1 Department of Obstetrics and Gynecology, The Second Hospital of Anhui Medical University, 230601 Hefei, Anhui, China
Abstract
Endometriosis is a chronic gynecologic disorder characterized by systemic inflammation, with growing evidence implicating gut microbial dysbiosis. However, the relationship between inflammatory cytokines and gut microbiota across disease stages remains unclear.
This retrospective cross-sectional study included 150 participants, divided into healthy controls (n = 40), benign gynecologic disease controls (n = 45), and patients with stage I–II (n = 25) or stage III–IV (n = 40) endometriosis. Levels of the serum cytokines interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α) were evaluated by enzyme-linked immunosorbent assay (ELISA). Gut microbiota was profiled via 16S rRNA sequencing, followed by assessment of microbial alpha diversity, beta diversity (Bray-Curtis), and genus-level taxonomic composition.
Serum IL-6 and TNF-α levels increased progressively with disease severity. IL-6 levels differed significantly across groups (Kruskal-Wallis p < 0.0001), with the stage III–IV endometriosis group showing a median level that was 12.8 pg/mL higher compared to healthy controls 95% confidence interval (CI: 10.7 to 13.8). Shannon diversity decreased significantly across groups, and principal coordinate analysis (PCoA) demonstrated distinct clustering of microbial communities according to disease status. Spearman correlation analysis revealed that the genus Prevotella was positively correlated with IL-6 (ρ = 0.33, q = 0.018), whereas Blautia was negatively correlated with TNF-α (ρ = –0.32, q = 0.026), with both remaining significant after correcting for the false discovery rate (FDR).
These findings suggest that systemic inflammation and gut microbiota alterations progress alongside endometriosis severity. Specific genera, such as Prevotella and Blautia, may serve as potential microbial markers and modulators of inflammatory status in endometriosis.
Keywords
- endometriosis
- gut microbiota
- cytokines
- inflammation
- biomarkers
Endometriosis is a common and chronic gynecological disorder characterized by the presence of endometrial-like tissue outside the uterus. Globally, it affects approximately 10% of women of reproductive age [1]. While traditionally viewed as a localized pelvic condition associated with dysmenorrhea, dyspareunia, and infertility, emerging evidence suggests that endometriosis is a complex systemic disease involving immunologic, inflammatory, and metabolic dysfunction [2]. Patients frequently present with symptoms that extend beyond the pelvis, including fatigue, gastrointestinal complaints and systemic inflammation, indicating the disease process is not confined to ectopic lesions but may reflect a broader dysregulation of host physiology [3].
Recent study has implicated the gut microbiota as a potential modulator of estrogen metabolism and immune responses [4], both of which are critical in the pathophysiology of endometriosis. A growing body of research indicates that imbalances in the gut microbiota are present in women with endometriosis, often characterized by decreased microbial diversity and alterations in the composition of specific bacterial genera. Notably, taxa such as Prevotella, Bacteroides, and Blautia have been consistently identified as differentially abundant in affected individuals [5]. Changes in gut microbial composition may influence systemic inflammation by impairing intestinal barrier function and regulating mucosal immune activity [6].
As well as changes in the gut microbiota, elevated circulating levels of
pro-inflammatory cytokines—including interleukin-6 (IL-6), tumor necrosis
factor-
Despite accumulating evidence implicating both gut microbial dysbiosis and systemic inflammation in endometriosis, a previous study has systematically integrated these two domains within the same analytic framework [8]. The absence of unified datasets limits our ability to understand whether specific microbial taxa are functionally linked to inflammatory signaling pathways, or whether they can serve as surrogate biomarkers for disease presence or severity [9]. Moreover, most available data are cross-sectional, underpowered, or focused on early-stage disease, thus limiting their generalizability for clinically significant endometriosis.
There is a critical need to define the immuno-microbial landscape of endometriosis using standardized, multi-dimensional approaches that combine the profiling of gut microbiota with quantitative assessment of circulating cytokines [10]. Investigating the associations between particular microbial taxa and inflammatory markers could provide new perspectives on disease mechanisms and facilitate the creation of comprehensive diagnostic or prognostic frameworks.
This study retrospectively analyzed changes in both gut microbial communities and circulating inflammatory cytokines in patients with moderate-to-severe endometriosis. Stool and blood samples were collected from patients who had previously undergone laparoscopic evaluation and had well-documented clinical diagnoses. 16S rRNA gene sequencing and multiplex cytokine assays were used to assess microbial diversity, taxonomic composition, and cytokine profiles, allowing clinical subgroups to be compared for these parameters. To identify potential biomarkers, a multivariable predictive model was constructed based on archived microbial and immunological data [11]. Correlations between microbial genera and cytokine levels were also explored to identify possible mechanisms underlying the gut-immune axis in endometriosis.
This study was designed as a single-center, cross-sectional, retrospective analysis conducted at the Second Hospital of Anhui Medical University in southern China. Clinical records from June 2022 to December 2023 were reviewed, and corresponding stored biospecimens were retrieved to explore associations between gut microbiota composition, circulating inflammatory cytokine profiles, and the severity of endometriosis across different clinical stages. This study was approved by the Institutional Ethics Committee of the Second Hospital of Anhui Medical University (Approval No. YX2024-164, dated October 2024). Biological samples (including serum and stool) were originally collected as part of routine clinical diagnosis and care between 2023 and 2025, with all patients providing written informed consent for biobanking and future research use. The archived and anonymized samples were retrospectively analyzed in the current study. An internal audit confirmed that all samples were collected under institutional protocols that were in effect prior to the formal 2025 study-specific approval.
A total of 150 premenopausal women aged 20 to 45 years were included based on available laparoscopic and histopathological data. Patients were stratified into four groups: healthy controls (n = 40), individuals with benign gynecologic conditions unrelated to endometriosis (e.g., uterine fibroids, ovarian cysts; n = 45), patients diagnosed with stage I–II endometriosis (n = 25), and patients with stage III–IV endometriosis (n = 40). The above classifications were based on criteria from the revised American Society for Reproductive Medicine (rASRM).
Patients with incomplete records, a history of confounding conditions (e.g., autoimmune/metabolic diseases, malignancies, pelvic infections, irritable/inflammatory bowel disease), or documented use of antibiotics or probiotics within six months prior to sample collection were excluded from analysis. Relevant clinical data, anthropometric measurements, and biospecimen information were obtained from institutional medical records and laboratory archives under standardized protocols.
