1 Graduate Institute of Clinical Medical Sciences, College of Medicine, Chang Gung University, 33302 Taoyuan City, Taiwan
2 Graduate Institute of Traditional Chinese Medicine, School of Chinese Medicine, College of Medicine, Chang Gung University, 33302 Taoyuan City, Taiwan
3 Department of Traditional Chinese Medicine, Chang Gung Memorial Hospital, 204 Keelung City, Taiwan
4 Institute of Traditional Medicine, School of Medicine, National Yang Ming Chiao Tung University, 11217 Taipei, Taiwan
5 Brion Research Institute of Taiwan, 23143 Taipei, Taiwan
Academic Editor: Salvatore Nesci
Abstract
Background: Non-alcoholic fatty liver disease (NAFLD) has become a prevalent issue and a consequence of metabolic syndrome impact on human health. Both of anti-atherosclerosis and anti-hepatic fibrosis capabilities of herbal medicine Ger-Gen-Chyn-Lian-Tang (GGCLT) has attracted attention, but their molecular regulatory mechanisms in a NAFLD model have not been elucidated. The aim of the present study was to explore the bioactivity of db/db mice following treatment with GGCLT. Methods: NAFLD phenotype of db/db mice were treated with GGCLT and lipogenesis, mitochondria dysfunction, mitophagy, macrophage polarization and adipose tissue browning were then evaluated using qRT-PCR and/or Western blot analysis, immunofluorescence, and immunohistochemistry assays, respectively. Results: GGCLT not only decreased serum levels of TG and free fatty acids, but glucose and insulin tolerance test in db/db mice. In parallel, GGCLT reduced lipogenesis and hypoxia-inflammation cascades in NAFLD progression. GGCLT reduced lipid accumulation and was accompanied by the enhanced mitochondria biogenesis, M2 macrophage, and decreased M1 macrophage. The latter two events contributing to the anti-inflammation are resulting from mitochondria dynamics, and the lipotoxicity lowering effect of GGCLT of NAFLD mice is mediated by promoting mitophagy in Parkin-dependent and -independent pathways, by mitochondrial fusion over fission manner. GGCLT also inactivated lipogenesis and decreased lipid accumulation in epididymal white adipose tissue with a higher M2/M1 macrophage ratio. Conclusions: Besides in the liver, modulating of mitochondrial biogenesis and adipose tissue browning were characterized by increased Tmem26, Tfam, and Prdm16 expression by GGCLT in EWAT also contributes to the beneficial action in NAFLD.
Keywords
- NAFLD
- Ger-Gen-Chyn-Lian-Tang
- lipotoxicity
- mitochondria biogenesis
- mitophagy
- adipose tissue browning
NAFLD (non-alcoholic fatty liver disease) prevalence is increasing constantly, with an increase in prevalence accompanied by obesity and type 2 diabetes (T2D) [1]. An excess flow of free fatty acids (FFA) arising due to dysfunctional/insulin resistant (IR) adipose tissue causing ectopic fat deposition in liver is closely the NAFLD development [2]. In NAFLD and IR, both of excess lipid droplets and insulin signaling play an important role in adipose tissue dysfunction [3]. The alterations in lipid metabolism are key to the development of NAFLD and its progression. In addition, the condition of high free fatty acids, oxidation stress, inflammatory response, oxidation stress, mitochondrial dysfunction, and macrophage polarization are observed in NAFLD [4].
Hepatic lipotoxicity, due to massive FFAs flux and lipogenesis, has been associated to NAFLD severity and comprises the processes of oxidative stress, autophagy, and inflammation processes [5]. In the liver, induction of CD36 translocation to the plasma membrane of the hepatocyte increases FFA uptake may be a determining factor in NAFLD patients with steatosis [5]. In keeping with these findings, attenuate mitochondrial oxidative stress and thereby protection against lipid-driven hepatotoxicity, therefore mechanisms and treatment strategies by modulating the patho-physiological feature of lipophagy is perhaps to halt NAFLD.
Hepatocytes accumulate excessive FFA due to increased availability of FFA beyond
their capacity for oxidation or export by lipoprotein particles in NAFLD [6].
Fatty acids flux was increased in hepatocytes results in enhanced fatty acid
import and oxidation of mitochondria in NAFLD. As oxidative stress signaling
promotes nuclear factor-
An interesting mechanism of hepatocyte sensitivity to hypoxic stress and mitochondrial dysfunction is the association NAFLD pathogenesis. In the liver and white adipose tissue (WAT), mitochondria is an energy metabolism center, which is maintained via combination ofmitochondrial respiratory chain (MRC) activity and fatty acid beta-oxidation [12, 13]. The clearance of damaged mitochondria is the responsible of mitophagy that is promote mitochondrial turnover [14, 15]. Mitophagy deregulation is also implicated in fatty liver disease, hepatic fibrosis, and liver cancer [16]. On the other hand, the WAT expansion is associated with body fat accrual leading to obesity. Fat is often noted for its energy-storing function, as in WAT; however, brown and beige adipocytes play a crucial role in whole-body energy homeostasis through futile catabolism of circulating fatty acids [17]. The uncoupling protein (UCP) is especially expressed in brown and beige fat, where it regulates thermogenesis by enabling mitochondria to acquire and oxidize a variety of extracellular metabolites that are associated with a variety of metabolic dysfunctions. Nevertheless, emerging evidence that the factors of biochemical and functional characteristics of thermogenic genes are including Ucp1, Cidea, Pgc1a, Cox7a, Prdm16, Cited1, Cd137, Tmem26 in beige adipocytes [18].
Ger-Gen-Chyn-Lian-Tang (GGCLT) is a standardized traditional Chinese herbal
medicine, which is composed of Puerariae radix, Scutellariae
radix, Coptidis rhizome, and Glycyrrhizae radix. GGCLT
attenuated atherosclerosis in an apolipoprotein E
GGCLT has the following four botanical components, the following herbs were
produced in an 8:3:3:2 weight ratio: (i) Puerariae Radix (roots of
Pueraria lobata, Ge Gen, 480 g); (ii) Scutellariae Radix (roots
of Scullellaria baicalensis, Huang Qin, 180 g); (iii) Coptidis
