1 Department of Obstetrics and Gynecology, Regional Training Research Hospital, Health Sciences University, Erzurum, Turkey
2 Department of Pathology, Faculty of Medicine, Ataturk University, Erzurum, Turkey
3 Department of Biochemistry, Faculty of Medicine, Erzincan Binali Yildirim University, Erzincan, Turkey
4 Department of Pharmacology, Faculty of Medicine, Erzincan Binali Yildirim University, Erzincan, Turkey
5 Department of Nursing, Faculty of Health Sciences, Erzincan Binali Yildirim University, Erzincan, Turkey
Abstract
Purpose: The aim of the study was to examine the possible effect of Rhodiola rosea root extract (RRE) on ovarian damage, infertility (IF), delay in conception (DC), decrease in the number of offspring (NO) in female rats that have been stressed with compulsory immobilization method (CIM) and on retardation of intrauterine physical development (IPD) in offspring. Materials and methods: Female Albino Wistar rats were divided like: healthy (HG), stress-induced control (SC), and stress + 50 mg/kg RRE (S + REG) groups including sixteen rats in each group. Six rats from each group were killed for conducting biochemical and histopathological examinations in ovarian tissues and others were observed for IF, DC, NO, and IPD. Results: It was found that RRE significantly prevented the increase in oxidative parameters and proinflammatory cytokine levels in ovarian tissue of animals stressed with CIM. RRE suppressed the increase in IF rate and DC whereas caused a decrease in NO and in IPD retardation. Conclusion: These results suggested RRE may have beneficial effects in the treatment of stress induced reproductive dysfunction. Content: The effect of RRE on oxidative and inflammatory changes in ovarian tissue and reproductive changes of animals exposed to stress with compulsory immobilization method.
Keywords
- Ovarian damage
- Stress
- Infertility
- Rhodiola rosea root
Infertility is known as the failure to achieve pregnancy after 12 months of unprotected intercourse in the literature [1]. Abnormalities such as tube damage, ovulation and implantation disorders are responsible for the infertility in women. These abnormalities leading to infertility are related with psychological stress [2]. Research has shown that chronic stress results in permanent and marked reproductive dysfunction, an increase in embryo resorption, and a decrease in conception rate [3, 4]. In fact, it is expressed in the literature that long-term or uncontrolled stress can adversely affect human health, including fertility [1]. It is also reported that stress-induced depression and anxiety can lead to infertility [5, 6]. Epidemiological studies have shown that depression and anxiety disorders occur simultaneously [7, 8]. Some of the studies show the increase of oxidative stress in depression and anxiety [9, 10]. In depressive disorders, an increase in the inflammatory response parallel to the increase in the production of proinflammatory cytokines is seen [11, 12]. Experimental studies have also shown that proinflammatory cytokines increase in anxiety disorders [13]. Therefore, oxidative stress and inflammation have been recognized among the basic reasons of reproductive dysfunction in many studies [14]. The act of oxidative stress and inflammation in pathologies like spontaneous abortions, embryopathies, preeclampsia, fetal growth restriction, preterm, and miscarriage affecting female reproductive processes is well known [15, 16]. This information suggests that an antioxidant, anti-inflammatory, antidepressant and anxiolytic agent may be effective in the treatment of stress-induced infertility and pathologies affecting reproductive processes. Rhodiola rosea root extract (RRE) used in the study is obtained from the root of Rhodiola rosea L. plant [17], which grows in the northern parts of Europe, Asia and the US. Preclinical studies have reported that RRE has anti-stress, neuropsychotropic, anxiolytic, antidepressant and neuroprotective effects [17, 18]. In addition, Rhodiola rosea extract has antioxidant and anti-inflammatory effects [19, 20]. No information was found in the literature about the effects of RRE in the treatment of stress-induced infertility and pathologies affecting reproductive processes. The present study aims to investigate the possible effect of RRE on ovarian damage, infertility (IF), delay in conception (DC), decrease in number of offspring (NO) and intrauterine physical development (IPD) retardation in experimental stress-induced rats.
