Academic Editor: Zhonghua Sun
This study aimed to prepare an anti-Vascular cell adhesion protein 1
(VCAM-1) nanoscale ultrasound microbubble contrast agent using the hyperbranched
self-assembly method for the molecular imaging diagnosis of atherosclerotic
vulnerable plaques in rabbits. Twenty-five rabbits with carotid atherosclerosis
were randomly divided into 5 groups, and the ear vein was injected with agents as
follows: Groups A and B: nanoscale ultrasound
microbubble contrast agent with and without anti-VCAM-1 agent; Groups C and D:
SonoVue ultrasonic microbubble contrast agent, with and without anti-VCAM-1
agent; Control group: saline. The molecular imaging diagnosis of the
atherosclerotic plaque, involved the examination of its vulnerability in the
rabbit carotid artery was performed using the contrast ultrasound mode. The
arrival and peaking time of the anti-VCAM-1 nanoscale ultrasound microbubble
contrast agent (Group A) for plaque occurred earlier than those of the other
groups (p
Stroke is the second leading cause of death due to cardiovascular and cerebrovascular diseases globally. Previous studies have reported that two important causes of ischemic stroke are occlusive stenosis and plaque rupture with associated thrombosis [1, 2, 3]. At present, there is no effective method for evaluating the susceptibility of atherosclerotic plaques to rupture [4, 5]. Thin fibrous cap, large lipid-rich necrotic cores, neovascularization, high infiltration of inflammatory cells, and plaque-related thrombosis are important for judging the susceptibility of an atherosclerotic plaque to rupture [6]. Neovascularization runs through the entire course of atherosclerotic plaque formation and development. New blood vessels are mostly composed of monolayer endothelial cells characterized by the lack of tight connections, poor continuity of the basement membrane, and lack of connective tissue, which provides support around the blood vessels, so they are brittle and permeable [7]. Inflammatory cells and fatty substances are more likely to infiltrate the plaque through gaps in the new blood vessels, leading to an inflammatory response within the plaque [8]. When the plaque is too large, ischemia and hypoxia occur easily, and the rupture of new blood vessels leads to intra-plaque bleeding [9]. This leads to a rapid increase in the volume of plaque and damage or rupture of the plaque surface [10]. These rupture-prone plaques are called “vulnerable plaques”. Therefore, neovascularization is a landmark characteristic of susceptibility to vulnerable plaques, and the detection of neovascularization in plaques guides clinical treatment. Neovascularization is difficult to detect using commercially available micron-scale ultrasound contrast media because of the extremely small diameter and tortuosity of the endothelial cells. Qualitative diagnosis of vulnerable plaques is still difficult at this stage.
Vascular cell adhesion molecule (VCAM)-1 is expressed by activated endothelial cells and plays a role in leukocyte rolling and adhesion by interacting with its receptor very late antigen-4 (VLA-4) on monocytes and lymphocytes [11, 12]. It has been reported that inflammatory activation of the endothelium, involving upregulation of VCAM-1, promotes the recruitment of monocytes into the arterial wall, precedes plaque formation, and plays a role in the occurrence and progression of plaque [12, 13]. Therefore, imaging techniques that assess changes in VCAM-1 expression could be utilized to determine the risk of atherosclerosis-related events [14, 15]. However, the short circulation time (minutes) and relatively large size (two–ten micrometers) of currently used microbubbles do not allow effective extravasation into tissue, preventing efficient targeting [16, 17]. Therefore, this study aimed to investigate the binding ability of self-made nanoscale microbubbles and VCAM-1 and compare the effects of self-made nanoscale microbubble contrast agent and SonoVue contrast agent on plaque ultrasound imaging. Accurate diagnosis of vulnerable plaques based on cellular and molecular imaging is predicted to guide the long-term clinical management and treatment options for high-risk stroke patients.
Forty adult male New Zealand white rabbits aged 3–5 months, with large ears and body mass of 2.0–2.5 kg, were provided by the Shanghai Fengxian Tengda Rabbit Industry [SCXK (Shanghai) 2014-0003]. The rabbits were fed under natural light, with alternating day and night cycles and unlimited drinking water. Each rabbit was kept in one cage and singly fed. Modeling experiment group (34 rabbits) were provided with high-fat diet (formula: 74.5% regular feed, 10% lard, 0.5% cholesterol, 10% egg yolk powder, 5% sucrose); 100 g daily for each rabbit. The control group (6 rabbits) were fed normally.
34 rabbits were used to construct a rabbit carotid atherosclerotic plaque model. The rabbit carotid atherosclerosis model was established by referring to the method of using high-fat feeding and modified silicone rubber ring reported by Li et al. [18]. Briefly, the left common carotid arteries were separated from the surrounding connective tissue after rabbits were anesthetized with 3 percent sodium pentobarbital. The left common carotid artery was sheathed with a silicone collar, and the collar-opening was closed with an adhesive until the end of the experiment. The skin was sutured after the opening was firmly closed.