Rhizoma (rhizomes of Coptis chinensis, Huang Lian, 180 g), and (iv)
Glycyrrhizae Radix (roots of Glycyrrhiza uralensis, Gan Cao,
120 g). The 960 g of the herbal components were steeped in 5 liters of dH
The six main index components of the GGCLT decoction were recognized by HPLC. A
Waters HPLC System (Waters Corporation, Milford, Massachusetts, United States)
with a 600 quaternary pump, a Sugai U-620 column temperature controller, a 717
plus autosampler, and a 996 photodiode array detector was used to perform the
HPLC-UV study. Puerarin, baicalin, berberine, baicalein, glycyrrhizic acid, and
wogonin in analytical-grade pure powder forms were used as standards for the HPLC
assay. Puerarin, baicalin, baicalein, glycyrrhizic acid, and wogonin were
purchased from Merck (Rahway, NJ, USA). Bererine was purchased from Selleckchem
(Burlington, MA, USA). The analytical conditions were set as follows: gradient
elution by the mixture of mobile phases A (mixture of 20 mM potassium dihydrogen
phosphate and 0.0085% phosphoric acid), B (acetonitrile) and C (water) at 0–30
min with a ratio of 90–75% A and 10–25% B; 30–40 min with a ratio of
75–65% A and 25–35% B; 40–55 min with a ratio of 65–0% A, 35–75% B and
0–25% C; 55–60 min with a ratio of 75–10% B and 25–90% C; 60–65 min with
a ratio of 0–90% A, 90–10% B and 90–0% C. The flow rate was kept constant
at 1.0 mL/min. The column temperature was controlled at 35 °C, the post
time was 15 min and the injection volume was 5
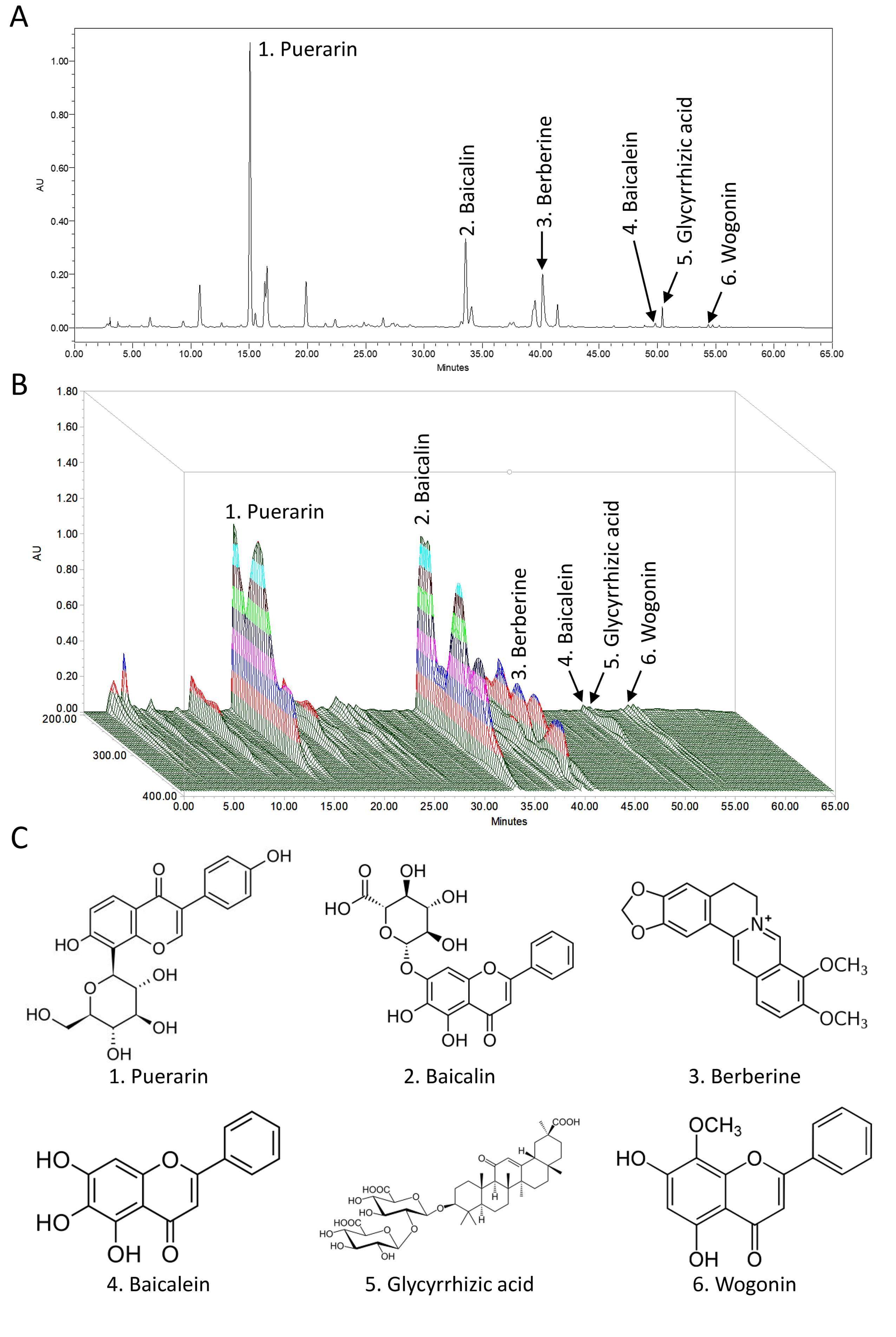 Fig. 1.
Fig. 1.Ger-Gen-Chyn-Lian-Tang (GGCLT) chromatogram determined by HPLC-UV. The panel shows the fingerprint of the HPLC fingerprint (detection wavelength 250 nm) (A) and three-dimensional structures of these six main index components (B). Structures of the compounds identified by HPLC-UV (C). Puerarin, baicalin, berberine, baicalein, glycyrrhizic acid, and wogonin, the six main index components of GGCLT.
The Traditional Chinese Medicine Integrative Database (TCMID) were used to screen the active compounds of GGCLT for herbal molecular mechanism analysis. Besides, literature analysis was performed to supplement the potential active ingredients and their pharmacological interactions. The fatty liver related genes were screened by Public Health Genomics and Precision Health Knowledge Base (v7.7). The intersection genes between targets of the active compounds of GGCLT and fatty liver related genes were overlapped by Venny 2.1.0.
The protein-protein interaction (PPI) network was constructed by Cytoscape 3.9.1 (Institute of Systems Biology, Seattle, Washington, USA). Network Analyzer was used for network diagrams. Construction of PPI network and screening of core targets fatty liver-related target proteins were analyzed by online STRING11.5 database to construct the PPI network. Protein type was set as “Homo sapiens” with the minimum interaction threshold of 0.4.
The Traditional Chinese Medicine Integrative Database (TCMID, Bioinformatics &
Drug Design group in Department of Pharmacy National University, Singapore) was
used to screen the GO function and KEGG pathway analysis that were performed to
determine the function of candidate genes in the drug-compound-disease-target
gene network. GO term, including biological process (BP), molecular functions
(MF), cellular components (CC) and KEGG pathway with p
The National Laboratory Animal Center (Taiwan) provided the C57BL/6J and
BKS.Cg-m
After a 6-hour fasting, a glucose load (1 g/kg of body weight) was administered by gavage or by intraperitoneal (i.p.) injection. Blood samples were collected from the tail at 0 (baseline), 15, 30, 60, 90 and 120 minutes after the glucose loads. Blood glucose levels were measured with a Blood Glucose Monitoring System (OneTouch® Ultra, Lifescan, Johnson & Johnson, USA). AUCs of glucose levels calculated according to Trapezoid rules with those from GTT.
After a 6-hour fasting, an insulin load (0.75 U/kg of body weight; Lilly, USA) was administered by gavage or by intraperitoneal (i.p.) injection. Blood samples were collected from the tail at 0 (baseline), 15, 30, 60, 90 and 120 minutes after the insulin loads. AUCs of glucose levels calculated according to Trapezoid rules with those from ITT. Plasma insulin levels were determined by ELISA kit.
The levels of fasting insulin and fasting glucose are used to compute the
homeostatic model assessment of insulin resistance (HOMA-IR). Scores were
calculated with the following equation: [fasting glucose (mmol/L)
Prior to biochemical examination, plasma was collected by centrifugation and kept at –20 °C. The Randox ATL and AST test was used to assess the plasma concentrations of alanine aminotransferase (ALT) and aspartate aminotransferase (AST) (Randox, Kearneysville, WV, USA).
Using the Randox triglycerides test and the NEFA Assay, the concentrations of plasma triglycerides (TG) and free fatty acids (FFA) were both directly assessed (Randox, Kearneysville, WV, USA).
Liver and EWAT were embedded in paraffin after being fixed overnight in 10%
Neutral buffered formalin. Then, xylene was used to deparaffinize portions that
were five micrometers thick, and a graded ethanol series was used to dehydrate
them. Hematoxylin (2 g/L) was used to stain the tissue slices for 15 minutes, and
eosin (0.1 percent in 0.0003 percent acetic acid) was used for 10 minutes at room
temperature. Masson stain used by Trichrome Stain (Masson) Kit (abcam, Cambridge,
USA). For immunohistochemistry, endogenous peroxidase activity was quenched with
3% H
| Antibodies | Identifier | Source | Dilution |
| MAB1501 | Millipore, Burlington, MA, USA | WB 1:1000 | |
| CCR7 | ab32527 | abcam, Cambridge, UK | IHC 1:100; WB 1:500 |
| CD11b | ab133357 | abcam, Cambridge, UK | IHC 1:100; WB 1:500 |
| CD11c | ab52632, ab11029 | abcam, Cambridge, UK | IHC 1:100; WB 1:500 |
| CD163 | ab182422 | abcam, Cambridge, UK | IHC 1:100; WB 1:500 |
| CD206 | ab64693 | abcam, Cambridge, UK | IHC 1:100; WB 1:500 |
| CD36 | SC-70644 | Santa Cruz Biotechnology, TX, USA | IHC 1:100; WB 1:500 |