Forty-eight female albino Wistar rats weighing 265-272 grams were used in this study. The rats were obtained from Ataturk University Experimental Application and Research Center. The rats were housed and fed at standard room temperature (22 ℃) before the experiment. The protocols and procedures were approved by the local Animal Experimentation Ethics Committee (Date: 27/06/2019, meeting no.: 7/120).
The ketamine was supplied from Pfizer (Turkey), and Rhodolia rosea root extract was supplied from Solgar (United States).
Experimental animals were grouped as: healthy (HG), stress control (SC), and stress + RRE (S + REG) groups.
Widely known compulsory immobilization method (CIM) was used in order to create a stress model for animals [21]. All animals (except for the HG group) were brought to the supine position, feet and arms were tied and held in the same position for 24 hours to implement CIM. 50 mg/kg of RRE was orally applied to S + REG (n-16) rats at the end of this period. 50 mg/kg of RRE has also showed biological activity in previous studies [20]. Distilled water was administered to the SC (n-16) and HG (n-16) group animals in the same way and in equal doses as in S + REG group. CIM procedure was applied for three times with 10-day intervals. RRE was given once a day for 30 days. Then, six rats (n-6) from each group were killed with high dose anesthesia (50 mg/kg thiopental sodium) and ovarian tissues were taken for biochemical and histopathological examinations. The remaining animals were kept with mature male rats for two months in appropriate laboratory conditions for reproduction. The rats being pregnant during experimental process were taken to separate cages and kept in a suitable environment. Rats that were not conceived and gave birth within two months were accepted as infertile. DC, NO and IPD were also investigated. Furthermore, delay in maternity period was calculated by deducting the pregnancy period (21 days) from the period as of the coitus of the female and male rats until the delivery (A) (A-21 = delay in maternity period). The decrease in the number of offspring (NO) obtained from the S+REG group was compared with HG. Weights of puppies born in SC and S + REG groups were compared with HG to assess the intrauterine physical development of puppies [21]. Experimental results were compared between groups and evaluated accordingly
All tissues were rinsed with phosphate buffered saline solution prior to dissection. The liver tissues were homogenized in ice-cold phosphate buffers (50 mM, pH 7.4) appropriate for the variable to be measured [22]. The tissue homogenates were centrifuged at 4,000 rpm for 30 min at 4 ℃, and the supernatants were extracted to analyze, malondialdehyde (MDA), Total Glutathione (tGSH), Total Anti-oxidant Status (TAS) and Total Oxidant Status (TOS), Nuclear Factor-Kappa B (NF-κB) and protein concentration. The protein concentration of the supernatant was measured using the method described by Bradford MM. The results were expressed by dividing to g protein [23]. All spectrophotometric measurements were performed via microplate reader (Bio-Tek, USA).
The method used by Ohkawa et al. was considered for MDA assessment [24]. This method is based on the spectrophotometric assessment of the absorption of the pink-colored complex created by thiobarbituric acid (TBA) and MDA at high temperature (95 ℃) at 532 nm wavelength. The amounts of MDA of the samples were determined by using the standard graphics formed with the MDA stock solution previously prepared considering the dilution coefficients.
The DTNB (5.5’-Dithiobis (2-nitrobenzoic acid)) in the assessment environment is a disulphide chromogen and is easily reduced by sulfhydryl group compounds. The resultant yellow color was spectrophotometrically assessed at 412 nm [25]. The GSH amounts of the samples were determined by using the standard graphics formed with the GSH stock solution previously prepared considering the dilution coefficients.
TOS and TAS levels of tissue homogenates were determined using a novel automated measurement method and commercially available kits (Rel Assay Diagnostics, Turkey) developed by Erel [26, 27]. The results were expressed as µmol Trolox equivalent/L. The percentage ratio of TOS to TAS was used as the oxidative stress index (OSI). OSI was calculated as TOS divided by 100 × TAS.
