- Academic Editor
†These authors contributed equally.
The substrates for arrhythmias in myocarditis and ischemic heart disease (IHD) are different, but it is yet to be determined whether there is a difference in outcomes following catheter ablation (CA) for ventricular tachycardia (VT) associated with these two conditions. This study aimed to compare outcomes after CA of VT in patients with myocarditis versus those with IHD.
Patients undergoing CA for sustained VT confirmed by endomyocardial biopsy as myocarditis, and patients with IHD experiencing sustained VT undergoing CA were retrospectively enrolled from February 2017 to March 2023. Initially, an endocardial approach was employed, reserving epicardial ablation procedures for non-responders. The primary endpoint was VT recurrence during follow up. All-cause mortality, repeat CA for VT and implantable cardioverter-defibrillator (ICD) implantation served as secondary endpoints. Kaplan-Meier curves compared outcomes between patient groups.
This study included 109 patients with IHD and 20 patients with myocarditis who underwent CA for sustained VT, from February 2017 to March 2023. Compared with IHD patients, myocarditis patients had a statistically significant lower complete short-term success rate of CA (60.0% vs. 85.3%, p = 0.013). During a follow-up period of 37 ± 21 months, 8 (40.0%) myocarditis patients experienced VT recurrence compared to 57 (52.3%) IHD patients, with no statistically significant difference between the two groups. During follow-up, 2 (10.0%) myocarditis patients died and 2 (10.0%) underwent repeat CA for VT recurrence, while 9 (8.3%) IHD patients died, 14 (12.8%) underwent a second CA for VT recurrence, and 8 (7.3%) received an ICD implantation. Additionally, there were no notable variations between the two groups regarding all-cause mortality, repeat CA for VT and ICD implantation.
It was demonstrated that the efficacy of CA in sustained VT in myocarditis patients was similar to that in IHD. For myocarditis patients with VT, CA might be equally effective.
Myocarditis, defined as inflammatory injury of the myocardium that can involve the cardiac conduction system and pericardial layers [1], affects approximately 10 to 22 people per 100,000 per year globally [2]. Previous studies have suggested that the probability of ventricular tachycardia (VT) after myocarditis is 6% [3] and the incidence of ventricular arrhythmias is as high as 55% in some specific types of myocarditis [4]. The VT may lead to an adverse short-term prognosis and is also a common mechanism of cardiac death [5, 6].
Recent study indicates that catheter ablation (CA) for VT is both effective and safe [7]. Studies in VT ablation in ischemic heart disease (IHD) patients have shown encouraging results [8, 9, 10]. However, data on myocarditis is scarce and most studies included a mix of patients without histological validation. The recurrence rates for VT in myocarditis patients who underwent CA range from 23–34% [11, 12]. The VT in myocarditis relates to inflammation, while that in IHD is linked to scarring [13, 14]. The arrhythmia substrates vary between these conditions. Therefore, there may be differences in ablation outcomes. However, fewer studies have compared CA outcomes for VT in myocarditis patients versus those with IHD. This study was conducted retrospectively to compare the outcomes after CA of VT in patients with myocarditis confirmed by histological validation and IHD.
This is a single-center retrospective study. This study consecutively enrolled myocarditis patients confirmed by endomyocardial biopsy (EMB) who underwent CA for sustained VT at the Fuwai Hospital, Chinese Academy of Medical Sciences from February 2017 to March 2023. Patients with IHD undergoing CA for sustained VT during the same period were included for comparison. Clinical presentations, family history, comorbidities, 12-lead electrocardiogram (ECG) results, and EMB of all patients were obtained from the electronic medical record system.
Sustained VT was defined as either lasting more than 30 seconds or necessitating termination within 30 seconds due to hemodynamic compromise [15]. The diagnosis of myocarditis was based on the pathological criteria of the current guideline [16]. EMB was performed via the right femoral vein to obtain myocardial tissue from the right ventricular septum. Myocarditis was pathologically diagnosed and staged according to the Dallas criteria [17]. In general, acute myocarditis showed myocardial cell necrosis and inflammation activation, while chronic myocarditis involved both destruction and remodeling. For patients with myocarditis, EMB was performed concurrently with CA using Jawz 2.2 mm Forceps, Maxi-Curved, 105 cm (Argon Medical Devices, Frisco, TX, USA). IHD was a cardiovascular condition characterized by diminished myocardial blood flow resulting from coronary artery disease [18].
Prior to the ablation procedure, all patients provided informed consent and were
prepared following the standard clinical protocol of our department. Mapping and
CA procedures were carried out under local anesthesia and sedation. ECG
monitoring was conducted continuously throughout the procedure. A decapolar
steerable electrode catheter was introduced into the coronary sinus through
femoral venous access, while a standard fixed-curve quadripolar catheter was
positioned in the right ventricle. Ventricular programmed or incremental
stimulation was used to induce clinical VT until the ventricular refractory
period was reached or VT onset occurred. For inducible and tolerable VTs,
activation mapping and entrainment mapping were performed to identify critical
isthmuses of reentry; for uninducible or unstable VTs, substrate mapping under
sinus rhythm were performed. Electroanatomic mapping was conducted using either
the CARTO 3D electroanatomical mapping system (Biosense Webster, Diamond Bar, CA,
USA) or the Ensite Precision 3D electroanatomical mapping system (Abbott
Laboratories, St. Paul, MN, USA). Endocardial mapping-guided ablation was
performed in all patients. For those in whom endocardial ablation failed,
epicardial mapping via subxiphoid pericardial puncture was considered. The
arrhythmogenic substrates comprised split electrograms, low voltage (
Ablation was conducted with radiofrequency energy, set at a target temperature of 45 °C and a maximum power of 50 W, utilizing irrigation at a flow rate of 12–30 cc/min. The ablation catheters used for catheter ablation were the FlexibilityTM ablation catheter (Abbott Laboratories, St. Paul, MN, USA) or the THERMOCOOL SMARTTOUCH ablation catheter (Biosense Webster, Diamond Bar, CA, USA). For ablations guided by activation mapping, the effectiveness of the ablation is evaluated post-procedure by repeating ventricular stimulation. If no VT can be induced, the ablation is defined as successful. If clinical VT cannot be induced, but other VT morphologies are inducible, the ablation is considered partially successful. If clinical VT is still inducible, the ablation is deemed a failure. For ablation guided by substrate mapping, the procedure target was eliminating all arrhythmogenic substrates.