To improve transparency and ensure reproducibility, the inclusion and exclusion criteria for each study group were explicitly defined. Participants in the healthy control group were premenopausal women aged 20 to 45 years who underwent laparoscopic procedures for non-endometriotic indications, such as elective tubal ligation or benign ovarian cysts. They had no history of chronic pelvic pain, dysmenorrhea, or infertility, and no visual or histologic evidence of endometriosis upon surgical evaluation. Individuals were excluded if they had a history of pelvic infection, gynecologic malignancy, or recent use (within 6 months) of antibiotics, probiotics, or hormone therapies.
For the healthy control group, participants were selected among women who underwent laparoscopic procedures for non-endometriotic indications, such as elective tubal ligation or benign ovarian cysts later confirmed to be non-endometriotic. All individuals were evaluated intraoperatively and showed no visible lesions suggestive of endometriosis, and no history of chronic pelvic pain, dysmenorrhea, or infertility. In addition, where indicated, histopathological examination confirmed the absence of ectopic endometrial tissue. This approach was used to minimize the likelihood of undiagnosed asymptomatic endometriosis in the control group.
The benign gynecologic group included patients diagnosed with non-endometriotic gynecologic conditions such as uterine fibroids or functional cysts, confirmed through imaging or surgery, and with no intraoperative or histological signs of endometriosis. Patients were excluded if they had overlapping endometriotic lesions, concurrent endocrine or autoimmune disorders, or had received hormonal or immunosuppressive treatment in the past 6 months.
Patients in the stage I–II endometriosis group were diagnosed according to rASRM criteria and based on laparoscopic findings and histopathological confirmation. They typically presented with symptoms such as pelvic pain or subfertility. Exclusion criteria included recent surgical treatment for endometriosis (within the previous year), incomplete staging, or coexisting severe pelvic pathologies.
The stage III–IV endometriosis group was comprised of women with confirmed advanced disease, as defined by rASRM staging during laparoscopic surgery, along with histological verification. These participants had not received hormonal therapy or antibiotics for at least 3 months prior to biospecimen collection. Women with deeply infiltrating endometriosis involving the bowel or bladder that precluded complete staging were excluded, as well as those receiving systemic immunomodulatory therapies.
The above criteria were designed to minimize confounding variables, while aligning with established diagnostic and microbiome research protocols, as reported in recent literature [12, 13].
Clinical assessments and biospecimen collections had been conducted as part of routine preoperative evaluations, or general gynecological examinations. Blood pressure measurements were recorded using a calibrated automated sphygmomanometer, with patients seated at rest. The documented value was the average of two successive readings.
Peripheral venous blood samples (5 mL) were collected in the early morning
(07:00–09:00) under fasting conditions into ethylenediaminetetraacetic acid (EDTA) tubes. After centrifugation at
3000 rpm for 10 minutes at 4 °C, serum was aliquoted into sterile
cryotubes and stored at –80 °C until cytokine analysis. Subsequent
quantification was focused on pro-inflammatory cytokines, including
interleukin-1
Stool samples (~200 mg) had been collected in sterile containers and delivered under cold-chain conditions to the institutional biobank. Samples were preserved in liquid nitrogen and stored at –80 °C prior to microbial DNA extraction. All biospecimens had been anonymized and linked to relevant clinical data through coded identifiers. The laboratory procedures adhered to institutional protocols for biosafety and biobanking.
For analytical purposes, primary group-wise comparisons were made between the healthy control group and the combined endometriosis cohort. Additional stratified analyses were conducted between stage I–II and stage III–IV endometriosis patients. The benign gynecologic group served as a reference for non-endometriotic perioperative factors, but was not used in the main statistical comparisons.
Approximately 200 mg of stool per sample was used for genomic DNA extraction with the QIAamp Fast DNA Stool Mini Kit (Cat# 51604; Qiagen, Hilden, Germany) following the supplier’s protocol. To enhance lysis of gram-positive bacteria, a bead-beating procedure was incorporated using 0.1 mm zirconia/silica beads. Taxonomic classification was conducted using a naïve Bayes model trained on the SILVA ribosomal RNA gene database project (SILVA) 138 reference dataset at 99% Operational Taxonomic Unit (OTU) similarity. To assess within-sample microbial diversity, alpha diversity metrics including Shannon and Chao1 indices were computed. Differences in microbial composition between groups were evaluated through beta diversity analysis based on Bray-Curtis dissimilarity, and visualized using principal coordinates analysis (PCoA). Group-level variation in microbial communities was statistically tested via permutational multivariate analysis of variance (PERMANOVA) with 999 permutations. Relative abundances of microbial taxa at the genus level were used for downstream comparisons and correlation analyses with clinical and inflammatory parameters.
Systemic inflammatory cytokine levels were measured with enzyme-linked
immunosorbent assay (ELISA) kits (MultiSciences Biotech Co., Hangzhou, Zhejiang,
China) following the manufacturer’s recommended protocols. After thawing on ice,
serum specimens were analyzed in duplicate using 96-well plates. The assays
targeted key pro-inflammatory cytokines, specifically IL-1
To ensure the reliability of results, intra- and inter-assay variability was
controlled, with coefficients of variation kept under 10% and 15%,
respectively. If duplicate readings varied by
Statistical processing was performed with R (v4.2.3, R Foundation for
Statistical Computing, Vienna, Austria) and Python, version 3.9 (Python Software
Foundation, Wilmington, DE, USA), utilizing established libraries such as
statsmodels, version 0.14.0 (Seabold, S. & Perktold, J.;
https://www.statsmodels.org/), scikit-learn, version 1.3.0 (Pedregosa,
F. et al.; https://scikit-learn.org/), ggplot2, version 3.4.2 (Wickham,
H.; R package; https://ggplot2.tidyverse.org/), vegan, version 2.6-4
(Oksanen, J. et al.; R package; https://cran.r-project.org/package=vegan),
seaborn, version 0.12.2 (Waskom, M.; https://seaborn.pydata.org/),
matplotlib, version 3.7.1 (Hunter, J. D.; https://matplotlib.org/).
Unless stated otherwise, statistical significance was defined as a two-tailed
p value
Normality of continuous variables was evaluated using the Shapiro-Wilk test.
