1 Department of Gynecology and Obstetrics, Women’s Hospital, Zhejiang University School of Medicine, 310006 Hangzhou, Zhejiang, China
†These authors contributed equally.
Abstract
Cervical polyps are often associated with localized inflammatory foci, which may be detected during pregnancy. In symptomatic cases, polypectomy currently represents the primary therapeutic intervention. However, the impact of cervical polyps on pregnancy outcomes and the clinical significance of cervical polypectomy remain subjects of ongoing debate. This study aimed to investigate the relationship between cervical polyps and pregnancy outcomes, focusing on spontaneous preterm birth (SPTB) and late miscarriage, and to evaluate the association of polypectomy with these outcomes. This retrospective study was conducted at a tertiary university-affiliated women’s hospital.
The study included 9990 consecutive women who underwent vaginal delivery, with or without cervical polyps, over a 12-month period from January to December 2021. All patients had undergone gynecological examination and transvaginal ultrasonography during early pregnancy. The diagnosis of cervical polyps in early pregnancy (4–12 gestational weeks) was determined through gross clinical inspection and confirmed by transvaginal ultrasound. Polypectomy should be considered in cases of heavy vaginal bleeding, secondary infection, excessively long polyps or prolapse of the vaginal orifice, and when cervical malignancy is strongly suspected. The associations of cervical polyps or polypectomy with late miscarriage and SPTB were evaluated using comparative analysis, as well as univariate and multivariate logistic regression.
A comparative analysis of pregnancy outcomes was performed between two groups: 94 (0.94%) cases with cervical polyps detected in the first trimester and 9896 cases without cervical polyps. The incidence of late miscarriage and SPTB was significantly higher in the polyp group than in the non-polyp group. Multivariate analysis revealed that cervical polyps in first trimester pregnancy was a significant independent risk factor for both late miscarriage (odds ratio [OR]: 96.94, 95% CI: 34.88–269.49, p < 0.001) and SPTB before 28 (OR: 31.48, 95% CI: 11.48–86.32, p < 0.001), 34 (OR: 26.13, 95% CI: 11.58–58.94, p < 0.001), or 37 (OR: 5.13, 95% CI: 2.59–10.17, p < 0.001) weeks of gestation. Our analysis demonstrated comparable pregnancy outcomes between the polypectomy and non-polypectomy groups, with no statistically significant association observed between cervical polypectomy and pregnancy outcomes in this cohort. Vaginal bleeding was identified as an independent protective factor for SPTB before 34 weeks of pregnancy in these patients (OR: 0.27, 95% CI: 0.09–0.83, p = 0.023).
Cervical polyps detected during the first trimester were associated with a significantly increased risk of both late miscarriage and SPTB; however, polypectomy did not significantly improve in pregnancy outcomes.
Keywords
- cervical polyps
- late miscarriage
- polypectomy
- spontaneous preterm birth
Cervical polyps, with an incidence of approximately 2%–5% in women of reproductive age, can also be identified during pregnancy [1]. They have the potential to trigger both miscarriage and premature delivery [2, 3], and their possible malignant transformation, which constitutes a significant clinical concern in reproductive medicine. We have consistently observed a notable association between cervical polyps in early pregnancy and subsequent occurrences of late miscarriage and preterm birth in obstetric practice. Cervical polyps in early pregnancy are a significant risk factor for spontaneous preterm birth (SPTB) in a retrospective study [3]. This raised concerns about polypectomy during pregnancy. However, opinions on whether to perform cervical polypectomy are different. According to a study that women who had cervical polypectomy during pregnancy had higher rates of SPTB compared with the general population [4]. Conversely, another study revealed that polypectomy procedures carry minimal risks of inducing miscarriage or preterm labor [5]. To date, the management of cervical polyps during pregnancy remains uncertain due to the lack of standardized clinical guidelines.