Patients were followed up through phone calls or clinic visits at 3, 6, and 12
months after discharge, and then annually thereafter. Regular telephone
interviews were conducted with patients or their family members as well. At each
follow-up, patients underwent 12-lead ECG and 24-hour Holter monitoring to
identify arrhythmias. For those with an implantable cardioverter-defibrillator
(ICD), device checks were performed every 6 months. The primary endpoint of this
study was recurrent VT. VT recurrence was defined as sustained VT (duration
The normality of the data was assessed using the Kolmogorov–Smirnov test.
Continuous variables were expressed as means
| Unmatched | Matched | ||||||||
| IHD | Myocarditis | p | SMD | IHD | Myocarditis | p | SMD | ||
| n = 109 | n = 20 | n = 20 | n = 12 | ||||||
| Male | 101 (92.7%) | 13 (65.0%) | 0.002 | 0.72 | 16 (80.0%) | 8 (66.7%) | 0.673 | 0.305 | |
| Age, years | 60 |
43 |
1.59 | 50 |
49 |
0.743 | 0.12 | ||
| Hypertension | 60 (55.0%) | 4 (20.0%) | 0.008 | 0.776 | 10 (50.0%) | 3 (25%) | 0.307 | 0.535 | |
| Diabetes | 29 (26.6%) | 0 | 0.02 | 0.851 | 6 (30.0%) | 0 | 0.102 | 0.926 | |
| CKD | 6 (5.5%) | 2 (10.0%) | 0.793 | 0.169 | 1 (5.0%) | 2 (16.7%) | 0.639 | 0.382 | |
| ICD history | 29 (26.6%) | 5 (25.0%) | 1 | 0.037 | 2 (10.0%) | 4 (33.3%) | 0.242 | 0.591 | |
| CA history | 14 (12.8%) | 3 (15.0%) | 1 | 0.062 | 3 (15.0%) | 2 (16.7%) | 1 | 0.046 | |
| Smoking | 67 (61.5%) | 6 (30.0%) | 0.018 | 0.666 | 11 (55.0%) | 4 (33.3%) | 0.41 | 0.447 | |
| Drinking | 43 (39.4%) | 3 (15.0%) | 0.065 | 0.571 | 6 (30.0%) | 3 (25.0%) | 1 | 0.112 | |
| LVEF, % | 48 |
54 |
0.016 | 0.559 | 52 |
53 |
0.883 | 0.051 | |
| NYHA III/IV | 14 (12.8%) | 3 (15.0%) | 1 | 0.062 | 0 | 1 (8.3%) | 0.793 | 0.426 | |
| VT with CHD | 98 (89.9%) | 17 (85.0%) | 0.797 | 0.149 | 17 (85.0%) | 10 (83.3%) | 1 | 0.046 | |
| Medicine | |||||||||
| Amiodarone | 61 (56.0%) | 12 (60.0%) | 0.929 | 0.082 | 8 (40.0%) | 6 (50.0%) | 0.854 | 0.202 | |
| β-blocker | 89 (81.7%) | 16 (80.0%) | 1 | 0.042 | 15 (75.0%) | 9 (75.0%) | 1 | ||
CA, catheter ablation; CHD, compromised hemodynamics; CKD, chronic kidney disease; ICD, implantable cardioverter defibrillator; IHD, ischemic heart disease; LVEF, left ventricular ejection fraction; NHYA, New York Heart Association; VT, ventricular tachycardia; SMD, standardized mean differences.