| DRP1 | sc-271583 | Santa Cruz Biotechnology, TX, USA | WB 1:500 |
| DRP1 (Tyr504) | PA5-105983 | Thermo Fisher Scientific, MA, USA | IHC 1:100; WB 1:500 |
| F4/80 | CI:A3-1 | Thermo Fisher Scientific, MA, USA | IHC 1:100; WB 1:500 |
| HIF1 | ab179483 | abcam, Cambridge, UK | IHC 1:100; WB 1:500 |
| Histone | SC-56616 | Santa Cruz Biotechnology, TX, USA | WB 1:500 |
| LC3B | GTX127375 | GeneTex, CA, USA | IHC 1:100; WB 1:500 |
| MCP-1 | ab7202 | abcam, Cambridge, UK | IHC 1:100; WB 1:500 |
| Mitofusin 1 | ab126575 | abcam, Cambridge, UK | IHC 1:100; WB 1:500 |
| MMP9 | AB19016 | Millipore, Burlington, MA, USA | IHC 1:100; WB 1:500 |
| Parkin | GTX39745 | GeneTex, CA, USA | IHC 1:100; WB 1:500 |
| PGC1 |
ab54481 | abcam, Cambridge, UK | IHC 1:100; WB 1:500 |
| SIRT1 | ab110304 | abcam, Cambridge, UK | IHC 1:100; WB 1:500 |
| SQSTM/p62 | GTX100685 | GeneTex, CA, USA | WB 1:500 |
| SQSTM/p62 (Ser403) | GTX128171 | GeneTex, CA, USA | IHC 1:100; WB 1:500 |
| SREBP-1 | SC-366 | Santa Cruz Biotechnology, TX, USA | IHC 1:100; WB 1:500 |
| Tmem26 | PA5-23477 | Thermo Fisher Scientific, Waltham, MA, USA | IHC 1:100; WB 1:500 |
| UCP1 | ab10983 | abcam, Cambridge, UK | IHC 1:100; WB 1:500 |
| VEGF | ab69479 | abcam, Cambridge, UK | IHC 1:100; WB 1:500 |
| Goat anti-mouse IgG | Ap124P | MilliporeSigma, USA | WB 1:5000 |
| Goat anti-rabbit IgG | 111-065-003 | Jackson ImmunoResearch Laboratories, PA, USA | WB 1:5000 |
| Goat anti-rat IgG | 112-065-003 | Jackson ImmunoResearch Laboratories, PA, USA | WB 1:5000 |
Tissues were homogenized in T-PER™ Tissue Protein Extraction
Reagent (0.30 mg tissue/200
Total RNA was extracted from 0.2 g liver and EWAT tissues using 1 mL of TRIzol
(Thermo Fisher Scientific, MA, USA). The cDNA was reverse transcribed from RNA
using a High-Capacity cDNA Reverse Transcription Kit (Thermo Fisher Scientific,
MA,USA) according to the manufacturer’s instructions. The cDNA samples were
amplified by qRT-PCR using SYBR Green I (Roche, Switzerland). For qPCR, the
following thermocycling conditions were applied: Initial denaturation at 95
°C for 5 min, followed by 35 cycles of denaturation at 94 °C
for 30 sec, annealing at 55 °C for 30 sec, and elongation at 72
°C for 0.5 min, before holding at 4 °C. By utilizing the
2
| Gene | Forward (5′→3′) | Reverse (5′→3′) | Accession No. | Reference |
| Srebp1 | actgtcttggttgttgatgagctggagcat | atcggcgcggaagctgtcggggtagcgtc | NM_011480 | [23] |
| Fas | tgtcattggcctcctcaaaaagggcgtcca | tcaccactgtgggctctgcagagaagcgag | NM_007988.3 | [23] |
| Scd1 | ccggagaccccttagatcga | tagcctgtaaaagatttctgcaaacc | NM_009127.4 | [23] |
| Acc1 | cccgccagcttaaggaca | tggatgggatgtgggca | NM_133360.3 | [24] |
| Cd36 | gcaaaacgactgcaggtcaac | tggtcccagtctcatttagcca | NM_001159558.1 | [23] |
| Fatp | gcttcaacagccgtatcctc | tcttcttgttggtggcactg | NM_011977.4 | [23] |
| Cpt-1 | ggacagagactgtgcgttcct | gcgatatccaacagtgcttga | NM_013495.2 | [23] |
| Cpt-2 | caactcgtatacccaaacccagtc | gttcccatcttgatcgaggacatc | NM_009949.2 | [25] |
| Tnf |
ttgacctcagcgctgagttg | cctgtagcccacgtcgtagc | NM_013693 | [23] |
| Ifn |
cctcaaacttggcaatactc | agcaacaacataagcgtcat | NM_008337.4 | [23] |
| Mcp1 | aggtccctgtcatgcttctg | tctggacccattccttcttg | NM_011333.3 | [23] |
| Il-1 |
aacctgctggtgtgtgacgttc | cagcacgaggcttttttgttgt | NM_008361.4 | [3] |
| Nlrp3 | agccttccaggatcctcttc | cttgggcagcagtttctttc | NM_145827.4 | [26] |
| Aim2 | ataggaggaacaacaacat | gccatcttctgctacata | NM_001013779.2 | [27] |
| Asc | gaagctgctgacagtgcaac | tgtgagctccaagccatacg | NM_023258.4 | [28] |
| Caspase1 | agatggcacatttccaggac | gatcctccagcagcaacttc | NM_009807.2 | [26] |
| Sirt1 | gcaacagcatcttgcctgat | gtgctactggtctcactt | NM_019812.3 | [29] |
| Pgc1 |
gactcagtgtcaccaccgaaa | tgaacgagagcgcatcctt | NM_008904.2 | [23] |
| Ucp1 | cctgcctctctcggaaacaa | tgtaggctgcccaatgaaca | NM_009463.3 | [23] |
| 20kDa/Complex I | ccagctgcgcagagttcatc | gagagagcttggggaccacg | NM_029272.4 | [30] |
| Ip/Complex II | tctaccgctgccacaccatc | aagccaatgctcgcttctcc | NM_023374.4 | [30] |
| Core II/Complex III | ccattggaaatgcagaggca | ggctggtgacttcctttggc | NM_025899.2 | [30] |
| Cox2/Complex IV | tcaaaagaagtgctggaaaaggtt | tctacctgagtgtctttgactgtg | NM_011198.4 | [31] |
| F1 |
atctatgcgggtgtacgggg | agggactggtgctggctgat | NM_007505.2 | [30] |
| Pink | gagcagactcccagttctcg | gtcccactccacaaggatgt | NM_026880.2 | [32] |
| Ndp52 | aaggactggattggcatcttta | aggtcagcgtacttgtctttc | NM_001271018.1 | [33] |
| Ambra1 | gggatgttgtgcctttgca | cctggtgtgggaagagagaaga | NM_172669.3 | [34] |
| Phb2 | cttggttccagtaccccattatc | cgagacaacactcgcaggg | NM_007531.2 | [35] |
| Bnip3 | cagcatgaatctggacgaag | atcttcctcagacagagtgc | NM_009760.4 | [35] |
| Nix | gagccggatactgtcgtcct | caatatagatgccgagcccca | NM_009761.3 | [36] |
| Fundc1 | tgtgatatccagcggcttcg | gccggctgttccttactttg | NM_028058.4 | [37] |
| Bcl2 | aaaccctgtgctgctatc | ctgtgttcttcatcgttacttc | NM_009741.5 | [37] |
| Tfam | ggaatgtggagcgtgctaaaa | tgctggaaaaacacttcggaata | NM_009360.4 | [3] |
| Cd137 | cgtgcagaactcctgtgataac | gtccacctatgctggagaagg | NM_011612.2 | [3] |
| Tmem26 | accctgtcatcccacagag | tgtttggtggagtcctaaggtc | NM_177794.3 | [3] |
| Prdm16 | cagcacggtgaagccattc | gcgtgcatccgcttgtg | NM_001291029.1 | [3] |
| Cox7a1 | cagcgtcatggtcagtctgt | agaaaaccgtgtggcagaga | NM_009944.3 | [3] |
| Cox8b | gaaccatgaagccaacgact | gcgaagttcacagtggttcc | NM_007751.3 | [3] |
| Zic1 | ctgttgtgggagacacgatg | cctcttctcagggctcacag | NM_009573.4 | [3] |
| Lxh8 | acacgagctgctacattaagga | cccagtcagtcgagtggatg | NM_010713.2 | [3] |
| Gadph | ccaatgtgtccgtcgtggatct | gttgaagtcgcaggagacaacc | NM_001289726.1 | [38] |
Crude mitochondrial fraction was extracted from liver and EWAT using Mitochondria Isolation Kit (Thermo Fisher Scientific, MA, USA) for Tissue followed by manufacturer’s instructions. With the assistance of the Bio-Rad Rapid Coomassie kit, the proteins were measured. For measurement of mitochondrial Complex I, III, and IV activity in liver and EWAT, using Mitochondrial Complex activity Assay Kit, respectively. For measurement of Citrate synthase activity and MDA using Colorimetric Assay kit followed by manufacturer’s instructions.