Blood (2 mL) of was sampled from the auricular vein at week 0, week 4, week 8,
and week 12 and centrifuged at a speed of 1118 g per 3–5 min at 4
Ultrasound examination of the common carotid artery in the rabbits was performed at 4, 8, and 12 weeks after surgery. The rabbits were anesthetized and immobilized, and the hair of the rabbits on the neck was shaved, with the head turned opposite to the side of the examination. The GE Vivid E9 ultrasound scanner (GE Healthcare Co., Milwaukee, WI, USA) with a line-of-array probe (8–10 MHz) was used for ultrasound examination. The scanning depth was adjusted to 3 cm, the grayscale gain was adjusted to 53%, and the two-dimensional ultrasound conditions were set to Mechanical index (MI) 1.2, Tis 0.6, Freq 4.0/8.0 MHz. Ultrasonic coupling agent was thickly coated as an in vitro sound transmission window. The common carotid artery was examined in the anterior followed by the lateral and transverse positions. The internal diameter of the carotid artery and the thickness of the medial membrane and the plaque were measured by local ZOOM. The size of the plaque, internal echo, smooth surface, and ulcer formation was observed, and the video was recorded. All the animal handling procedures and protocols were approved by the Animal Ethics Committee at East China Normal University.
To evaluate the histological characteristics of atherosclerotic plaques, the
left common carotid artery was taken from the rabbit for HE staining. The HE
staining was carried out in accordance with standard procedures. The left carotid
artery was fixed with 4% formaldehyde and embedded in paraffin. Then, the
paraffin-embedded tissue was cut into 5
Hydrophobic hyperbranched polyether hyperbranched
poly(3-ethyl-3-oxetanemethanol) (HBPO) was obtained from a cationic ring-opening
polymer using the “one-pot two-step method” [19, 20]. The ring-opening
polymerization of ethylene oxide with HBPO was further realized as core to obtain
the target product, HBPO-star- poly(ethylene oxide) (HBPO-star-PEO) (Fig. 1). Two
hundred mg of HBPO-star-PEO was added to 10 mL of phosphate buffered saline
(PBS, Sangon Biotech Co., Ltd., Shanghai, China) and stirred to
dissolve. Sulfur hexafluoride (SF6, Shanghai Mi Zheng Gas Co., Ltd., Shanghai,
China) was bubbled into the above solution. The ultrasonic cell disruptor
conditions for stirring were set under ventilation, as follows: ventilation speed
10 mL/min, temperature 25
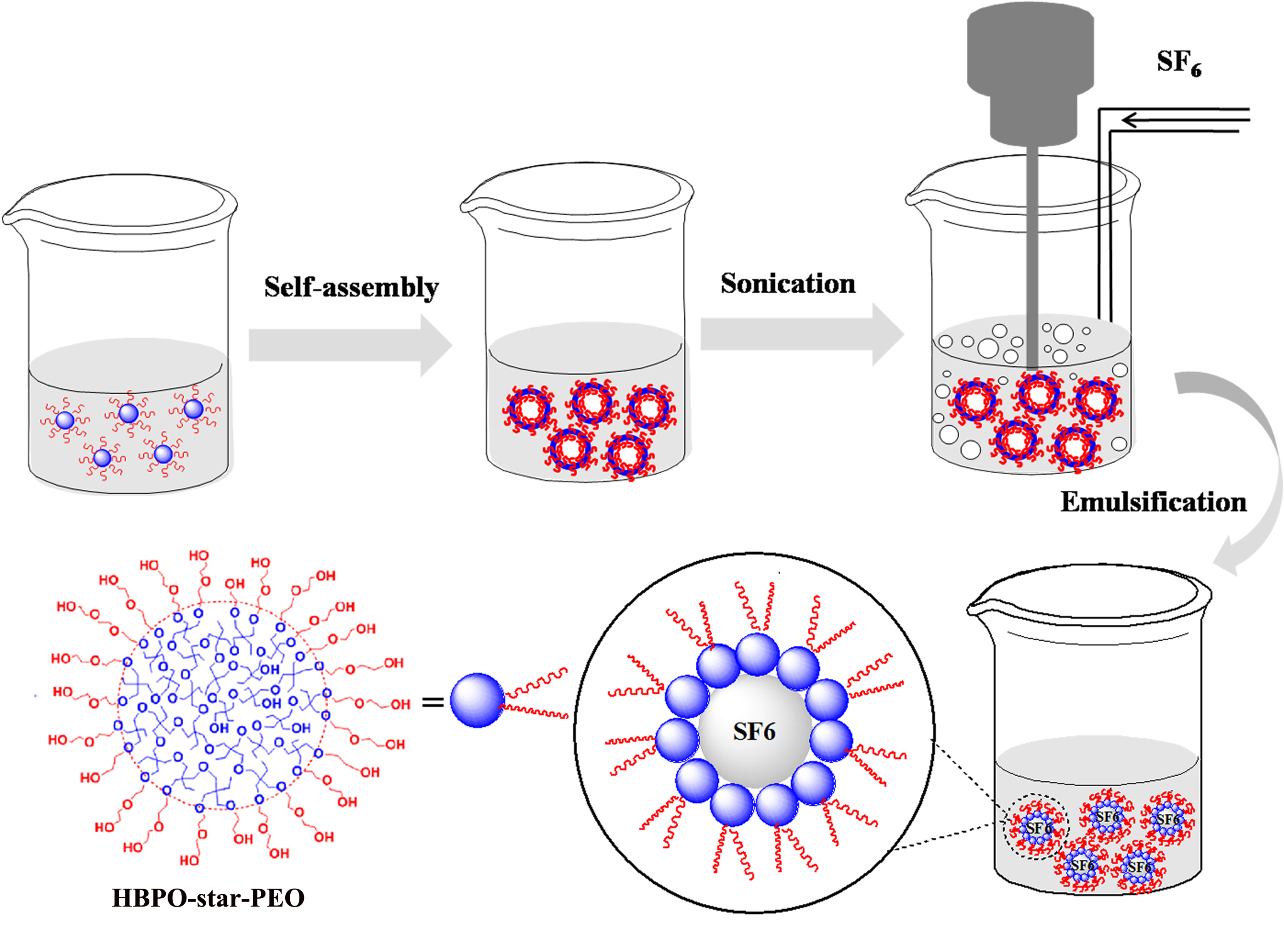 Fig. 1.