Data conforming to a normal distribution were summarized as the mean
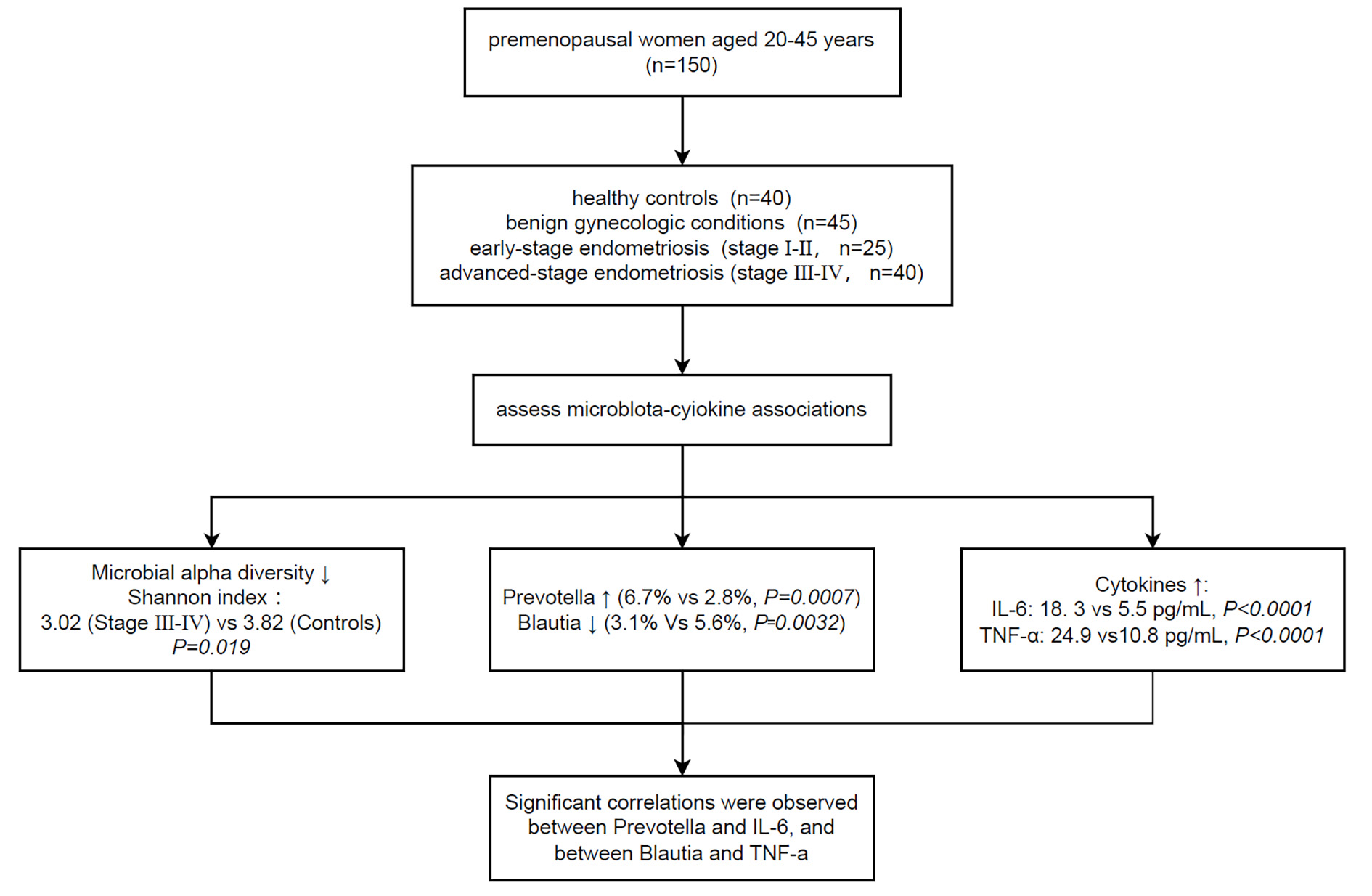 Fig. 1.
Fig. 1.
Study design and microbiota-cytokine association framework. This flowchart illustrates the structure of the study cohort and the analytic
workflow. Clinical data and biospecimens from 150 premenopausal women aged 20–45
years were retrospectively analyzed and categorized into four subgroups according
to prior laparoscopic and histopathological records: healthy controls (n = 40),
patients with benign gynecologic conditions (n = 45), early-stage endometriosis
(stage I–II, n = 25), and advanced-stage endometriosis (stage III–IV, n = 40).
Microbial alpha diversity (Shannon index) was reduced in the advanced-stage group
(p = 0.019), accompanied by increased Prevotella and decreased
Blautia abundance. Circulating interleukin-6 (IL-6) and tumor necrosis
factor-
Differences in the median between groups (e.g., healthy controls vs. stage III–IV endometriosis) were estimated using the Hodges-Lehmann method, a non-parametric approach for assessing central tendency differences. Associated 95% confidence intervals (CIs) were calculated using the exact distribution of Walsh averages, as implemented in the scipy.stats package (Python v3.9; https://www.python.org/). This method was used to complement Kruskal-Wallis tests when interpreting median differences between groups.
The study cohort comprised of 150 premenopausal women, categorized into four groups: healthy controls (n = 40), individuals with benign gynecologic disorders (n = 45), patients diagnosed with stage I–II endometriosis (n = 25), and patients with advanced-stage (stage III–IV) endometriosis (n = 40). Age distribution did not differ significantly across groups (p = 0.234; data not shown).
To address disease-specific effects, the analysis was structured in two tiers. First, comparisons were made between healthy controls and all patients with endometriosis in order to capture the overall differences in gut microbiota composition and inflammatory cytokines. Second, subgroup analyses comparing patients with early-stage (I–II) and advanced-stage (III–IV) endometriosis were performed to explore changes associated with disease progression. Although the benign gynecologic group was included to control for perioperative, hormonal, or hospitalization-related confounders, it was not incorporated in testing of the primary hypothesis. This approach allowed the preservation of data integrity, while focusing analytical power on disease-specific contrasts.
Notable trends were observed for several inflammatory and metabolic markers
(Table 1). The median serum IL-6 level increased progressively with disease
severity, from 5.5 pg/mL (IQR: 4.1–6.7) in healthy controls to 18.3 pg/mL
(14.3–20.5) in patients with stage III–IV endometriosis (p
| Variable | Healthy control | Benign GYN | EM I–II | EM III–IV | p value | Effect size (vs. HC) | 95% CI |
| IL-6 (pg/mL) | 5.5 (4.1–6.7) | 8.4 (7.2–10.3) | 11.8 (10.7–12.9) | 18.3 (14.3–20.5) | +12.8 pg/mL | 10.7 to 13.8 | |
| TNF-α (pg/mL) | 10.8 (7.8–12.0) | 14.7 (11.4–16.7) | 18.7 (15.4–21.4) | 24.9 (21.8–28.0) | +14.1 pg/mL | 12.0 to 16.1 | |
| Creatinine (μmol/L) | 58.4 (56.1–61.5) | 61.9 (57.7–64.4) | 65.3 (61.5–68.7) | 70.5 (63.1–75.5) | +12.1 μmol/L | 9.2 to 15.5 | |
| BMI (kg/m2) | 20.7 (19.9–21.9) | 22.1 (21.0–23.4) | 23.2 (22.6–24.2) | 24.8 (23.6–25.8) | +4.1 kg/m2 | 3.5 to 4.8 | |
| Diastolic BP (mmHg) | 70.6 (65.9–74.2) | 70.5 (68.6–76.2) | 72.9 (69.8–78.3) | 76.2 (71.4–78.5) | 0.0048 | +5.6 mmHg | 1.4 to 7.2 |
Data are presented as median (interquartile range, IQR). p values are based on Kruskal-Wallis test. Effect sizes reflect median difference from healthy controls, estimated using the Hodges-Lehmann method.