Examinations of serum TNF alpha, MCP-1 and IL-1
All data are expressed as means
A typical GGCLT HPLC-UV chromatography fingerprint profile was obtained and six major constituents were identified (Fig. 1). The well-separated peaks with retention times of 65 min and perentage of compound in GGCLT (lyophilizate, %) were identified as follows: (1) Puerarin (15.07 min, 28.90%); (2) Baicalin (33.55 min, 11.67%); (3) Berberine (40.15 min, 7.77); (4) Baicalein (49.81 min, 0.54%); (5) Glycyrrhizic acid (50.42 min, 1.22); (6) Wogonin (54.35 min, 0.28%) (Table 3). Puerarin is the bioactive ingredient of Puerariae Radix; baicalin, baicalein, wogonin are the bioactive ingredient of Scutellariae Radix; berberine is the bioactive ingredient of Coptidis Rhizoma; and glycyrrhizic acid is the bioactive ingredient of Glycyrrhizae Radix. Based on the results, Puerarin was found to be the most abundant compound of GGCLT (lyophilizate), followed by baicalin, berberine, glycyrrhizic acid, baicalein, and wogonin. In addition, the 3D-HPLC fingerprint and the structure of compounds are shown (Fig. 1).
| No. | Compound | Retention time (min) | Percentage of compound in GGCLT (lyophilizate, %) | Weight of compound in GGCLT (lyophilizate, mg/g) |
| 1 | Puerarin | 15.07 | 28.90 | 289.0 |
| 2 | Baicalin | 33.55 | 11.67 | 116.7 |
| 3 | Berberine | 40.15 | 7.77 | 77.7 |
| 4 | Baicalein | 49.81 | 0.54 | 5.4 |
| 5 | Glycyrrhizic acid | 50.42 | 1.22 | 12.2 |
| 6 | Wogonin | 54.35 | 0.28 | 2.8 |
A total of 26 genes overlapped between 512 target genes of the active compounds
of GGCLT and 163 fatty liver related genes (Fig. 2A). Then, a PPI network was
constructed based on 26 overlapped genes between target proteins and fatty liver
relate genes (Fig. 2B). We found the network contained 26 nodes with interaction
score
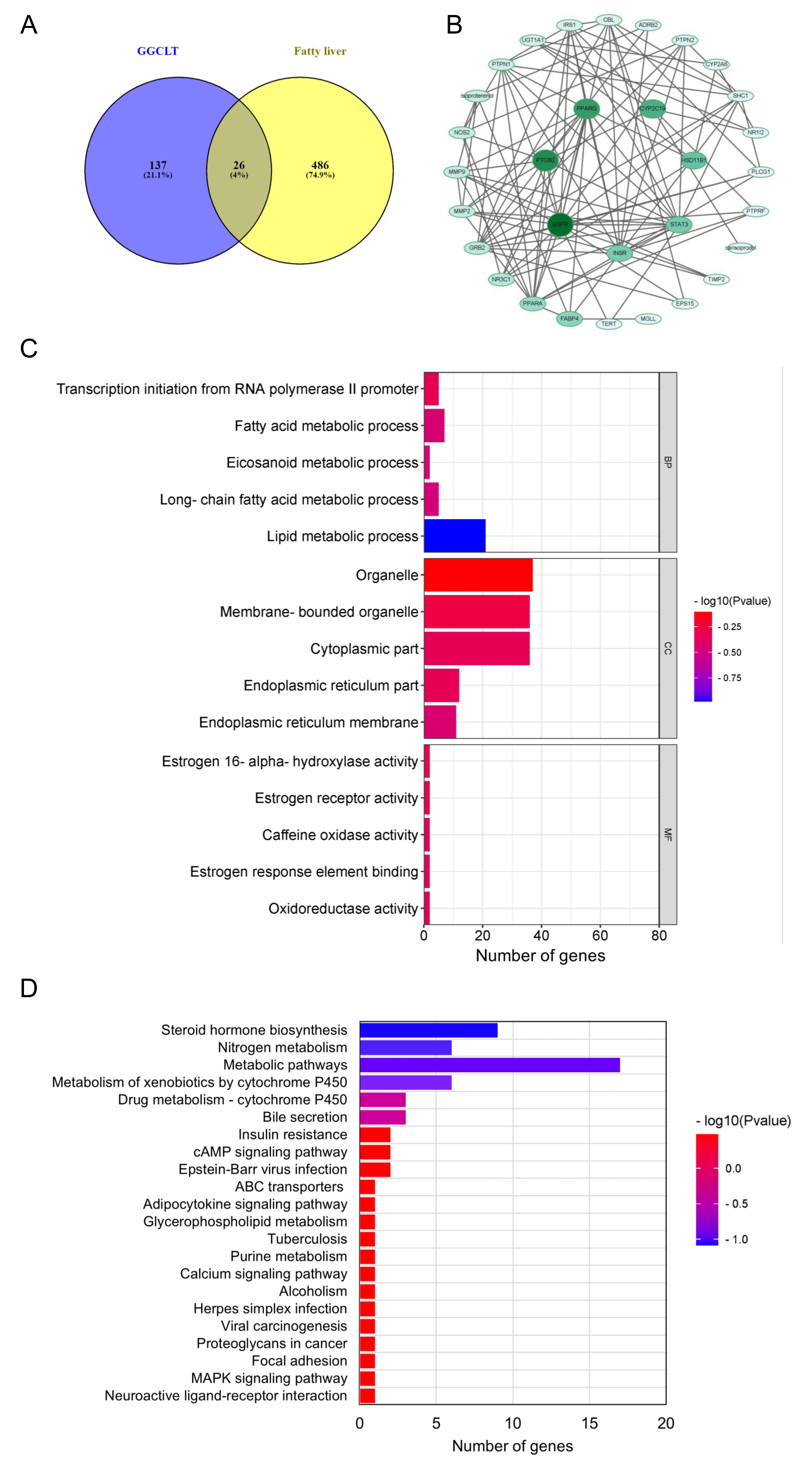 Fig. 2.
Fig. 2.GGCLT target prediction, creation of the PPI network, and analysis of enriched GO and KEGG pathways. Venn plot of the overlapped genes between targets of the active compounds of GGCLT and fatty liver related genes (A). The hub genes in the protein-protein interaction (PPI) network (B). PPI network of the 26 overlapped genes between target proteins of the active compounds of GGCLT and fatty liver related genes. Data show the top 5 remarkably enriched items in the cell component (CC), biological process (BP), molecular function (MF) (C). Data show the top 22 remarkably enriched items in the KEGG pathways (D).
When compared to normal mice, db/db animals had a higher body weight and liver weight/body weight ratio (Fig. 3A,B). GGCLT 150 mg/kg dramatically lowered body weight and the liver/body weight ratio in db/db mice (Fig. 3A,B). In addition, plasma ALT and AST were significantly decreased in db/db mice treated GGCLT compared with db/db mice (Fig. 3C,D). After 28 days GGCLT treatment, the glucose and GTT/ITT, fasting glucose, fasting insulin, and HOMA-IR were decreased compared with db/db mice (Fig. 3E–K), suggesting that GGCLT improved whole-body insulin sensitivity. GGCLT significant reduced plasma TG and FFA level compared with db/db mice (Fig. 3J,K), as well as reduced liver steatosis and fibrosis (Fig. 3L). As shown in Fig. 3L–O, the SREBP-1c and downstream target genes, Fas, Scd1 and Acc1 mRNA expression were significantly increased as well as fatty acid transporters (Cd36, Fatp, Cpt1 and Cpt2) were significantly increased in the liver of db/db mice (Fig. 3L–O). Moreover, GGCLT significantly reduced hepatic lipogenesis therefore decreased fatty acid uptake in NAFLD model.
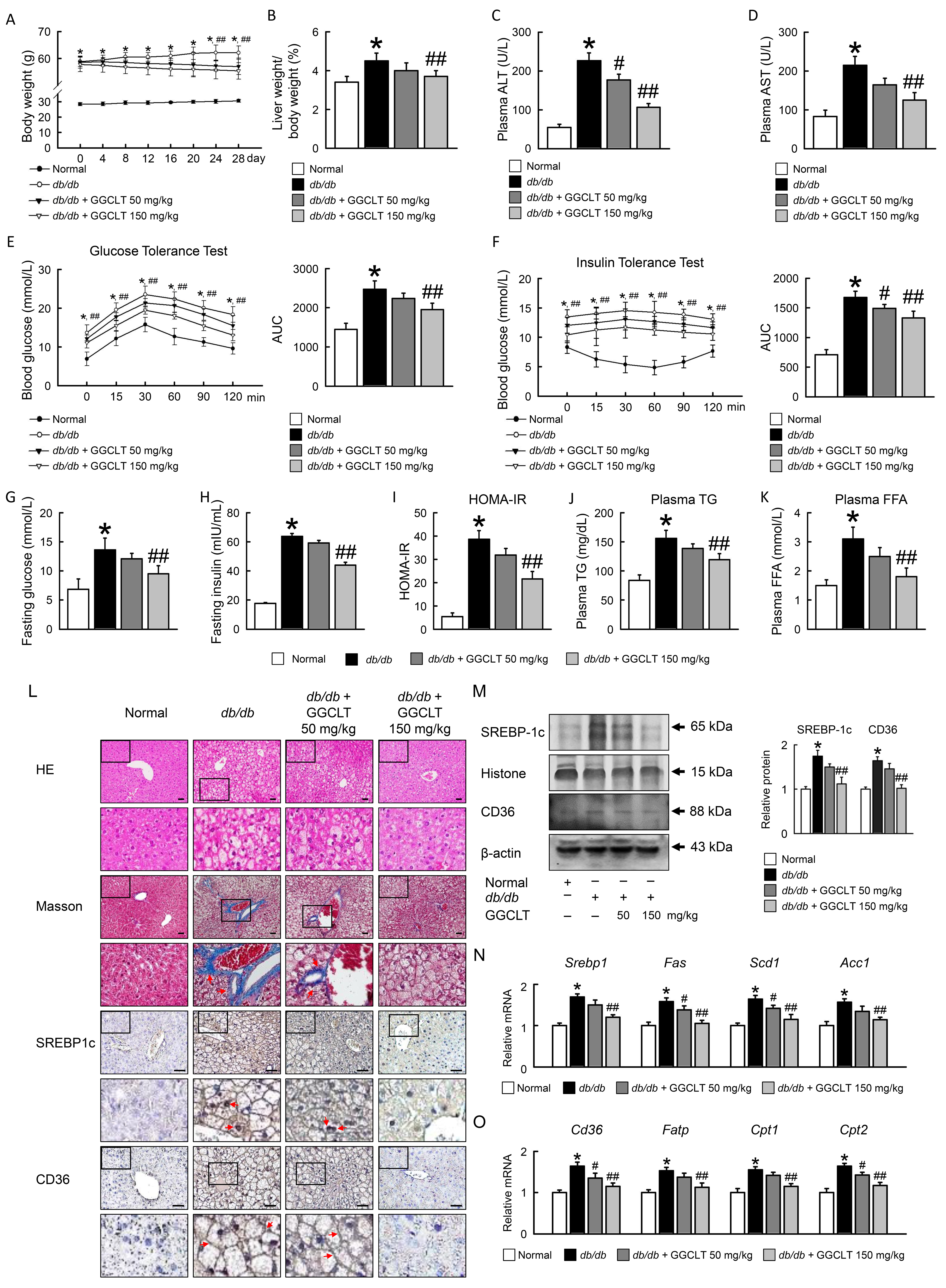 Fig. 3.