At our medical center, we observed that many women diagnosed in pregnancy of first trimester with cervical polyps exhibit higher rates of late miscarriage and SPTB. This pattern warrants investigation of a potential causal relationship. Notably, this study aimed to investigate the association between cervical polyps identified in early pregnancy and adverse pregnancy outcomes, specifically late miscarriage and SPTB (before 28, 34, or 37 weeks of gestation). Additionally, the study sought to evaluate the impact of cervical polypectomy on these pregnancy outcomes.
Patients were recruited from the Women’s Hospital, Zhejiang University, between
January 2021 and December 2021. The inclusion criteria were as follows: (1) Women
aged
All patients underwent gynecological examination and transvaginal ultrasonography during early pregnancy. Other clinical information was also collected, including age, body mass index (BMI), reproductive history (gestation, parturition, abortion, and preterm birth), history of conization, vaginal bleeding, and bacterial vaginosis. The diagnosis of cervical polyps in early pregnancy (4–12 gestational weeks) was made through gross inspection by the clinician and transvaginal ultrasound evaluation, including assessment of polyp location, morphological features, and vascularity to exclude endocervical polyps. Cervical polyps are soft, elastic growths that arise from the endocervical canal. They can vary in size, ranging from a grain of rice to a thumbnail.
The procedure settings were determined at the initiation of the case. In this retrospective study, women with cervical polyps in pregnancy of first trimester were categorized into a surgery group or a non-surgery group based on the interventions received. The surgical intervention comprised transvaginal cervical polypectomy. Before the procedure, the surgeon assessed each patient’s condition by considering clinical symptoms and ultrasonography findings, particularly the location and morphology of the polyps. Patients presenting with more severe clinical manifestations—such as significant vaginal bleeding, symptomatic bacterial vaginosis, and easily accessible polyps—were more likely to undergo polypectomy. All surgeons participating in this study had received rigorous standardized training, had been similarly evaluated, and had more than five years of experience performing this procedure. All polyp specimens removed during polypectomy were subjected to standard histopathological examination.
Cervical polyps were identified before 12 weeks of gestation. Prenatal assessments were conducted every 4 weeks until 28 weeks, every 2 weeks thereafter, and weekly from 36 weeks until delivery. The primary outcomes were gestational age at vaginal delivery and birth weight. Late abortion was clinically defined as spontaneous pregnancy loss occurring between 12 and 23 + 6 weeks of gestation. SPTB was defined as delivery occurring between 24 weeks and 36 + 6 weeks of gestation, resulting from either spontaneous onset of labor or preterm premature rupture of membranes, excluding medically indicated preterm deliveries. In addition, SPTB was divided into before 28 weeks, 34 weeks and 37 weeks. Because our hospital has limited capacity to rescue preterm births before 24 weeks of gestation, we excluded preterm births before 24 weeks.
For descriptive analysis, proportions were used for categorical variables and
means
The recruitment process is illustrated in Fig. 1. The retrospective study
included 10,344 women who delivered vaginally at the Women’s Hospital, School of
Medicine, Zhejiang University, between January 2021 and December 2021. Women with
multiple pregnancies (n = 116), intrauterine fetal death (n = 63), major fetal
malformations (n = 65), or major fetal complications (n = 110) were ruled out.