Fig. 1.Schematic diagram of sonic emulsification method. 200 mg of HBPO-star-PEO was added to 10 mL of phosphate buffered saline (10 mL/min) and stirred to dissolve. Sulfur hexafluoride was bubbled into the above solution, and the ultrasound was enhanced with an ultrasonic cell disruptor under ventilation.
The GE Vivid E9 ultrasound scanner (GE Healthcare Co., Milwaukee, Wisconsin, USA) with a line-of-array probe (8–10 MHz) was used for ultrasound examination. The condition was set to MI 1.5 and the image acquisition was performed in the Contrast mode. The peripheral venous channel was established by the ear veins, and the rabbit heart was scaned. First, the rabbit heart was scanned with ultrasound before injecting nano-scale ultrasound microbubbles and the images were recorded. Then, 1 mL of nanoscale ultrasound microbubbles were injected into the heart, and the needle was immediately flushed with 1 mL of saline to observe the perfusion of the heart.
The VCAM-1 antibody (CAT. No. Ab98954, Abcam, Cambridge, UK) was gradually
reacted with the fluorescein isothiocyanate (FITC, ab102884, Abcam, Cambridge,
UK) according to the manufacturer’s instructions to obtain the FITC/VCAM-1
fluorescence antibody. A PBS buffer was added and diluted to 1 mL, and the 500 uL
antibody diluent was absorbed and added into the self-made nanoscale microbubbles
suspension, incubated at room temperature for 30 min, the nanoscale microbubbles
are floated and the liquid under the nanoscale microbubbles is discarded. The
nanoscale microbubbles were washed with the PBS buffer several times until there
was clear and transparent in the lower aqueous solution, and the self-made
nanobubbles were directly combined with the VCAM-1 antibody to obtain the
anti-VCAM-1 nanometer ultrasonic nanoscale microbubbles. Fluorescent reagents and
SonoVue microbubbles were mixed to prepare fluorescence-labeled anti-VCAM-1
SonoVue microbubbles. The concentrations of the two types of microbubbles were
adjusted to 3
Twenty-five rabbits with carotid atherosclerotic plaques were randomly divided into five groups: Group A (n = 5): injected with 2 mL of the self-made anti-VCAM-1 nanometer ultrasonic microbubble contrast agent; Group B (n = 5): injected with 2 mL of the self-made nanoscale ultrasonic microbubble contrast agent; Group C (n = 5): injected with 2 mL of the anti-VCAM-1 + SonoVue ultrasonic microbubble contrast agent; Group D (n = 5): injected with 2 mL of the SonoVue ultrasonic microbubble contrast agent; Control group (n = 5): injected with 2 mL saline.
