Frontiers in Bioscience-Landmark (FBL) is published by IMR Press from Volume 26 Issue 5 (2021). Previous articles were published by another publisher on a subscription basis, and they are hosted by IMR Press on imrpress.com as a courtesy and upon agreement with Frontiers in Bioscience.
1 State Key Laboratory of Stem Cell and Reproductive Biology, Institute of Zoology, Chinese Academy of Sciences, Beijing, 100101, China
2 University of Chinese Academy of Sciences, Beijing, 100049, China
Abstract
Enhancer of zeste homolog 2 (EZH2), a catalytic component of polycomb repressive complex 2 (PRC2), epigenetically regulates chromatin structure and gene expression through trimethylation at histone H3K27 and recruitment of DNA methyltransferases for gene silencing. Despite extensive studies of the role of EZH2 in cancer progression and malignancy, increasing evidences suggest that EZH2 plays a critical role in stem cells renewal, maintenance, and differentiation into specific cell lineages. Here, we reviewed the update information regarding how EZH2 contributes to stem cell maintenance and cell lineage determination (including gonadogenesis, neurogenesis, myogenesis, osteogenesis, hematopoiesis, lymphopoiesis, adipogenesis, epidermal differentiation and hepatogenesis), and the regulation of EZH2 by phosphorylation and different signaling pathways.
Keywords
- Organogenesis
- PCR2
- H3K27me3
- Cell Fate Decision
- Embryonic Development
- Review
EZH2 is a bona fide histone methyltransferase, methylating histone 3 at lysine 27 (H3K27) leading to a selective suppression of gene transcription through a change in chromatin formation (1). It is an integral component of the polycomb repressive complex (PRC) 2. Polycomb proteins are an evolutionarily conserved family of chromatin regulators known best for their function in establishing and maintaining epigenetic memory during development. Besides EZH2, PRC2 consists of additional two core components: embryonic ectoderm development (EED), and suppressor of zeste 12 (SUZ12) (Figure 1).
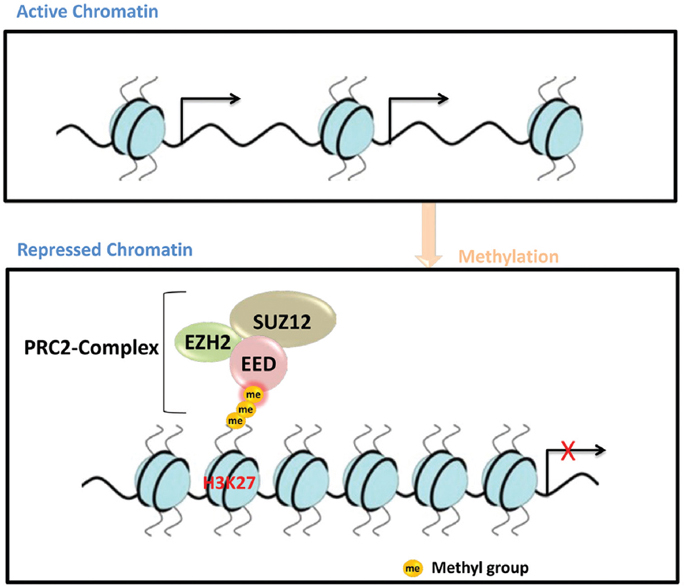 Figure 1
Figure 1Schematic diagram showing three core components of PRC2 complex and suppression of gene transcription via methylating H3K27.
Since EZH2 displays histone methyltransferase activity, epigenetic mechanisms are frequently invoked in explaining developmental and physiological consequences upon its loss or overexpression. The role of EZH2 in cancer progression and malignancy has been extensively studied in the last decade (2, 3). Moreover, growing evidences demonstrate that the PRC2 proteins are critical for the regulation of gene expressions in stem cells maintenance and lineage specification (4). Here, we focus on the recent progress regarding the roles of EZH2 in ontogenesis, pathways underlying cell fate decision and its regulatory mechanisms.
During ontogenesis, cell proliferation, differentiation, and morphogenesis have to be tightly coordinated. This process involves extensive changes in gene expression, which entails epigenetic mechanisms such as DNA methylation, nucleosome remodeling, and post-translational modifications of the histones (5). Epigenetic modifications mark the genome as regions that are either accessible or closed for the transcription machinery (Figure 1). Since these modifications can be inherited through cell divisions, epigenetic control is thought to maintain identity and behavior of a given cell type. Accordingly, fate switches from a proliferative multipotent progenitor cell to a non-dividing terminally differentiated cell type are accompanied and potentially controlled by changes in epigenetic information.