Event-free survival was assessed using the Kaplan–Meier method and analyzed
with the log-rank test. Cox proportional hazard modeling was then performed,
incorporating potential confounders identified from significant univariate
associations (p
During the study period, 109 IHD patients and 20 myocarditis patients with
proven EMB were enrolled (Fig. 1). The mean age was 57
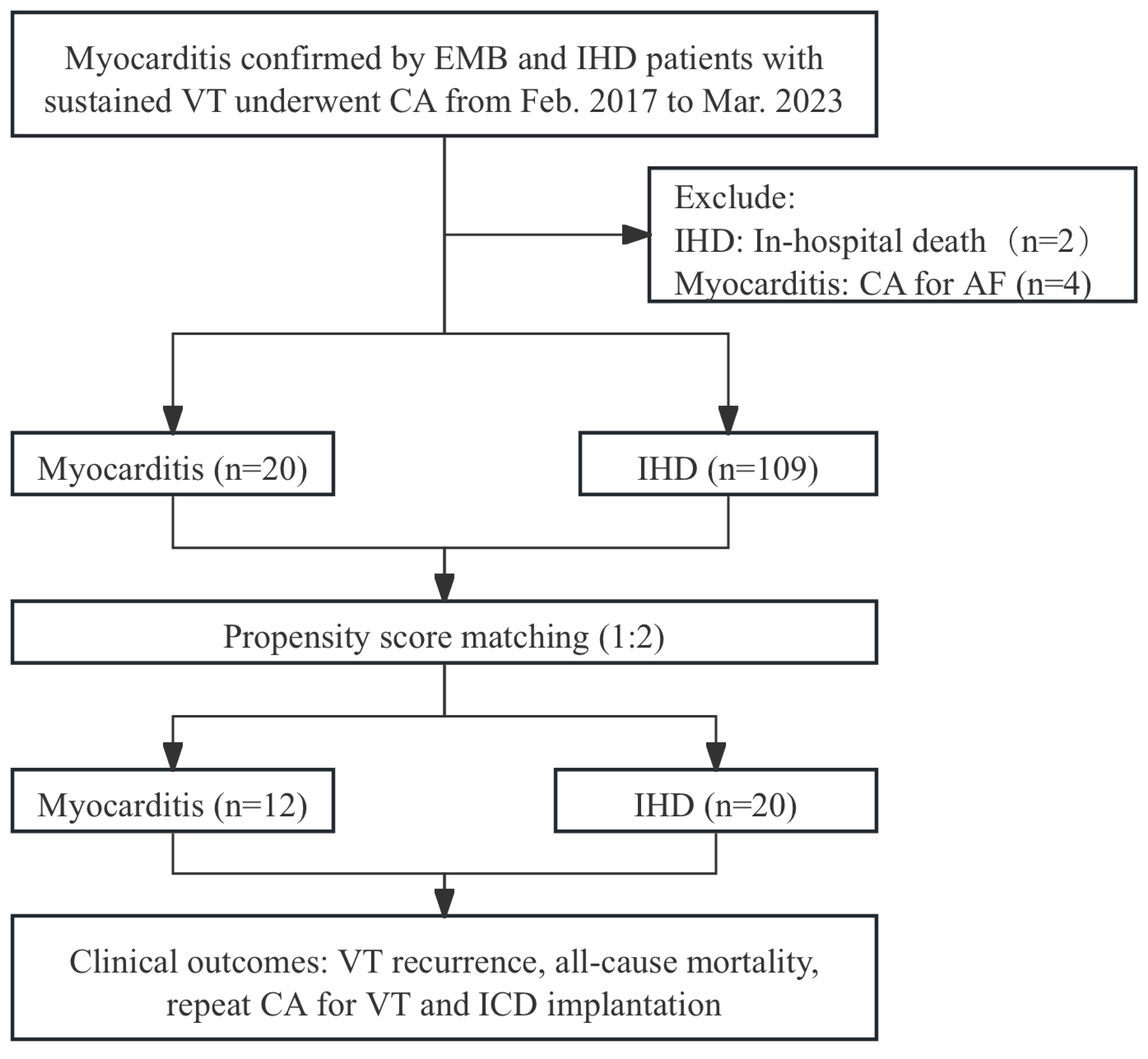 Fig. 1.
Fig. 1.
Study flowchart. AF, atrial fibrillation; CA, catheter ablation; EMB, endomyocardial biopsy; ICD, implantable cardioverter-defibrillator; IHD, ischemic heart disease; VT, ventricular tachycardia.
In this study, activation mapping was performed in 21 (16.3 %) patients and substrate mapping was performed in 100 (77.5%) patients. 8 (6.2%) patients underwent epicardial mapping due to failed endocardial ablation. During the procedures, 20 (15.5%) patients experienced hemodynamically unstable VT requiring defibrillation. Table 2 presented the electrophysiological findings and the specific origin of VT was in Supplementary Table 1. Out of the 129 patients, 105 (81.4%) achieved complete success with CA, 20 (15.5%) experienced partial success, and 4 (3.1%) encountered failure. 121 (93.8%) patients underwent endocardial CA and 8 (6.2%) patients underwent epicardial CA. Figs. 2,3 show the cardiac electrophysiological findings of a patient with myocarditis and a patient with IHD, respectively. In terms of CA complications, 3 patients developed pericardial effusion, with 1 requiring pericardial drainage. During hospitalization, 13 (10.1%) patients underwent ICD implantation following CA. Patients declined ICD implantation primarily because of economic concerns, but also out of fear of potential complications.