The rest of 9990 women were enrolled in our study. They were divided into two
groups: 94 women with cervical polyps in pregnancy of first trimester (P group)
and 9896 women absence of cervical polyps (non-P group). Women with cervical
polyps were further divided into the surgery group (n = 13) and the non-surgery
group (n = 81). The baseline obstetric characteristics of these women are
presented in Table 1. There was no significant difference between the non-polyp
and polyp groups with respect to age, BMI, multipara, previous late-term
abortion, SPTB before 34 weeks, or conization (p
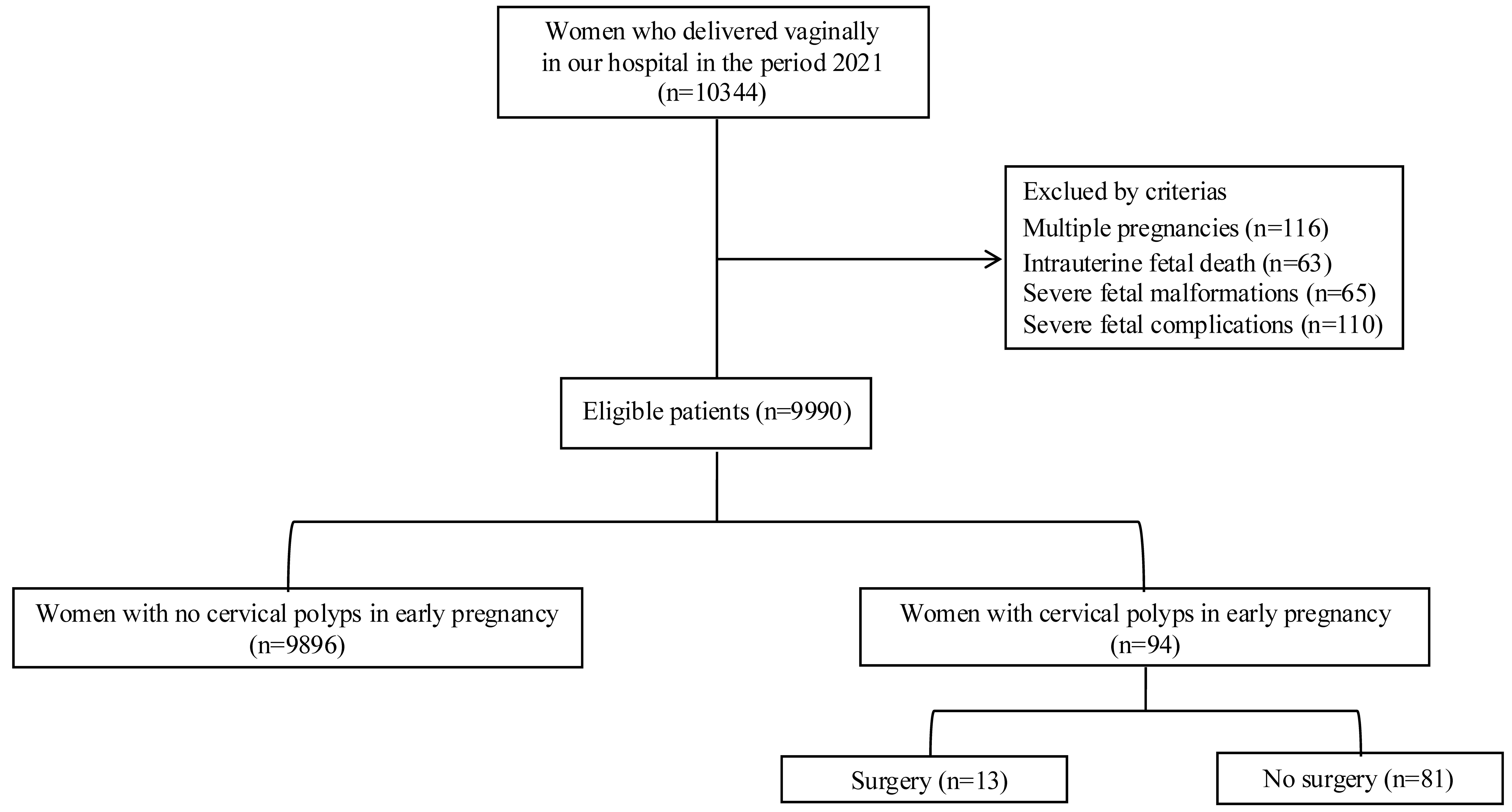 Fig. 1.
Fig. 1.