EZH2 is essential for the development of embryos and loss of EZH2 leads to the embryonic lethality in mice (6). EZH2 deficient embryos show impaired growth potential and defection in the establishment of embryonic stem cells and in the adhesion and differentiation of trophoblast cells into giant cells. EZH2 is expressed prior to embryo implantation in the early stages. Furthermore, EZH2 defection induces significant reduction in transcriptional level of the pluripotency related marker but the up-regulated expression of genes involved in germ layer differentiation (7), suggesting EZH2 is essential for the early development of preimplantation embryos through epigenetic modifications and regulation of apoptosis. EZH2 deficient embryos displayed impaired growth potential, preventing development of embryonic stem cells and the onset of differentiation of trophectoderm cells (7-9).
Spermatogenesis is the fundamental of male fertility and is under critical controls by a variety of epigenetic regulators. EZH2 plays a ‘non-classical’ role in the regulation of spermatogonial differentiation and apoptosis in mouse spermatogenesis (10). It might be important for spermatogonial stem cells self-renewal and maintenance of pluripotency (11) suggested by its high expression in spermatogonial stem cells. Our recent knockout study demonstrated that EZH2 deletion promotes spermatogonial differentiation and apoptosis (10). Mu W et al. suggest that only the combined loss of EZH1 and EZH2 caused a depletion of global H3K27me3 marks and meiotic arrest in spermatocytes (12). The distribution of EZH2 in germ cells is highly regulated with its localization predominantly restricted to round spermatids in the perinuclear acrosome region that could be a specialized epigenetic region where methylation of histones serves a role in the spermiogenic chromatin remodeling inferring that EZH2 might be a key effector of this event (13). Moreover, an expression profile of testicular biopsies shows that EZH2 is a germ cell-specific epigenetic gene upregulated before meiosis (14).
Female fertility and embryonic developmental competencies rely on a proper maturation of the oocyte, which includes meiosis and accurate alignment of chromosomes. EZH2 is uncovered to play an important role in the control of oocyte meiotic maturation, maintains the chromosome accurate alignment and oocyte euploidy. Ectopic expression of EZH2 results in meiotic maturation of oocyte during the MI stage, followed by chromosomal dislocation and aneuploidy (15). In the early mouse embryo development, EZH2 was detected as a maternally inherited protein in the oocytes (8, 9). Lack of maternal EZH2 resulted in severe growth retardation of neonates (8), suggesting the importance of EZH2 in the control of female reproduction.
Adult neurogenesis is a process that includes the proliferation of neural stem/progenitor cells, the differentiation of neurons, and the integration of new neurons into the existing neural circuitry (15). Neurogenesis of neural stem cells is promoted by inactivating PRC2 protein by knocking out EZH2 (16). Cortex-specific deletion of EZH2 at embryonic day 12 disrupts cortical neurogenesis and alters the timing of cortical development (17). EZH2 also plays a role in pushing the neural stem cells toward a neuronal lineage during the development. Overexpression of EZH2 inhibits astrocyte differentiation but promotes oligodendrocyte differentiation (18). EZH2 in neural stem cells is crucial for proliferative activity and maintenance of neural stemness (19). As a candidate gene, mutations in EZH2 cause Weaver’s syndrome, which is characterized by learning disabilities and general overgrowth (20, 21). Conditional knockout of EZH2 results in impairments in spatial learning and memory, contextual fear memory, and pattern separation (22).
Skeletal myogenesis provides a useful biological context within which to examine EZH2 regulation. The expression of EZH2 is negatively correlated with myogenesis. In undifferentiated myoblasts, EZH2 targets the promoter region of muscle specific genes to inhibit their expression (23). Induction of skeletal muscle cells differentiation leads to the dissociation of EZH2 from chromatin, the recruitment of developmental modulators and the activation of the miR-214 transcription. Then the miR-214 negatively regulates EZH2 through a negative feedback mechanism, resulting in accelerated skeletal muscle cells differentiation (24). Adult skeletal muscle represents a dynamic tissue in our body that possesses a remarkable capacity to regenerate in response to injury (25, 26). Recent Studies reported that the abnormal expression of EZH2 results in muscle disease. TNF treatment induces NF-κB expression by recruiting the EZH2 and DNMT3b to inhibit skeletal muscle cells differentiation by epigenetic transcriptional silencing (27).