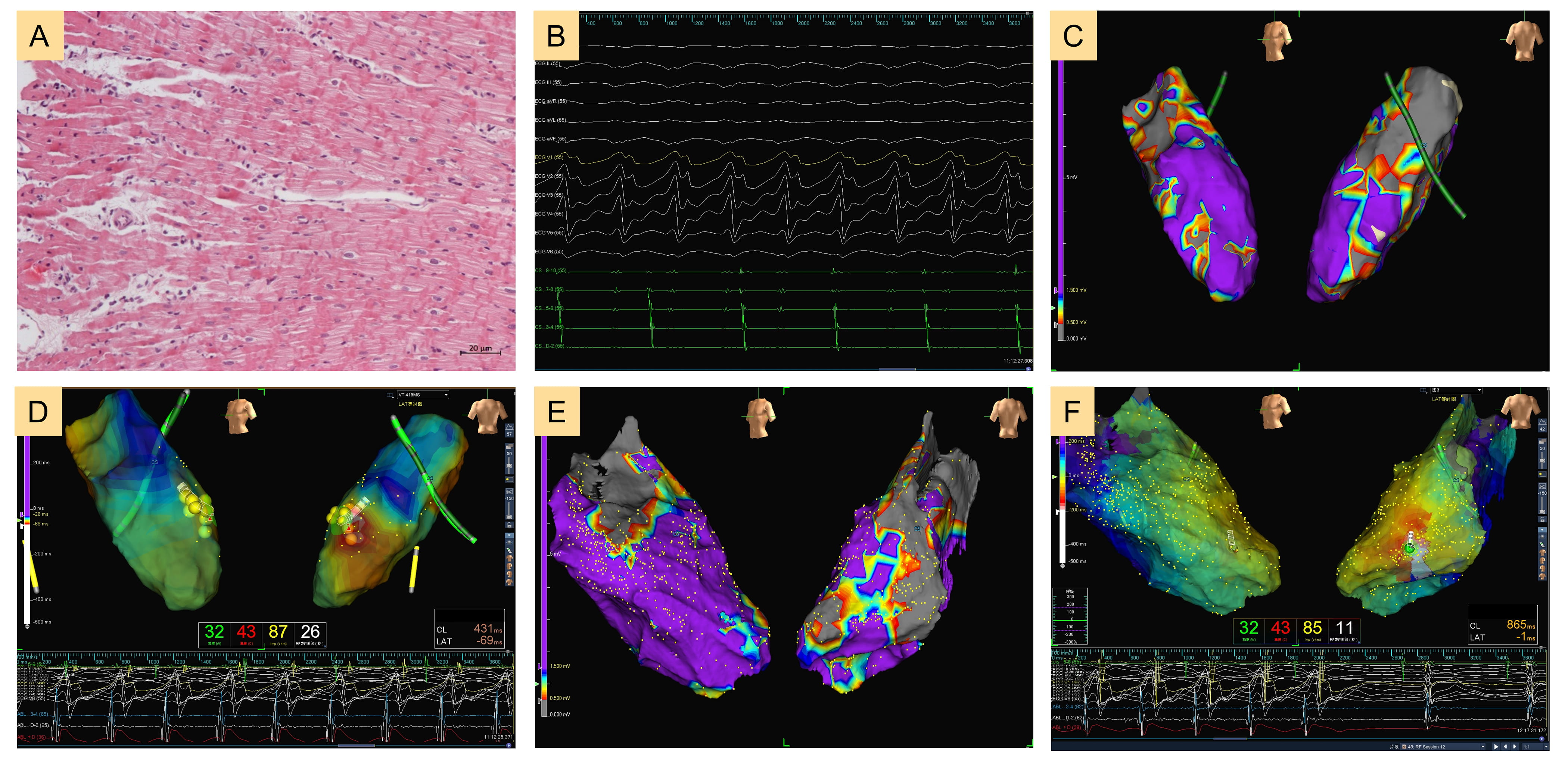 Fig. 2.
Fig. 2.
Sustained ventricular tachycardia in a patient with myocarditis. (A) The endomyocardial biopsy finding. Scale bar: 20 µm. (B) The electrocardiogram of sustained ventricular tachycardia. (C) Left ventricular endocardial substrate mapping results. (D) Activation mapping results and failure of ventricular tachycardia ablation. (E) Left ventricular epicardial substrate mapping results. (F) Activation mapping results and success of ventricular tachycardia ablation. ABL, ablation; CL, cycle length; LAT, local activation time; ECG, electrocardiogram; CS, coronary sinus.
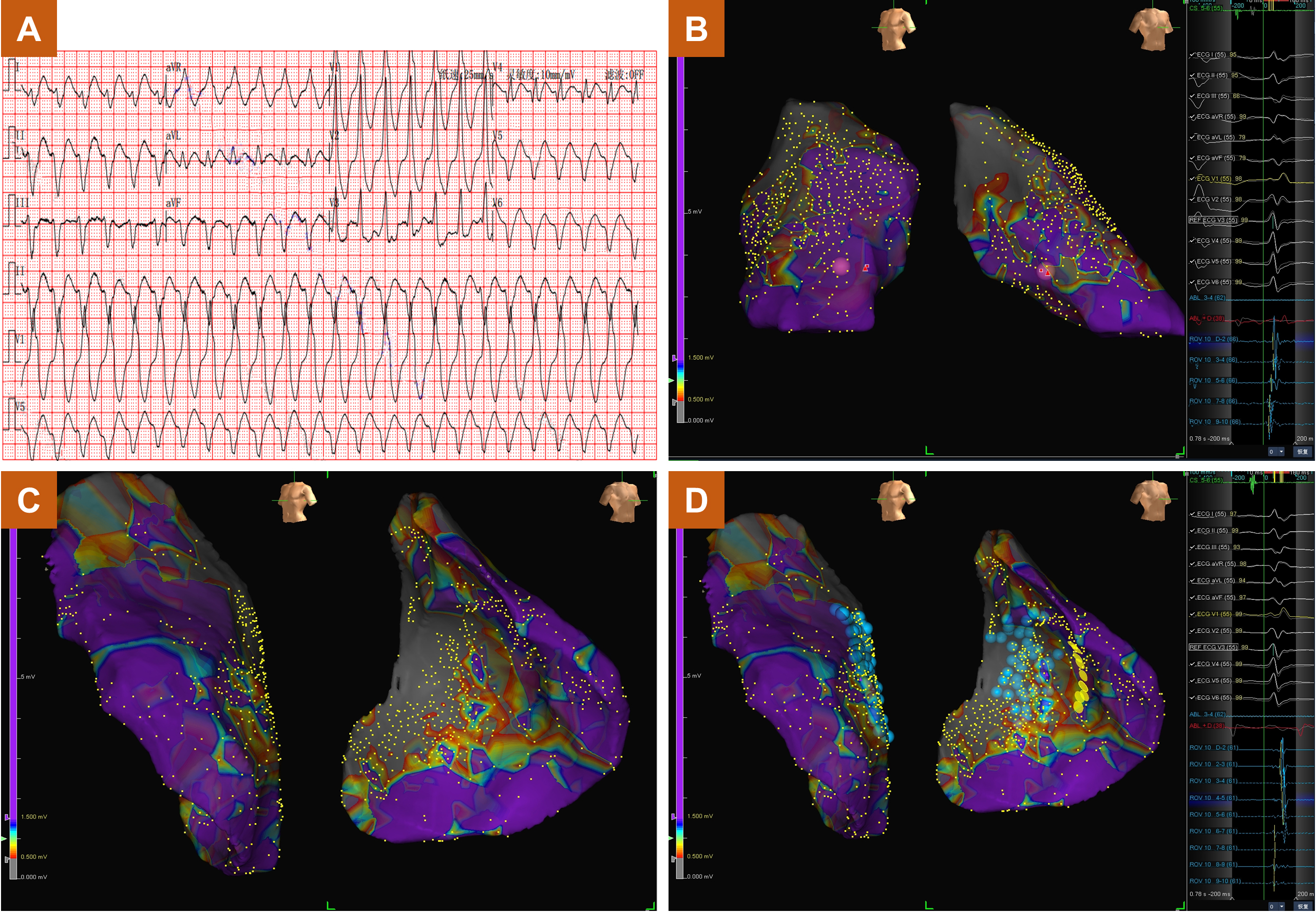 Fig. 3.
Fig. 3.
Sustained ventricular tachycardia in a patient with ischemic heart disease. (A) The electrocardiogram of sustained ventricular tachycardia. (B) The endocardial substrate mapping results. (C) The epicardial substrate mapping results. (D) Successful ablation based on epicardial delayed potentials. ABL, ablation; ECG, electrocardiogram; REF, reference electrode; CS, coronary sinus.
| IHD | Myocarditis | p | IHD | Myocarditis | p | ||
| n = 109 | n = 20 | n = 20 | n = 12 | ||||
| Activation mapping, n (%) | 9 (8.3%) | 12 (60.0%) | 3 (15.0%) | 7 (58.3%) | 0.002 | ||