The experimental animals were anesthetized and immobilized, and their neck hair was shaved. The GE Vivid E9 ultrasonic diagnostic instrument (GE Healthcare Co., Milwaukee, WI, USA) and 9L linear array probe (8–10 MHz) (GE Healthcare Co., Milwaukee, WI, USA) were used for observation; two-dimensional ultrasound was used to identify the carotid artery before switching to the imaging model (MI 0.14, 0.0, Tis Freq 3.2/6.4 MHz). Contrast agents were injected into the marginal vein of the ear, and the entire contrast-agent filling process was observed and recorded.
To observe the development of the common carotid artery plaque and the lumen enhancement, 4 levels were classified based on the degree of enhancement of the contrast-enhanced ultrasound (CEUS): level I, plaques without enhancement; level II, a dot of enhancement of the plaque; level III, a short linear enhancement of the plaque; level IV, enhancement of plaques throughout or in most parts of the sample.
The CEUS image was exported, and a time-intensity curve was analyzed offline with the EchoPAC software v203 (GE Healthcare Co., Milwaukee, WI, USA). The carotid plaque area was selected as region of interest, and the arrival time, time to peak, basic intensity (BI), peak intensity (PI), and enhancement intensity (EI, EI = PI – BI) were analyzed and compared. The arrival time (in seconds) was defined as the first point of the curve clearly above the baseline intensity, followed by a rise. The time to peak (in seconds) was defined as the time from the start of the injection to the maximum intensity of the curve. The peak intensity (in decibels) was defined as the maximum intensity.
After the experiment, the rabbits were sacrificed, and immunohistochemical
staining was performed on the left common carotid artery. The paraffin-embedded
tissue was cut into sections with a thickness of 5
The data were expressed as mean
Thirty-four rabbits were used to construct rabbit carotid atherosclerotic plaque models, and 25 rabbits were successfully modeled. In 25 rabbits, blood lipid levels increased with feeding time, and the difference of total cholesterol, triglyceride, and low density lipoprotein concentrations between week 8 and 0 was statistically significant (Table 1). There was no statistical difference between week 12 and week 8 (Table 1).
| week 0 | week 4 | week 8 | week 12 | p-value | p-value | |
| week 8 vs. week 0 | week 12 vs. week 0 | |||||
| TC (mmol/L) | 2.36 |
6.13 |
9.62 |
9.39 |
t = –30.88, p |
t = –1.47, p = 0.16 |
| TG (mmol/L) | 1.95 |
5.86 |
9.85 |
10.33 |
t = –40.70, p = 0.00 | t = –0.89, p = 0.38 |
| LDL (mmol/L) | 1.70 |
5.56 |
8.98 |
9.41 |
t = –32.33, p = 0.00 | t = –1.77, p = 0.09 |
| IMT (mm) | 0.11 |
0.14 |
0.38 |
0.52 |
t = –24.96, p = 0.00 | t = –9.047, p = 0.00 |
| TC, total cholesterol; TG, triglyceride; LDL, Low Density Lipoprotein;
IMT, intima-media thickness. Note: Difference among groups was compared by one-way analysis of variance with. * indicates p | ||||||
In order to verify the presence of atherosclerotic plaques, HE staining and contrast-enhanced ultrasound (CEUS) examination were used. In the experimental groups, foam cells were observed under the lesioned intima. The surface fibrous cap structure was broken, the elastic fibers were damaged and thinned, the thickess of media was increased with augmanted smooth muscle cells number and presence of foamy cells, and plaques were observed protruding into the lumen (Fig. 2A). The rabbit endothelial cells in the control group were intact (Fig. 2B).