Flowchart of patients’ enrollment.
| Characteristics | Non-polyp (n = 9896) | Polyp (n = 94) | p |
| Age (years) | 30.27 |
30.95 |
0.078 |
| BMI (kg/m2) | 26.85 |
25.09 |
0.705 |
| Parous | 2785 (28.14) | 24 (25.53) | 0.575 |
| Late-term abortion history | 1000 (10.11) | 8 (8.51) | 0.609 |
| History of spontaneous preterm birth (SPTB) before 34 weeks | 900 (9.09) | 7 (7.45) | 0.580 |
| History of conization | 330 (3.33) | 3 (3.19) | 0.939 |
| Bacterial vaginosis | 231 (2.33) | 8 (8.51) |
Data are represented as means
**p
The comparison of pregnancy outcomes between the two groups is presented in
Table 2. Both gestational age (p
| Outcomes | Non-polyp (n = 9896) | Polyp (n = 94) | p |
| Gestational age at delivery, week | 38.90 |
33.46 |
|
| Birth weight, g | 3273.21 |
2365.28 |
|
| Late abortion | 12 (0.12) | 12 (12.77) | |
| SPTB before 28 weeks | 16 (0.16) | 8 (8.51) | |
| SPTB before 34 weeks | 55 (0.56) | 16 (17.02) | |
| SPTB before 37 weeks | 205 (2.07) | 15 (15.96) |
Data are represented as means
**p
We next compared factors associated with late abortion or SPTB by univariate and
multivariate analyses as presented in Table 3. Logistic regression revealed that
a late-term abortion history, previous SPTB before 34 weeks, and the presence of
cervical polyps were significant risk factors for adverse pregnancy outcomes.
Specifically, a late-term abortion history (OR: 62.14, 95% CI:
22.94–168.03, p
| Univariate analysis | Multivariate analysis | ||||
| OR (95% CI) | p | OR (95% CI) | p | ||
| Late abortion | |||||
| Age | 1.09 (0.93, 1.20) | 0.093 | |||
| Parous | 1.05 (0.40, 42.54) | 0.909 | |||
| Late-term abortion history | 74.32 (32.79, 168.47) | 62.14 (22.94, 168.03) | |||
| Previous SPTB before 34 weeks | 44.56 (17.20, 115.43) | 14.05 (4.63, 42.60) | |||
| History of conization | 0.00 | 0.997 | |||
| Bacterial vaginosis | 3.73 (0.84, 15.96) | 0.076 | |||
| Cervical polyps | 120.54 (52.60, 276.20) | 96.94 (34.88, 269.49) | |||
| SPTB before 28 weeks of pregnancy | |||||
| Age | 1.04 (0.94, 1.15) | 0.476 | |||
| Parous | 1.83 (0.81, 4.12) | 0.145 | |||
| Late-term abortion history | 52.53 (23.17, 119.04) | 33.11 (13.34, 82.22) | |||
| Previous SPTB before 34 weeks | 55.80 (22.47, 138.61) | 24.59 (8.62, 70.21) | |||
| History of conization | 11.67 (1.54, 88.67) | 0.018** | 0.94 (0.07, 12.89) | 0.981 | |
| Bacterial vaginosis | 5.89 (1.74, 19.88) | 0.004** | 5.93 (1.62, 21.67) | 0.007* | |
| Cervical polyps | 57.44 (23.94, 137.74) | 31.48 (11.48, 86.32) | |||
| SPTB before 34 weeks of pregnancy | |||||
| Age | 1.02 (0.94, 1.06) | 0.490 | |||
