e., locomotion while simultaneously executing another task such as talking, reading, or planning) on real-world surfaces are frequently required for healthy older adults [14, 15]. According to the capacity-sharing theory [16], simultaneously performing two tasks may impair performance in one or both tasks because of limited cognitive resources. Thus, executive function is crucial for successfully performing dual tasks because this cognitive process may contribute to the effective allocation of neural resources between both tasks by suppressing irrelevant responses and decreasing task-switch costs [17]. However, healthy older adults often showed lower gait speed and greater stride time variability (STV) during dual-task walking than during single-task walking [18, 19, 20]. Furthermore, older adults who exhibited lower prefrontal cortical excitability showed more impaired executive functions with lower dual-task performances [21]. Modulating cortical excitability across key cognition-related brain areas may enhance dual-task walking performances in healthy older adults.
Transcranial direct current stimulation (tDCS) may be effective way for advancing dual-task walking performance in older adults [22, 23, 24]. The potential mechanisms underlying tDCS-induced neuromodulation posited that anodal stimulation may facilitate cortical excitability by depolarizing the membrane, while cathodal stimulation may suppress cortical excitability by hyperpolarizing the membrane [25, 26, 27]. Further, the excitatory and inhibitory effects caused by tDCS protocols may be sustained for a few hours [28]. A recent meta-analysis study confirmed a possibility of positive effects on dual-task performances after tDCS in older adults [29]. However, these meta-analytic findings did not focus on healthy older adults (e.g., one out of three studies including mild cognitive impairments) and failed to report effects on single-task performance. Potentially, these findings could be affected by confounding bias because of clinical characteristic of participants and task difficulty [30]. Thus, this systematic review and meta-analysis aimed to examine the effects of tDCS protocols on walking performance in healthy older adults. We focused on healthy older population who had no motor and cognitive deficits, and further estimated tDCS effects on both single- and dual-task walking performances.
Based on Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines, a systematic review and meta-analysis was performed [31]. The PRISMA checklist is shown in Supplementary Material. We established five inclusion criteria using the Population, Intervention, Comparison, Outcomes and Study (PICOS) framework for establishing reliable selection criteria: (a) Population: healthy older adults aged 60 years or over; (b) Intervention: tDCS protocols; (c) Comparison: controls with sham stimulation; (d) Outcome: gait performance; (e) Study design: randomized control trials (RCT) using a parallel or crossover design [32]. Consistent with previous meta-analyses [33, 34, 35], we excluded studies involving participants diagnosed with neurological diseases or significant medical, psychiatric, or cognitive deficits. Further, studies that did not explicitly state the exclusion of participants with such conditions were also excluded. We removed review articles, case studies, animal studies, and studies that failed to provide sufficient data for computing effect sizes. Using three databases including the PubMed (https://pubmed.ncbi.nlm.nih.gov/), Web of Science (https://www.webofscience.com/wos/woscc/basic-search), and Cochrane Library (https://www.cochranelibrary.com/), two researchers (BJC and HL) independently performed a literature search. All articles published by August 1, 2024 were carefully screened. The following keywords were used: (older adults OR elderly OR aged) AND (non-invasive brain stimulation OR NIBS OR transcranial electrical stimulation OR TES OR transcranial direct current stimulation OR tDCS) AND (gait OR walking OR timed up and go OR Timed up and Go test (TUG) OR locomotion OR mobility).
For conducting meta-analysis procedures, we applied the Comprehensive Meta-Analysis software version 4.0 (Biostat, Englewood, NJ, USA). All effect sizes were estimated by calculating the standardized mean difference (SMD). For RCT using a parallel design, individual effect sizes were quantified by comparing mean and standard deviation values of gait performance between tDCS and sham stimulation groups. For RCT using a crossover design, individual effect sizes were calculated by conducting paired analysis that used sample size and mean difference values with standard error [36, 37]. The analysis may decrease possibility of disguising clinically important heterogeneity induced by incorporating crossover design as if parallel design [38].
The greater values of SMD indicated more improvements in gait performance after receiving active tDCS protocols than those for sham tDCS protocols. We used the random-effects model for minimizing inherent heterogeneity caused by different experimental conditions such as participants and study protocols [39]. A separate random-effect meta-analysis was performed to determine effects of tDCS protocols on gait performance during single- and dual-task walking tasks.
For single- and dual-task performances, we performed three moderator variable analyses to examine effects of tDCS protocols based on the following conditions: (a) gait variable, (b) targeted brain areas, and (c) stimulation timing. Additional meta-regression analyses were conducted to identify relationship between tDCS effects and demographic characteristics (i.e., mean age and female ratio) and tDCS parameter (i.e., stimulation intensity, duration, and session), respectively.
Using Higgins and Green’s I-squared, we estimated levels of heterogeneity across
individual effect sizes [40]. Specific ranges of heterogeneity include: (a) low
I-squared (
Two researchers (BJC and HL) conducted independent methodological quality assessment for each qualified study based on the Cochrane risk of bias assessment tool version 2 [43]. The tool comprised six domains: (a) randomization process, (b) timing of identification or recruitment of participants, (c) deviations from intended intervention, (d) missing outcome data, (e) measurement of the outcome, and (f) selection of the reported results [44]. Consistent with the protocols in the Cochrane risk bias assessment tool [45], the risk of bias for each domain was determined: (a) low risk of bias, (b) some concern, and (c) high risk of bias.
Initially, we found 826 studies involving 221 from the PubMed, 169 from the Web of Science, and 436 from the Cochrane Library. After removing 229 duplicated studies, we additionally excluded 164 studies (i.e., 40 review articles, 13 case studies, 46 protocol studies, one animal study, and 64 studies that irrelevant to our topic). After screening, 422 studies that did not fulfill the inclusion criteria were removed: (a) 298 studies that did not recruit healthy older adults (i.e., 63 studies that did not focus on older adults, 94 studies involving patients with neurodegenerative disorders, 89 studies involving patients with cerebrovascular disorders, 12 studies involving patients with musculoskeletal disorders, 40 studies involving patients with motor and cognitive disorders), (b) 48 studies that did not apply tDCS protocol, (c) 65 studies that did not assess gait performances, and (d) 11 studies that reported insufficient data. Finally, 11 studies were included in this study [22, 23, 24, 46, 47, 48, 49, 50, 51, 52, 53]. The PRISMA flow diagram describes specific study identification procedure (Fig. 1).