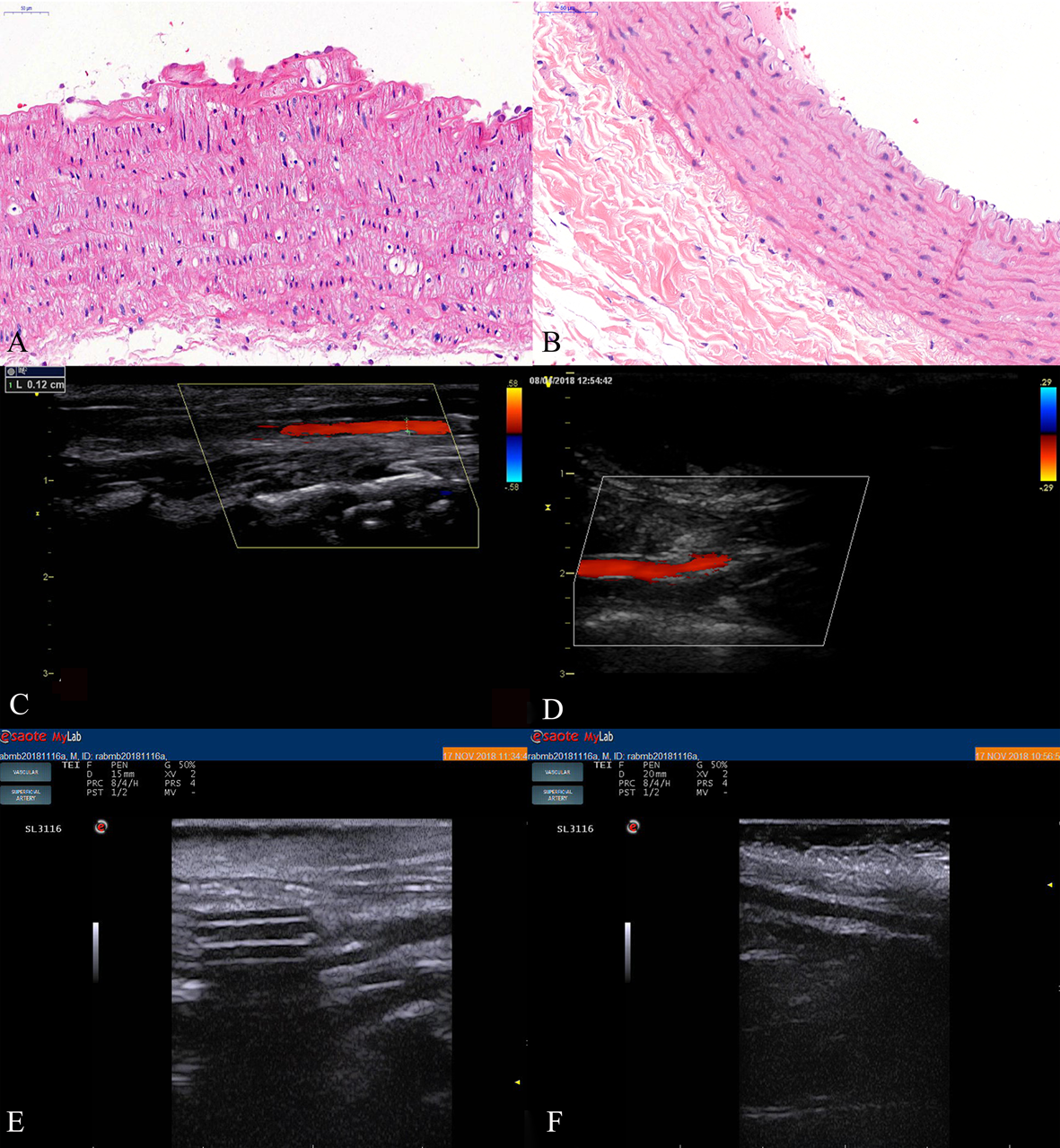 Fig. 2.
Fig. 2.Formation of vulnerable plaque in rabbit atherosclerosis. (A) HE
staining of carotid atherosclerotic plaque in rabbits. (B) HE staining of carotid
artery in rabbits of control group (400
CEUS showed that the carotid artery
intima-media thickness (IMT) was thickened 0.03 mm, and the color Doppler flow
signal was well-filled at week 4. At week 8, the carotid artery IMT was thickened
more, and some rabbits had small plaques protruding into the lumen. Color Doppler
flow imaging (CDFI) showed a filling defect of blood flow at the plaque at the
week 8 and week 12, and the diameter stenosis rate was less than 30% (Fig. 2C,D).
Compared with the preoperative findings, the difference was statistically
significant (t = 24.96, p
TEM and DLS data showed that the self-made nanoscale microbubbles have a
particle size of about 517.3