Tissue-homogenate NF-κB concentrations were measured using rat-specific sandwich enzyme-linked immunosorbent assay Rat Nuclear Factor- Kappa B ELISA immunoassay kits (Cat. No.: 201-11-0288, SunRed). Analyses were performed according to the instructions of the manufacturer. Briefly, monoclonal antibody specific rat NF-κB was coated onto the wells of the micro plates. The tissue homogenate, standards and biotinylated monoclonal antibody specific and streptavidin-HRP were pipetted into these wells and then incubated at 37 ℃ for 60 min. After washing, chromogen reagent A and chromogen reagent B were added acting by the bound enzyme to produce a blue color. It was incubated at 37 ℃ for 10 min. Then H2SO4 was added. The intensity of yellow color formed was directly proportional to the concentration of rat NF-κB present in the original specimen. The well plates were read at 450 nm and the absorbance of the samples was calculated with formulations using standard graphics at the end of the course.
All of the tissue samples were first fixed in a 10% formaldehyde solution for light microscope assessment. Following the identification process, tissue samples were washed under tap water in cassettes for 24 hours. Samples were then treated with conventional grades of alcohol (70%, 80%, 90%, and 100%) to remove the water within tissues. Tissues were then passed through xylol and embedded in paraffin. Four-to-five micron sections were cut from the paraffin blocks and hematoxylin-eosin staining was administered. Their photos were taken following the Olympus DP2-SAL firmware program (Olympus® Inc. Tokyo, Japan) assessment. Histopathological assessment was carried out by the pathologist blind for the study groups.
All statistical analyses were performed using “SPSS statistical software” (Version 18) and p values < 0.05 were considered significant. The results were expressed as “mean ± standard error of mean” (x ± SEM). The significance of the difference between the groups was determined using one-way analysis of variance (ANOVA) test followed by post-hoc Tukey test.
1)The results of MDA and tGSH
As can be seen in Figure 1, the amount of MDA significantly increased in the ovarian tissue of the animals treated with immobilization method compared to healthy group and REG group (p < 0.001). The amount of MDA in REG group was not statistically significant according to healthy controls (p > 0.05). Stress significantly decreased the amount of tGSH in the ovarian tissue of the animals compared to healthy and REG group (p < 0.001). The amount of tGSH in REG group was statistically insignificant compared to healthy controls (p > 0.05).
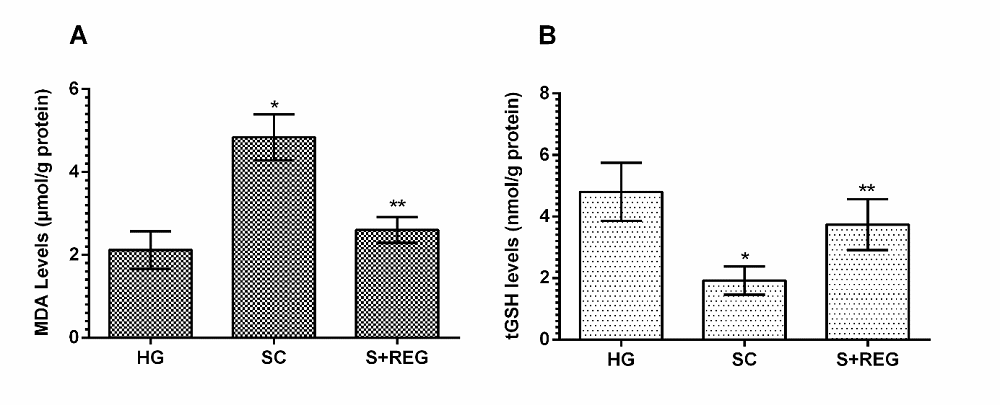 Figure 1.
Figure 1.— MDA and tGSH levels in ovarian tissues of experimental groups. * p < 0.001, according to HG and S + REG group, ** p > 0.05, according to HG group.
