1 Postgraduate Program in Genetic Resources and Productivity-Genetics, Colegio de Postgraduados, Texcoco, Mex. 56264, Mexico
2 Mexico Valley Experimental Field, National Institute of Forestry, Agricultural and Livestock Research, Coatlinchán, Texcoco, Mex. 56250, Mexico
3 International Maize and Wheat Improvement Center, Texcoco, Mex. 56237, Mexico
†These authors contributed equally.
Abstract
Leaf rust caused by Puccinia triticina Erikss. is a widely distributed wheat (Triticum aestivum L.) disease. Using wild relatives, such as Triticum spelta L., as a source of desirable traits represents a good strategy for developing wheat varieties, as T. spelta L. has shown tolerance to various types of biotic and abiotic stresses. This study aimed to determine the genetic basis of resistance to leaf rust in the accession Triticum spelta 109 (PI 355580).
The resistant genotype T. spelta 109 was crossed with the bread wheat variety Roelfs F2007, and 135 F3 families were generated to analyze the genetics of resistance to the MBJ/SP leaf rust race. The families were classified into three groups: (i) homozygous-resistant; (ii) homozygous-susceptible; (iii) segregating. A χ2 test was performed to compare whether the expected and observed segregation ratios fit and to determine the number of genes involved in the resistance of T. spelta 109.
The seedling tests in the F1 generation showed susceptibility in all plants, indicating that the resistance is conferred by a recessive gene(s). The results of the χ2 test revealed that the observed segregation ratios of the F3 families followed the expected values, suggesting that a recessive gene confers the leaf rust resistance present in T. spelta 109. According to our results and the reported recessive genes identified among the T. spelta accessions, the identified recessive gene in T. spelta 109 (PI355580) is different and most likely a novel leaf rust resistance gene.
The genetic resistance to leaf rust of T. spelta 109 (PI 355580) is conferred by a single recessive gene. The importance and usefulness of searching for rust resistance genes from different sources and incorporating them into the genetic base of wheat breeding programs to provide diversity is confirmed.
Keywords
- Puccinia triticina Erikss.
- Triticum spelta
- genetic resistance
- recessive gene
Leaf rust, caused by the fungus Puccinia triticina Erikss. is a widely distributed wheat disease that, when present, can cause significant yield losses [1]. This fungus, due to its biotrophic nature, can produce urediniospores that, disseminated by the wind, infect plants hundreds of kilometers away, often resulting in epidemics on a continental scale [2]. Depending on the severity of the damage to the foliage, the most impactful effect of the disease is reflected in the drastic decrease in the number of grains per head and their weight [2].
Genetic resistance represents the most widely used method to reduce economic and ecological losses caused by leaf rust; therefore, most wheat breeding programs worldwide employ this strategy [3]. Wheat originated in the Middle East and evolved through several natural crosses between related wild species [4]. Subsequently, the use of species related to wheat has made it possible to replicate this process of evolution in different ways [5], becoming, in recent years, one of the most successful ways of sourcing and taking advantage of the genetic diversity of related and wild species, which provide new genes to the wheat populations presently used in food and agriculture to promote high production, quality, disease resistance, and the necessary safety requirements [6].
Wheat resistance to leaf rust can be classified into two types: (i) race-specific and (ii) partial or slow-developing [7]. Race-specific resistance genes are effective at all stages of plant development. However, race-specific genes expressed at the adult plant stage and poorly expressed in seedlings [7] have also been characterized. Conversely, partial resistance is optimally expressed in adult plants, as seedlings may be susceptible [7]. Resistance to rusts commonly presented by wheat genotypes is known as race-specific; however, the pathogen can overcome such resistance due to continuous evolution toward new physiological races, which is why the constant search for new sources of resistance remains important.
More than 80 leaf rust resistance genes have been catalogued [8], of which almost 50% were derived from wild or cultivated species closely related to T. aestivum L. [9]. Compared to domesticated species, wild relatives retain high levels of genetic diversity [10]; therefore, utilizing the gene pool of their ancestors represents a viable strategy for incorporating desirable traits and developing superior varieties of bread wheat [11].
Triticum spelta L. is a hexaploid species (2n = 6x = 42, AABBDD) considered the ancestor of T. aestivum [12, 13]. T. spelta plants are characterized by being tall, with brittle rachis, with long and lax spikes in which the glume is not easily detached, making threshing difficult. Although the presence of these undesirable traits may limit their use as a cultivated species, the production of T. spelta plants is preserved due to their tolerance to various types of biotic and abiotic stresses. Meanwhile, T. spelta plants have been utilized in the field of genetic improvement as a parent for agronomic traits [14]. There are T. spelta accessions in which resistance to diseases has been detected, and upon identification of these resistance genes, the genes were successfully transferred to bread wheat. The resistance genes for leaf rust from T. spelta have been catalogued in the Wheat Gene Catalogue [8]: Lr44 [15], Lr65 [16], and Lr71 [17]. A recent study [18], evaluating spelt wheat genotypes for leaf rust resistance, has also identified the presence of Lr1 and Lr10. However, additional resistance factors remain undetected by the currently employed molecular markers. These unidentified factors may include both race-specific resistance genes and non-race-specific adult plant resistance genes [18].
