Streptococcus pneumoniae is the most common pathogenic bacterium of pneumonia and can cause death. Here, we examined the impact of Baicalein on mouse mortality caused by Streptococcus pneumoniae infection. Baicalein protected mice from Streptococcus pneumoniae-induced deaths. Baicalein reduced alveolocapillary membrane permeability, lung infiltration of inflammatory cells, inflammatory cytokine production and pulmonary cell apoptosis. These evidences show that Baicalein is useful in the therapy of a severe form of pneumonia that can cause deaths.
Pneumonia, an inflammatory disease of the lung, can be caused by the infection of multifarious pathogens such as fungi, viruses and bacteria (1). Pneumonia is a major health problem in the elderly with massive socioeconomic costs and high morbidity (2, 3). Streptococcus pneumoniae is a Gram-positive bacterium and the most common pathogen of pneumonia, accounting for approximately 27% of all pneumonia cases globally (3). Current therapeutic strategies such as antimicrobial regimens, antibiotics, and adjunctive therapies are difficult to achieve the accurate diagnosis and appropriate therapy for pneumonia due to various factors such as antimicrobial resistance, immunosuppression and polypharmacy (4, 5). Considering the challenges and massive disease burdens in the management of pneumonia, it is imperative to seek for alternative approaches to alleviate pneumonia-induced tissue damages and deaths.
Baicalein (5,6,7-trihydroxyflavone) is a nature flavonoid isolated from the roots of Oroxylum Indicum and Scutellaria Baicalensis Georgi, and it has been proposed to treat cerebral ischemia, cancers, and inflammatory diseases (6-8). For example, Baicalein has been found to facilitate cell apoptosis by upregulating DEPP (decidual protein induced by progesterone) and Gadd45α (growth arrest and DNA damage-inducible 45α) as well as activating MAPK pathway in colon cancer (9). Baicalein was also found to ameliorate 2, 4, 6-trinitrobenzene sulfonic acid (TNBS)-induced colitis by reducing myeloperoxidase (MPO) activity and the expression of pro-inflammatory cytokines such as interleukin (IL)-1β and tumor necrosis factor (TNF)-α in mice (10).
In the present study, we found that Baicalein treatment reduced the mortality of Streptococcus pneumoniae-infected mice. Baicalein alleviated Streptococcus pneumoniae-induced pneumonia by inhibiting the production of pro-inflammatory cytokine production and infiltration of pro-inflammatory cells in the mouse lung.
Male C57BL/6J mice (8-10 weeks old) were purchased from Henan Experimental Animal Center (Zhengzhou, China). All animal experiments were performed with the approval of Experiment Animal Management and Ethics Committee of Xinxiang Medical University and the National Guide for the Care and Use of Laboratory Animals. Streptococcus pneumoniae (ATCC49619, American Type Culture Collection, Rockville, MD, USA) and Baicalein (Sigma-Aldrich, St. Louis, MO, USA) was suspended in phosphate-buffered saline (PBS). To explore the effects of Baicalein on Streptococcus pneumoniae-induced pneumonia, mice were intranasally-administered with Streptococcus pneumoniae (1×107 colony forming unit (CFU) per mouse) and intraperitoneally injected with PBS or Baicalein (25, 50, or 100 mg/kg body weight) at the indicated time points post-infection. Each experimental group included 20 mice.
Inflammation in the mouse lung was evaluated as previously described (11). Briefly, mouse lung was homogenized in Hank’s buffered salt solution. The slurry was then centrifuged, and TNF-α, IL-6, interferon (IFN)-γ, IL-1β, C-X-C motif chemokine ligand (CXCL)1 or CXCL2 levels in the supernatant were assessed by ELISA using corresponding commercial kits (R&D systems, Minneapolis, MN, USA).
Mouse broncho-alveolar lavage fluid (BALF) was collected by intratracheal lavage as previously described (12). Total protein concentration in each BALF sample was determined using Pierce™ BCA Protein Assay Kit (Thermo Fisher Scientific, Rockford, IL, USA). Cells in the BALF samples were collected by centrifugation and resuspended in PBS. Total cell count was determined using a hematology counter (Sysmex SF 3000, Sysmex Co., Kobe, Japan), and the abundance of neutrophils or macrophages in the BALF sample was assessed by flow cytometry. Neutrophils were identified by the double staining of PE/CY7-GR1 (RB6-865, eBioscience, San Diego, CA, USA) and PE-CD11b (M1/70, eBioscience) antibodies, while macrophages were determined using allophycocyanin (APC)-F4/80 (eBioscience) antibody. MPO activity was determined using a Myeloperoxidase Colorimetric Activity Assay Kit (Sigma-Aldrich) following the instructions of manufacturer.