| Substrate mapping, n (%) | 95 (87.2%) | 5 (25.0%) | 15 (75.0%) | 3 (25.0%) | 0.006 | ||
| Epicardial mapping, n (%) | 5 (4.6%) | 3 (15.0%) | 0.107 | 2 (10.0%) | 2 (16.7%) | 0.620 | |
| VTs in the procedure | |||||||
| 1 | 77 (70.6%) | 9 (45.0%) | 0.038 | 16 (80.0%) | 6 (50.0%) | 0.119 | |
| 2 | 14 (12.8%) | 5 (25.0%) | 0.174 | 2 (10.0%) | 3 (25.0%) | 0.338 | |
| 18 (16.5%) | 6 (30.0%) | 0.208 | 2 (10.0%) | 3 (25.0%) | 0.338 | ||
| VT CL, ms | 334 |
331 |
0.682 | 321 |
340 |
0.156 | |
| VT origin | |||||||
| Left ventricular | 99 (90.8%) | 8 (40.0%) | 16 (80.0%) | 5 (41.7%) | 0.053 | ||
| Right ventricular | 5 (4.6%) | 9 (45.0%) | 2 (10.0%) | 5 (41.7%) | 0.073 | ||
| Epicardial origin | 5 (4.6%) | 3 (15.0%) | 0.107 | 2 (10.0%) | 2 (16.7%) | 0.620 | |
| Defibrillation | 19 (17.4%) | 1 (5.0%) | 0.120 | 3 (15.0%) | 0 | 0.274 | |
IHD, ischemic heart disease; VT, ventricular tachycardia; VT CL, ventricular tachycardia cycle length.
Patients with myocarditis more commonly underwent activation mapping and less
frequently undergo substrate mapping, compared with IHD patients (Table 2). Of
note, myocarditis patients had a significantly lower rate of complete success
than IHD patients [12 (60.0%) vs. 93 (85.3%), p = 0.013] (Table 2).