2)The results of TOS and TAS analysis
It was found that the balance between TOS and TAS levels in the ovarian tissue of stress-treated animals changed in favor of TOS. The TOS level in the stress-treated group increased significantly compared to the healthy group and REG group (p < 0.001), whereas the TAS level decreased significantly (p < 0.001). As seen in Figure 2, the TOS levels in the ovarian tissue of RRE administered animals were not statistically significant compared to healthy group (p > 0.05). Moreover, the TAS levels in the ovarian tissue of RRE administered animals were close to healthy group (p = 0.048).
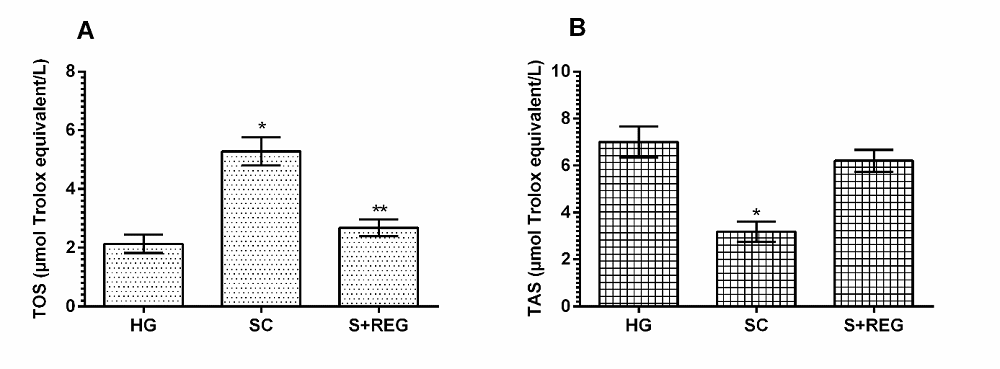 Figure 2.
Figure 2.— Oxidant/antioxidant balance in the ovarian tissues of experimental groups. * p < 0.001, according to HG and S + REG group, ** p > 0.05, according to HG group.
3)NF-κB analysis results
Stress caused an increase in NF-κB levels in the ovarian tissue of the animals. NF-κB levels significantly increased in the ovarian tissue of the stressed group compared to healthy and REG group (p < 0.001) as can be seen in Figure 3.
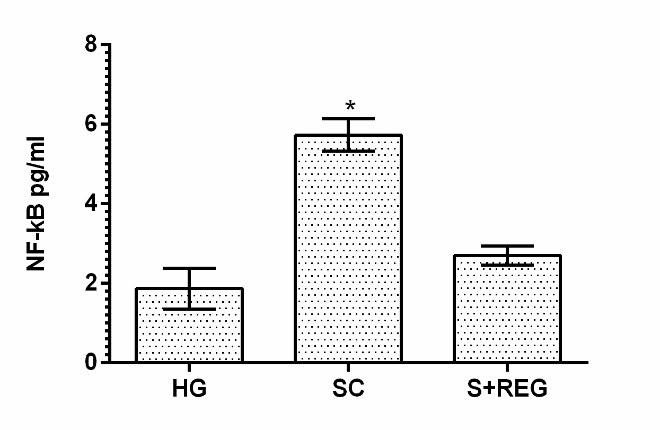 Figure 3.
Figure 3.— NF-κB levels in in the ovarian tissues of experimental groups. * p <0.001, according to HG and S + REG group.
As shown in Table 1, two of the ten reproductive female rats in SC group gave birth whereas the remaining 8 female rats did not give birth in two months. The mean duration of maternity of the animals in the SC group was 42 days in SC group and 27 days in S + REG group. 7 of 10 female rats gave birth and 3 of them were infertile. All female rats in HG group gave birth within 24 days.
| Groups | Male pups | Female pups | Total weight | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n | Mean ± SD | p* | p** | n | Mean ± SD | p* | p** | n | Mean ± SD | |||
| HG | 31 | 5.20 ± 0.47 | - | <0.001 | 54 | 4.90 ± 0.66 | - | <0.001 | 85 | 5.01 ± 0.62 | ||
| SC | 10 | 3.90 ± 0.42 | < 0.001 | - | 5 | 3.70 ± 0.61 | < 0.001 | - | 15 | 3.83 ± 0.48 | ||
| S + REG | 30 | 5.00 ± 0.79 | 0.629 | <0.001 | 26 | 4.70 ± 0.44 | 0.605 | 0.001 | 56 | 4.91 ± 0.66 | ||
HG: healthy group, SC: stress control group, S + REG: stress + RRE group, * Pearson Chi-Square test.