Mouse lung tissue samples were immersed in optimal cutting temperature compound and snap-froze in liquid nitrogen. For histopathological examination, the samples were sliced into 4 micrometer sections and stained with hematoxylin and eosin (H&E) as previously described (13). The extent of lung damage was evaluated using a histological scoring scheme as previously described (13). Cell apoptosis in the lung tissue samples was assessed by TUNEL assay using an in situ Cell Death Detection Kit (Sigma-Aldrich). TUNEL positive cell percentage was counted in 10 random fields.
Data analysis was performed using SPSS 21.0 software (SPSS Inc., Chicago, IL, USA) with the results expressing as mean ± standard deviation. For significance test, Dunnett’s test was performed in Figure 1, and Student’s t-test was performed in the rest of the figures. A difference was considered statistically significant when p < 0.05.
 Figure 1
Figure 1Baicalein protected mice from Streptococcus pneumoniae-induced deaths. (A) Brief outline of our experimental design. Mice were intranasally infected with about 1×107 CFU Streptococcus pneumoniae and intraperitoneally injected with PBS (vehicle) or Baicalein (Bai, 25, 50, 100 mg/kg) at 12 h intervals for a total of 48 h. (B) The mortality of mice was monitored every 12 h. *p < 0.05.
To investigate the potential protective effect of Baicalein on pneumonia death, mice were infected with Streptococcus pneumoniae and administered with PBS or Baicalein at the indicated time points (Figure 1A). Mortality of Baicalein- or vehicle-treated mice were then monitored at 12-, 24-, 36- or 48-hours post-infection. We did not observe any mouse death in the initial 24 h upon the infection of Streptococcus pneumoniae (Data not presented). At 36- or 48-h post-infection, mortality of vehicle-treated mice was over 20% and over 60%, respectively, which was significantly decreased by Baicalein treatment in a dose dependent manner (Figure 1B). These data suggested that Baicalein could protect mice from Streptococcus pneumoniae-induced deaths.
Inflammation is implicated in pneumonia-related tissue damage and death. To evaluate the influence of Baicalein treatment on the inflammatory responses in Streptococcus pneumoniae-infected mice, we analyzed the pro-inflammatory cytokine levels in mouse lung. Our ELISA results showed that treatment with Baicalein markedly reduced the levels of TNF-α, IL-6, IL-1β and IFN-γ in the mouse lung at 36 h and 48 h after Streptococcus pneumoniae infection (Figure 2A-2C). We also found that Baicalein treatment resulted in a significant reduction in CXCL1 and CXCL2 levels in mouse lung at 36 h and 48 h post-infection (Figure 2E and 2F).
 Figure 2
Figure 2Baicalein inhibited inflammatory cytokine production during acute Streptococcus pneumoniae infection. (A-F) Mice were intranasally infected with about 1×107 CFU Streptococcus pneumoniae and intraperitoneally injected with PBS or Baicalein (100 mg/kg) for 36 h or 48 h. Then, ELISA assay was performed to measure TNF-α, IL-6, IFN-γ, IL-1β, CXCL1 or CXCL2 levels in the supernatants of mouse lung homogenates. *p < 0.05.
Previous studies showed that pro-inflammatory cytokines such as TNF-α, IL-6, and IL-1β could be secreted by macrophages (14, 15), while IFN-γ, CXCL1 and CXCL2 could be produced by neutrophils (16-18). We next investigated the effects of Baicalein treatment on the abundance of macrophages and neutrophils in the lung of Streptococcus pneumoniae-infected mice. As displayed in Figure 3A-3C, the number of total cells, macrophages and neutrophils was significantly reduced in the BALF of Streptococcus pneumoniae-infected mice by baicalein treatment. We also found that the activity of MPO (a marker of neutrophil activation) in the Streptococcus pneumoniae-infected mouse lung was reduced by Baicalein treatment (Figure 3D). These data suggested that Baicalein inhibited the inflammatory responses in Streptococcus pneumoniae-infected mice, and reduced the redisposition of macrophages and neutrophils to the lung.