GYN, gynecology; EM, endometriosis; HC, healthy control; BMI, body mass index; BP, blood pressure; CI, confidence interval.
Other systemic indicators such as serum creatinine and body mass index (BMI)
also showed significant between-group differences. Patients with stage III–IV
endometriosis had higher creatinine levels (median: 70.5 µmol/L) and BMI
(24.8 kg/m2) compared with healthy controls (58.4 µmol/L and 20.7
kg/m2, respectively). Both indicators were significantly different between
groups (p
The baseline characteristics and pairwise effect estimates are summarized in Table 1.
Circulating levels of inflammatory cytokines demonstrated a clear stepwise
increase across the four clinical groups. IL-6 concentrations differed
significantly among clinical groups (Kruskal-Wallis p
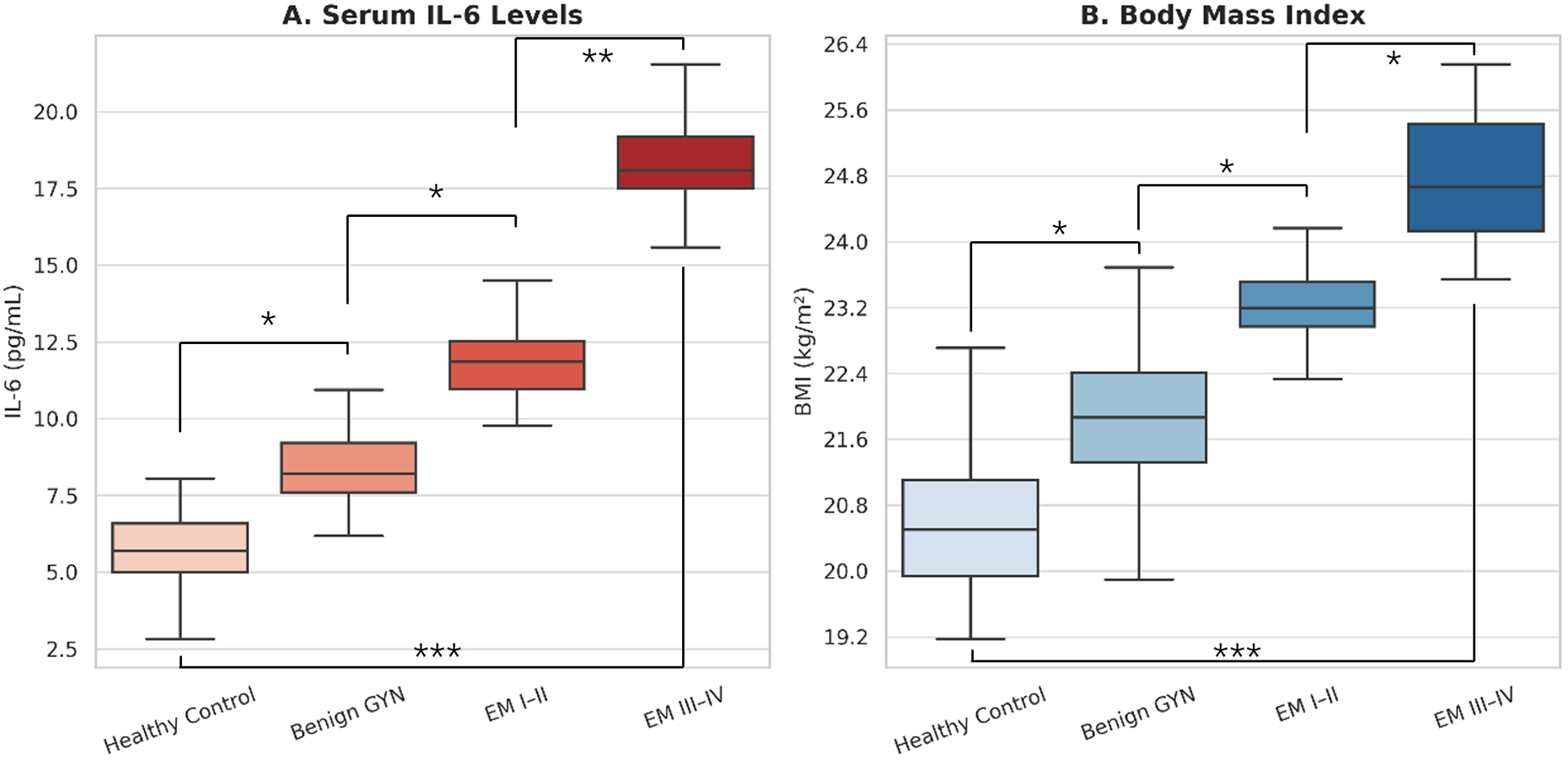 Fig. 2.
Fig. 2.
Group-wise comparisons of IL-6 levels and BMI across disease
severity. (A) IL-6 concentrations across four participant groups: healthy
controls, patients with benign gynecologic conditions, and patients with stage
I–II or stage III–IV endometriosis. IL-6 levels showed a progressive increase
with disease severity. (B) BMI across the same clinical groups, with higher BMI
observed in patients with advanced endometriosis. Each box represents the
IQR, with the median denoted by a horizontal line. Whiskers
indicate 1.5
| Variable | Kruskal-Wallis H Statistic | p value |
| IL-6 (pg/mL) | 136.11 | |
| TNF- |
132.05 | |
| Creatinine (µmol/L) | 45.23 | |
| BMI (kg/m2) | 58.77 | |
| Diastolic BP (mmHg) | 10.62 | 0.0048 |
Note: Data were analyzed using the Kruskal-Wallis test to assess non-parametric differences across four clinical subgroups: healthy controls, benign gynecological conditions, early-stage endometriosis (stage I–II), and advanced-stage endometriosis (stage III–IV). All variables demonstrated statistically significant group-level differences.