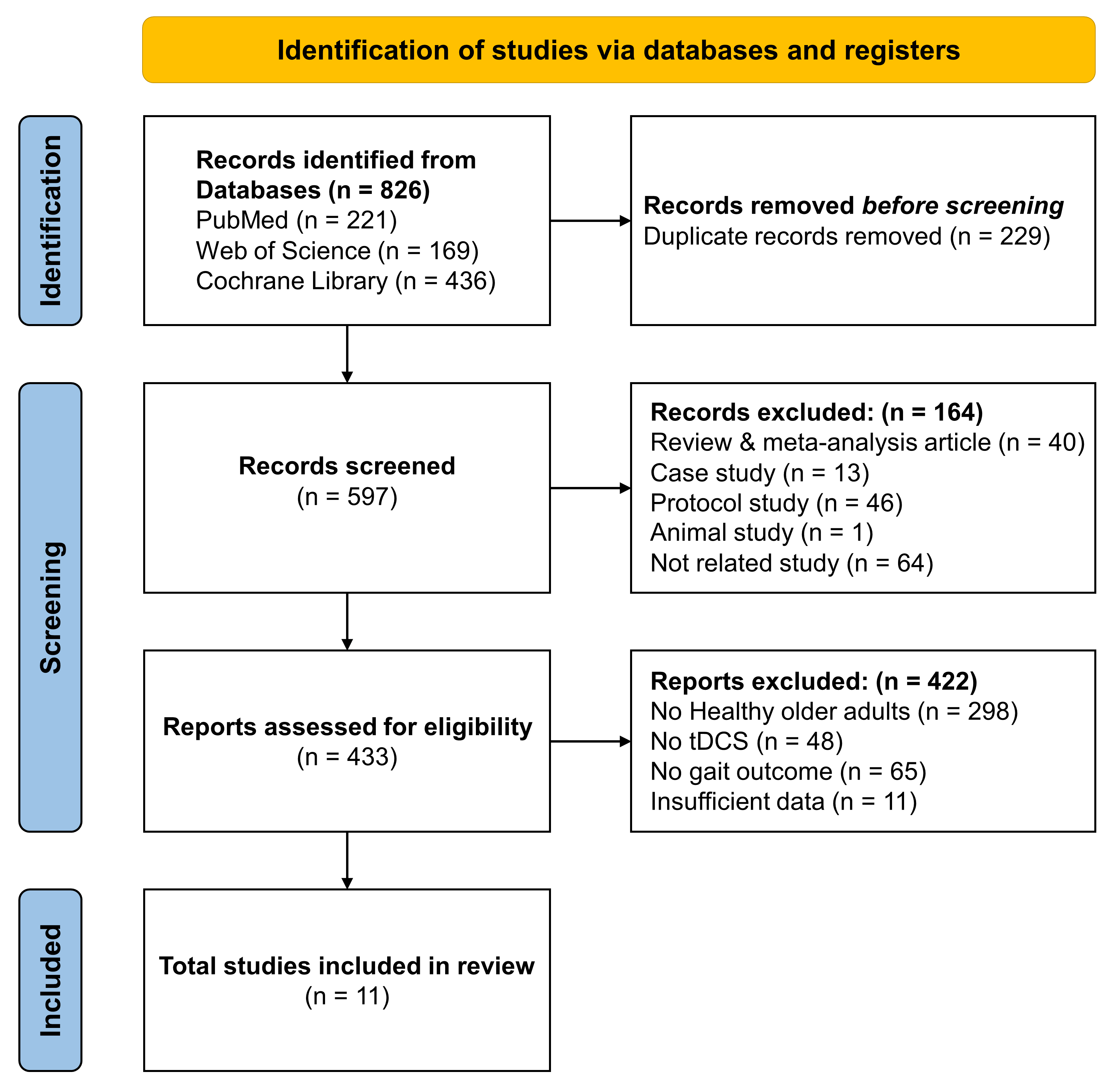 Fig. 1.
Fig. 1.
PRISMA flowchart for the study identification procedure. tDCS, transcranial direct current stimulation; PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-analyses.
The 11 included studies focused on 338 healthy older adults who have no motor and cognitive deficits (range of mean age = 61.0–78.8 years, body mass index (BMI) = 24.8–31.6 kg/m2, and female ratio = 45–80%). Seven studies reported levels of motor or cognitive functions at baseline: (a) activities-specific balance confidence scale from two studies (a range of mean = 77.1–83.9) [54], (b) Berg balance scale from two studies (a range of mean = 46.6–50.1) [55], and (c) Montreal cognitive assessment from four studies (a range of mean = 25.8–28.3) [56]. Table 1 (Ref. [22, 23, 24, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57]) show more specific details for demographic information about participants.
| Study | Study design | Total | Age | Gender | Height | Weight | BMI | Motor and cognitive function | |||
| (N) | (yrs) | (F/M) | (m) | (kg) | (kg/m2) | ||||||
| Chatterjee et al. [46] 2023 | Parallel | Real: 18 | 76.1 |
11 F, 7 M | NA | NA | NA | ABC | 81.5 |
BBS | 48.6 |
| Sham: 15 | 73.7 |
11 F, 4 M | 83.9 |
50.1 | |||||||
| Clark et al. [47] 2021 | Parallel | Real: 7 | 75.4 |
5 F, 2 M | NA | NA | 31.5 |
ABC | 81.5 |
BBS | 46.6 |
| Sham: 5 | 70.6 |
4 F, 1 M | 31.6 |
77.1 |
48.8 | ||||||
| Manor et al. [22] 2016 | Crossover | Total: 37 | 61.0 |
25 F, 12 M | 1.7 |
71 |
NA | NA | |||
| Orcioli-Silva et al. [48] 2021 | Parallel | Real: 10 | 66.0 |
5 F, 5 M | 1.71 |
71.0 |
NA | MoCA | 28.2 |
FES-I | 17.6 |
| Sham: 8 | 69.9 |
7 F, 1 M | 1.67 |
73.6 |
28.3 |
18.1 | |||||
| Rodrigues et al. [49] 2023 | Parallel | Real: 14 | 71.3 |
8 F, 6 M | NA | NA | NA | MMSE | 27.6 | ||
| Sham: 13 | 70.9 |
10 F, 3 M | 26.1 | ||||||||
| Rostami et al. [50] 2020 | Parallel | Real: 16 | 68.8 |
8 F, 8 M | NA | NA | NA | NA | |||
| Sham: 16 | 67.3 |
8 F, 8 M | |||||||||
| Sayig-Keren et al. [23] 2023 | Crossover | Total: 20 | 72.6 |
9 F, 11 M | NA | NA | 26.3 |
MoCA | 25.8 |
CCI | 3.4 |
| Schneider et al. [51] 2021 | Crossover | Total: 25 | 73.9 |
20 F, 5 M | NA | NA | 26.6 |
MoCA | 26.6 | ||
| Yi et al. [52] 2021 | Parallel | Real: 31 | 78.1 |
21 F, 10 M | 1.56 |
62.2 |
25.5 |
NA | |||
| Sham: 26 | 78.8 |
17 F, 9 M | 1.57 |
61.5 |
24.8 |
||||||
| Zhou et al. [53] 2018 | Crossover | Total: 20 | 61.0 |
NA | 1.58 |
61.0 |
NA | NA | |||
| Zhou et al. [24] 2021 | Crossover | Total: 57 | 75.0 |
43 F, 14 M | 1.6 |
74.3 |
NA | MoCA | 26.5 | ||
Data are mean
Four studies administered tDCS protocols during gait assessments (i.e., on-line timing) and seven studies applied tDCS before gait assessment (i.e., off-line timing). Eight out of 11 studies administered a single tDCS session (i.e., only one session during the experiment) and three studies provided multiple sessions of tDCS protocol (i.e., 5–18 sessions). For targeted brain areas of stimulation, anodal stimulation was applied to: (a) dorsolateral prefrontal cortex (DLPFC) from five studies, (b) primary motor cortex (M1) from three studies, and (c) multiple regions including DLPFC–M1 and prefrontal cortex (PFC)–M1 from two studies. Moreover, one study applied three different protocols stimulating DLPFC, M1, and DLPFC–M1, respectively [24]. Specific information for tDCS protocols is shown in Table 2 (Ref. [22, 23, 24, 46, 47, 48, 49, 50, 51, 52, 53, 58]).