There is increasing evidence that shows a reciprocal relationship between adipogenesis and osteogenesis. Besides other molecules, recent studies report that the switch between adipogenesis and osteogenesis can be epigenetically regulated by EZH2 as followings: The histone H3K27me3 methyltransferase activity of EZH2 promotes adipogenesis by disrupting the WNT/β-Catenin signaling through direct binding to the promoters of Wnt genes including WNT1, -6, -10a, and -10b to repress their expression (28). On the contrary, it has recently been reported that suppression of methyltransferase activity via phosphorylation of EZH2 by cyclin dependent kinase 1 (CDK1) results in hMSCs differentiation into osteoblasts (Figure 2) (29). It has also been demonstrated that dissociation of EZH2 from the promoter of myocyte enhancer factor-2 interacting transcriptional repressor (MITR) gene, also named as histone deacetylase 9c (HDAC9c), increases its expression and interaction with PPARγ2 in the nucleus to interrupt PPARγ2 activity and prevent adipogenesis, and thus, promoting hMSC osteogenic differentiation (30).
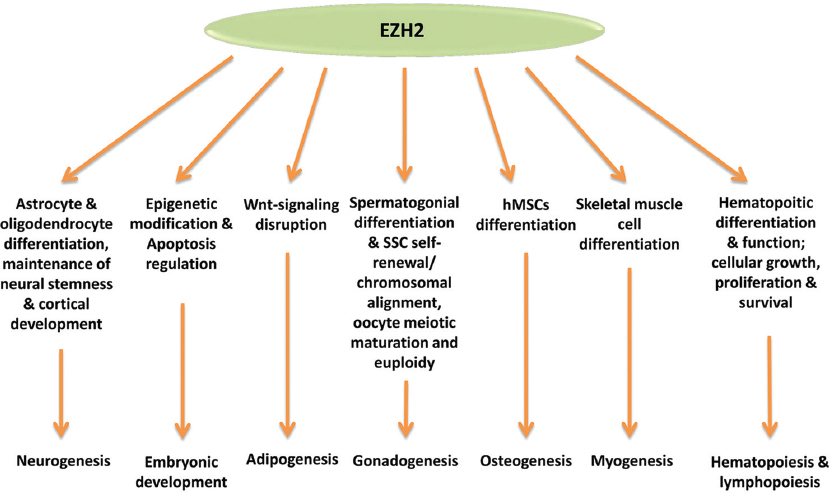 Figure 2
Figure 2Roles of EZH2 in cell lineage determination during development.
The skeletal defects described in heterozygous deletion of EZH2 in early mesenchyme tissue is similar to that seen in human patients who carry autosomal dominant mutations in EZH2 in a condition known as Weaver syndrome (21, 31). The bones of people with Weaver syndrome grow and develop more quickly, both before and after birth, and adults are generally taller, display clinodactyly, a larger head (macrocephaly), and craniosynostosis (21). Their general overgrowth and advanced bone age suggests that the skeletal stem cell pool undergoes premature cellular senescence and maturation, as we have shown for cultured human BMSCs after knockdown of EZH2 (31). Therefore, the EZH2 heterozygous mouse could provide a promising murine model for Weaver syndrome, which could allow us to understand the role of EZH2 during skeletal aging. In vivo, complete ablation of EZH2 in early limb bud mesenchymal cells resulted in limb and cranial patterning defects related to altered expression of the Hox and zinc finger genes; depression and accelerated expression of developmental, bone-related extracellular matrix proteins; and cyclin-dependent kinase inhibitors critical for post-proliferative cell growth arrest (33, 34). A recent study identified that EZH2 ablation resulted in trabecular bone patterning throughout the entire hindlimbs of newborn and 4-week old mice, whereas the trabeculae in heterozygous and wild-type controls were restricted to the metaphyseal regions of the bone (35). These observations suggest that ablation of EZH2 not only affects the size of the limbs but also affects the localization of trabeculae within the hindlimb bones and is highly dependent on the levels of EZH2 present during embryonic and postnatal development. Further studies investigating the mechanism of how EZH2 regulates endochondral ossification and intramembranous ossification will be critical for a greater understanding of how bone development is epigenetically regulated.