Fig. 3.GGCLT prevents NAFLD development in db/db mice. Body
weight (g) (A). Liver weight/ body weight (%) (B). Plasma ALT (U/L) (C). Plasma
AST (U/L) (D). Glucose tolerance test (E). Insulin tolerance test (F). Fasting
glucose (mmol/L) (G). Fasting insulin (mIU/mL) (H). HOMA-IR (I). Plasma TG
(mg/dL) (J). Plasma FFA (mmol/L) (K). Representative HE, Masson, SREBP-1c and
CD36 staining of liver (L). Red arrow highlights the positive staining. Scale
bar: 50
The serum and hepatic TNF-
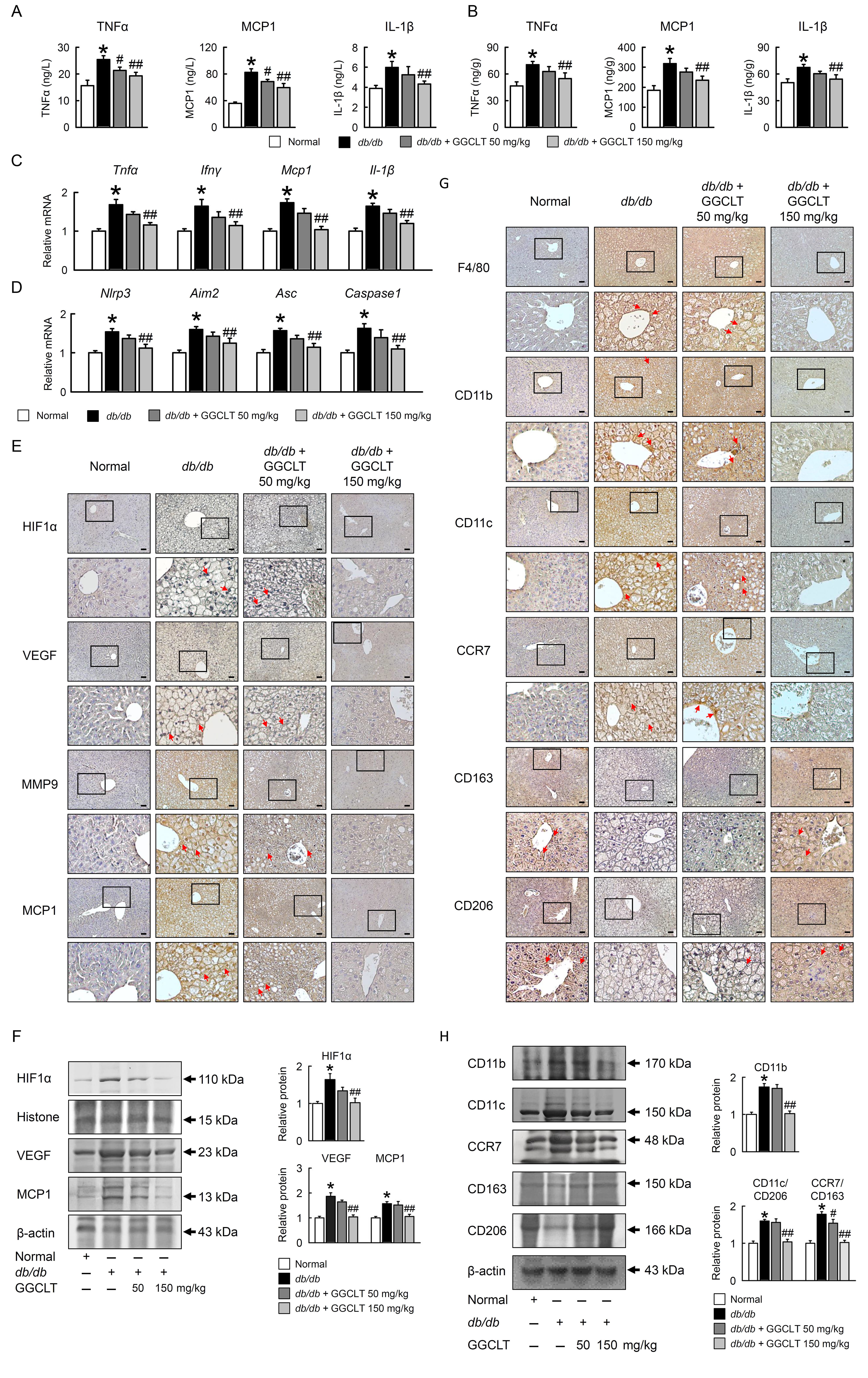 Fig. 4.
Fig. 4.GGCLT reduced hepatic inflammation and M1 polarization in
db/db mice. Serum concentration of TNF
F4/80-positive macrophage infiltration in the db/db mce was considerably enhanced in the liver, which corresponded to the level of serum inflammatory cytokine expression. Macrophage infiltration in the group administered GGCLT 150 mg/kg was significantly lower than that in the db/db mice (Fig. 4G,H). Next, we examined whether macrophagy polarization is involved in NAFLD in db/db mice by assesing M1/M2 marker expression. Immunohistochemistry analysis showed a significant reduction in F4/80, CD11b, CD11c, CCR7 and increase in CD163, CD206 protein level in db/db mice treated with GGCLT 150 mg/kg compared to db/db mice (Fig. 4G,H).