However, the CA failure rates of the two groups of patients did not show a
statistically significant difference. There was no notable contrast in terms of
CA complications and ICD implantation when comparing patients with IHD to those
with myocarditis (all p
| IHD | Myocarditis | p | IHD | Myocarditis | p | |||
| n = 109 | n = 20 | n = 20 | n = 12 | |||||
| In-hospital | ||||||||
| CA result | ||||||||
| failure | 4 (3.7%) | 0 | 1 | 0 | 0 | - | ||
| partial success | 12 (11.0%) | 8 (40.0%) | 0.003 | 1 (5.0%) | 4 (33.3%) | 0.053 | ||
| complete success | 93 (85.3%) | 12 (60.0%) | 0.013 | 19 (95.0%) | 8 (66.7%) | 0.053 | ||
| CA complication, n (%) | 1 (0.9%) | 2 (10.0%) | 0.095 | 0 | 1 (8.3%) | 0.793 | ||
| ICD implantation, n (%) | 12 (11.0%) | 1 (5.0%) | 0.677 | 1 (5.0%) | 1 (8.3%) | 1 | ||
| Follow-up | ||||||||
| VT recurrence | 57 (52.3%) | 8 (40.0%) | 0.843 | 15 (75.0%) | 5 (41.7%) | 0.515 | ||
| Re-CA for VT recurrence | 14 (12.8%) | 2 (10.0%) | 0.799 | 8 (40.0%) | 1 (8.3%) | 0.257 | ||
| Death | 9 (8.3%) | 2 (10.0%) | 0.188 | 0 | 1 (8.3%) | 0.157 | ||
| Cardiac death | 7 (6.4%) | 2 (10.0%) | 0.145 | 0 | 1 (8.3%) | 0.157 | ||
| Anti-tachycardia pacing | 21 (19.3%) | 0 | 0.115 | 3 (15.0%) | 0 | 0.248 | ||
| ICD implantation | 8 (7.3%) | 0 | 0.312 | 3 (15.0%) | 0 | 0.281 | ||
| LVEF*, % | 47 |
54 |
0.077 | 55 |
51 |
0.449 | ||
| LVEF change*, % | 0 (–2, 3) | 2 (–1, 7) | 0.532 | 0 (–2, 2) | 0 (–1, 7) | 0.586 | ||
CA, catheter ablation; ICD, implantable cardioverter defibrillator; IHD, ischemic heart disease; LVEF, left ventricular ejection fraction; Re-CA, repeat-catheter ablation; VT, ventricular tachycardia. *, 93 patients had transthoracic echocardiography data during the follow-up.
In a follow-up with an average duration of 37
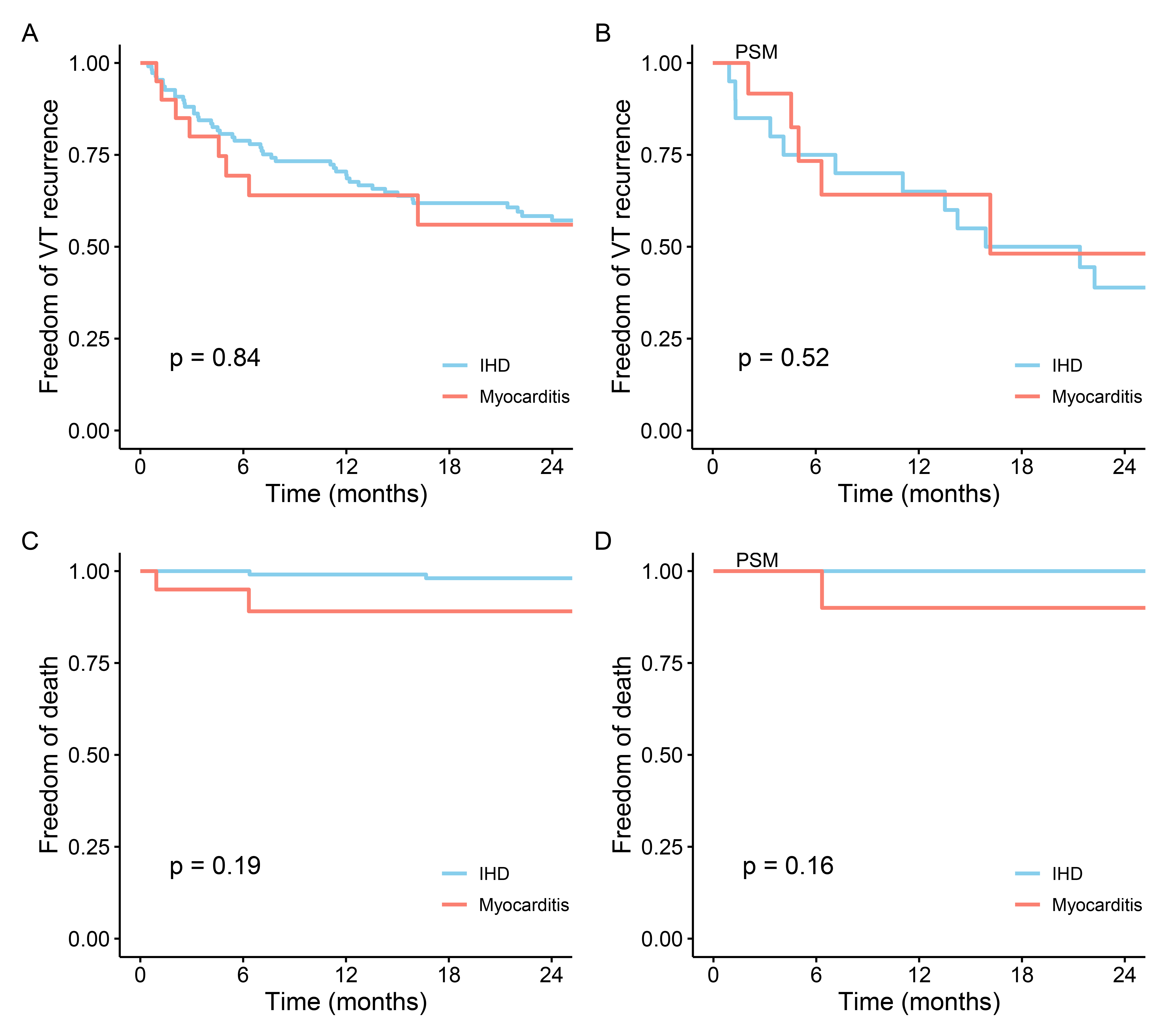 Fig. 4.