| Parous | 1.39 (0.94, 2.27) | 0.184 | |||
| Late-term abortion history | 66.59 (40.57, 109.32) | 63.40 (35.14, 114.39) | |||
| Previous SPTB before 34 weeks | 64.44 (36.22, 114.63) | 50.40 (23.24, 109.16) | |||
| History of conization | 7.96 (1.88, 33.70) | 0.005** | 0.88 (0.09, 8.30) | 0.914 | |
| Bacterial vaginosis | 3.14 (1.25, 7.86) | 0.015* | 2.62 (0.90, 47.20) | 0.062 | |
| Cervical polyps | 36.70 (20.14, 66.85) | 26.13 (11.58, 58.94) | |||
| SPTB before 37 weeks of pregnancy | |||||
| Age | 0.94 (0.94, 1.03) | 0.593 | |||
| Parous | 0.93 (0.71, 1.29) | 0.788 | |||
| Late-term abortion history | 28.23 (19.76, 40.34) | 25.13 (17.23, 36.66) | |||
| Previous SPTB before 34 weeks | 25.64 (15.79, 41.65) | 19.37 (11.12, 33.73) | |||
| History of conization | 2.48 (0.59, 10.37) | 0.213 | |||
| Bacterial vaginosis | 1.48 (0.51, 2.61) | 0.743 | |||
| Cervical polyps | 8.94 (5.04, 15.86) | 5.13 (2.59, 10.17) | |||
| Cervical cerclage | |||||
| Age | 1.04 (0.94, 1.11) | 0.209 | |||
| Parous | 1.04 (0.61, 1.92) | 0.774 | |||
| Late-term abortion history | 57.42 (33.23, 99.19) | 2.92 (0.74, 11.71) | 0.135 | ||
| Previous SPTB before 34 weeks | 29.94 (14.54, 61.71) | 4.53 (0.54, 38.13) | 0.205 | ||
| History of conization | 15.71 (4.68, 52.64) | 2.74 (0.51, 15.14) | 0.367 | ||
| Bacterial vaginosis | 2.28 (0.71, 7.34) | 0.167 | |||
| Cervical polyps | 15.84 (6.98, 35.93) | 1.50 (0.23, 14.33) | 0.765 | ||
*p
For further analysis, we compared pregnancy outcomes between the polypectomy
group and the non-polypectomy group. The baseline characteristics of these women
with cervical polyps in early pregnancy are presented in Table 4. No significant
difference was observed between the two groups in age, BMI, parity, a late-term
abortion history, previous SPTB before 34 weeks, previous conization, bacterial
vaginosis, vaginal bleeding, polyp size, or cervical cervix ligature (p
| Non-polypectomy group (n = 81) | Polypectomy group (n = 13) | p | |
| Age (years) | 30.74 |
31.46 |
0.520 |
| BMI (kg/m2) | 25.16 |
24.88 |
0.789 |
| Parous | 20 (24.69) | 5 (38.46) | 0.207 |
| Late-term abortion history | 8 (9.88) | 1 (7.69) | 0.735 |
| History of SPTB before 34 weeks | 8 (9.88) | 0 (0.00) | 0.213 |
| History of conization | 5 (6.17) | 0 (0.00) | 0.195 |
| Bacterial vaginosis | 9 (11.11) | 1 (7.69) | 0.614 |
| Vaginal bleeding | 48 (59.26) | 11 (84.62) | 0.065 |
| Polyp size ( |
38 (46.91) | 10 (76.91) | 0.045* |
| Polyp size (cm) | 1.56 |
2.09 |
0.117 |
| Cervical cervix ligature# | 5 (6.17) | 1 (7.69) | 1.000 |
Data are represented as means
#Yates’ corrected chi-squared test.