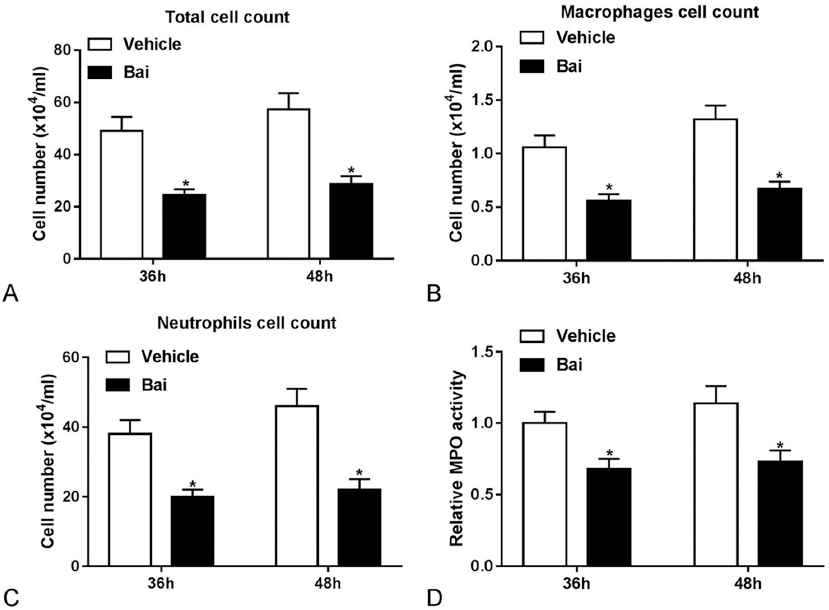 Figure 3
Figure 3Baicalein inhibited the accumulation of inflammatory cells and reduced MPO activity in Streptococcus pneumoniae-infected mouse lung. (A-D) Mice were intranasally infected with about 1×107 CFU Streptococcus pneumoniae and intraperitoneally injected with PBS or Baicalein (100 mg/kg) for 36 h or 48 h, followed by the determination of total cell number, macrophage number, neutrophil number and MPO activity. *p < 0.05.
Next, histopathological analysis was performed to further explore the effects of Baicalein treatment on Streptococcus pneumoniae-induced lung injury. As displayed in Figure 4A-4C, the introduction of Baicalein resulted in the disintegration of lung parenchyma (Figure 4A), the accumulation of alveolar architecture (Figure 4A), the reduction of histological scores (Figure 4B) and the decrease of infiltrative cell number (Figure 4C) in the lung of mice infected with Streptococcus pneumoniae. Moreover, total protein levels in the BALF were measured to evaluate the influence of Baicalein treatment on alveolocapillary membrane permeability (19), and the results suggested that Baicalein treatment reduced the release of total proteins in the BALF of mice infected with Streptococcus pneumoniae, indicating that Baicalein could attenuate the increase in alveolocapillary membrane permeability caused by Streptococcus pneumoniae infection (Figure 4D). The TUNEL assay results showed that Baicalein treatment significantly reduced cell apoptosis in the Streptococcus pneumoniae-infected mouse lung (Figure 4E and 4F). These data showed that Baicalein treatment protected mice from Streptococcus pneumoniae-induced lung damage.
 Figure 4
Figure 4Baicalein protected mice from Streptococcus pneumoniae-induced lung damage. (A-F) Mice were intranasally infected with about 1×107 CFU Streptococcus pneumoniae and intraperitoneally injected with PBS or Baicalein (100 mg/kg) for 48 h. (A) The representative photographs of H&E staining of lung tissues. (B) Histology score of H&E staining results. (C) Analysis of infiltrative cell number in the H&E staining results. (D) Protein concentration was determined in the BALF of treated mice. (E) TUNEL apoptosis detection of lung tissue samples from PBS and baicalein-treated groups. (F) The quantification of TUNEL positive cell percentage. *p < 0.05.