| Study | Group | Targeted Brain Area | Intensity | Duration | Timing | Surface | Session | Gait Variables | ||
| Anodal | Cathodal | (Period) | Single Task | Dual Task | ||||||
| Chatterjee et al. [46] 2023 | Trt | R-DLPFC | L-DLPFC | 2 mA | 20 min | On | 35 cm2 | 1 | Gait speed | NA |
| Clark et al. [47] 2021 | Trt | R-DLPFC | L-DLPFC | 2 mA | 20 min | On | 35 cm2 | 18 | Gait speed, Figure-8 walk time | NA |
| (6 weeks) | ||||||||||
| Manor et al. [22] 2016 | Trt | L-DLPFC | R-supraorbital ridge | 2 mA | 20 min | Off | 35 cm2 | 1 | Gait speed | Gait speed, Gait speed cost |
| Orcioli-Silva et al. [48] 2021 | Trt | L-PFC, M1 | R-mastoid | 0.6 mA | 20 min | On | 9 cm2 | 1 | NA | STV cost |
| Rodrigues et al. [49] 2023 | Trt | L-DLPFC | R-supraorbital ridge | 2 mA | 20 min | Off | 25 cm2 | 16 | TUG | NA |
| (8 weeks) | ||||||||||
| Rostami et al. [50] 2020 | Trt | L-M1 | R-supraorbital ridge | 1 mA | 20 min | Off | 55.25 cm2 | 5 | TUG, Figure-8 walk time | NA |
| (1 week) | ||||||||||
| Sayig-Keren et al. [23] 2023 | Trt | L-DLPFC | R-APC, L-LPC, L-MPC | 2 mA | 20 min | Off | 16 cm2 | 1 | Gait speed, STV | Gait speed, Gait speed cost, STV |
| Schneider et al. [51] 2021 | Trt | L-DLPFC, M1 | R-APC, L-LPC, L-MPC, L-S1, | 1.5 mA | 20 min | On | 3.14 cm2 | 1 | NA | Gait speed cost |
| Yi et al. [52] 2021 | Trt | M1 | L-M1, R-M1 | 1.98 mA | 20 min | Off | 24 cm2 | 1 | Gait speed, TUG | NA |
| Zhou et al. [53] 2018 | Trt | M1 | R-supraorbital ridge | 2 mA | 20 min | Off | 35 cm2 | 1 | TUG | NA |
| Zhou et al. [24] 2021 | Trt 1 | L-DLPFC | R-APC, L-LPC, L-MPC | 1.5 mA | 20 min | Off | 3.14 cm2 | 1 | Gait speed, STV | Gait speed, Gait speed cost, STV, STV cost |
| Trt 2 | M1 | R-APC, L-MPC, L-S1 | ||||||||
| Trt 3 | L-DLPFC, M1 | R-APC, L-LPC, L-MPC, L-S1 | ||||||||
APC, anterior prefrontal cortex; DLPFC, dorsolateral prefrontal cortex; L, left; LPC, lateral premotor cortex; M1, primary motor cortex; MPC, medial premotor cortex; R, right; S1, primary somatosensory cortex; STV, stride time variability; Trt, treatment; PFC, prefrontal cortex; TUG, Timed up and Go test [58].
Nine studies assessed single-task walking performances: (a) gait speed from two studies, (b) gait-related functional mobility (i.e., timed up and go (TUG) [58] and Figure-eight walk time) from three studies, (c) both gait and gait-related functional mobility from two studies, and (d) both gait speed and STV from two studies. Furthermore, five studies evaluated gait performances during dual-task walking: (a) gait speed cost (i.e., differences in gait speed from single-task walking to dual-task walking; lower values of cost indicating better dual-task walking performance) from one study, (b) both gait speed and gait speed cost from one study, (c) STV cost (i.e., differences in STV from single-task walking to dual-task walking; lower values of cost indicating better dual-task walking performance) from one study, (d) one study evaluated gait speed, gait speed cost, STV, and (e) one study assessed all gait speed, gait speed, STV, and STV cost.
The Cochrane risk-of-bias revealed a low risk of methodological biases for each domain (Fig. 2): (a) randomization process (all studies with low risk), (b) timing of identification or recruitment of participants (all studies with low risk), (c) deviations from intended intervention (all studies with low risk), (d) measurement of the outcome (10 studies with low risk and one study with some concern), and (e) selection of the reported result (all studies with low risk). For the missing outcome data, we found a relatively moderate methodological biases (three studies with some concerns) because missingness in the outcome because of the dropout of participants.