*p
| Non-polypectomy group (n = 81) | Polypectomy group (n = 13) | p | |
| Gestational age at delivery, week | 33.46 |
33.85 |
0.842 |
| Birth weight, g | 2370.14 |
2358.08 |
0.972 |
| Late-term abortion | 12 (14.81) | 1 (7.69) | 0.349 |
| SPTB before 28 weeks of pregnancy | 9 (11.11) | 1 (7.69) | 0.614 |
| SPTB before 34 weeks of pregnancy | 15 (18.52) | 3 (23.08) | 0.620 |
| SPTB before 37 weeks of pregnancy | 15 (18.52) | 1 (7.69) | 0.192 |
Data are represented as means
Table 6 presents the risk factors for late abortion and SPTB in the presence of
cervical polyps, confirmed by the univariate and the multiple logistic regression
analyses. A late-term abortion history was an independent risk factor for the
late-term abortion in these patients (OR: 7.03, 95% CI: 1.33–37.17, p =
0.022). Conversely, vaginal bleeding was identified as an independent protective
factor for SPTB before 34 weeks in these patients (OR: 0.27, 95% CI: 0.09–0.83,
p = 0.023). Additionally, no significant association was observed
between polyp size, polypectomy and pregnancy outcomes complicated by cervical
polyps (p
| Univariate analysis | Multivariate analysis | ||||
| OR (95% CI) | p | OR (95% CI) | p | ||
| Late abortion | |||||
| Age | 1.09 (0.93, 1.30) | 0.272 | |||
| Parous | 1.11 (0.27, 4.58) | 0.889 | |||
| Late-term abortion history | 7.03 (1.33, 37.17) | 0.022* | 7.03 (1.33, 37.17) | 0.022* | |
| Previous SPTB before 34 weeks | 4.17 (0.67, 26.05) | 0.127 | |||
| History of conization | 0.00 | 1.000 | |||
| Bacterial vaginosis | 0.00 | 0.991 | |||
| Vaginal bleeding | 1.02 (0.27, 3.77) | 0.982 | |||
| Polyp size ( |
2.88 (0.71, 11.64) | 0.139 | |||
| Polypectomy | 0.57 (0.07, 4.77) | 0.594 | |||
| SPTB before 28 weeks of pregnancy | |||||
| Age | 1.09 (0.90, 1.32) | 0.358 | |||
| Parous | 1.86 (0.41, 8.48) | 0.423 | |||
| Late-term abortion history | 1.81 (0.19, 17.24) | 0.606 | |||
| Previous SPTB before 34 weeks | 2.20 (0.23, 21.56) | 0.498 | |||
| History of conization | 5.71 (0.46, 71.14) | 0.176 | |||
| Bacterial vaginosis | 1.53 (0.16, 14.29) | 0.709 | |||
| Vaginal bleeding | 4.48 (0.53, 38.14) | 0.170 | |||
| Polyp size ( |
7.72 (0.91, 65.58) | 0.061 | |||
| Polypectomy | 0.83 (0.09, 7.39) | 0.870 | |||
| SPTB before 34 weeks of pregnancy | |||||
| Age | 0.98 (0.84, 1.14) | 0.793 | |||
| Parous | 0.96 (0.28, 3.35) | 0.955 | |||
| Late-term abortion history | 4.04 (0.81, 20.19) | 0.088 | |||
| Previous SPTB before 34 weeks | 2.50 (0.47, 15.03) | 0.316 | |||
| History of conization | 2.43 (0.20, 28.21) | 0.486 | |||
| Bacterial vaginosis | 1.62 (0.29, 8.87) | 0.579 | |||
| Vaginal bleeding | 0.27 (0.09, 0.83) | 0.023* | 0.27 (0.09, 0.83) | 0.023* | |
| Polyp size ( |
0.37 (0.12, 1.16) | 0.087 | |||
| Polypectomy | 1.48 (0.36, 6.12) | 0.591 | |||
| SPTB before 37 weeks of pregnancy | |||||
| Age | 1.00 (0.86, 21.57) | 0.989 | |||
| Mul parous | 0.74 (0.19, 3.02) | 0.701 | |||
| Late-term abortion history | 0.00 | 1.000 | |||
| Previous SPTB before 34 weeks | 1.09 (0.12, 10.13) | 0.938 | |||
| History of conization | 0.00 | 1.000 | |||
| Bacterial vaginosis | 0.76 (0.09, 6.69) | 0.803 | |||
| Vaginal bleeding | 1.05 (0.32, 3.44) | 0.936 | |||
| Polyp size ( |
0.68 (0.21, 2.13) | 0.503 | |||
| Polypectomy | 0.41 (0.05, 3.44) | 0.411 | |||
*p
Cervical polyps are common gynecologic conditions in reproductive-age women, typically arising from glandular epithelial hyperplasia and are commonly benign. To our knowledge, the association between the cervical polyps detected before 12 weeks of gestation and reproductive outcomes has not been well elucidated, and no standardized clinical pathways exist for managing cervical polyps during pregnancy. This study included a large cohort of women who delivered vaginally with or without complications from cervical polyps, and followed their reproductive outcomes—specifically gestational age and birth weight, which are crucial factors for couples preparing for childbirth.