Further resistance genes present in T. spelta could be identified and utilized to maintain the diversity of effective genetic resistance in bread wheat germplasm; therefore, additional genetic testing of T. spelta is necessary to detect new genes and characterize them according to their genetic action [3].
Due to the constant evolution of the fungus that causes leaf rust towards new races and the need to find new sources of resistance for its control, this study aimed to determine the genetics of a possible new gene toward leaf resistance present in an accession of T. spelta (T. spelta 109 = PI 355580)
From a collection of Triticum spelta obtained from the germplasm bank of the International Maize and Wheat Improvement Center (CIMMYT), the accession “T. spelta 109 (PI 355580)” from Switzerland was selected for its resistance to Puccinia triticina Erikss. at the seedling stage against the rust race MBJ/SP according to the nomenclature proposed by Long and Kolmer [19]. Another reason for this selection was that leaf tip necrosis (Ltn) was observed at the adult plant stage [20], which is associated with the Lr34/Ltn1 [21], Lr46/Ltn2 [22], Lr67/Ltn3 [23], and Lr68/Ltn4 genes [24].
“Roelfs F2007” is a spring-growing bread wheat variety developed from the cross Tacupeto F2001/Kukuna, created by CIMMYT and released by the National Institute of Forestry, Agricultural and Livestock Research (INIFAP). Roelfs F2007 is recommended for the autumn–winter production cycles in the northwestern region of Mexico [25]. Moreover, Roelfs F2007 exhibits moderate resistance in the field [25], which is attributed to the presence of adult plant resistance genes Lr46, Lr68, and LrB1 (unpublished data). In this study, Roelfs F2007 was susceptible to Puccinia triticina Erikss. race MBJ/SP at the seedling stage, so it was used as a susceptible parent to form a segregating population with T. spelta 109.
The cross between T. spelta 109 and the cultivar Roelfs F2007 was made at the Norman Borlaug Experimental Agricultural Station (CENEB), Ciudad Obregón, Sonora, during the autumn/winter 2018–2019 growing cycle. A total of 20 seeds were obtained from the crosses; of these, 10 were sown, and five F1 plants were selected. Three of the selected plants were used to advance to the next generation, while the remaining two were retained as reserves. The F1 seed was sown at El Batán, Mexico, during the 2019 summer cycle to produce the F2 seed. From each F1 plant, 50 F2 seeds were sown at a density of 10 seeds per linear meter in the CENEB field during the 2019–2020 autumn–winter cycle, to obtain 50 individual F2 plants. During the 2020 summer at El Batán, Mexico, seeds from the F2 plants were used to generate 50 F3 families, resulting in a total of 150 families. Of these 150 families, 135 were used for the genetic analysis.
The resistant genotype T. spelta 109, the susceptible variety Roelfs F2007, the F1 generation, and the 135 F3 families obtained were used for the genetic analysis of resistance. F2 generation plants were not used to assess resistance; the evaluation of F3 segregating families was preferred because it represents a more reliable generation, since more than one plant can be observed and counted to determine the type of infection [3].
The evaluation was conducted at the National Laboratory of Rusts and Other Wheat
Diseases (LANARET) of National Institute of Forestry, Agricultural and Livestock
Research (INIFAP) - Mexico Valley Experimental Field, Coatlinchán, Texcoco,
Mexico State 56250, Mexico. The 135 F3 families, the parents: T.
spelta 109 and Roelfs F2007, and the F1 seeds obtained from the crossing of
the latter, were sown in plastic trays of 20 cm
Inoculation was performed 12 DAP using the MBJ/SP leaf rust race of P. triticina Erikss. whose avirulence/virulence formula is: Lr2a, 2b, 2c, 3ka, 9, 16, 18, 19, 21, 24, 25, 26, 28, 29, 30, 32, 33, 36/1, 3, 3bg, 10, 11, 13, 15, 17, 20, 23, 27+31, 44. A suspension of urediniospores was sprayed in light mineral oil (Soltrol 170, Chevron Phillips Chemical Company, The Woodlands, TX, USA) at a concentration of 5 mg/mL using an atomizer connected to an electric vacuum pump (FE-1500L, Fabricantes Feligneo, Zapopan, Jalisco, Mexico). The plants were left to dry for 15 minutes to allow the excess oil to evaporate, and then were placed in a dew chamber in darkness at 22 °C for 16 hours. Then, the plants were moved to a greenhouse at 24 °C and provided with additional lamp light.