To evaluate disease-associated alterations in gut microbiota, 16S rRNA
sequencing was performed on fecal samples from all participants. Alpha diversity,
assessed by the Shannon index, showed a progressive reduction across disease
severity groups. The median diversity value declined from 3.15 (IQR: 2.95–3.42)
in healthy controls to 2.78 (2.60–2.92) in stage III–IV endometriosis patients
(p
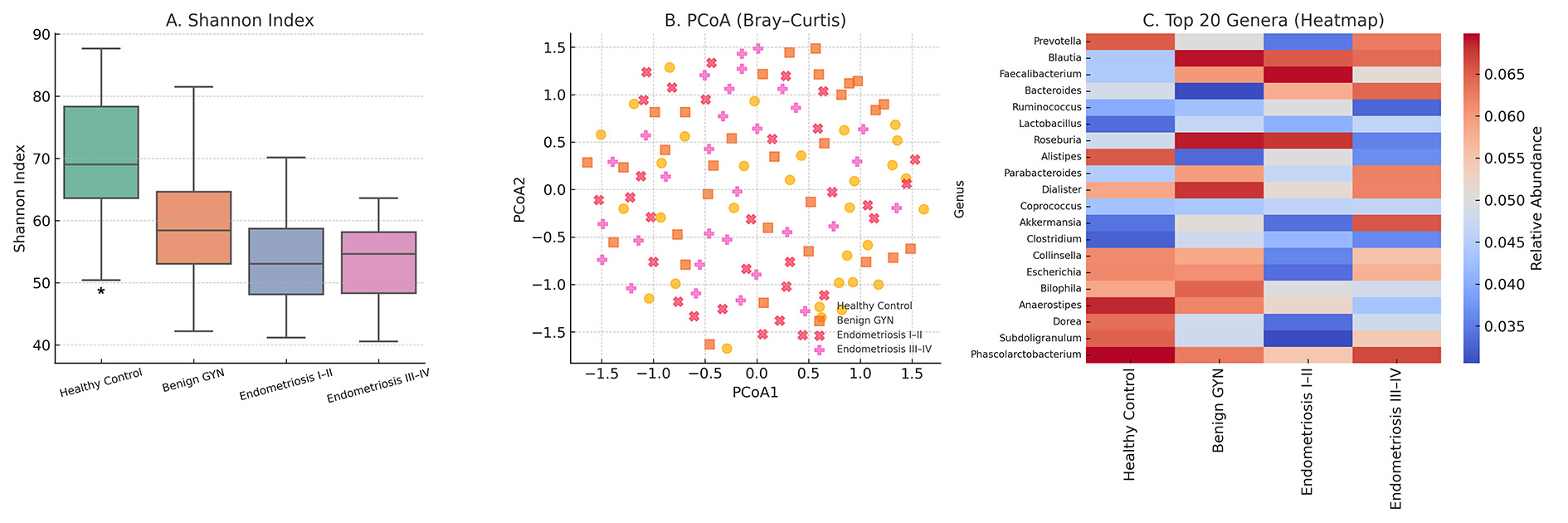 Fig. 3.
Fig. 3.
Gut microbiota diversity and composition across disease stages.
(A) Boxplot showing decreasing alpha diversity (Shannon index) across four
groups: healthy controls, benign gynecologic conditions, stage I–II
endometriosis, and stage III–IV endometriosis. (B) Principal Coordinates
Analysis (PCoA) based on Bray-Curtis dissimilarity demonstrates distinct
clustering of gut microbial communities by disease group. (C) Heatmap displaying
the relative abundance of the top 20 most prevalent bacterial genera, averaged
across participants within each group. Each box in (A) represents the
IQR, with the median indicated by a horizontal line;
whiskers extend to 1.5
Beta diversity analysis using Bray-Curtis dissimilarity and PCoA revealed distinct clustering by disease stage, with a clear separation between advanced endometriosis and non-disease groups (Fig. 3B). This compositional shift was confirmed by PERMANOVA (R2 = 0.12, p = 0.002), suggesting a significant structural divergence in microbial communities.
At the genus level, relative abundance profiles identified several taxa with stage-associated trends. Prevotella was progressively enriched and reached the highest level in stage III–IV disease. In contrast, Blautia, an anti-inflammatory short-chain fatty acid-producing genus, was markedly depleted in more advanced disease. A hierarchical heatmap of the top 20 genera illustrates these shifts (Fig. 3C), highlighting a disease-stage-dependent gradient in microbial signatures.
These results demonstrate that both microbial diversity and taxonomic composition are significantly altered in women with endometriosis, with the most profound dysbiosis observed in patients with advanced-stage disease.
Serum concentrations of key pro-inflammatory cytokines were analyzed across the
four clinical groups using Kruskal-Wallis tests, followed by post hoc comparisons
to determine group-wise differences. As shown in Fig. 4, a clear upward trend was
observed for IL-6, which increased progressively from healthy individuals
(median: 5.5 pg/mL, IQR: 4.1–6.7) to patients with stage III–IV endometriosis
(18.3 pg/mL, 14.3–20.5). Statistically significant differences in the IL-6 level
were detected between all clinical stages based on the Kruskal-Wallis test
(p
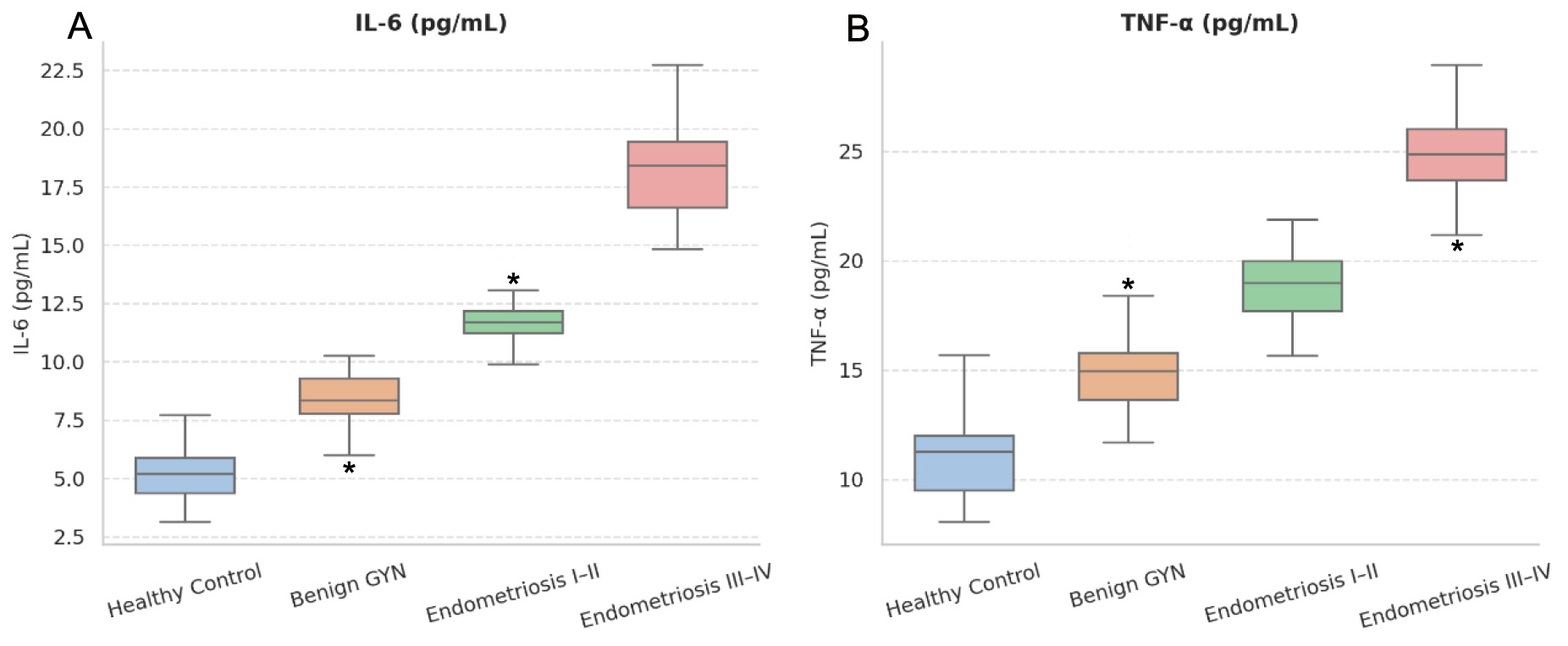 Fig. 4.