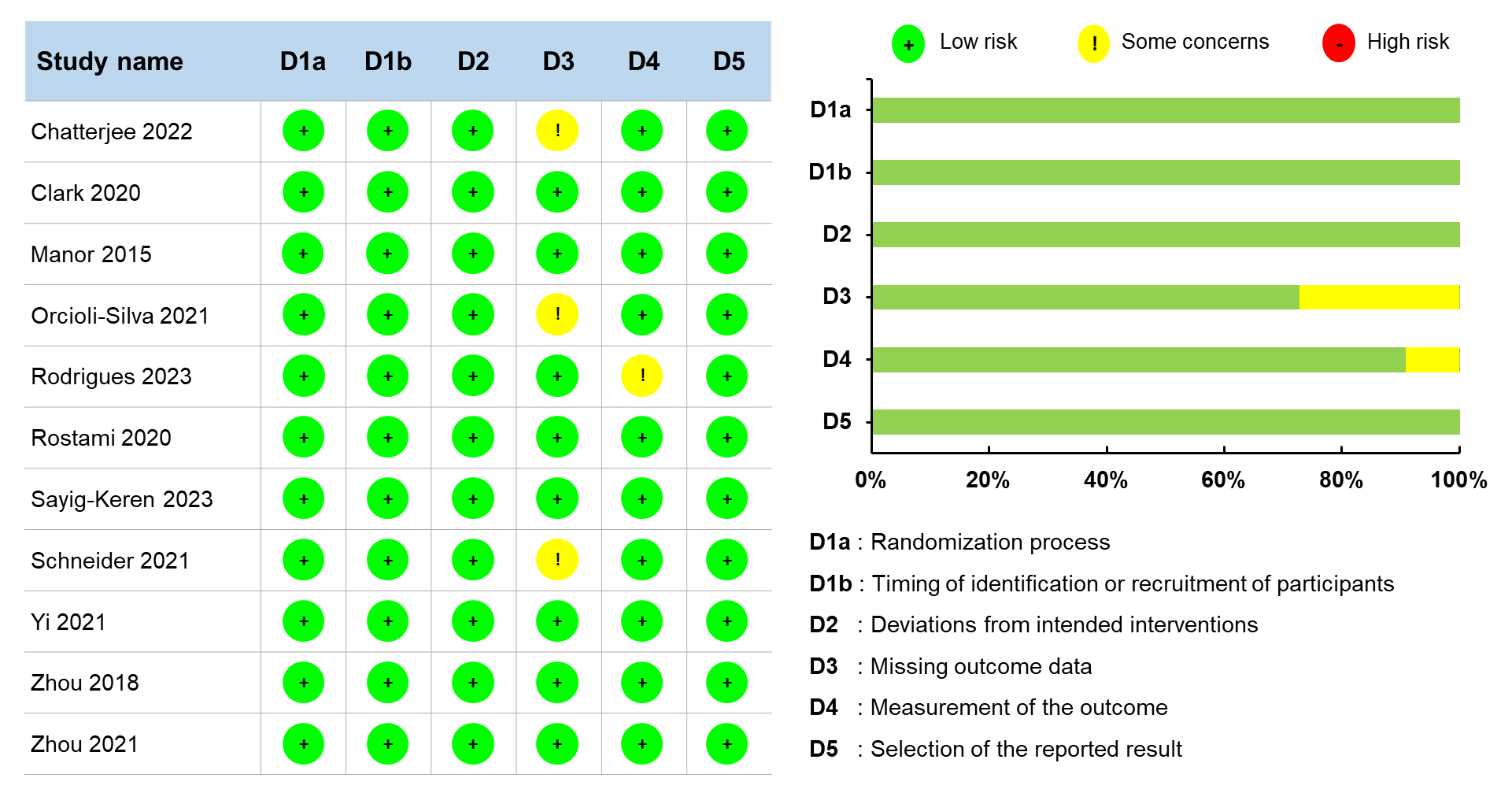 Fig. 2.
Fig. 2.
Methodological quality estimation.
A random-effects meta-analysis revealed that tDCS significantly enhanced
single-task walking performances in healthy older adults (18 comparisons reported
by nine studies; SMD = 0.142; standard error = 0.064; 95% confidence
interval (CI) = 0.016 to 0.269; Z = 2.209; p = 0.027; I-squared =
38.9%; Egger’s
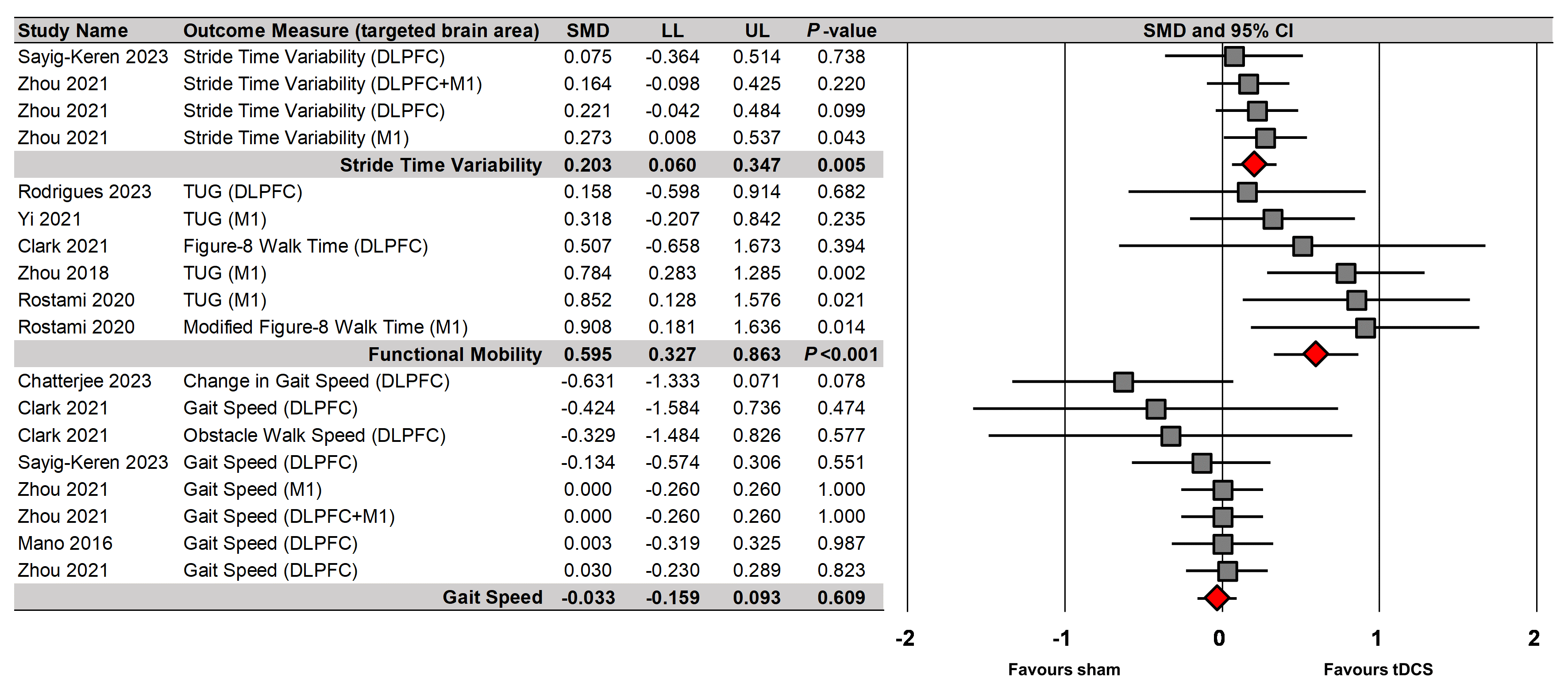 Fig. 3.
Fig. 3.
tDCS effects on different gait variable during single-task walking. CI, confidence interval; LL, lower limit; SMD, standardized mean difference; UL, upper limit.