The SIRT1, PGC1
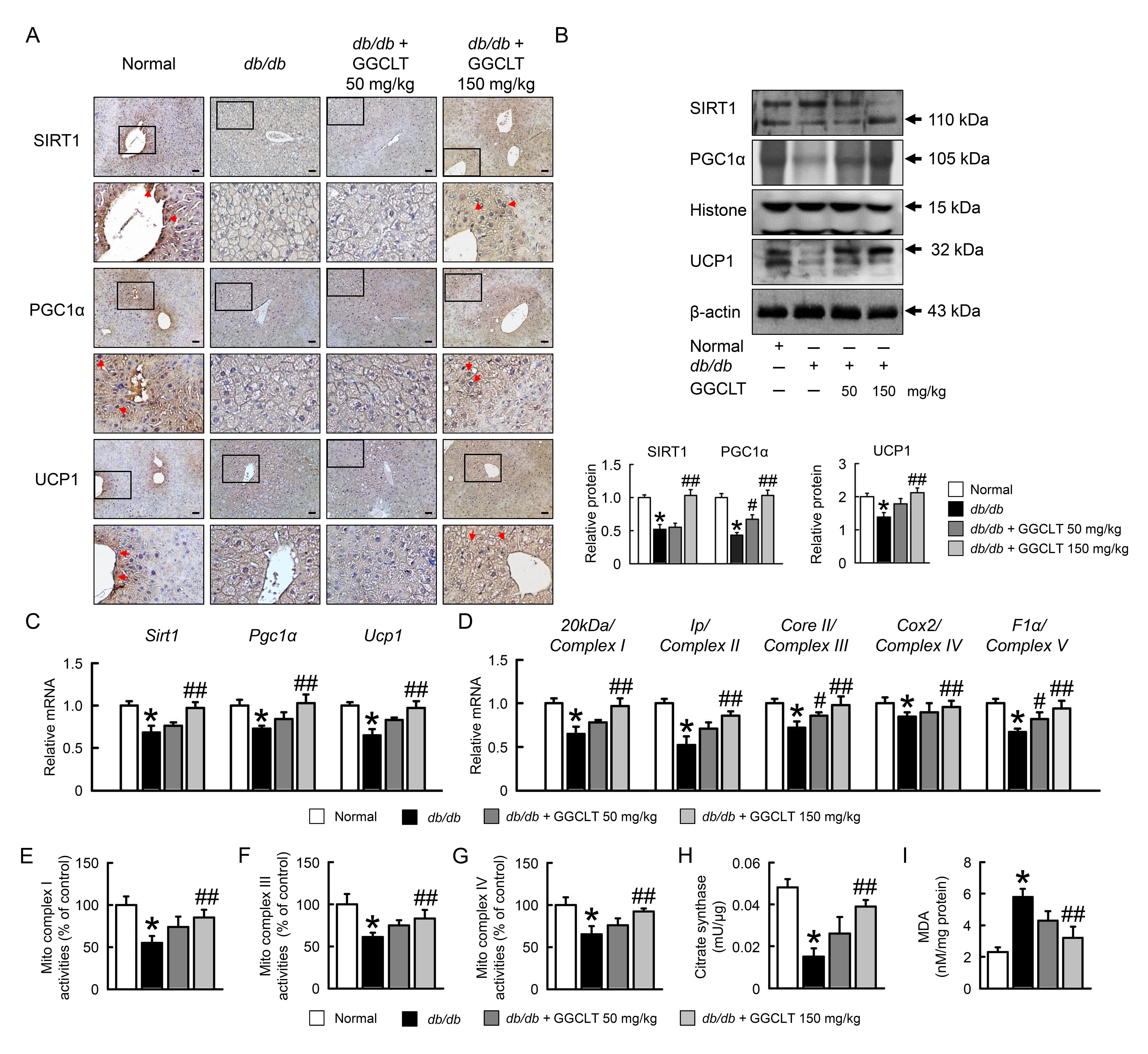 Fig. 5.
Fig. 5.GGCLT improved hepatic mitochondrial biogenesis in
db/db mice. Representative SIRT1, PGC1
Compared to normal mice, the protein expression levels of Parkin, LC3B, phospho (p)-p62, and mitofusin-1 (MNF1) in the liver of db/db mice were decreased but phospho-dynamin-related protein (p-Drp1) expression was increased (Fig. 6A,B). The GGCLT administration for 28 days, the opposite expression patterns were observed (Fig. 6A,B). Moreover, GGCLT effectively enhanced the expression of Parkin-dependent/independent mitophagy pathway, including Pink, calcium binding and coiled-coil domain 2 (Ndp52/Calcoco2), prohibitin 2 (Phb2), autophagy and beclin 1 regulator 1 (Ambra1), B-cell lymphoma 2 (Bcl2), BCL2 interacting protein 3 (Bnip3), FUN14 domain containing 1 (Fundc1) compared to db/db mice (Fig. 6C,D).
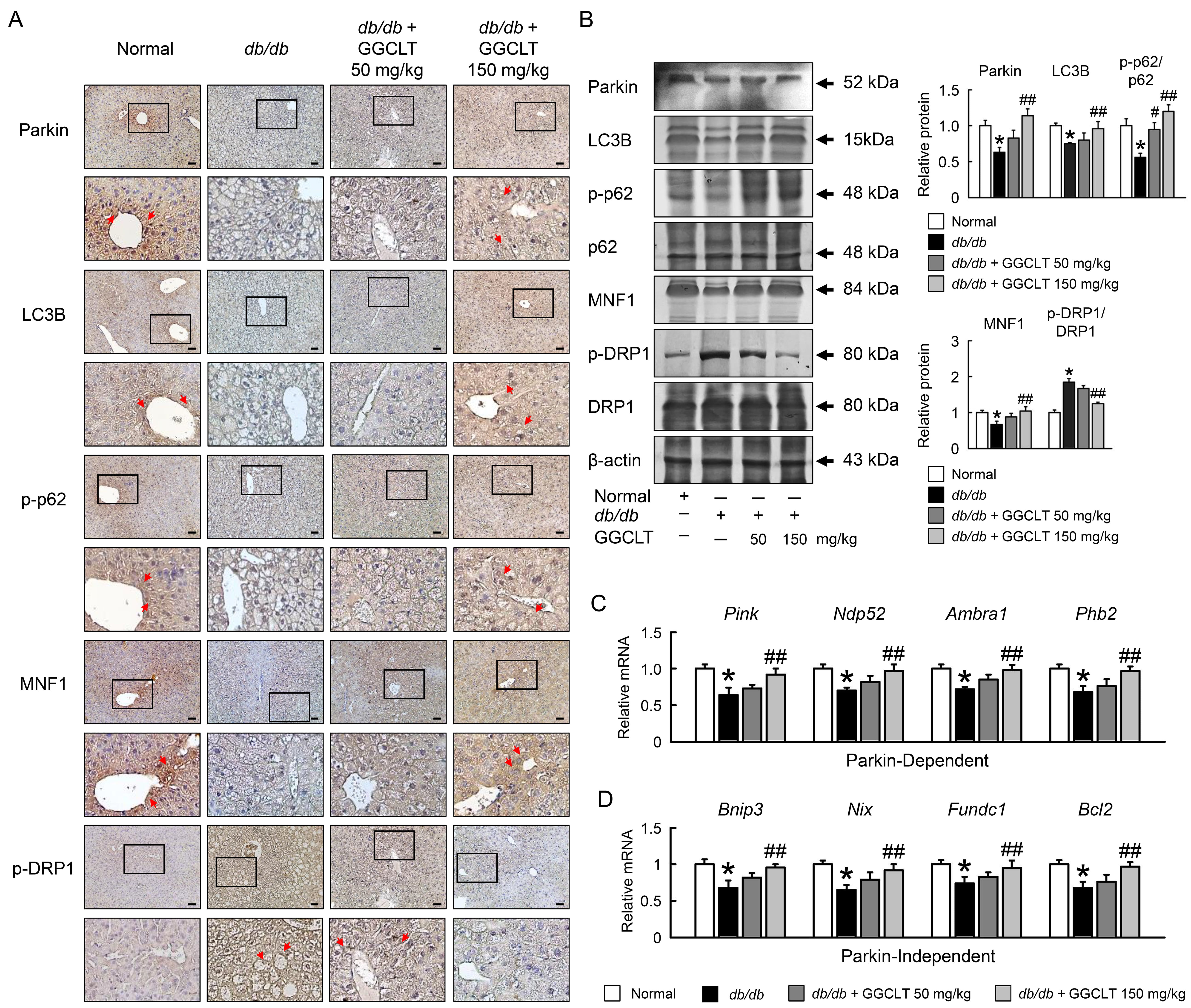 Fig. 6.
Fig. 6.GGCLT enhanced hepatic mitophagy in db/db mice.
Representative Parkin, LC3B, p-p62, MNF1, and p-DRP1 staining of liver. Red arrow
highlights the positive staining (A). Scale bar: 50
The shape of EWAT and adipocyte diameter size were large in db/db compared with normal mice (Fig. 7A,B), and GGCLT 150 mg/kg significant reduced EWAT size and adipocyte diameter size. GGCLT resulted in dramatically reduced the SREBP-1c and target genes, Fas, SCD1, Scd1, as well as fatty acid transporters (Cd36, Fatp, Cpt1 and Cpt2) mRNA expression in EWAT of db/db mice (Fig. 7C–F). Moreover, GGCLT significantly reduced hepatic lipogenesis and fatty acid transporters, suggesting that GGCLT regulated lipid homeostasis in db/db mice.