EZH2 plays a major role in normal hematopoiesis by promoting pluripotency maintenance and self-renewal of adult stem cells (36, 37). As EZH2, EED or SUZ12 loss-of-function mutations increase the activity of hematopoietic stem cells (HSCs) and progenitor cells, PRC2 could contribute to their negative control (38). EZH2 overexpression in HSCs prevents exhaustion of their long-term repopulating potential during serial transplantation (39-41). Therefore, EZH2 could prevent stem cell senescence (38) by regulating adult HSC differentiation, but not their self-renewal capacity (41-43). EZH2 also increases the pool of quiescent HSCs by supporting their proliferation and inhibiting apoptosis. However, EZH2 inactivation has opposite effects on apoptosis and proliferation in multipotent progenitors compared with long-term HSCs (42). In long-term HSCs, EZH2 is associated with hematopoietic differentiation and function, cellular growth and proliferation and survival (43). Moreover, depletion of EZH2 in highly proliferating fetal stem cells resulted in the failure in hematopoiesis and cardiogenesis even in the presence of EZH1 (41). It is still not clear how EZH1 and EZH2 coordinate to regulate H3K27 methylation and transcription during development.
EZH2 helps regulate lymphocyte production of HSCs. During lymphopoiesis, EZH2 is strongly expressed in proliferating cells, such as human germinal center B cells, cycling T and B lymphocytes and plasmablasts, suggesting an important role in cell cycle regulation and in lymphocyte division (44-46). Conditional knockout studies in mice have enabled dissection of the roles of EZH2 in lymphopoiesis. In the lymphoid lineages, EZH2 deletion resulted in impairment of B-lymphoid development (41, 43, 46), germinal center B-cell formation (47), and terminal differentiation of naive CD41 T-cells (48), but it was largely dispensable for T-lymphoid development in the thymus (49). Although several studies have shown that conditional inactivation of EZH2 appears to have minimal impact on lymphocyte function in adult mice, the role of EZH2 in fetal lymphopoiesis is less clear.
In addition to the described cell lineages above, EZH2 is involved in the regulation of epidermal differentiation and hepatogenesis. In epidermal progenitor cells, EZH2 is highly expressed to control proliferation by repressing CDK-inhibitors, which suppress cell cycle progression. During epidermal differentiation, decreased level of EZH2 reduces cell proliferation and removes histone H3K27me3 to recruit AP1 transcriptional factor to the structural genes that are required for epidermal differentiation (50). Knockdown of EZH2 significantly impairs the proliferative and self-renewal capability of murine hepatic progenitor cells and promotes the differentiation and terminal maturation of hepatocytes as measured by detection of the metabolic enzyme genes. These results demonstrate that EZH2 is critical and essential in proliferative and self-renewal capacity of hepatic progenitor cells and prevents them from differentiation (51).
STAT3 signaling is considered to play important roles in various stem cells (52, 53). A series of reports showed that histone methyl transferases such as SET7/9 can regulate signaling pathways through direct methylation of p53, NF-κB, and STAT3 (54-57), raising the possibility that EZH2 might have such a property. Previous study has reported that the overexpression of EZH2 induces STAT3 methylation and promoted STAT3 activity (Figure 3); and AKT signaling is required for the EZH2-STAT3 interaction and STAT3 methylation (58). Inhibition of AKT signaling decreases STAT3 activity via EZH2 phosphorylation, implicating that PI3K/AKT signaling is an upstream mediator of the EZH2-STAT3 interaction (58). Activation of the PI3K/AKT pathway in glioma is associated with adverse clinical outcome and various PI3K/AKT inhibitors are being tested in clinical trials (59-61). Therefore, it would be interesting to examine EZH2 expression as a biomarker to predict therapeutic responses to PI3K/AKT targeted therapies.
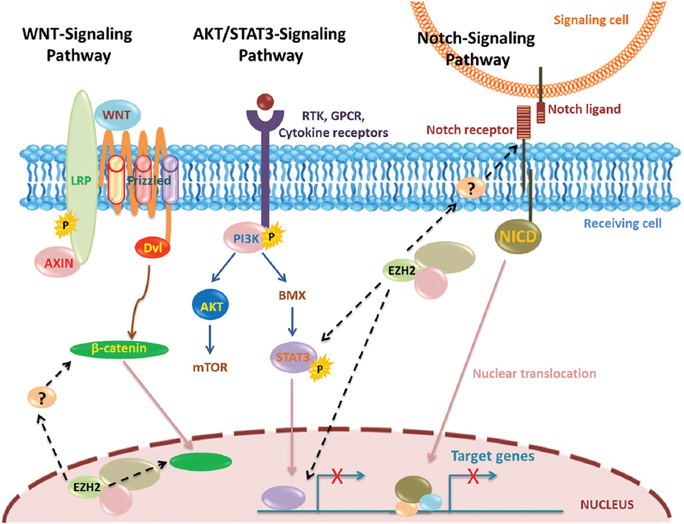 Figure 3
Figure 3Involvement of EZH2 in suppressing transcription via different signaling pathways.