For targeted brain regions, moderator variable analysis indicated that tDCS
targeting the M1 significantly enhanced gait variables (Fig. 4) (six comparisons
reported by four studies; SMD = 0.424; standard error = 0.150; 95% CI =
0.129 to 0.719; Z = 2.818; p = 0.005; I-squared = 63.3%; Egger’s
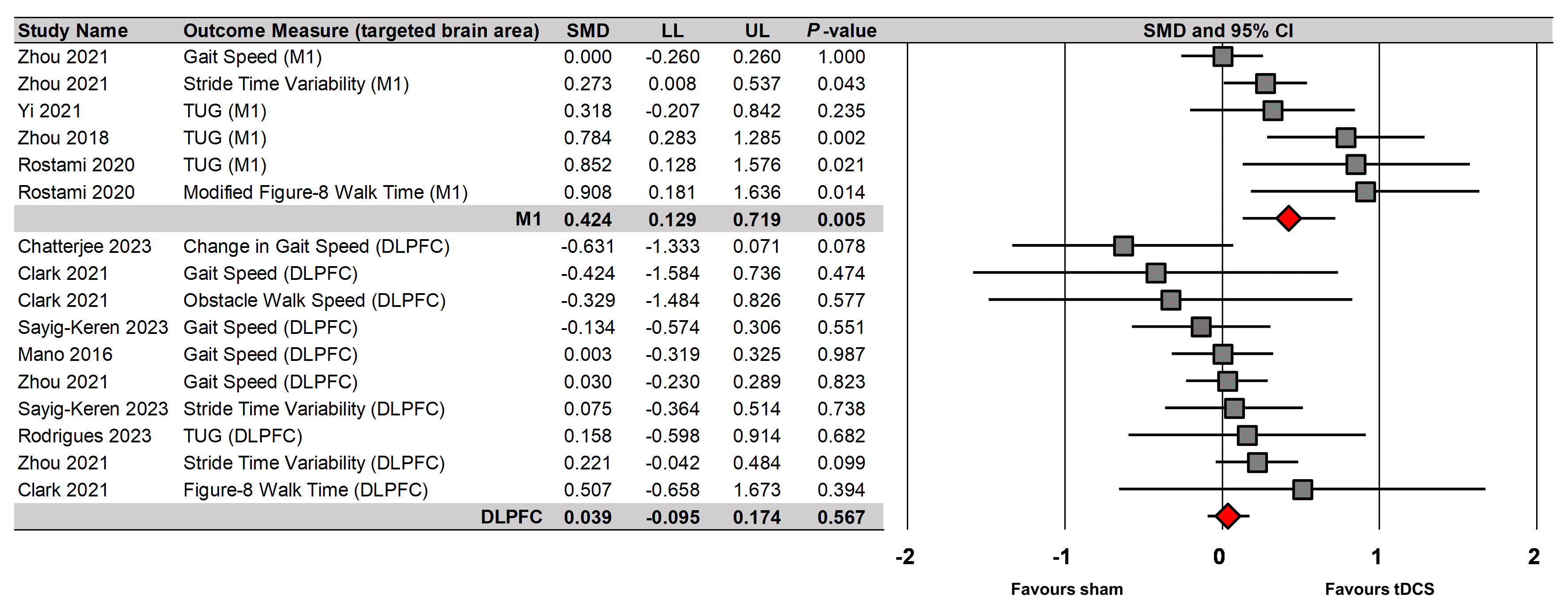 Fig. 4.
Fig. 4.
tDCS effects on single-task walking performances based on targeted brain regions.
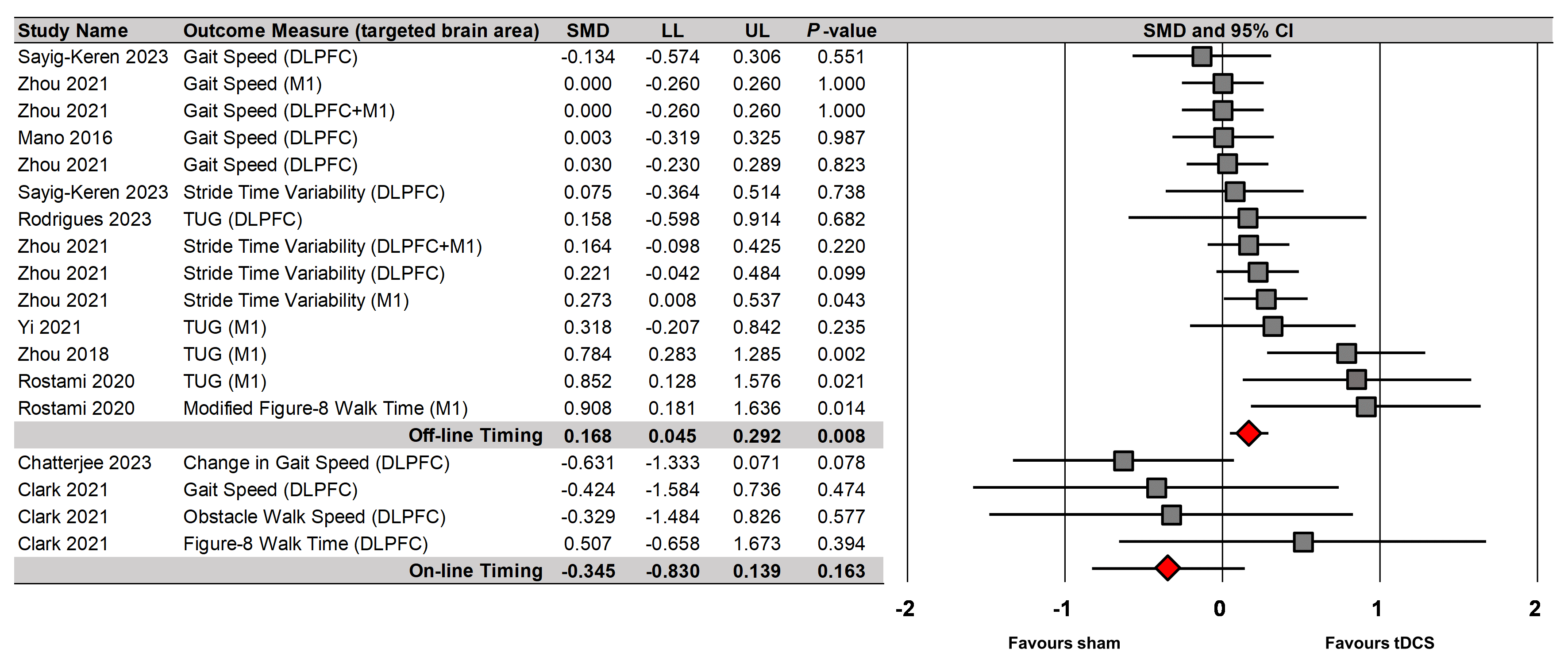 Fig. 5.