Fig. 4.
Group-wise distribution of serum inflammatory cytokines. (A)
IL-6 and (B) TNF-
A similar trend was noted for TNF-
Post hoc comparisons with the Kruskal-Wallis test confirmed that IL-6 and
TNF-
Based on Linear Discriminant Analysis Effect Size (LEfSe) analysis, 8 genera
demonstrated significant group-level differences in relative abundance across the
disease stages. Prevotella and Escherichia were progressively
enriched in patients with advanced-stage endometriosis, while Blautia,
Ruminococcus, Subdoligranulum and Bacteroides showed
decreasing trends. These taxa met the threshold for false discovery rate (FDR)
correction (q
To explore the interplay between gut microbiota and systemic inflammation, Spearman correlation analyses were performed between genus-level relative abundances, Shannon diversity, and serum cytokine levels in all participants. The resulting correlation matrix is presented in Fig. 5A.
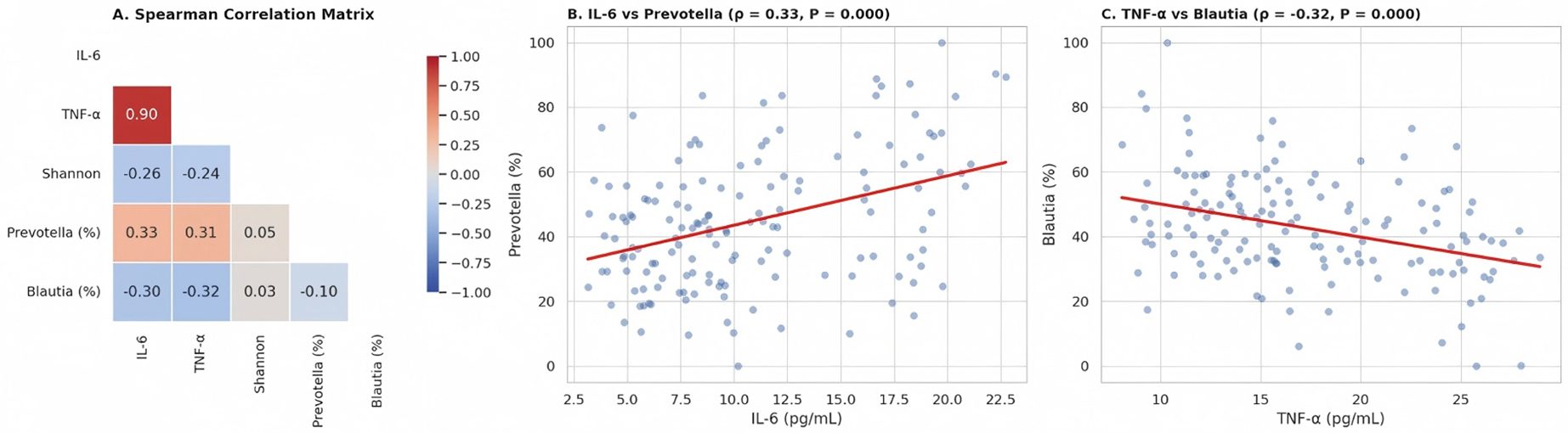 Fig. 5.
Fig. 5.
Correlation between gut microbiota, alpha diversity, and
systemic inflammation. (A) Spearman correlation matrix showing pairwise
associations between serum inflammatory cytokines (IL-6, TNF-
Among the significant associations, the abundance of prevotella showed
a moderate positive correlation with IL-6 (
Other genera such as Ruminococcus and Faecalibacterium
displayed weaker, non-significant correlations with IL-8 and IL-1
We next examined whether the association between endometriosis severity and inflammatory burden was independent of metabolic and lifestyle factors by constructing generalized linear models (GLMs) using IL-6 concentration and microbial diversity (Shannon index) as continuous outcomes. These models were adjusted for BMI, dietary pattern, and history of antibiotic use (Fig. 6A,B).