Fig. 4.
Comparison of follow-up outcomes after catheter ablation for IHD and myocarditis. (A) Comparison of recurrence of VT after catheter ablation in IHD and myocarditis among all patients. (B) Comparison of recurrence of VT after catheter ablation in IHD and myocarditis in the PSM cohort. (C) Comparison of death after catheter ablation in IHD and myocarditis among all patients. (D) Comparison of death after catheter ablation in IHD and myocarditis in PSM cohort. IHD, ischemic heart disease; VT, ventricular tachycardia; PSM, propensity score matching.
| Univariate | Multivariate | ||||
| HR (95% CI) | p | HR (95% CI) | p | ||
| Female | 0.487 (0.246, 0.965) | 0.039 | 0.498 (0.252, 0.985) | 0.045 | |
| NYHA III/IV | 1.253 (0.636, 2.468) | 0.515 | - | 0.931 | |
| Myocarditis | 1.078 (0.510, 2.278) | 0.844 | - | 0.774 | |
| CA outcomes | |||||
| Failure vs. complete success | 1.202 (0.291, 4.961) | 0.799 | - | 0.990 | |
| Partial vs. complete success | 0.666 (0.314, 1.412) | 0.289 | - | 0.143 | |
| Length of VT, ms | 0.995 (0.990, 1.000) | 0.056 | 0.995 (0.990, 1.000) | 0.034 | |
| Number of VT | 1.111 (0.936, 1.320) | 0.228 | - | 0.952 | |
| ICD history | 1.655 (0.977, 2.804) | 0.061 | 1.769 (1.042, 3.004) | 0.035 | |
| CA history | 1.368 (0.671, 2.788) | 0.388 | - | 0.340 | |
CA, catheter ablation; CI, confidence interval; HR, hazard ratio; ICD, implantable cardioverter defibrillator; NHYA, New York Heart Association; VT, ventricular tachycardia.
In the myocarditis cohort, 1 patient in each of the acute phase subgroup and
chronic phase subgroup suffered death, with no statistical difference observed
(p = 0.335). In the chronic myocarditis, 5 patients (33.3%) experienced
VT recurrence compared to 60.0% VT recurrence in the acute myocarditis, although
without statistical significance (p = 0.165). In addition, the
recurrence rates of VT in acute myocarditis and chronic myocarditis showed no
statistically significant difference compared to IHD (both p
There is limited data on the short- and long-term outcomes of VT ablation in myocarditis compared to IHD. This retrospective study compared VT catheter ablation outcomes in EBM-identified myocarditis and IHD patients and revealed similar results for both conditions. In addition, being male and having a longer VT cycle length were protective factors against VT recurrence.
Myocarditis is defined as inflammatory injury of the myocardium that can involve the cardiac conduction system and pericardial layers, and is generally mild and self-limited [16, 19]. However, patients can develop a temporary or permanent impairment of cardiac function including acute cardiomyopathy with hemodynamic compromise or severe arrhythmias. Ventricular arrhythmias were associated with sudden cardiac death [20]. According to current guidelines, patients diagnosed with myocarditis and experiencing VT may be considered for implantation of an ICD [15]. Complications associated with ICD implantation and inappropriate discharges should not be overlooked. Appropriate shocks result in discomfort, diminish quality of life, shorten device lifespan, and potentially elevate mortality rates [21]. Furthermore, many individuals were unable to undergo ICD implantation due to economic reasons [22, 23]. Catheter ablation is increasingly recognized as an effective treatment option for such arrhythmias, despite limited data on ventricular arrhythmias in the context of myocarditis [24].
In patients with myocarditis, the EMB showed an inflammatory infiltrate, along with necrosis or degeneration of neighboring myocytes [17]. The immune response may lead to electrophysiological or structural changes, causing abnormalities in action potential conduction or repolarization, thereby promoting the development of arrhythmias. The arrhythmogenic substrate of IHD is typically scar-related and commonly tends to be subendocardial (thus readily accessible for ablation) [7]. In myocarditis, the arrhythmogenic substrate is commonly found in an anteroseptal or inferolateral pattern, frequently affecting perivalvular, intramural, or epicardial areas, and the coronary arteries are typically patent [7]. In this study, the CA outcomes for both groups were similar, despite their different mechanisms of arrhythmogenic substrate formation. It has been reported that CA of VT in patients with non-ischemic cardiomyopathy (NICM) has been reported to have less favorable outcomes and higher VT recurrence rates as compared to IHD patients [25]. Previous study reported that myocarditis had superior outcomes than other kinds of NICM after adjusting for potential covariates [26]. In this study, there was no statistically significant difference observed in clinical outcomes after CA between the IHD cohort and the myocarditis cohort. For myocarditis patients with drug-refractory VT, CA is equally effective. Further research is needed to understand the specific mechanism.