Once the susceptible parent showed visible signs of infection (10 days after inoculation), the infection type of all families was determined using a 0–4 scale [26]. In this scale, “0” depicts immunity, as no uredia or other macroscopic sign of infection is observed; “;” is almost immune because there are no uredia but there are chlorotic or necrotic flecks that indicate hypersensitivity; “1” is very resistant, since small uredia are surrounded by necrosis; “2”, is moderately resistant, since the uredia observed can be small or medium, often surrounded by chlorosis or necrosis and a chlorotic or necrotic border may surround a green island; “X” represents a heterogeneous reaction, i.e., uredia of variable size are randomly distributed on a single leaf. Within the scale, “3” is considered moderately susceptible because there are medium-sized uredia that may be associated with chlorosis; “4” is where large uredia without chlorosis are observed [26]. The scale also included variations or combinations of the infection type, such as “0;”, “;1”, “11+”, or “1+3c”, etc., since it is a relative visual scale.
The families were classified into three groups: (i) homozygous-resistant, with
an infection similar to that of the resistant parent; (ii)
homozygous-susceptible, with an infection similar to that of the susceptible
parent; (iii) segregating, which included plants with an infection similar to
those of the two previous groups. Finally, the number of each family in each
group was determined, and a
Contrasting infection types (ITs) were observed in the parents; T. spelta 109 showed “;1”, which is a quasi-immune response characterized by chlorotic or necrotic flecks that indicate hypersensitivity. Roelfs F2007 showed “3” where medium uredia were observed, and some were associated with chlorosis. F1 also showed the “3” IT (Fig. 1).
 Fig. 1.
Fig. 1.
Parents and F1 infection types. T. spelta 109 (resistant parent) with infection type “;1”, Roelfs F2007 (susceptible parent) with infection type “3”, and F1 with infection type “3” in seedlings. National Laboratory of Rusts and Other Wheat Diseases (LANARET) of INIFAP-Mexico Valley Experimental Field, Coatlinchán, Texcoco, Mexico State, Mexico, 2024.
Table 1 presents the number of F3 families registered in each group. Homozygous-resistant families exhibited a type of infection equal to that of the resistant parent, i.e., IT = 0; (Fig. 2a). Similarly, homozygous-susceptible families showed one type of infection equal to the susceptible parent, IT = 3 (Fig. 2b), and segregating families showed two types of infection: “;1” and “3” (Fig. 2c).
| Family type | O | E | ||
| Homozygous-susceptible | 34 | 33.75 | 0.0019 | |
| Segregating | 69 | 67.5 | 0.0333 | |
| Homozygous-resistant | 32 | 33.75 | 0.0907 | |
| Total | 135 | 135 | 0.1259 | 5.99 |
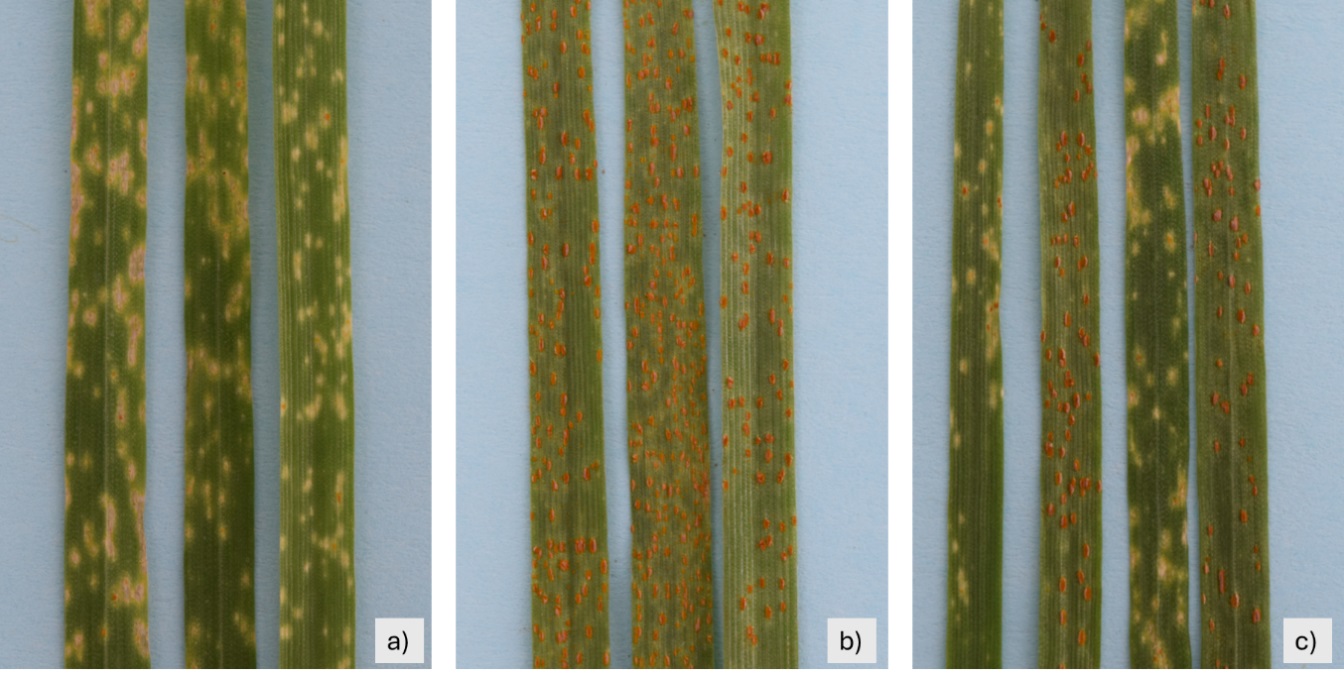 Fig. 2.