NOTCH receptors and ligands are single-pass transmembrane proteins that play an important role in cell fate decisions during embryonic and postnatal development (62, 63). NOTCH receptors are overexpressed in many kinds of cancers (64). Previously it was shown that NOTCH1 was decreased when EZH2 was suppressed in cancers (65). Recent study found that EZH2 regulated NOTCH1 expression by directly interacting with the NOTCH1 promoter; evidently supporting the existence of EZH2-NOTCH1 signaling pathway in the malignant gliomas (66). EZH2 acts as an activator of the NOTCH1 promoter and signaling to expand the stem cell pool, leading to accelerated cancer initiation and growth. Interestingly this function is independent of EZH2 histone methyltransferase activity and of its PRC2-binding partners, paving the way for novel therapeutic strategies (67).
The WNT signaling pathways, activated by at least nineteen WNT isoforms, are considered to play very important roles in many cellular processes, such as proliferation, differentiation and apoptosis (68, 69). Additionally, the WNT pathway also regulates critical aspects during embryonic development (68). The WNT signaling pathways include four branches: the WNT/β-Catenin (canonical) pathway, the planar cell polarity (PCP) pathway, the WNT/Ca2+ pathway and the protein kinase A pathway (68-70). Among these, WNT/β-Catenin signaling pathway has been most thoroughly studied. This signaling pathway is activated after the WNT proteins (including WNT3a) binding to the Frizzleds (FZS)/low-density lipoprotein receptor-related protein 5/6 (LRP5/6) complex on the cell membrane. Then the activation of the pathway leads to the stabilization and accumulation of β-Catenin in the cytoplasm, which will translocate into the nucleus and stimulate the transcription of downstream target genes (Figure 3). Several studies reported that EZH2 contributed to constitutive activation of WNT/β-Catenin signaling pathway directly (71-73) and/or indirectly (74). Previously down regulation of EZH2 inhibited the expression of WNT3a and β-Catenin significantly, and it markedly promoted the expression of GSK-3β (glycogen synthase kinase-3β), an inhibitor of the WNT/β-Catenin pathway (75).
EZH2 and WNTs are both important regulators of development. A previous study demonstrated a direct, functional link between these two types of developmental regulators and show that EZH2 and its H3K27 methyltransferase activity are required for adipogenesis (28). Further, EZH2 appears to regulate the size of the developing midbrain both by direct repression of cell cycle inhibitors and, indirectly, by sustaining canonical WNT/β-Catenin signaling pathway (76). Recent evidence has reported that EZH2 inhibits the osteogenic differentiation of MSCs by repressing the WNT gene, which leads to the inactivation of WNT/β-Catenin signaling pathway (28). EZH2 is required to initiate skeletal muscle differentiation by modulating WNT signaling through the direct repression of Wnt antagonists (77).
Genome-wide profiling studies revealed the enrichment of H3K27 methyltransferase EZH2 and associated H3K27me3 on WNT genes (78-80). However, it was unclear from these studies whether EZH2 represses WNT expression, as knockdown of EZH2 in human embryonic fibroblasts failed to increase WNT expression, which could be due to insufficient knockdown and/or the functional redundancy between EZH1 and EZH2 (80, 81). Deletion of EZH2 in embryonic stem cells led to depression of many EZH2 target genes. However, its effect on WNT expression remains unclear (78, 81-84).
The PRC2 proteins are known to epigenetically regulate chromatin structure and stepwise modulate gene expression profiles to maintain stem cell property or enable stem cell differentiation into specific cell lineages. In this article, we have described the update information regarding EZH2, a catalytic component of PRC2, in pluri- or multi-potency of stem cells and cell lineage commitment by targeting specific gene sets, as well as the regulation of EZH2. Although much has been known from genome-wide mapping the EZH2-targeted genes in different types of cell lines, it will be more meaningful to extend these studies to homogeneous cell populations from specific developmental stages and tissues. Additionally, little is known about the EZH2-interacting proteins between undifferentiated and differentiated stem cells and in specific cell lineages. Proteomics approaches will be useful to genome-widely identify these EZH2-interacting proteins, which will help further identify key modulators of EZH2 in stem cell maintenance and lineage specification. More understanding of the roles of EZH2 in specific cell lineage determination will be beneficial for application of stem cells as therapeutics for injuries or diseases.
The authors delightedly acknowledge Young Elite Scientists Sponsorship Program by CAST (grant no. YESS20160118); National Natural Science Foundation of China (grant no. 31471352 and 31501198); CAS-TWAS President’s Fellowship program for PhD.