Fig. 5.
tDCS effects on single-task walking performances based on timing of tDCS.
The meta-regression analyses failed to report significant relationships between enhancements in single-task walking performances after tDCS and following demographic characteristics and tDCS parameters: (a) age (18 comparisons reported by nine studies; Y = 1.472–0.018X; p = 0.150), (b) female ratio (17 comparisons reported by eight studies; Y = 0.162–0.079X; p = 0.873), (c) intensity of stimulation (18 comparisons reported by nine studies; Y = 0.712–0.349X; p = 0.104), and (d) session of stimulation (19 comparisons reported by five studies; Y = 0.128–0.003X; p = 0.841). Meta-regression analyses were not conducted on the duration of stimulation in single-task walking performances because all studies applied same tDCS protocols.
A random-effects meta-analysis denoted that tDCS significantly enhanced
dual-task walking performances in healthy older adults (19 comparisons reported
by five studies; SMD = 0.281; standard error = 0.061; 95% CI = 0.162 to
0.400; Z = 4.612; p
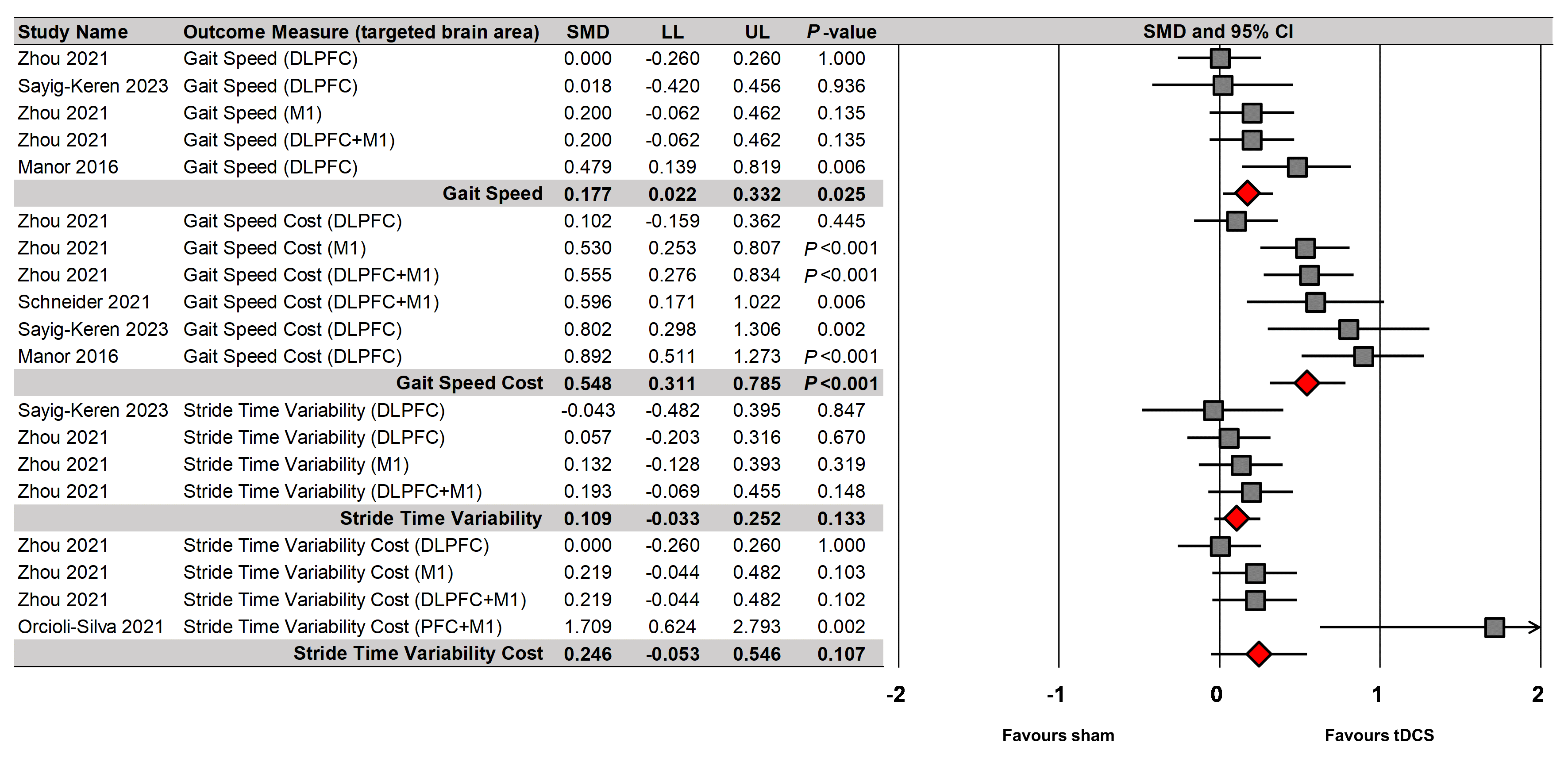 Fig. 6.
Fig. 6.
tDCS effects on different gait variables during dual-task walking.
For targeted brain regions, moderator variable analysis denoted significant
effects of tDCS on two brain area (Fig. 7): (a) DLPFC (nine comparisons reported
by three studies; SMD = 0.231; standard error = 0.106; 95% CI = 0.023
to 0.438; Z = 2.181; p = 0.029; I-squared = 72.0%; Egger’s
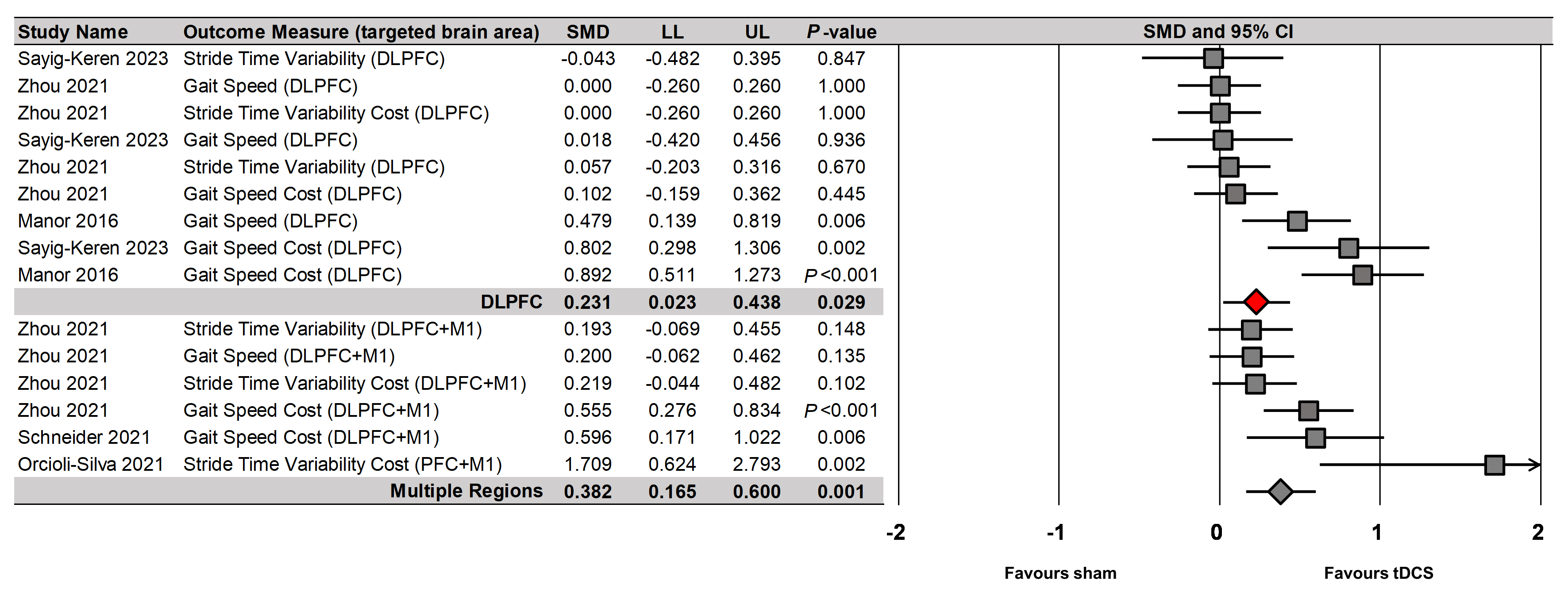 Fig. 7.