Streptococcus pneumoniae, also named pneumococcus, is normally colonized in the upper airways of the host (20). Streptococcus pneumoniae can transmit, invade and colonize into other sites such as lung and bloodstream by evading or utilizing the host’s immunological and inflammatory responses, causing multiple diseases such as septicemia, meningitis and pneumonia (20, 21). Moreover, the resistance of Streptococcus pneumoniae to multiple antibiotics such as macrolides and β-lactams is increasingly becoming a severe challenge in the management of Streptococcus pneumoniae-related diseases (22, 23). Streptococcus pneumoniae pneumonia is a global devastating disease which mainly affects children and the elderly (24, 25). Furthermore, pneumonia induced by bacteria such as Streptococcus pneumoniae is a common fatal complication during influenza pandemics such as the 2009 influenza pandemic (26, 27). Although great advances have been made in the management of pneumonia, it is still the leading cause of infectious deaths worldwide (3, 5, 28).
Baicalein has been shown to possess multiple biological activities such as anti-oxidation, anti-infection and anti-inflammation (20, 21). Our study aimed to investigate whether Baicalein could be used to treat Streptococcus pneumoniae-induced pneumonia, or whether it could be used to ameliorate pneumonia-induced lung damage. We found that Baicalein at the concentration of 50 or 100 mg/kg body weight could protect mice from Streptococcus pneumoniae infection-induced deaths at 36 h and 48 h after infection. The dysregulation of inflammatory responses plays vital roles in the pathogenesis of pneumonia (29, 30). Macrophages and neutrophils have been identified as vital mediators of inflammatory responses against Streptococcus pneumoniae (31-33). Also, previous studies showed that macrophages stimulated with Streptococcus pneumoniae could secret some pro-inflammatory cytokines such as TNF-α, IL-6, IL-1α, and IL-1β (14, 15). Moreover, IFN-γ could be generated by neutrophils during Streptococcus pneumoniae-induced pneumonia (18). Additionally, neutrophils-derived chemokines, including CXCL1 and CXCL2, were strikingly increased in mice following the infection of Streptococcus pneumoniae (16, 17). Hence, the effects of Baicalein on the production of these cytokines and chemokines were further tested in the Streptococcus pneumoniae-infected mouse lung. Our results showed that Baicalein treatment reduced the level of these pro-inflammatory cytokines and chemokines elevated by Streptococcus pneumoniae-infection in the mouse lung. Also, the number of total cells, macrophages and neutrophils was notably down-regulated in BALF of Streptococcus pneumoniae-infected mice after Baicalein treatment. Moreover, Baicalein treatment reduced MPO activity in lung of Streptococcus pneumoniae-infected mice, indicating the downregulation of overall neutrophil activity. Taken together, our data indicated that inflammatory cells, cytokines and chemokines might cause lung tissue damage during Streptococcus pneumoniae-infection, which could be alleviated by Baicalein treatment. In consistent with our results, Burnley et al. showed that corticotropin-released hormone could protect mice against Streptococcus pneumoniae-induced pneumonia by inhibiting the activation of neutrophils and CXCL1 production (34). The introduction of TNF-α improved mouse susceptibility to Streptococcus pneumoniae-induced pneumonia (35). Also, Yende et al. showed that elderly patients with higher TNF-α and IL-6 levels had an increased risk of pneumonia (36). However, some studies pointed out that these inflammatory cells and cytokines could enhance host defense against Streptococcus pneumoniae. For instance, macrophages-derived cytokines such as IL-1α, IL-1β, TNF-α and IL-6 have been reported as essential mediators in protecting host from systemic Streptococcus pneumoniae infection (35, 37-39). Also, IFN-γ contributed to enhance host defense against Streptococcus pneumoniae (18). Additionally, our finding further revealed that the introduction of Baicalein protected mice from Streptococcus pneumoniae-induced lung damage.
Collectively, our data showed that Baicalein protected mice from Streptococcus pneumoniae-induced lung damage and deaths by the regulation of inflammatory cells and cytokines, providing a potential therapeutic drug for Streptococcus pneumoniae pneumonia.
This study is supported by grants from National Natural Science Foundation of China (NSFC) (No.81570624 and 81770721) and Science and technology innovation talent program of Xinxiang (No:CXRC17002).