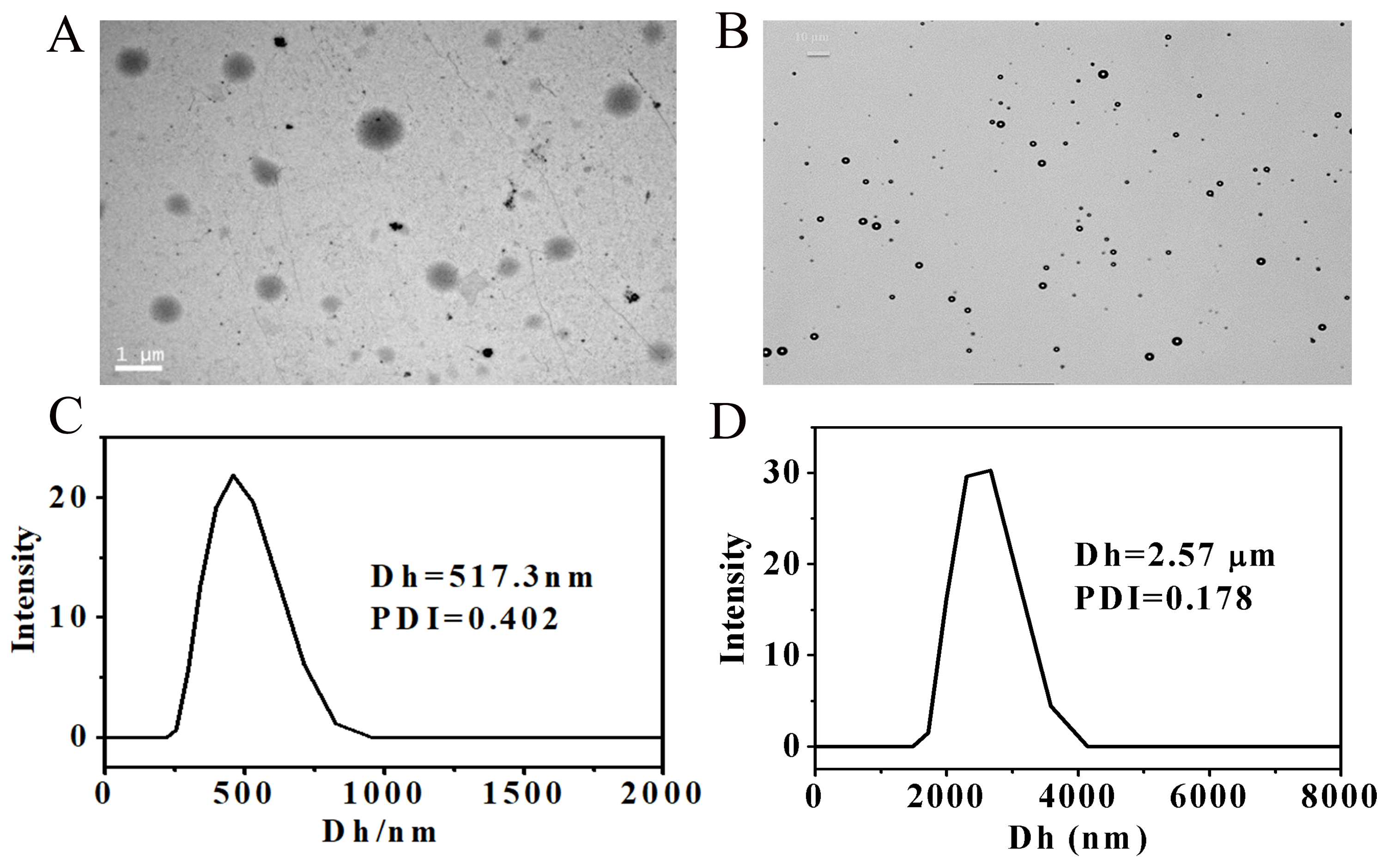 Fig. 3.
Fig. 3.Self-made microbubble particle size quantitative determination. (A) Self-made microbubble observed by transmission electron microscope. (B) SonoVue microbubble observed by transmission electron microscope. (C) The particle size of the self-made microbubbles is measured by DLS. (D) The particle size of the SonoVue microbubbles is measured by DLS.
Before the contrast agent was injected, the echo signal value inside the heart was very low, and its internal structure cannot be observed (Fig. 4A). After the nano-contrast agent was injected, the rabbit heart was quickly perfused, and the internal atrioventricular structure and valve activity were clearly visible (Fig. 4B). The average imaging time was 50–60 s, indicating that the self-assembled nanoscale ultrasound microbubble contrast agent hasa certain ability imaging in vivo.