In our study, we identified that cervical polyps in early pregnancy negatively impacted pregnancy outcomes, as reflected in gestational age and birth weight, consistent with previous studies [2, 3, 4]. Furthermore, we demonstrated that a late-term abortion history, previous SPTB before 34 weeks, and the presence of cervical polyps were independent risk factors for late-term abortion and SPTB occurring before 28, 34, and 37 weeks, respectively. In addition, bacterial vaginosis was an obvious independent risk factor for SPTB before 28 weeks.
We hypothesized that the elevated risk of miscarriage and preterm delivery in
patients with cervical polyps may be mediated by the following mechanisms. First,
cervical polyps may trigger chronic inflammation in the cervical and uterine
microenvironment, stimulating pro-inflammatory mediators [for example,
interleukin (IL)-6, tumor necrosis factor-
In this study, a history of cervical conization was not identified as a risk factor for late-term miscarriage or SPTB, but this conclusion may be subject to potential bias. This is because our analysis only included data on cervical cerclage among patients with cervical polyps, without a comprehensive statistical comparison of cervical cerclage status in most patients without polyps. A late-term abortion history and previous SPTB were identified as independent risk factors for the recurrence of late-term abortion and SPTB in the current gestation. No significant association was found between the placement of cervical cerclage and the following factors: A late-term abortion history, occurrence of SPTB before 34 weeks of gestation, previous cervical conization procedures, or presence of cervical polyps. The main causes of late miscarriage include cervical insufficiency, infections, structural abnormalities of the uterus, placental disorders, fetal anomalies, endocrine disturbances, immune factors, and adverse lifestyle behaviors. SPTB is primarily caused by preterm labor (accounting for 40%–50% of cases) or premature rupture of membranes (20%–30%) and occasionally by cervical insufficiency or placental abruption. However, we did not conduct a detailed individual analysis of these specific factors, which may introduce potential biases into our findings. Previous research has demonstrated that cervical polyps served as significant risk factors of cervical incompetence, often necessitating cerclage of the cervix [9]. According to international clinical guidelines, cervical cerclage is recommended for singleton pregnancies in cases with previous SPTB before 34 weeks coupled with cervical length shortening to less than 25 mm before 24 weeks [10]. Further robust, large-scale research is warranted to optimize cervical cerclage strategies and evaluate their efficacy in improving perinatal outcomes, with a focus on patient selection criteria, timing of intervention, and long-term maternal and neonatal benefits.
Currently, treatment approaches vary due to the lack of standardized guidelines for cervical polyps in early pregnancy. In clinical practice, polypectomy is considered in cases of heavy vaginal bleeding, secondary infection, excessively long polyps or prolapse at the vaginal orifice, or highly suspected cervical malignant transformation. These criteria also guided the surgical indications in this study.
Our results revealed no significant correlation between undergoing polypectomy
or polyp size in pregnancy and the occurrence of the late-term abortion or SPTB.
In contrast, the proportion of cervical ligature was comparable between the
polypectomy and non-polypectomy groups. Several studies have suggested that
polypectomy during pregnancy may adversely affect pregnancy outcomes [4].