Fig. 7.
tDCS effects on dual-task walking performances based on targeted brain regions.
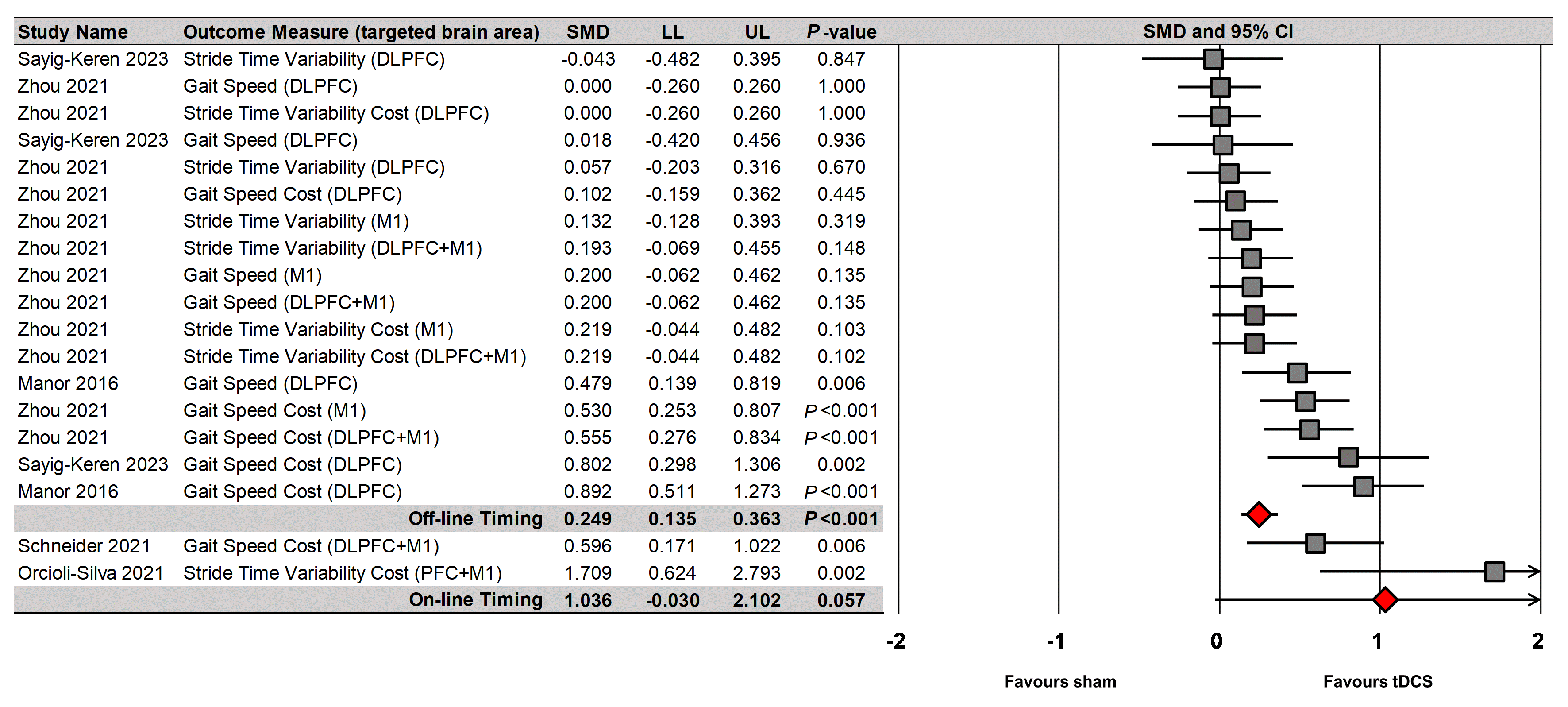 Fig. 8.
Fig. 8.
tDCS effects on dual-task walking performances based on timing of tDCS.
The random-effect meta-regression analysis confirmed enhancements in dual-task walking performances after tDCS were significantly correlated with decreased age (19 comparisons reported by five studies; Y = 3.007–0.037X; p = 0.003; Supplementary Fig. 9). However, the analysis found no significant relationships between enhancements in dual-tasks after tDCS and following two variables; (a) female ratio (19 comparisons reported by five studies; Y = 0.583–0.425X; p = 0.498) and (b) intensity of stimulation (19 comparisons reported by five studies; Y = 0.454–0.110X; p = 0.598). Meta-regression analyses were not conducted on the session and duration of stimulation in dual-task walking performances because all studies applied same tDCS protocols.
This meta-analysis investigated the effects of tDCS on gait performance during single- and dual-task walking in healthy older adults. The findings revealed that tDCS significantly improved overall gait performance during single- and dual-task walking. Specifically, improvements in STV, TUG, and figure-eight walk time were observed for single-task performances, while increases in gait speed and reductions in speed cost were observed for dual-task performances. For targeted brain regions, tDCS stimulating M1 significantly advanced the gait performance during single-task walking, and tDCS protocols stimulating multiple areas including prefrontal cortex were beneficial for dual-task walking performance. For both single- and dual-task walking performances, off-line tDCS protocols were effective. Improvements in dual-task walking performances after tDCS decreased with increasing age for older adults.