** One-way analysis of variance (ANOVA) test followed by post-hoc Tukey test.
As can be seen in Table 2, two female rats gave birth to 10 males, with the average weight of 3.9 g, and 5 female offspring weighing 3.7 g in the SC group. 7 female rats who were pregnant gave birth to 3 males with the average weight of 5.0 g and 26 female pups with the average weight of 4.7 grams in the REG group. In the SG animal group, 10 pregnant rats gave birth to an average of 5.2 grams, 31 male and 54 female pups weighing 4.9 grams.
| Groups | Non-infertile rats n (%) | Infertile rats n (%) | p* | Delay in maternity period (days) | p** |
|---|---|---|---|---|---|
| HG (n = 6) | 10 (100) | 0 (0.0) | 0.001 | 24 21 = 3 | 0.000 |
| SC (n = 6) | 2 (20) | 8 (80) | 42 21 = 21 | ||
| S + REG (n = 6) | 7 (70) | 3 (30) | 27 21 = 6 |
HG: healthy group, SC: stress control group, S + REG: stress + RRE group, SD: standard deviation.
*according to HG group, ** according to SC group.
Figure 4A shows normal corpus luteum, developing follicular structures, and subcapsular area in ovarian tissue of the healthy group. In the ovarian tissue of the stressed animals, diffuse congestion, hemorrhage, sporadic accumulation of fluid and inflammatory cell clusters/infiltrates in the subcapsular area, and necrotic areas of the stroma in the parenchyma were detected (Figure 4B). Mild congestion was observed in the ovarian tissue of the RRE treated group. Fluid accumulation and inflammatory cell infiltrates in the subcapsular area were not observed. In addition, mild necrotic areas were found in the stroma in the parenchyma, follicle structure, and corpus luteum (Figure 4C).
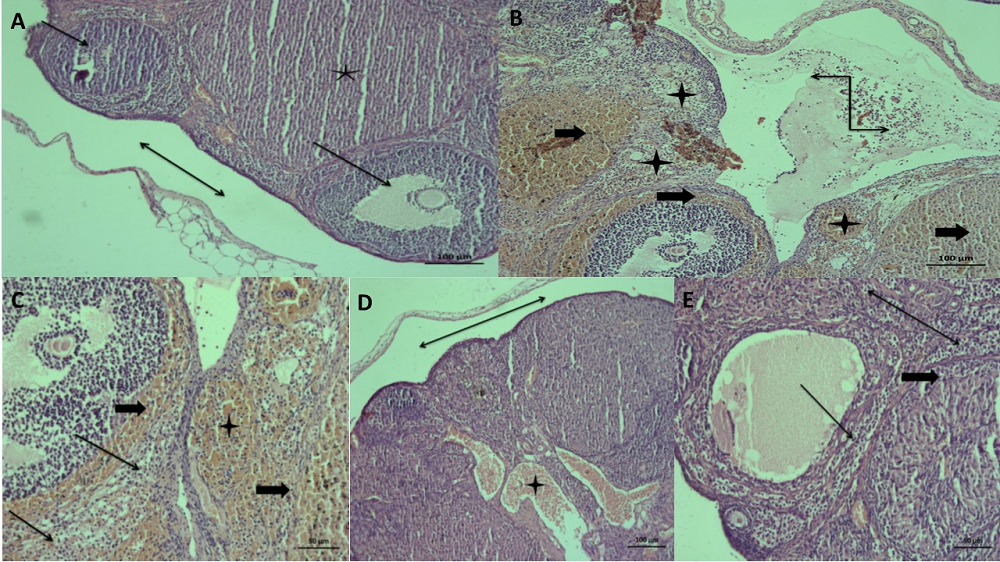 Figure 4.