In this study, complete elimination of any VT was achieved in 60.0% of myocarditis patients compared with 84.5% of the IHD patients. The complete short-term success rate of CA in myocarditis was lower than that in IHD patients. Of note, the success rate of CA in patients with chronic myocarditis was comparable to that in IHD patients, which hinted that acute myocarditis patients might be with a more complex arrhythmogenic substrate. Peretto Giovanni et al. [12] found that CA in the acute phase was a risk factor for early VT recurrence through an observational study. In this study, the short-term success rate following VT ablation in acute myocarditis was lower compared to chronic cases, and the recurrence of VT in acute myocarditis was higher than in chronic cases, although these differences did not reach statistical significance. For myocarditis patients with concomitant VT, a delayed CA strategy could be considered if clinically feasible. It is noteworthy that the follow-up outcomes for partial success and complete success were similar, with no statistically significant difference. Although pursuing complete elimination of all inducible VTs was desirable, ablation of the clinical VT only might be acceptable when achieving complete success was challenging. No significant difference in CA failure was observed between the two groups. These findings align with earlier studies and demonstrate relatively high immediate success rates for both myocarditis and IHD [24, 26].
Myocarditis patients had a significantly lower rate of substrate mapping, such as low voltage region, delay potential distribution and fragmentation potential distribution. The short-term success rates among different mapping methods were indistinguishable in the overall cohort and in myocarditis patients. No research study or meta-analysis has demonstrated superior outcomes with the conventional approach when contrasted with substrate-based ablation. For patients who failed endocardial ablation, equivalent therapeutic effects could be achieved through epicardial ablation. Therefore, a more sophisticated ablation strategy that integrates substrate mapping with reentry circuit characterization through activation mapping should be employed in myocarditis to improve short-term success rates. Our findings support the idea of considering epicardial ablation as a subsequent step if VT remains inducible following endocardial ablation in patients with myocarditis. Given the increased risk of complications associated with epicardial ablation, this approach may be more judicious than a mandatory combined endocardial and epicardial strategy, as recommended by some authors.
At long-term follow-up after CA, 47.8% of IHD patients and 60% of myocarditis patients were free from VT and the majority of VT recurrence occurred within one year after CA. This outcome aligns with already published data in the biggest multicenter trial [24]. In the Multicenter Thermocool Ventricular Tachycardia Ablation trial, the reported VT recurrence was 47% at 6 months [27]. Arenal Ángel et al. [10] found that CA decreased the composite endpoint of cardiovascular death, appropriate ICD shock, hospitalization for heart failure, or severe treatment-related complications compared to antiarrhythmic drugs (AADs). In this study, although a high rate of VT recurrence was observed, no significant association was found between VT recurrence and mortality. Not every VT recurrence was lethal. Furthermore, the necessity of repeat ablation and ICD implantation for patients experiencing VT recurrence may not be immediate. This may be related to the relatively good LVEF of the study population.
In this study, female gender was an independent risk factor for recurrent VT
after CA during follow-up. Distinct variations exist between women and men in the
manifestation, etiology, and therapeutic response to specific arrhythmias. An
international multicenter study suggested that women with structural heart
disease exhibit poorer VT-free survival post-ablation compared to men, despite
presenting more favorable baseline characteristics such as younger age, higher
LVEF, lower incidence of VT storm, and fewer medical comorbidities [28]. In this
study, the poorer prognosis seen in women might be attributed to a more complex
arrhythmia substrate, because women had a higher number of VTs than men (2.1
It is important to acknowledge that while VT ablation can modify the existing substrate temporarily, it may not prevent the ongoing progression of the underlying disease or the development of new substrate and triggers over time. The idea of a “fixed” morphological substrate may hold true in the context of post-infarction cardiomyopathy, but in cases of myocarditis, there are unidentified factors that contribute to the evolution and alteration of the arrhythmia substrate over time. Utilizing CA and AADs remains a crucial strategy to reduce the occurrence of VT and improve clinical symptoms in patients with myocarditis and VT. Not every patient with myocarditis and concurrent VT may require an ICD, and strict patient selection criteria are needed for ICD implantation. For myocarditis patients who already have an ICD, the indication for ICD removal can be assessed based on the occurrence of VT and ICD discharges during follow-up. Epicardial biomaterials, as a potential therapeutic approach, may also play a role in the treatment of VT in patients with myocarditis in the future [29].
There exists limitations. Firstly, this study is a single-center, retrospective, nonrandomized study. In addition, the sample size included in this study was relatively small, and the follow-up period of 37 months was relatively short. Secondly, this study represents a population in the earlier stages of left ventricular remodeling and impaired systolic function. Consequently, the results for patients receiving ablation therapy at advanced stages of the disease may differ from those reported in this study. The influence of the stimulation site on scar localization was not considered. The inability to induce VT with programmed stimulation both at the start and end of the ablation procedure in certain patients creates uncertainty around the definition of short-term success in these cases and may affect the overall short-term outcomes.
Although the short-term success rates after VT ablation in myocarditis was significantly lower than that in IDH, the follow-up outcomes were similar. Less substrate mapping and more epicardial mapping was performed in myocarditis patients. For myocarditis patients with VT, CA might be equally effective.
The datasets used during the current study are available from the corresponding author on reasonable request.
This study was designed by SS and LL. XP, LKZ, ZXZ, YLX, ZHZ and MTX were responsible for data collation and statistical analysis. SS, LL, XP, LKZ and MTX wrote the first draft. ZXZ, YLX, ZHZ and YY reviewed it critically for important intellectual content. YY interpretated the data and reviewed the work. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was approved by the Ethics Committee of Fuwai Hospital (ethics approval number is 2017-950) and obeyed the Declaration of Helsinki. And a waiver of informed consent was granted because this study was retrospective.
Not applicable.
This research was funded by Clinical and Translational Medicine Research Project of Chinese Academy of Medical Sciences, grant number 2022-LC04.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/RCM25604.
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