Positive effects of tDCS on gait-related performances during single- and dual-task walking in healthy older adults reinforce the previous meta-analytic findings that tDCS improved dynamic balance and postural control in elderly people [59, 60]. To the best our knowledge, this meta-analysis was the first to show tDCS effects on gait performance improvements by focusing on healthy older adults who had no physical and cognitive impairments. Some studies reported inconsistent results in motor improvements after tDCS protocols because of ceiling effects potentially caused by characteristics of participants (e.g., healthy older adults) and task difficulty (e.g., single-task walking) [61, 62, 63]. Importantly, our findings suggest that applying tDCS protocols transiently may improve gait performances in healthy older adults regardless of how challenging a task is to complete.
For dual-task walking, the meta-analytic results revealed significant improvements in gait speed-related outcome variables (i.e., speed and speed cost) although these improvements were not observed in single-task walking. Dual-task costs during gait performance (i.e., altered gait functions from single task to dual task) may increase because of insufficient neural resources necessary for simultaneously completing both cognitive and motor task requirements (e.g., walking while checking watch or phone) [12, 24]. Moreover, age-related cognitive impairments may interfere with effective allocation of neural resources in the brain, leading to greater reduction of gait speed while performing cognitive tasks [12, 23]. Previous meta-analysis studies reported that tDCS protocols facilitated neural excitability across prefrontal cortical regions, resulting in improved cognitive functions (e.g., faster reaction time and better executive function) in older adults [29, 64, 65]. Perhaps, these findings indicate that the contribution of tDCS protocols to cognition-related neural plasticity may enhance dual-task walking performances in healthy older adults.
In fact, our moderator variable analysis identified that tDCS stimulating M1 significantly advanced single-task walking performances, while tDCS protocols stimulating multiple areas including prefrontal cortex improved dual-task walking performances. These findings support a proposition that altered motor cortical excitability by tDCS may affect gait performances requiring lower level of cognitive resources, whereas tDCS protocols targeting prefrontal cortical regions may be effective for improving daily walking on the ground that normally requires greater cognitive resources [51, 52, 66]. Motor improvements in healthy older adults (i.e., single- and dual-task gait performances) with tDCS protocols that stimulated cognition-related regions may be associated with cognitive-motor integration [67]. Cognitive-motor integration refers to concurrent interaction between cognitive processes and motor control systems through neural networks for goal-directed actions [68]. Previous studies reported that distinct brain areas predominately involved in cognitive and motor tasks were integrated into a single network when both tasks were performed simultaneously [69, 70]. For example, a study using functional magnetic resonance imaging revealed increased excitability in both M1 and DLPFC during dual tasks (i.e., a stepping task combined with a serial subtraction task or verbal fluency) as compared with excitability level of the cortical regions during single task (i.e., executing motor and cognitive task, separately) [71]. These findings support a proposition that dual-task walking performance may require the involvement of motor cortical regions as well as cognition-related brain areas such as prefrontal cortical regions. Presumably, applying tDCS protocols may modulate cortical excitability in a specific region that can further influence anatomically or functionally connected areas contributing to neural communications across cognitive and motor networks. For example, anodal stimulation on the left DLPFC improved both cognitive function (i.e., Montreal Cognitive Assessment score) and motor performance (i.e., gait and standing postural sway) compared with sham tDCS condition [72]. Moreover, Lee and colleagues [67] reported positive effects of anodal stimulation with DLPFC on isometric pinch force control capabilities. Taken together, healthy aging population may receive beneficial effects on gait performances by reinforcing their cognitive functions with tDCS protocols.
Interestingly, we found that off-line tDCS protocols were effective for improving both single- and dual-task walking performances, which is consistent with previous findings that off-line tDCS protocols significantly advanced motor and cognitive functions [73, 74]. These findings support a proposition that tDCS protocols may be suitable for clinical application in healthy older adults because participants can concentrate and naturally execute walking practice with minimized distraction and discomfort in off-line tDCS condition. However, no significant effects of on-line tDCS protocols may be attributed to the insufficient number of studies (e.g., two studies for single- and dual-task walking). Thus, further studies will be necessary to confirm on-line tDCS effects. Interestingly, the meta-regression analysis identified that greater enhancements in dual-task walking performances after tDCS occurred in younger elderly individuals. Previous studies raised a possibility that tDCS-induced neural plasticity may be affected by age [75, 76]. Potentially, tDCS effects on dual-task walking decreased with altered excitatory effects of anodal stimulation for older adults.
Despite the positive effects of tDCS on single- and dual-task performances in healthy older adults, caution is necessary when interpreting these findings. First, the positive overall effects of tDCS protocols on gait functions were acquired from a insufficient number of included studies (e.g., 2–6 studies). For dual-gait performances, five out of 11 qualified studies assessed dual-task performances before and after tDCS protocols. Second, tDCS protocols used for each study were inconsistent. Further, our meta-regression findings confirmed different tDCS parameters were not correlated with overall effects so that optimal parameters (e.g., targeted brain area, intensity, frequency, and sessions) for improving gait performance in healthy older adults are still inclusive. Thus, further studies that focused on healthy older adults with standardized tDCS protocols are necessary to support current findings. In addition to the transient effects observed in this meta-analysis, administering multiple sessions of tDCS protocols may lead to retention effects on gait performances [46, 72]. Perhaps, home-based tDCS training program may be effective for long-term of gait rehabilitation in aging population because of the safety and feasibility of tDCS protocols [77, 78]. Finally, tDCS effects may be influenced by different tDCS parameters [79, 80] so that future studies should explore individualized tDCS parameters for optimizing gait performances in older adults.
This systematic review and meta-analysis confirmed that tDCS significantly enhanced single-and dual-gait performances in healthy older adults. Specifically, applying tDCS protocols enhanced gait STV and gait-related mobility (i.e., TUG and Figure-eight walk time) in single tasks and improved gait speed in dual tasks. Further, tDCS stimulating motor cortical regions advanced single-gait performances, whereas tDCS targeting prefrontal cortical regions was beneficial for improving dual-gait performances. Off-line tDCS protocols showed positive effects on both single-and dual-gait performances. These findings indicate that tDCS protocols may be a promising tool to support mobility and reduce gait-related challenges in healthy aging populations.
All data points generated or analyzed during this study are included in this article and there are no further underlying data necessary to reproduce the results.
BJC and HL contributed to acquisition of data and statistical analyses. BJC, HL, and NK performed data interpretation, manuscript drafts and editorial changes. NK conceived and designed the study. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This work was supported by Incheon National University Research Grant in 2024 (2024-0059) to NK.
The authors declare no conflict of interest. Nyeonju Kang is serving as one of the Guest editors of this journal. We declare that Nyeonju Kang had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Yoshihiro Noda.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/JIN36636.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.