Figure 4.— Histopathological changes of the experimental groups. (A) The ovarian tissue section of healthy group: the corpus luteum (star), developing follicular structures (arrows), subcapsular area (bidirectional arrow) (H&E, × 100). (B) Ovarian tissue of the stressed group section: diffuse congestion (stars) and hemorrhage (block arrows), fluid accumulation and inflammatory cell infiltrate in subcapsular area (black zigzag arrows), (H&E, × 100). (C) Ovarian tissue of the stressed group section: necrotic changes (arrow), congestion (star) and hemorrhage (block arrow) (H&E, × 200) (D) The ovarian tissue of the RRE-treated group: mild congestion (asterisk), subcapsular area without fluid accumulation and inflammatory cell infiltrate (bidirectional arrow) (H&E, × 100). (E) Ovarian tissue section of the RRE-treated group showing mild necrosis in the parenchymal stroma (bidirectional arrow), follicular structure (arrows) and corpus luteum (block arrow) (H&E, × 200).
The possible effects of RRE on ovarian damage and reproductive process disorders were experimentally analyzed in female rats in the study. Our biochemical results showed that MDA, TOS and NF-κB levels in ovarian tissue of animals stressed with CIM increased whereas tGSH and TAS levels decreased compared to healthy and RRE groups. These results indicate that experimental stress changes the oxidant/antioxidant balance in the ovarian tissue in favor of oxidants. This situation is expressed as oxidative stress in the literature [28, 29]. MDA being high in ovarian tissue of the stressed group is a toxic product of lipid peroxidation initiated by reactive oxygen species (ROS) causing cellular damage [30, 31]. It has been reported that serum MDA levels of patients with anxiety and various psychological disorders are higher than that of healthy controls [32, 33] and they return to normal with citalopram treatment [10]. The amount of tGSH in the ovarian tissue of the stressed group with elevated MDA was found to be lower compared to healthy and RRE groups in the present study. GSH which is an endogenous antioxidant molecule in tripeptide form detoxifies hydrogen peroxide and organic peroxides and protects cells from ROS damage [34]. A decrease in plasma antioxidant system has been reported in rats exposed to stress [35]. The oxidant/antioxidant balance in the ovarian tissue of the stressed group was also shown to be impaired in favor of oxidants by measuring TOS and TAS levels. TOS is used to determine the cumulative oxidative effects of various oxidants in biological systems [27], while TAS is used to determine the cumulative antioxidative effects of antioxidants [26]. In the present study, it was also observed that NF-κB levels significantly increased in ovarian tissue of stressed animals compared to healthy and RRE groups. As is known, NF-κB is an inducible transcription factor that plays a central role in immune and inflammatory responses [36]. Activated by excessive mechanical stress, NF-κB stimulates the release of inflammatory cytokines such as IL-1β, TNF- and IL-6 in cells [36, 37]. These cytokines increase in stress, anxiety and depressive disorders, and also increase the inflammatory response in the ovarian tissue [11-13]. Oral administration of RRE antagonized the stress-induced increase of MDA, TOS and NF-κB in ovarian tissue and decrease of tGSH and TAS. This effect of RRE may be due to its anti-stress, antidepressant and anxiolytic properties. In a clinical study, it was observed that the stress response decreased in patients treated with RRE [38]. In another clinical study, it was reported that RRE had an antidepressant effect [39]. It has also been found in animal models that RRE has an anxiolytic effect [17]. In the literature, it has been reported that the antioxidant effect of RRE is due to the active ingredient of oligomeric proanthocyanide in the content of Rhodiola rosea [40]. The fact that RRE inhibited stress-induced NF-κB increase in ovarian tissue indicates that it has both antioxidant and anti-inflammatory effect. It is known that inflammatory response increases parallel to the increase in production of cytokines in stress, depression and anxiety disorders [11-13].