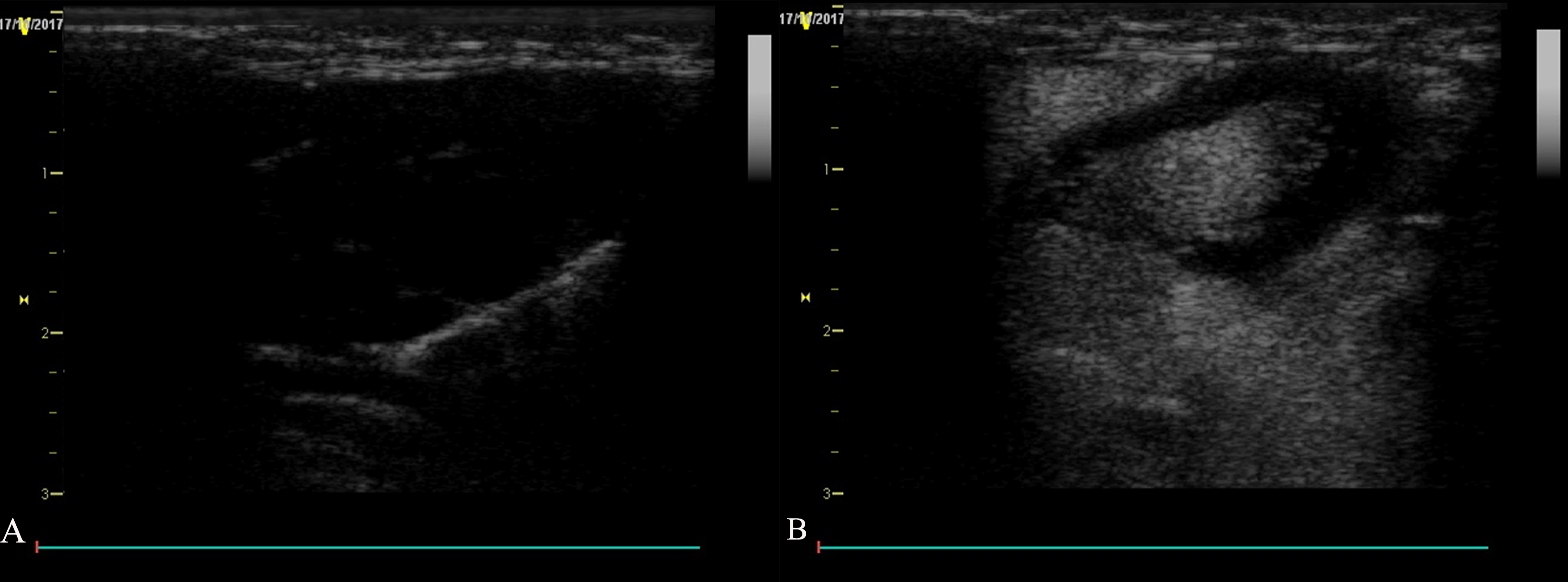 Fig. 4.
Fig. 4.Rabbit heart radiography. (A) Image before injection of nano microbubbles. (B) Image after injection of nano-microbubbles.
Under the same determination conditions, the fluorescence intensities of the
anti-VCAM-1 with a known fluorescence and the ultrasonic microbubble contrast
agent solution with an unknown fluorescence concentration of the VCAM-1 antibody
were measured, and the working curve of fluorescence intensity - concentration
was drawn. The concentration of the fluorescent anti-VCAM-1 ultrasound
microbubble contrast agent was calculated according to the working curve. The
antibody loads of the self-made microbubble contrast agent and SonoVue were 3.56
 Fig. 5.
Fig. 5.Quantitative analysis of antibodies carried by microbubbles. VCAM-1 load of the self-made microbubble contrast agent was measured using the fluorescence spectrophotometer.
After the injection of the ultrasound contrast agent, contrast agent signal was observed in the carotid lumen at 3 seconds after injection in the four groups except for the control group (Fig. 6). The arrival time inside the plaque was later than that of the the carotid lumen, and strengthening began at the plaque base and gradually progressed to the more internal portions or short linear strengthening, which was predominantly level II or level III. After the enhancement lasted for several minutes, the enhancement effect at the plaque gradually weakened, and the contrast agent gradually cleared.
 Fig. 6.
Fig. 6.Rabbit carotid artery contrast-enhanced ultrasound. (A) Image showing the anti-VCAM-1 nanometer contrast agent group with short linear enhancement in the plaque. (B) Image showing the nanoscale contrast agent group with dot enhancement in the plaques. (C) Anti-VCAM-1 sonovi group. (D) SonoVue group; dot enhancement was observed in all plaques. (E) Control group; no contrast agent sonogram is observed in the lumen.
There was a statistically significant difference in the arrival time of the
plaques between group A and group B (p
| Group | A | B | C | D | Control | F | p |
| Arrival time (s) | 9.15 |
10.06 |
10.93 |
10.31 |
- | 20.67 | |
| Time to peak (s) | 11.23 |
11.51 |
12.50 |
12.33 |
- | 13.72 | |
| Enhancement intensity (dB) | 11.50 |
10.50 |
5.99 |
6.88 |
- | 26.78 | |
In order to verify the results of the ultrasound examination, the left common carotid artery of the rabbit was collected and analyzed by immunohistological examination. Immunohistochemical detection results showed that the anti-VCAM-1 nanoscale ultrasound contrast agent group had brown staining of the surface and the internal portions of the carotid plaques. The anti-VCAM-1+SonoVue contrast group also showed minimal dyeing on the surface of the plaques; the other three groups did not express VCAM-1 in the vascular plaque. This indicates that the target ultrasound contrast agent against VCAM-1 may specifically bind to the plaque, and the combined effect of the anti-VCAM-1 nanometer ultrasound contrast agent group was the best (Fig. 7).
 Fig. 7.