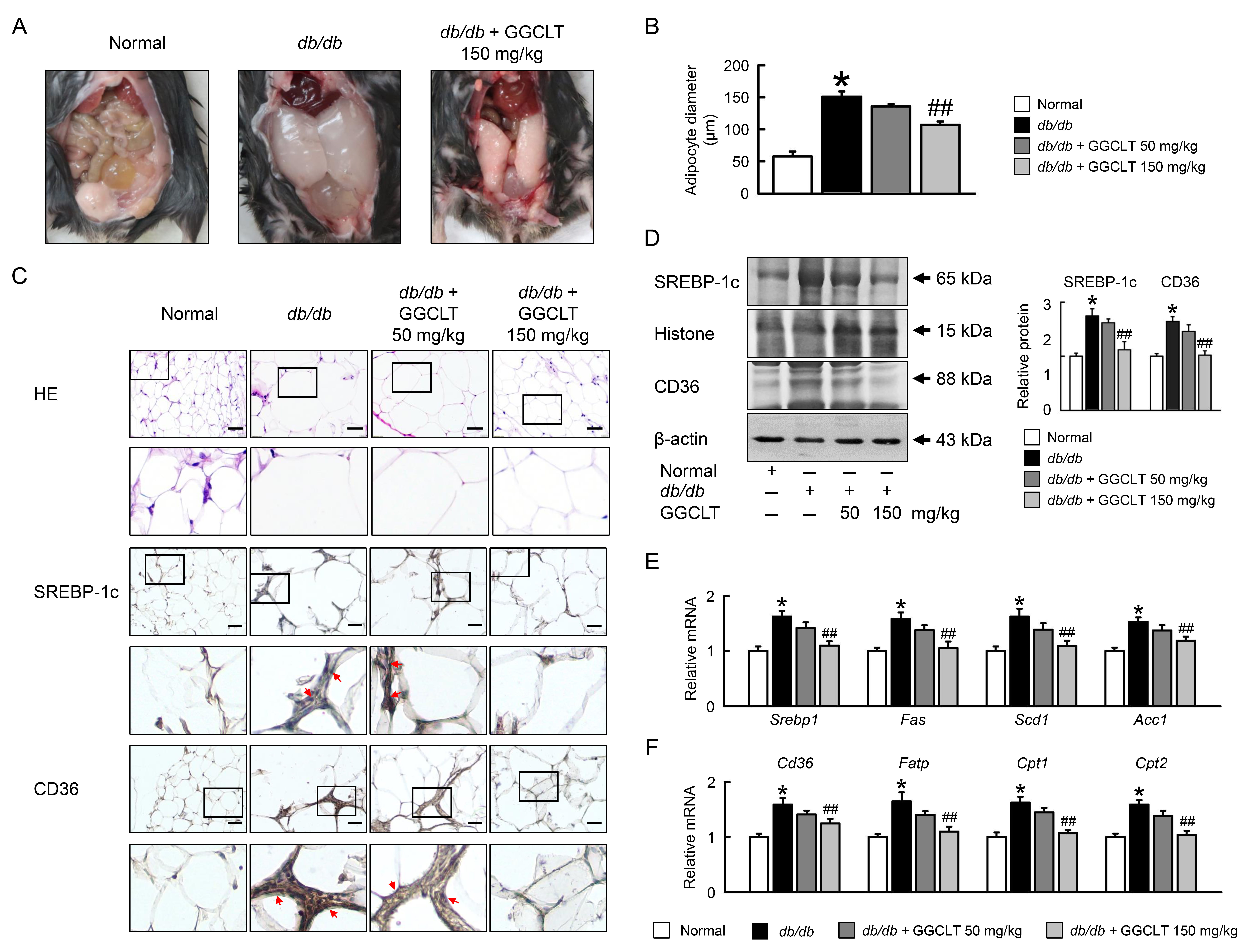 Fig. 7.
Fig. 7.GGCLT attenuated lipogenesis in EWAT of db/db mice.
Representative pictures of EWAT in mice (A). Adipocyte diameter (
db/db mice had higher Tnf
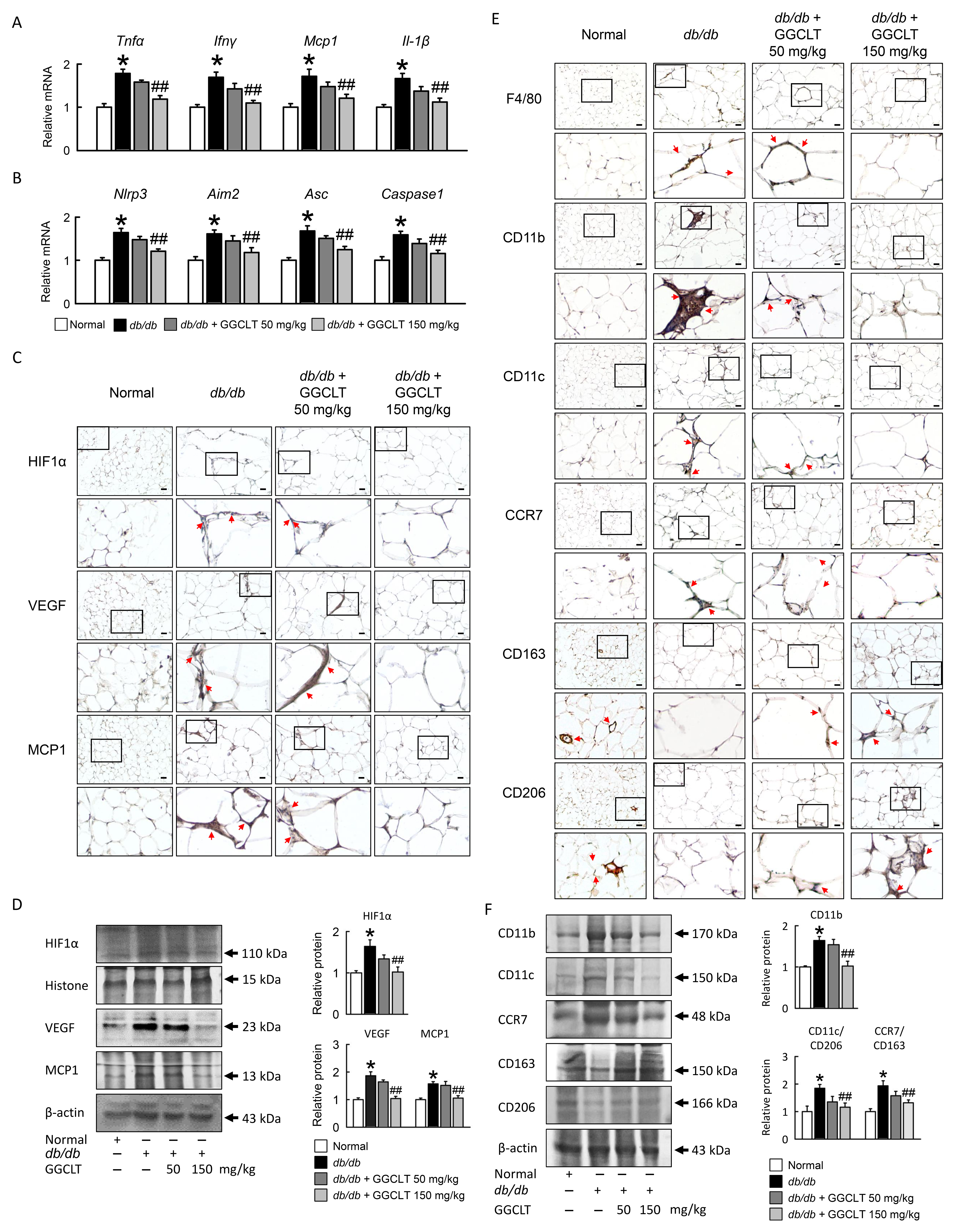 Fig. 8.
Fig. 8.GGCLT reduced inflammation and M1 polarization in EWAT of
db/db mice. Quantification of Tnf
The EWAT dramatically boosted F4/80-positive macrophage infiltration in the db/db mce. Macrophage infiltration was considerably lower in the GGCLT group than in the db/db mice (Fig. 8E). Macrophages were increased in EWAT of db/db mice were due to an increase in CD11b, CD11c, and CCR7 compared with normal mice. GGCLT reduced macrophage infiltration (F4/80, CD11b) and M1 macrophage markers (CD11c, CCR7) in EWAT of db/db mice (Fig. 8E,F). On the contrary, GGCLT 150 mg/kg enhanced M2 macrophage markers (CD163, CD206) protein levels in EWAT of db/db mice (Fig. 8E,F).