It has been shown that stress results in reproductive dysfunction, increased embryo resorption, and decreased pregnancy rate in many studies [3, 4]. In our study, infertility developed in 80% of the stress group animals with high oxidant and NF-κB levels. Administration of RRE reduced stress-induced infertility in animals by 70%. These experimental results are consistent with the following literature data. Previous studies have shown that psychological disorders, oxidative stress and inflammation are associated with infertility [1, 5, 6, 14]. Aksoy et al. have showed that oxidative stress is one of the main causes of infertility due to ovarian I/R injury [41]. Unlubilgin et al. have stated that proinflammatory cytokines play a role in the development of infertility in rats [42]. The benefit of Rhodiola rosea extract in the treatment of various psychological disorders is well known [17, 18]. In addition, RRE has been shown to have antioxidant and anti-inflammatory effect, and inhibitory effect on IL-1β, TNF-α and IL-6 production [19, 20, 42]. Data showing that psychological disorders negatively affect duration of conception are available in the literature [43]. In our study, duration of conception was prolonged in the stress group where oxidative and proinflammatory cytokine levels elevated compared to the healthy and RRE treated groups. This shows that oxidative stress and inflammation associated with psychological stress delay conception time. Aksoy et al. also showed that oxidative stress adversely affected the duration of conception [41]. Unlubilgin et al. have showed that the increase in proinflammatory cytokine levels in ovarian tissue prolongs duration of conception [42]. In the present study, it was found that the decrease in the number of offspring in the stress group was directly proportional to the increase in oxidant and proinflammatory parameters. The fact that the decrease in the number of offspring is related to oxidative stress is supported by previous studies [44]. Moreover, significant IPD retardation was found in animals exposed to stress with CIM in the present study. Weights of the pups born in the stressed group were found to be significantly lower compared to that of healthy and RRE-treated groups. Birth weight is considered to be an indicator of IPD [45, 46]. It has been reported that high oxidant parameters and low antioxidant parameters in ovarian tissue negatively affect IPD [40]. Furthermore, the role of oxidative stress and inflammation in pathologies such as embryopathies and fetal growth restriction is well known [15, 16]. The fact that oxidant and proinflammatory cytokine levels in ovarian tissue of animals treated with RRE is close to that of the healthy group and that the birth weight of the offspring is less affected by stress coincides with the literature. Diffuse congestion, hemorrhage, subcapsular fluid accumulation and inflammatory cell infiltrates, and necrotic areas in the parenchymal stroma were detected in the ovarian tissue of the stress group where oxidative stress and inflammation markers showed a significant increase. However, ovarian tissue treated with RRE was found to be normal except for mild congestion and mild necrotic areas. Oxidative stress and proinflammatory cytokine elevation have been reported to cause hemorrhage, congestion, follicle degeneration, inflammatory cell infiltration and necrosis in ovarian tissue. In addition, it has been noted that this histopathological damage causes infertility and delayed conception in animals [41].
In conclusion, we have demonstrated that oxidative and inflammatory damage develops in ovarian tissue of animals exposed to stress with CIM by using biochemical and histopathological methods. CIM-related stress negatively affected fertility, duration of conception, number of offspring and IPD in female rats. RRE reduced the severity of stress-related oxidative and inflammatory damage in the ovarian tissue. In addition, RRE antagonized the negative effect of stress on reproductive function and IPD. These results suggest that RRE may be useful in the treatment of ovarian damage and reproductive dysfunction due to psychological stress.
CIM, Compulsory immobilization method; DC, Delay in conception; IF, Infertility; IPD, Intrauterine physical development; MDA, Malondialdehyde; NF-κB, Nuclear Factor- Kappa B; NO, decrease in the number of offspring; tGSH, Total glutathione; GSHRd, Glutathione reductase; RRE, Rhodiola rosea root extract; TAS, Total anti-oxidant status; TOS, Total oxidant status.
We would like to express my gratitude to all those who helped me during the writing of this manuscript. Thanks to all the peer reviewers and editors for their opinions and suggestions.
The authors declare no conflict of interest.