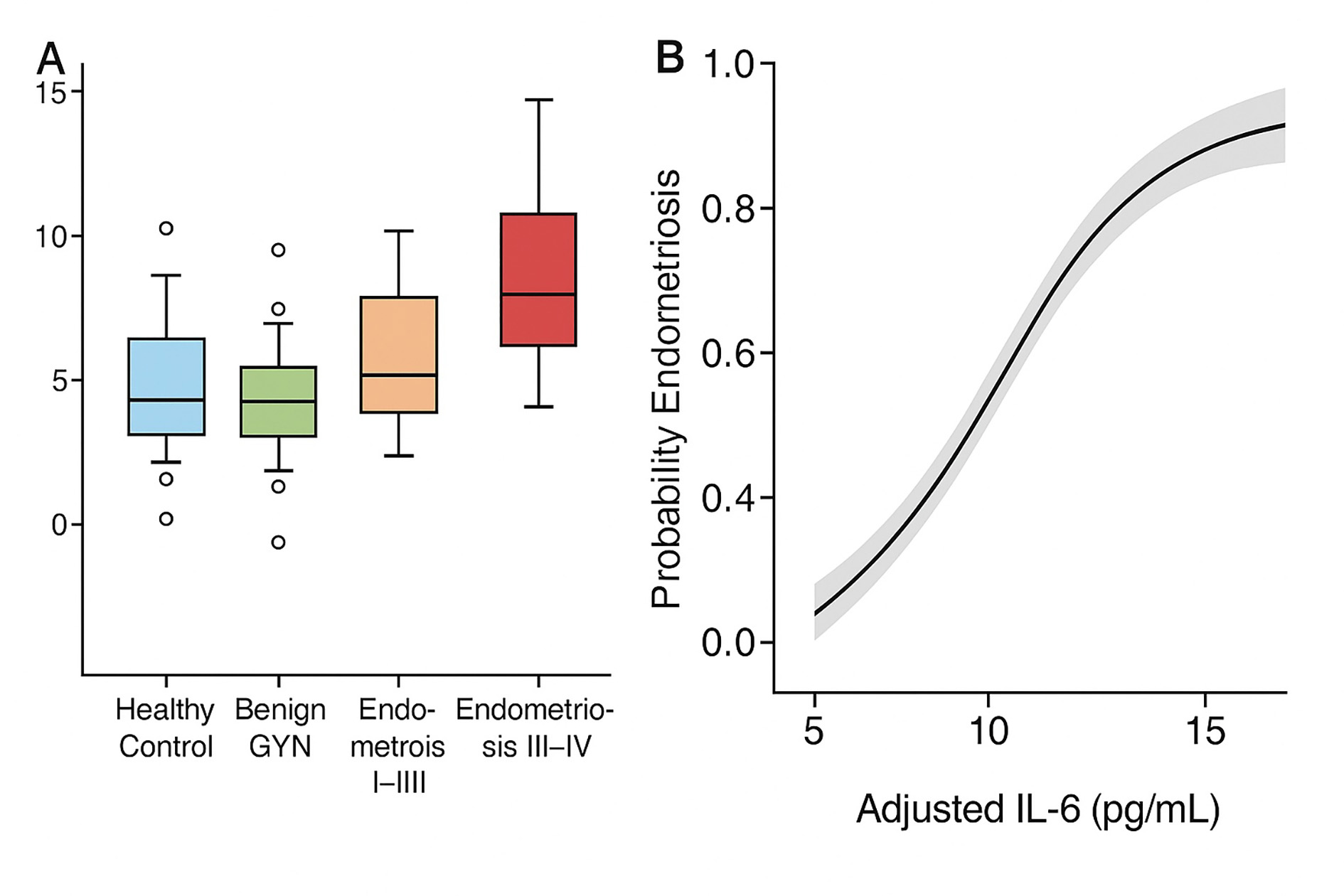 Fig. 6.
Fig. 6.
Adjusted IL-6 levels and predictive model estimates across disease groups. (A) Adjusted serum IL-6 concentrations across four participant groups (healthy controls, benign gynecologic conditions, stage I–II endometriosis, and stage III–IV endometriosis), based on generalized linear model controlling for BMI, dietary pattern, and antibiotic use history. Error bars represent 95% confidence intervals. (B) Model-predicted IL-6 values across disease stages derived from the same adjusted model. The shaded area indicates the 95% confidence band for predicted means. Open circles in panel A represent individual outlier values identified by boxplot analysis.
After multivariable adjustment, IL-6 remained significantly elevated in both the
stage I–II (
The IL-6 values predicted by the model across different disease stages (Fig. 6B) mirrored the observed trends, demonstrating progressive inflammatory activation with increasing disease severity, even after adjustment for covariates (Table 3).
| Variable | p value | ||
| Outcome: IL-6 (pg/mL) | |||
| Endometriosis I–II | 4.82 (3.25 to 6.40) | ||
| Endometriosis III–IV | 9.84 (8.13 to 11.56) | ||
| BMI (per 1 kg/m2 increase) | 0.47 (0.15 to 0.80) | 0.005 | |
| Non-omnivorous diet | –1.12 (–2.58 to 0.34) | 0.131 | |
| Prior antibiotic use ( |
0.92 (–0.45 to 2.29) | 0.185 | |
| Outcome: Shannon index | |||
| Endometriosis I–II | –0.31 (–0.49 to –0.14) | 0.001 | |
| Endometriosis III–IV | –0.56 (–0.74 to –0.38) | ||
| BMI (per 1 kg/m2 increase) | –0.04 (–0.08 to 0.01) | 0.082 | |
| Non-omnivorous diet | 0.12 (–0.05 to 0.30) | 0.168 | |
| Prior antibiotic use | –0.15 (–0.32 to 0.01) | 0.067 | |
Note: Omnivorous diet = reference group; Healthy control = reference
group for disease stage. Models adjusted for all listed covariates. Variance
inflation factors (VIF) values
This cross-sectional study of 150 women across four clinically distinct groups
identified a multidimensional biological signature associated with the severity
of endometriosis. The levels of inflammatory cytokines, such as IL-6,
TNF-
Rather than evaluating microbial or immune alterations in isolation, it is important to note that in the current study both domains were integrated within a unified analytic framework. By simultaneously quantifying taxonomic diversity and cytokine burden, as well as their interrelationship, we provide evidence that endometriosis may be characterized by a disrupted gut-immune axis, particularly in the advanced stages. These results support the notion that endometriosis is not just a localized gynecologic disorder, but rather a systemic and immunologically active condition, with potential implications for early detection, risk stratification, and novel therapeutic targeting.
Our findings align with and expand upon previous studies reporting gut microbial
dysbiosis and elevated systemic inflammation in women with endometriosis. Several
prior investigations have documented decreased microbial diversity and
overrepresentation of Prevotella in endometriosis patients [5], often
alongside the depletion of short-chain fatty acid (SCFA)-producing genera such as
Blautia and Ruminococcus. Increased circulating levels of IL-6
and TNF-
Our study is among the first to integrate quantitative cytokine profiling with
gut microbiota sequencing in a stratified surgical cohort, thereby allowing
detailed resolution across disease stages. The use of a four-group
design—including healthy controls and benign gynecologic comparators—helps to
delineate changes that are specific to endometriosis rather than to general
pelvic pathology [17]. Furthermore, the statistically significant correlations
observed between Prevotella and IL-6, and between Blautia and
TNF-
The observed associations between specific gut microbial taxa and systemic
inflammatory cytokines in the current study supports the notion of a functional
interaction between microbial dysbiosis and immune activation in endometriosis
[18]. The enrichment of Prevotella and depletion of Blautia in
patients with advanced disease stages may have mechanistic implications.