Fig. 2.
Infection types observed in the different family groups. (a) Leaves of an F3 homozygous resistant family at the seedling stage. (b) Leaves of an F3 susceptible homozygous family at the seedling stage. (c) Leaves of an F3 segregating family at the seedling stage. National Laboratory of Rusts and Other Wheat Diseases (LANARET) of INIFAP-Mexico Valley Experimental Field, Coatlinchán, Texcoco, Mexico State, Mexico, 2024.
Under the assumption that a major recessive gene promotes resistance, the expected frequencies in F3 families are 1:2:1, homozygous-resistant, segregating, and homozygous-susceptible, respectively.
When performing the
In all the evaluated seedlings from the F1 generation, an IT of “3” was observed, indicating susceptibility (Fig. 1). This suggests that the resistance is conferred by a recessive gene(s). Other leaf rust resistance genes identified in T. spelta accessions, such as Lr65 and Lr71, exhibit a dominant gene action type [28, 29]. Conversely, Lr44 is reported to have partial or recessive genetic action [15]. However, Lr44 is ineffective against the leaf rust race MBJ/SP used in this study.
Recessive catalogued genes conferring leaf rust resistance are not very common. However, Lr30 is one of the first reported leaf rust recessive genes [30]. Recently, other recessive genes, such as LrTs276-2, have been identified that confer resistance to Puccinia triticina Erikss. [28] and the MlHubel gene, which confers resistance to Blumeria graminis f. sp. tritici [31], both have been identified in accessions of T. spelta. Lr83 is also a recessive gene that confers resistance to leaf rust from the Triticum dicoccoides species [32].
Among the stripe rust resistance genes cataloged, most are dominant, and a few, such as Yr3a [33], Yr6 [34], Yr51 [35], and a temporarily designated YrZ15-1949 [36], are recessive. A similar situation occurs for stem rust catalogued resistance genes, in which only Sr17 has been reported as recessive [37]. Molecular analysis of the F3 population is currently being performed to identify a molecular marker linked to the resistance gene temporarily designated LrTspelta109 (PI 355580). According to our results and the reported recessive genes identified among the T. spelta accessions, the identified recessive gene in T. spelta 109 = PI355580 is different and most likely a novel leaf rust resistance gene which, when combined with other effective and available resistance genes against leaf rust will significantly contribute in developing, through breeding, new and more durable resistant varieties with a broad spectrum against the leaf rust pathogen P. triticina Erikss.
Spelt wheat possesses undesirable agronomic traits for modern agricultural systems due to the presence of the Rht-B1a, Rht-D1a, and q alleles, which result in taller plants and tenacious glumes, a vernalization requirement, and photoperiod sensitivity, thereby increasing lodging and hindering mechanical harvesting and processing [38]. Although some of these traits were observed in the germplasm used in this study, future backcrossing to the Roelfs F2007 parent and other bread wheats should be conducted to develop lines that no longer express these undesirable traits.
The genetic resistance to rust of the leaf of T. spelta 109 (PI 355580) is conferred by a single recessive gene. The importance and usefulness of searching for rust resistance genes from different sources and incorporating them into the genetic base of wheat improvement programs to provide diversity was confirmed.
All data reported in this paper will be shared by the corresponding author upon reasonable request.
JHE, YRVR, IBR, and SD designed the research study. Data collection and analysis: YRVR and JHE. Research project conception and manuscript preparation: YRVR, JHE, IBR, ASV and CMC. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The material used for this study was obtained from CIMMYT (International Maize and Wheat Improvement Center) germplasm bank.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