Consistent with the histological features, GGCLT enhance mitochondria biogenesis
and thermogenesis markers, SIRT1, PGC-1
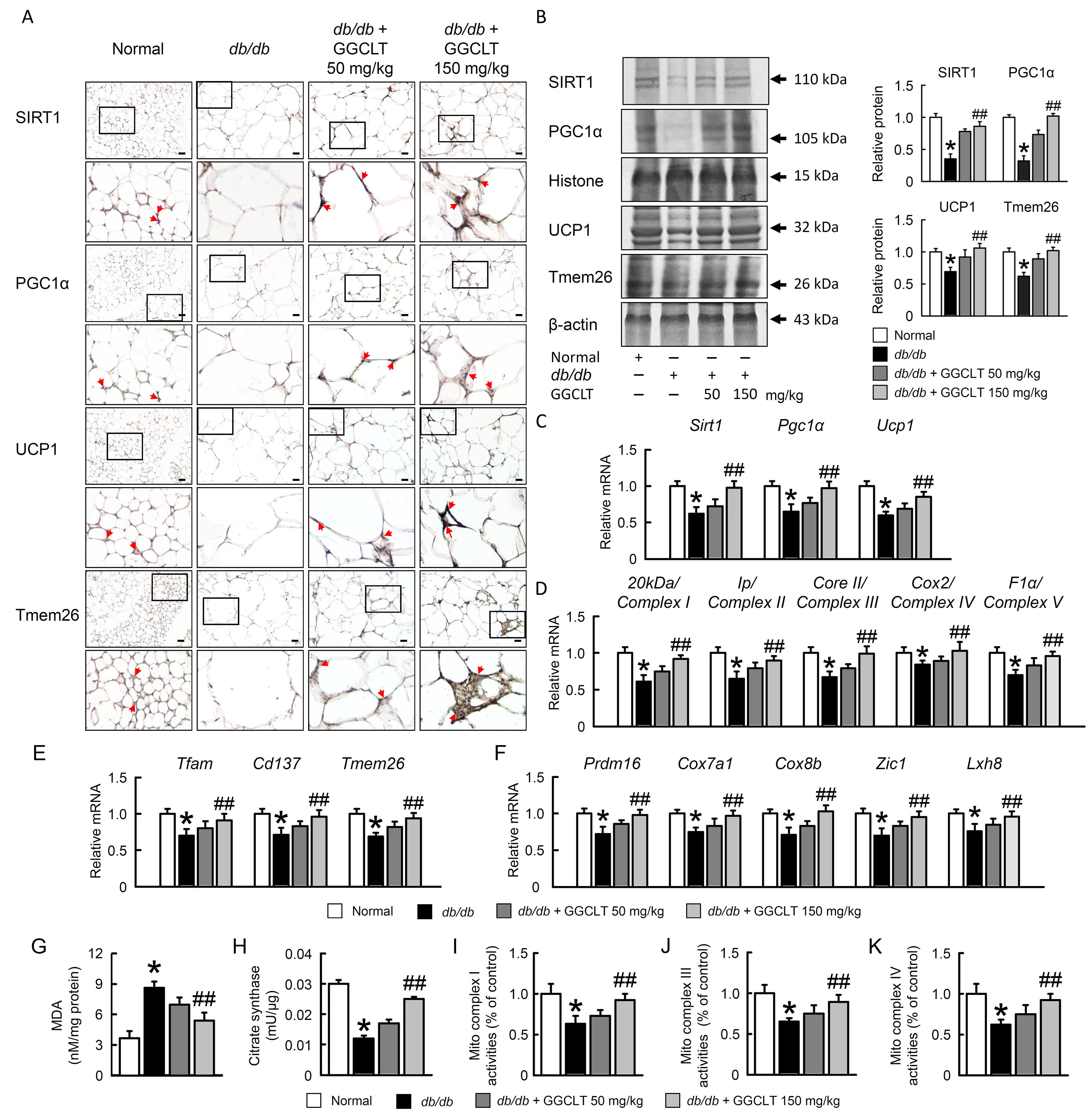 Fig. 9.
Fig. 9.GGCLT improved mitochondrial biogenesis in EWAT of
db/db mice. Representative SIRT1, PGC1
NAFLD remains a health concern due to its noticeable complexities in disease progression and complications. For the management of NAFLD, it is imperative to use the best available treatment. The traditional Chinese herbal formula (i.e., GGCLT) contains several plants, some of which contain more than one phytochemical component, and as a result, several plants work in conjunction symbiotically with each other in order to maximize the therapeutic benefits. The beneficial effects of GGCLT on the various medicinal purposes may be due to its wide spectrum of pharmacological properties such as vasodilation, cardioprotection, neuroprotection, antioxidant, anticancer, antiinflammation, alleviating pain, and attenuating insulin resistance [19, 20].
Anti-diabetic property of puerarin enhanced the performance of mitochondria in
muscle and promoted the oxidation of fatty acids, which thus prevented the
accumulation of intramyocellular lipids in diabetic rats [39]. Glycyrrhizin
(GL) significantly attenuated serum bile acid accumulation partially by restoring
inflammation-mediated hepatic farnesoid X receptor inhibition in
choline-deficient diet (MCD) induced nonalcoholic steatohepatitis (NASH) mice
[40]. The preclinical evidence suggested that berberine might be an effective and
promising drug for treating NAFLD/NASH [41]. Berberine can significantly improve
blood lipids and liver function in patients with NAFLD and has good advantage in
reducing blood glucose in patients with NAFLD [42]. Wogonine improves
hyperglycemia and dyslipidemia by activation of PPAR
In this condition, circulating free fatty acid (FFA) levels are elevated despite the increased concentration of insulin due to tissue resistance to its action [46]. In addition, FFA is among most common molecules suspected for lipotoxicity. Hepatic FAs derive from the adipose tissue lipolysis and are synthesized in the liver, or are released from lysosomes after autophagy. Consequently, surplus FFA that are not incorporated in more complex forms are lipotoxic to hepatocytes, they result in endoplasma reticular stress, mitochondrial dysfunction and oxidative stress, followed by the formation of reactive oxygen species (ROS). These processes activate the proinflammatory and profibrotic pathways, causing the disease to progress to NASH [4, 47]. In this study, lipotoxicity, inflammation, and IR are a contributing factor in the NAFLD suite. GGCLT mitigated inflammation and RI induced by lipotoxicity, which decreased lipolysis of adipocytes and improved lipotoxicity. In addition, GGCLT with each condition improve each other and shows a downward trend in the development of NAFLD in the presence of lipotoxicity, IR and inflammation form a vicious cycle.
Hepatic CD36 overexpression corresponds to a marked increase in uptake of
hepatic FFAs and a decrease in
Adipocytes hypertrophy and hyperplasia in obesity, allowing these cells to compartmentalize triglycerides. Furthermore, insulin resistant adipocytes produce an excess of free fatty acids, which may cause liver inflammation [53]. In the current model, the improved metabolic profile of GGCLT was associated with a lower influx in macrophage depolarization. The development of inflammation in the liver and EWAT in db/db mice was preceded by macrophage activation and proinflammatory gene expression. GGCLT treatment significantly reduced M1/M2 levels of macrophage and inflammation-associated genes in both liver and adipose tissue in this study. In our study, we found that EWAT inflammation is important in the development of insulin resistance and NAFLD in db/db mice. In this study, the gene expression analysis of EWAT revealed that genes related to adipose browning and macrophage depolarization were up-regulated, and mitochondria biogenesis correlated with hepatic contents. These findings suggest that GGCLT may have an anti-inflammatory effect on peripheral white adipose tissue browning and may also play an important role in the pathogenesis of NAFLD.
Previous research has shown that GGCLT reduces lipid accumulation through
primary hepatocytes, implying that activation of AMPK and PPAR in hepatocytes,
causing a reduction in lipid formation, contributes to GGCLT’s beneficial action
in ApoE
WAT browning was linked to a decline in parkin-mediated mitophagy, and parkin expression inhibited WAT browning [54]. One of the underlying causes of NAFLD is mitochondrial dysfunction caused by a failure of the quality control processes [55]. Extreme mitochondrial fission is a precursor to hepatocyte death that causes mitochondrial dysfunction. Autophagy targets depolarized mitochondria for degradation in response to mitochondrial damage (mitophagy) [56]. One of the underlying reasons of NAFLD is mitochondrial dysfunction caused by a breakdown of quality control procedures. As a result, mitochondria are active organelles that constantly fusion and fission. Abnormal mitochondrial fission is a precursor to hepatocyte apoptosis that causes mitochondrial malfunction.
GGCLT enhances
Crosstalk between the liver and adipose tissue is a crucial factor in the
emergence of NAFLD. Large amounts of physiologically active tissue may be found
in adipose tissue, which has a major impact on liver disease. Adipose tissue
rather than liver tissue itself may be the best target for treating hepatic
illness. In addition, activating mitochondria biogenesis has been demonstrated in
our study and the main treatment foundation for NAFLD, diabetes, and obesity is
thought to be GGCLT. We established the same mitochondria
HML, CHW, and TYL designed the research study. HML and CHW performed the research. HML, ZYC, and CHH provided help and advice on validation, formal analysis and investigation. HML, MCL, and CHW analyzed the data. HML, CHW, and TYL wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
All of the animal experiments were performed in accordance with the Animal Care and Use Committee of Chang Gung University Institutional Animal Care and Use Committee (CGU107-265).
Not applicable.
This research was funded by grants from the Ministry of Science and Technology, Taipei, Taiwan (MOST 102-2320-B182-015-MY3, 103-2320-B182-002-MY3, and 106-2320-B-182-005-MY3, 109-2320-B-182-023-MY3) and the Chang Gung Memorial Hospital, Linkou, Taiwan (CMRPD1B0261, CMRPD1B0262, CMRPD1D0351, CMRPD1D0352).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.