Fig. 7.Immunohistochemical staining. (A) Immunohistochemical staining of
plaque in the anti-VCAM-1 nanocontrast agent group. (B) The nanometer contrast
agent group. (C) The anti-VCAM-1 SonoVue contrast agent group. (D) The SonoVue
contrast agent group. (E) The control group. Magnification 400
Atherosclerotic plaque rupture is the main cause of cardiovascular and cerebrovascular events, and the instability and vulnerability of plaques are important risk factors for major adverse cardiovascular events [22, 23]. The mechanism underlying the formation of vulnerable plaques is complex. Studies have found that the inflammatory response is related to the vulnerability of atherosclerotic plaques, and it persists throughout all stages of plaque progression. Plaque inflammation should have occurred in patients in a subclinical state [24, 25]. Neovascularization in atherosclerotic plaque is also a characteristic of vulnerable plaques. When the lipid core of the plaque is hypoxic, macrophages release hypoxic induction factors, which induce neovascularization [26]. Under normal circumstances, the oxygen in the vascular cavity diffuses to the wall of the vessel. When arterial plaque is formed, oxygen cannot diffuse to the adventitia of the blood vessel, and then neovascularization can offset the deficit of oxygen supply and demand [27]. New blood vessels can cause inflammatory cells and lipids to enter the plaque, thicken and enlarge the plaque, and aggravate the narrowing of the arterial lumen. Due to the lack of support from endothelial cells and vascular smooth muscle cells, blood vessels are highly permeable and brittle, and are prone to rupture and bleeding [28]. Therefore, CEUS is important for detecting neovascularization and vascular inflammation during the early stages.
The ideal commercial ultrasound contrast agent should meet the basic
requirements of small particle size, long in vivo half-life, a soft and
elastic shell of the contrast agent, good biocompatibility, and a simple
preparation method [29, 30]. With the development of ultrasonic molecular
imaging, contrast agents must bind specific antibodies or ligands to become
targeted contrast agents. Currently, molecular ultrasound imaging technology
based on targeted ultrasound contrast agents has become a research hotspot, and
scholars have applied this imaging technology to the evaluation of microvascular
endothelial inflammation [31]. With the introduction of new materials, nanoscale
ultrasound contrast agents have emerged. Nanoscale ultrasound contrast agents
have many advantages over microscale ultrasound contrast agents [32]. First,
nanoscale ultrasound contrast agents have stronger penetrating power, and
microbubbles with a particle size of
In previous experiments, we found that the hyperbranched polymer microbubble
preparation method was simple; the microbubble could be self-assembled into
vesicles in solution. Vesicles of various sizes can be prepared by changing the
self-assembly conditions, and the particle sizes can be freely adjusted from
nanometers to microns. Self-made vesicles have stable structures and properties
similar to those of biofilms [34, 35]. Most importantly, the hyperbranched
polymer terminal functional groups are abundant and can serve as binding sites
for carrying antibodies or genes [36, 37]. In this study, we prepared
microbubbles with a particle size of 517.3
At present, targeted ultrasound contrast agents have been used to study the cellular and molecular characteristics of ischemia-reperfusion injury, transplant rejection, thrombosis, tumor angiogenesis, and other diseases. Still, they have not been used to target the arterial system with high shear force [39, 40]. The immunohistochemical results of this study showed that the plaques of the anti-VCAM-1 contrast agent group were stained, and it can be concluded that the targeted microvesicles could specifically bind to the VCAM-1 targeted site on the wall of the common carotid artery. This may be because the blood flow in the artery is pulsed, and the diastolic blood flow is relatively slow, which provides a buffer time for the targeted antibody binding target sites and the loading of the contrast agent surface with a large and firm amount of the VCAM-1 antibody [14]. Therefore, the self-made small particle size anti-VCAM-1 targeted ultrasound microbubble contrast agent may be used to diagnose early inflammation of atherosclerotic plaques.
In this study, hyperplastic polymer HBPO-STAR-PEo/HBpo-star-PDMAEMA was
self-assembled and harmonically emulsified in solution to prepare an ultrasonic
contrast agent, with a particle size controlled at 517.3
Conception and design of the research—SLY; acquisition of data—CW, XXC; analysis and interpretation of data—CW, QQH; statistical analysis—CW, KZ, JH; drafting the manuscript—CW; revision of manuscript for important intellectual content—CW, SLY. All authors reviewed the manuscript.
All the animal handling procedures and protocols were carried out in accordance with relevant guidelines and regulations as well as in accordance with ARRIVE guidelines. All the animal handling procedures and protocols were approved by the Animal Ethics Committee at East China Normal University (Rb20200402).
We would like to express our gratitude to all those who helped us during the writing of this manuscript.
This study was funded by general program of Shanghai Municipal Health and Family Planning Commission (No. 201740053) and the guided project of Shanghai Municipal Science and Technology Commission (No. 18411970000).
The authors declare no conflict of interest.
The data analyzed in this study is available from the corresponding author upon reasonable request.