Similarly, Zhang et al. [11] highlighted that the use of vaginoscopy
reduced the incidence of preterm birth and premature rupture of membranes
compared to conventional polypectomy techniques in women with symptomatic
cervical polyps. Notably, the timing of polypectomy may be a critical factor
influencing clinical outcomes. Fukuta et al. [4] reported that
polypectomy at
We observed that vaginal bleeding emerged as an independent protective factor against SPTB before 34 weeks of gestation in patients with cervical polyps. This unexpected finding, which deviates from conventional clinical expectations, may be explained by heightened clinical vigilance and the more aggressive tocolytic interventions typically employed when managing patients with cervical polyps accompanied by vaginal bleeding. It is also possible that the sample size of patients with pregnancy complicated by cervical polyps in this study was relatively small, leading to potential deviations. Further large-scale, prospective studies are warranted to validate the precise impact of vaginal bleeding on pregnancy outcomes and to establish evidence-based clinical guidelines for its management in antenatal care.
Our observed cervical polyp prevalence of 0.94% was indeed lower than population estimates, while the 54.25% adverse outcome rate appeared elevated. This may be partially attributed to the experience of the initial examining physician, as cervical polyps can occasionally be misidentified as simple inflammatory hyperplasia during routine gynecological examination, resulting in missed diagnoses. However, even when the cervix was solely inflamed or when polyps spontaneously resolved or were no longer visible during pregnancy, these conditions may still elevate the risk of late miscarriage and SPTB [2]. That’s to say, non-bleeding polyps may have been misclassified as inflammatory changes during initial exam. On the other hand, symptomatic patients were more likely to be referred to our tertiary center. The differences in prevalence and outcome rates likely reflected selection bias inherent in our clinical cohort. Besides, our primary inclusion criterion was vaginal delivery, with subsequent stratification into polyp (n = 94) and non-polyp (n = 9896) groups based on documented clinical and ultrasound findings. The lack of polyp size/symptom specifications represents a study limitation, which might introduce a degree of selection bias and manifest as a crucial limitation. Specifically, the proportion of polyps larger than 1.2 cm in the polypectomy group was significantly higher than that in the non-polypectomy group (p = 0.045). To mitigate the influence of confounding factors, we conducted a multivariate logistic analysis to explore factors associated with late abortion and SPTB, thereby improving accuracy and reliability by simultaneously considering multiple variables affecting the outcomes. We adjusted for obstetric history and vaginal bleeding in the multivariate analysis, while other potential confounding were unmeasured, such as cervical inflammation, intrauterine infection, cervical incompetence. Further rigorous contrivable studies are required to verify our findings and the impact of these factors on reproductive outcomes in patients with cervical polyps.
This study has some limitations. First, existing evidence from prior studies
indicates a potential association between cervical polyps during pregnancy and
increased susceptibility to cervical incompetence in specific female populations.
Unfortunately, we did not investigate the cervical lengths, Bishop scores, and
cervical cerclage among these patients, which limited our capacity to thoroughly
evaluate the potential impact of cervical polyps on cervical function. Moreover,
the degree of cervical inflammation was not quantified or assessed using specific
inflammatory markers, such as IL-6 and TNF-
In summary, our findings suggested that cervical polyps detected in the first trimester significantly compromise pregnancy outcomes, as evidenced by reduced gestational age, increased late miscarriage and SPTB, and lower birth weight. Notably, subsequent polypectomy intervention failed to mitigate these adverse effects. An adequately powered randomized controlled trial is required to confirm our findings and to conduct a more comprehensive evaluation of the factors influencing clinical decision-making, including baseline clinical presentations, polyp characteristics (such as size, morphology, and location) and detailed histopathological subtypes.
The data that support the findings of this study are not publicly available due to privacy reason but are available from the corresponding author upon reasonable request.
Concept or design: TF; Acquisition of data: TF, TW; Analysis or interpretation of data: TF, TW; Drafting of the article: TF, TW; Critical revision for important intellectual content: TF. Both authors contributed to editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was carried out in accordance with the guidelines of the Declaration of Helsinki and was approved by the Ethics Committee of Women’s Hospital, Zhejiang University School of Medicine (no. IRB-20230022-R). The written consent was signed by the patients or their families.
The authors acknowledge all patients for their cooperation during the follow-up period.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