Prevotella species are known to synthesize lipopolysaccharide (LPS), a
strong endotoxin that activates signaling pathways through Toll-like receptor 4
(TLR4), ultimately promoting the secretion of inflammatory cytokines such as IL-6
and TNF-
The imbalance between pro-inflammatory and anti-inflammatory microbial
metabolites may contribute to a systemic inflammatory milieu that promotes the
progression of endometriotic lesions. LPS from Gram-negative bacteria like
Prevotella can stimulate macrophages in the peritoneal cavity [20],
thereby increasing the secretion of IL-6 and TNF-
Collectively, these findings support the hypothesis of a disrupted gut-immune axis in endometriosis, where microbial dysbiosis contributes to systemic inflammation, creating a feedback loop that sustains disease progression. Further clarification of such microbiota-immune interactions could lead to the development of new treatment approaches for endometriosis in which immune activity is regulated through modulation of the gut microbiome [21].
Beyond the mechanistic insights, our findings suggest potential clinical
applications for improving the diagnosis and management of endometriosis. The
clear stratification of inflammatory and microbial profiles across disease stages
suggests these biological markers may serve as adjuncts to current diagnostic
paradigms. In particular, the consistent elevation of IL-6 and TNF-
Given the frequent diagnostic delay in endometriosis, often exceeding 7 years from symptom onset, there is an urgent need for non-invasive, biomarker-based risk stratification tools. Our results suggest that commonly available clinical indices could be interpreted alongside immune and microbial signatures to improve earlier diagnostic evaluation in symptomatic women and inform decisions regarding surgical triage [22]. Moreover, the identification of taxon-specific inflammatory associations raises the possibility of microbiota-targeted interventions, including probiotic supplementation, dietary modulation, or fecal microbiota transplantation. Such adjunct strategies could attenuate systemic inflammation and potentially modulate disease activity [23]. These applications merit further investigation in prospective or interventional studies.
Several methodological constraints in this study should be acknowledged. Most notably, its cross-sectional nature restricts the ability to draw causal conclusions, making it unclear whether the identified changes in gut microbiota and cytokine profiles are contributing factors to endometriosis, or outcomes of the disease. Longitudinal studies are needed to evaluate temporal relationships and responses to intervention. Second, although participants with antibiotic or probiotic exposure within the past six months were excluded to minimize confounding, earlier exposures and dietary variation may still have influenced the microbial composition. Third, this was a single-center study with a moderate sample size, potentially limiting the generalizability of its findings to broader populations and healthcare settings [24]. Lastly, while 16S rRNA sequencing allows high-throughput taxonomic characterization, this technique lacks functional resolution. Multi-omics approaches—such as shotgun metagenomics, metabolomics, or host transcriptomics—will be required to elucidate microbial function and host–microbe interactions at a mechanistic level.
This study has several limitations. First, due to its retrospective design, the timing of biological sample collection was not standardized according to the menstrual phase. Although most samples were obtained during the early follicular phase, as per routine clinical scheduling, the exact cycle staging was not systematically recorded. Given the hormone sensitivity of both cytokine levels and gut microbiota composition, this may represent a source of biological variability. Second, no formal a priori sample size calculation was performed, as patient inclusion was based on the availability of biological samples. Nevertheless, the observed group-level differences in inflammatory markers and microbial metrics were statistically robust, suggesting adequate statistical power. Third, while modest increases in diastolic blood pressure were observed among patients with advanced endometriosis, its clinical and mechanistic relevance remains unclear. Earlier studies suggested potential links between endometriosis, vascular dysfunction, and cardiometabolic risk, possibly mediated by chronic inflammation and altered estrogen signaling [25, 26]. Further prospective research is warranted to explore these associations.
Finally, the cross-sectional nature of the study and the lack of functional assays—such as microbial metabolite profiling or SCFA quantification—limit causal interpretation. While statistically significant correlations were found between specific bacterial genera and cytokine levels, these do not establish mechanistic relationships. Further studies that integrate longitudinal sampling and metabolomic analyses are needed to validate these microbiota–immune interactions.
In summary, this study provides evidence that moderate-to-severe endometriosis is associated with both gut microbial dysbiosis and systemic immune activation. The identification of coordinated alterations in specific genera and inflammatory cytokines supports the concept of a disrupted gut-immune axis in endometriosis. These findings may inform the development of novel diagnostic tools and therapeutic strategies targeting microbial-immune interactions in this complex and burdensome disease.
This study demonstrates that endometriosis, particularly in its
moderate-to-severe forms, is associated with a distinct pattern of gut microbial
dysbiosis and systemic immune activation. We identified a progressive loss of
microbial diversity, specific alterations in genera such as Prevotella
and Blautia, and elevated levels of inflammatory cytokines including
IL-6 and TNF-
These findings support the concept that endometriosis is a systemic inflammatory condition involving gut-immune crosstalk, rather than being a purely localized pelvic disorder. By integrating microbial and cytokine data within a unified analytic framework, this study provides novel insights into the pathophysiology of endometriosis and suggests that such signatures may serve as early biomarkers of disease severity or progression.
Future research should aim to validate these associations in larger, longitudinal cohorts and to explore whether targeted modulation of the gut microbiota provides therapeutic benefit in endometriosis. Research into the gut-immune axis is a promising frontier for gaining a better mechanistic understanding of this burdensome and often underdiagnosed disease, as well as for precision intervention.
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
HW and WW conceived and designed the study and developed the methodology. HW, WW, and WZ conducted the investigation and curated the data. HW and WW implemented the software and processed the data; formal statistical analyses were performed by HW, WW, and SM. WW managed the project and obtained funding; WW and WZ provided resources. HW, WW, and WZ validated the results and prepared the visualizations. HW and WZ drafted the original manuscript, and WW and SM critically reviewed and edited the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The current study was approved by the Ethics Committee of the Second Hospital of Anhui Medical University (approval number YX2024-164). Written informed consents from all patients were obtained in any experimental work with humans. The study was carried out in accordance with the guidelines of the Declaration of Helsinki.
We would like to express our gratitude to all those who helped us during the writing of this manuscript.
This research received no external funding.
The authors declare no conflict of interest.
During the preparation of this work, the authors used ChatGPT (OpenAI, San Francisco, CA, USA) to assist with grammar checking and language polishing. After using this tool, the authors carefully reviewed and edited the content as needed and take full responsibility for the final content of the manuscript.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/CEOG44257.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






